Cloning and Expression Analysis of a PISTILLATA Homologous Gene from Pineapple (Ananas comosus L. Merr)
Abstract
:1. Introduction
2. Results and Discussion
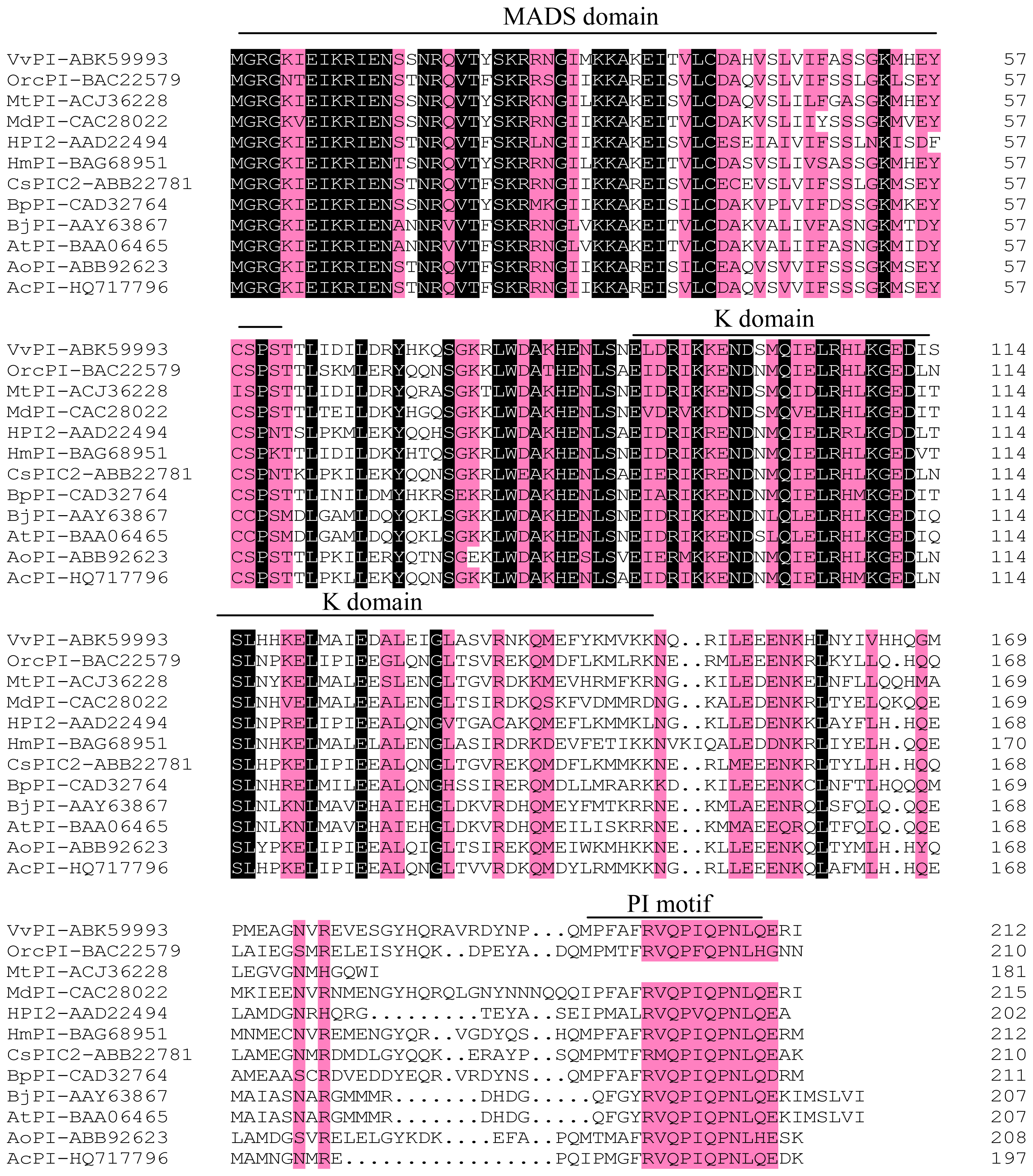
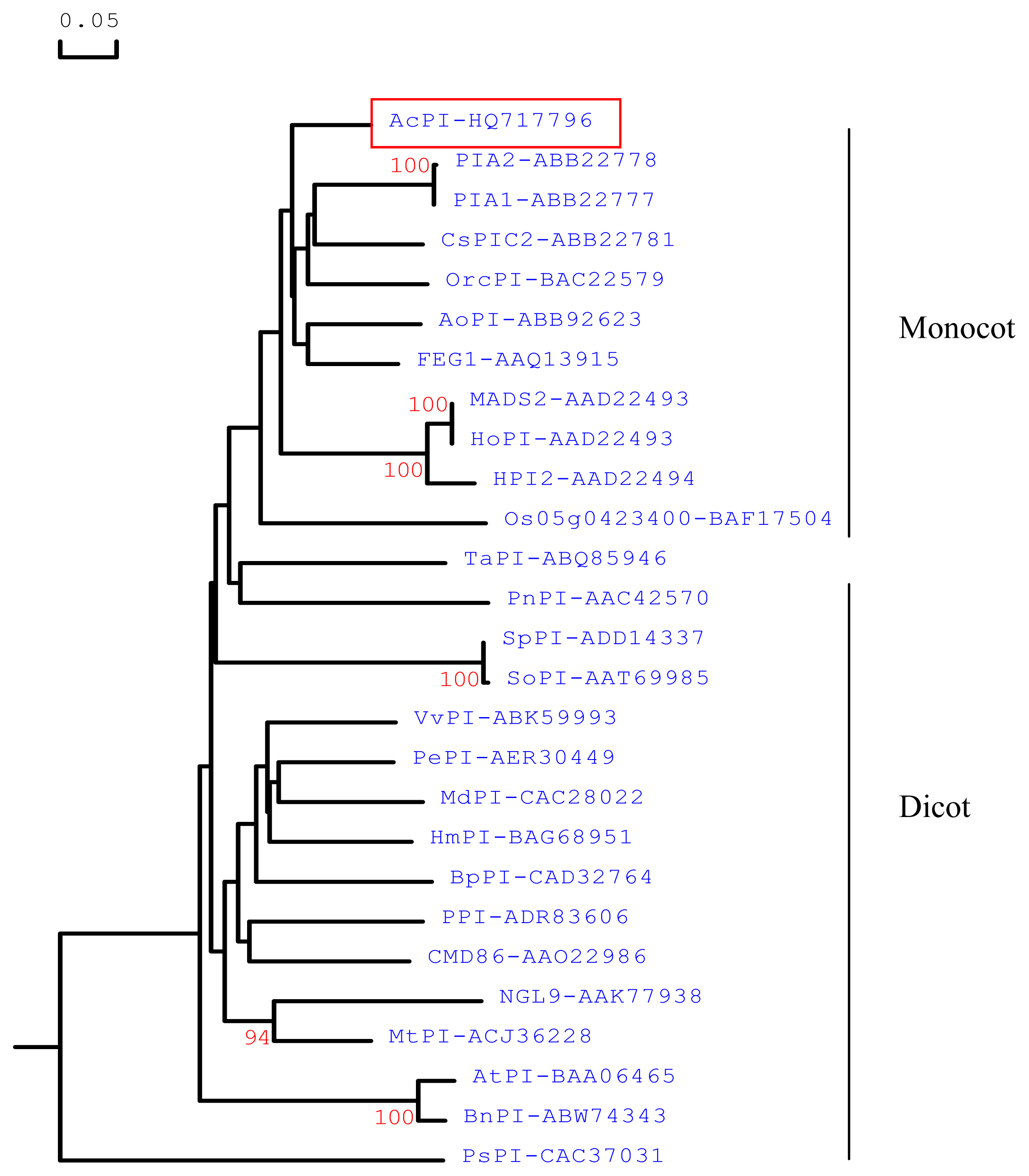
2.1. Cloning and Sequence Analyses of AcPI
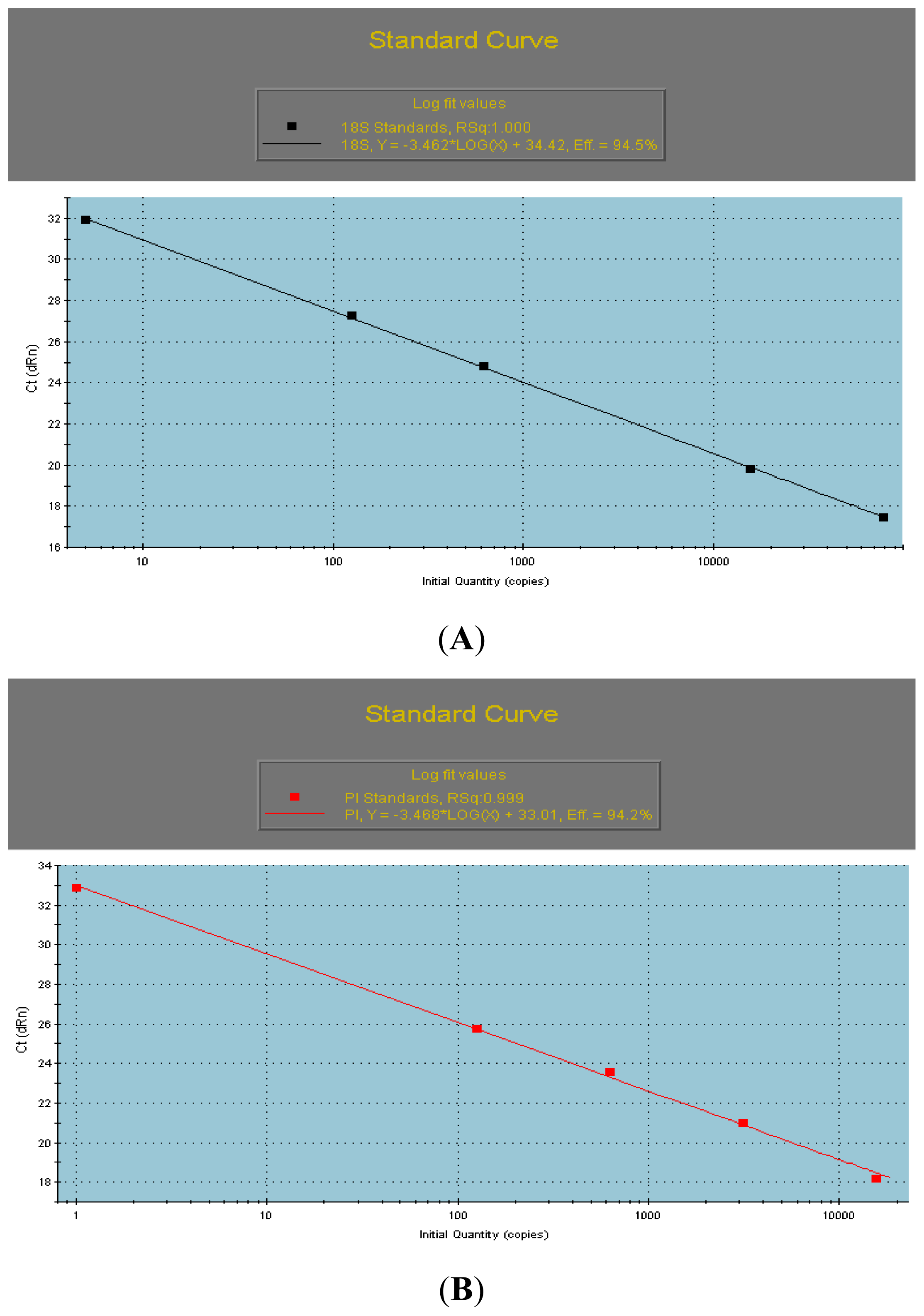
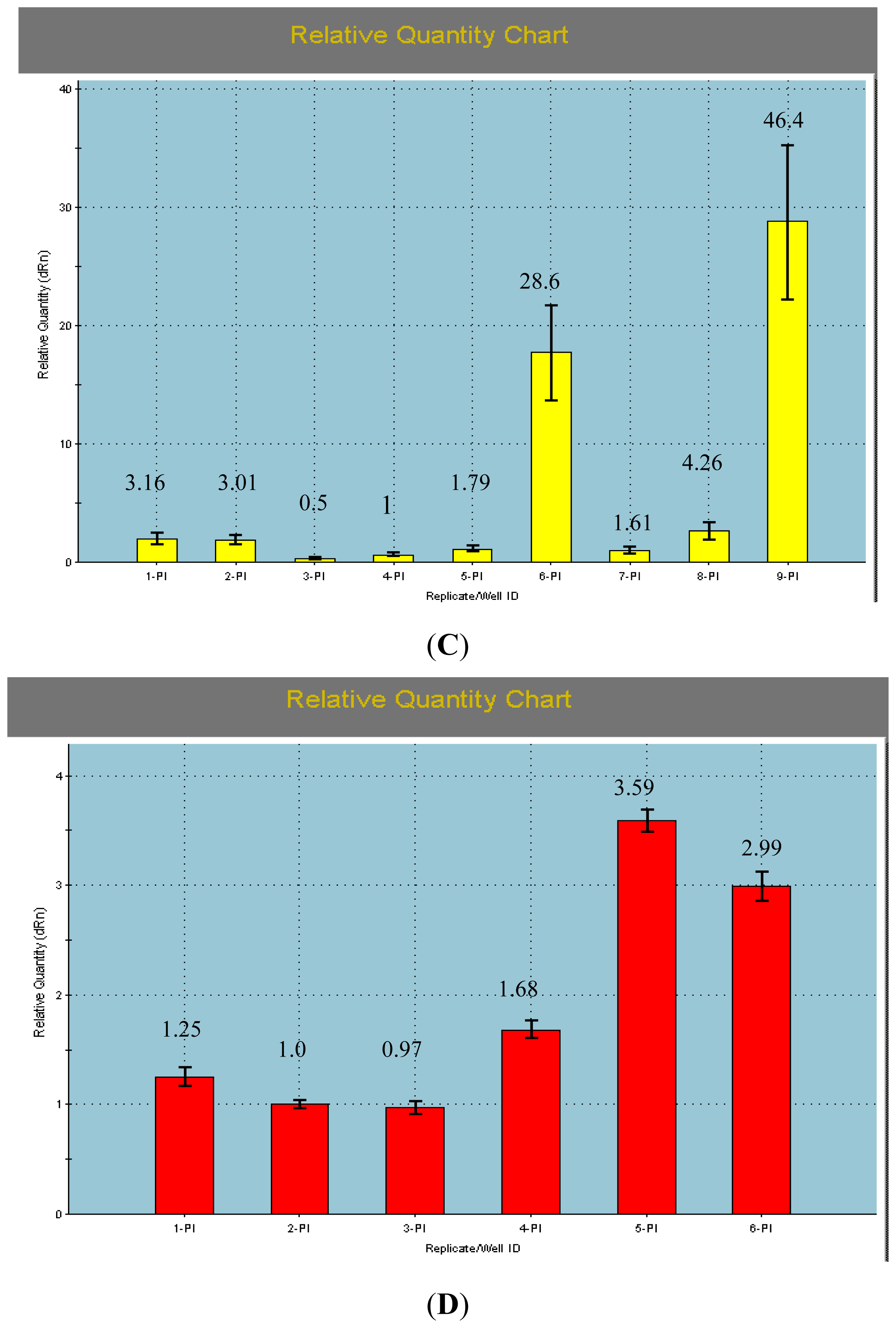
2.2. The qRT-PCR Analyses of AcPI
2.3. Ectopic Expression of AcPI in Arabidopsis
2.4. Discussion
3. Materials and Methods
3.1. Plant Materials and Treatments
3.2. Cloning of AcPI cDNAs
3.3. Sequence and Phylogenetic Analyses
3.4. qRT-PCR Analyses of the AcPI Gene
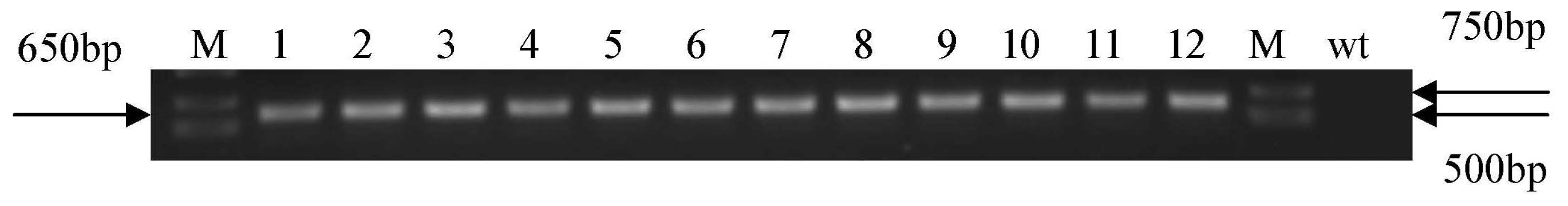
3.5. Arabidopsis Transformation and PCR Analysis of Transgenic Plants
4. Conclusions
Acknowledgments
- Conflict of InterestThe authors declare no conflict of interest.
References
- Meyerowitz, E.M.; Smyth, D.R.; Bowman, J.L. Abnormal flowers and pattern formation in floral development. Development 1989, 106, 209–217. [Google Scholar]
- Coen, E.S. The role of homeotic genes in floral development and evolution. Plant Mol. Biol 1991, 42, 241–279. [Google Scholar]
- Weigel, D.; Meyerowitz, E.M. The ABCs of floral homeotic genes. Cell 1994, 78, 203–209. [Google Scholar]
- Schwarz-Sommer, Z.; Huijser, P.; Nacken, W.; Saedler, H.; Sommer, H. Genetic control of flower development by homeotic genes in Antirrhinum majus. Science 1990, 250, 931–936. [Google Scholar]
- Bowman, J.L.; Smith, D.R.; Meyerowitz, E.M. Genetic interactions among floral homeotic genes of Arabidopsis. Development 1991, 112, 1–20. [Google Scholar]
- Coen, E.S.; Meyerowitz, E.M. The war of the whorls: Genetic interactions controlling flower development. Nature 1991, 353, 31–37. [Google Scholar]
- Sommer, H.; Beltrán, J.P.; Huijser, P.; Pape, H.; Lönnig, W.E.; Saedler, H.; Schwarz-Sommer, Z. Deficiens, a homeotic gene involved in the control of flower morphogenesis in Antirrhinum majus: The protein shows homology to transcription factors. EMBO J 1990, 9, 605–613. [Google Scholar]
- Goto, K.; Meyerowitz, E.M. Function and regulation of the Arabidopsis floral homeotic gene PISTILLATA. Genes Dev 1994, 8, 1548–1560. [Google Scholar]
- Becker, A.; Kaufmann, K.; Freialdenhoven, A.; Vincent, C.; Li, M.A.; Saedler, H.; Theissen, G. A novel MADS-box gene subfamily with a sister-group relationship to class B floral homeotic genes. Mol. Gen. Genom 2002, 266, 942–950. [Google Scholar]
- Bowman, J.L.; Smith, D.R.; Meyerowitz, E.M. Genes directing flower development in Arabidopsis. The Plant Cell 1989, 1, 37–52. [Google Scholar]
- Krizek, B.A.; Meyerowitz, E.M. The Arabidopsis homeotic genes APETALA3 and PISTILLATA are sufficient to provide the B class organ identity function. Development 1996, 122, 11–22. [Google Scholar]
- Ng, M.; Yanofsky, M.F. Function and evolution of the plant MADS-box gene family. Nat. Rev. Genet 2001, 2, 186–195. [Google Scholar]
- Shore, P.; Sharrocks, A.D. The MADS-box family of transcription factors. Eur. J. Biochem 1995, 229, 1–13. [Google Scholar]
- Theiben, G.; Strater, T.; Fisher, A.; Saedler, H. Structural characterization of chromosomal location and phylogenetic evaluation of two pairs of AGAMOUS-like MADS-box genes from maize. Gene 1995, 156, 155–166. [Google Scholar]
- Theiben, G.; Kim, J.T.; Saedler, H. Classification and phylogeny of the MADS-box multigene family suggest defined roles of MADS-box gene subfamilies in the morphological evolution of eukaryotes. J. Mol. Evol 1996, 43, 484–516. [Google Scholar]
- Chung, Y.Y.; Kim, S.R.; Kang, H.G.; Noh, Y.S.; Park, M.C.; Finkel, D.; Gynheung, A. Characterization of two rice MADS box genes homologous to GLOBOSA. Plant Sci 1995, 109, 45–56. [Google Scholar]
- Mena, M.; Mandel, M.A.; Lerner, D.R.; Yanofsky, M.F.; Schmidt, R.J. A characterization of the MADS-box gene family in maize. Plant J 1995, 8, 845–854. [Google Scholar]
- Kang, H.G.; Noh, Y.S.; Chung, Y.Y.; Costa, M.A.; An, K.; An, G. Phenotypic alterations of petal and sepal by ectopic expression of a rice MADS box gene in tobacco. Plant Mol. Biol 1995, 29, 1–10. [Google Scholar]
- Ambrose, B.A.; Lerner, D.R.; Ciceri, P.; Padilla, C.M.; Yanofsky, M.F.; Schmidt, R.J. Molecular and genetic analyses of the Silky1 gene reveal conservation in floral organ specification between eudicots and monocots. Mol. Cell 2000, 5, 569–579. [Google Scholar]
- Kyozuka, J.; Kobayashi, T.; Morita, M.; Shimamoto, K. Spatially and temporally regulated expression of rice MADS box genes with similarity to Arabidopsis calss A, B and C genes. Plant Cell Physiol 2000, 41, 710–718. [Google Scholar]
- Munster, T.; Wingen, L.U.; Faigl, W.; Werth, S.; Saedler, H.; Theissen, G. Characterization of three GLOBOSA-like MADS-box genes from maize: Evidence for ancient paralogy in one class of floral homeotic B-function genes of grasses. Gene 2001, 262, 1–13. [Google Scholar]
- Fornara, F.; Parenicova, L.; Falasca, G.; Pelucchi, N.; Masiero, S.; Ciannamea, S.; Lopez-Dee, Z.; Altamura, M.M.; Colombo, L.; Kater, M.M. Functional characterization of OsMADS18, a member of the AP1/SQUA subfamily of MADS-box genes. Plant Physiol 2004, 135, 2207–2219. [Google Scholar]
- Van Tunen, A.J.; Eikelboom, W.; Angenent, G.C. Floral organogenesis in Tulipa. Flower. Newsl 1993, 16, 33–38. [Google Scholar]
- Wu, X.P.; Shi, J.S.; Xi, M.L.; Luo, Z.W.; Hu, X.H. A B functional gene cloned from Lily encodes an ortholog of Arabidopsis PISTILLATA (PI). Plant Mol. Biol. Rep 2010, 28, 684–691. [Google Scholar]
- Guo, B.; Saiyin, Hexige; Zhang, T.; Jon, K. Pittman; Chen, D.H.; Ming, F. Cloning and characterization of a PI-like MADS-box gene in Phalaenopsis orchid. J. Biochem. Mol. Biol 2007, 40, 845–852. [Google Scholar]
- Kalivas, A.; Pasentsis, K.; Polidoros, A.N.; Tsaftaris, A.S. Heterotopic expression of B-class floral homeotic genes PISTILLATA/GLOBOSA supports a modified model for crocus (Crocus sativus L.) flower formation. DNA Seq 2007, 18, 120–130. [Google Scholar]
- Theissen, G. Development of floral organ identity: Stories from the MADS house. Curr. Opin. Plant Biol 2001, 4, 75–85. [Google Scholar]
- Richmann, J.L.; Meyerowitz, E.M. MADS domain proteins in plant development. Biol Chem 1997, 378, 1079–1101. [Google Scholar]
- Ma, H.; Yanofsky, M.F.; Meyerowitz, E.M. AGL1-AGL9, an Arabidopsis gene family with similarity to floral homeotic and transciption factor genes. Gene Dev 1991, 5, 484–495. [Google Scholar]
- Kramer, E.M.; Dorit, R.L.; Irish, V.F. Molecular evolution of genes controlling petal and stamen development: Duplication and divergence within the APETALA3 and PISTILLATA MADS box gene lineages. Genetics 1998, 149, 765–783. [Google Scholar]
- Yang, Y.; Fanning, L.; Jack, T. The K domain mediates heterodimerization of the Arabidopsis floral organ identity proteins, APETALA3 and PISTILLATA. Plant J 2003, 33, 47–59. [Google Scholar]
- Angenent, G.C.; Colombo, L. Molecular control of ovule development. Trends Plant Sci 1996, 1, 228–232. [Google Scholar]
- Kanno, A.; Saeki, H.; Kameya, T.; Saedler, H.; Theissen, G. Heterotopic expression of class B floral homeotic genes supports a modified ABC model for tulip (Tulipa gesneriana). Plant Mol. Biol 2003, 52, 831–841. [Google Scholar]
- Jack, T.; Brockman, L.L.; Meyerowitz, E.M. The homeotic gene APETALA3 of Arabidopsis thaliana encodes a MADS box and is expressed in petals and stamens. Cell 1992, 68, 683–697. [Google Scholar]
- Schwarz-Sommer, Z.; Hue, I.; Huijser, P.; Flor, P.J.; Hansen, R.; Tetens, F.; Lönnig, W.E.; Saedler, H.; Sommer, H. Characterization of the Antirrhinum floral homeotic MADS-box gene deficiens: Evidence for DNA binding and autoregulation of its persistent expression throughout flower development. EMBO J 1992, 11, 251–263. [Google Scholar]
- Tröbner, W.; Ramirez, L.; Motte, P.; Hue, I.; Huijser, P.; Lönnig, W.E.; Saedler, H.; Sommer, H.; Schwarz-Sommer, Z. GLOBOSA: A homeotic gene which interacts with DEFICIENS in the control of Antirrhinum floral organogenesis. EMBO J 1992, 11, 4693–4704. [Google Scholar]
- Busi, M.; Victoria; Bustamante, C.; Angelo, C.; Hidalgo-Cuevas, M.; Boggio, S.B.; Valle, E.M.; Zabaleta, E. MADS-box genes expressed during tomato seed and fruit development. Plant Mol. Biol 2003, 52, 801–815. [Google Scholar]
- Kim, S.T.; Koh, J.; Yoo, M.J.; Kong, H.Z.; Hu, Y.; Ma, H.; Soltis, P.S.; Soltis, D.E. Expression of floral MADS-box genes in basal angiosperms: Implications for the evolution of floral regulators. Plant J 2005, 43, 724–744. [Google Scholar]
- Turnbull, C.G.; Sinclair, E.R.; Anderson, K.L.; Nissen, R.J.; Shorter, A.J.; Lanham, T.E. Routes of ethephon uptake in pineapple (Ananas comosus) and reasons for failure of flower induction. J. Plant Growth Regul 1999, 18, 145–152. [Google Scholar]
- BLAST Home Page. Available online: http://www.ncbi.nlm.nih.gov/BLAST.cgi accessed 18 January 2012.
- Compute pI/Mw Tool. Available online: http://web.expasy.org/compute_pi/ accessed 18 January 2012.
- Chen, H.; Nelson, R.S.; Sherwood, J.L. Enhanced recovery of transformants of Agrobacterium tumefaciens after freeze-thaw transformation and drug selection. Biotechniques 1994, 16, 664–670. [Google Scholar]
- Clough, S.J.; Bent, A.F. Floral dip: A simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J 1998, 16, 735–743. [Google Scholar]

© 2012 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Lv, L.-L.; Duan, J.; Xie, J.-H.; Liu, Y.-G.; Wei, C.-B.; Liu, S.-H.; Zhang, J.-X.; Sun, G.-M. Cloning and Expression Analysis of a PISTILLATA Homologous Gene from Pineapple (Ananas comosus L. Merr). Int. J. Mol. Sci. 2012, 13, 1039-1053. https://doi.org/10.3390/ijms13011039
Lv L-L, Duan J, Xie J-H, Liu Y-G, Wei C-B, Liu S-H, Zhang J-X, Sun G-M. Cloning and Expression Analysis of a PISTILLATA Homologous Gene from Pineapple (Ananas comosus L. Merr). International Journal of Molecular Sciences. 2012; 13(1):1039-1053. https://doi.org/10.3390/ijms13011039
Chicago/Turabian StyleLv, Ling-Ling, Jun Duan, Jiang-Hui Xie, Yu-Ge Liu, Chang-Bin Wei, Sheng-Hui Liu, Jian-Xia Zhang, and Guang-Ming Sun. 2012. "Cloning and Expression Analysis of a PISTILLATA Homologous Gene from Pineapple (Ananas comosus L. Merr)" International Journal of Molecular Sciences 13, no. 1: 1039-1053. https://doi.org/10.3390/ijms13011039





