Characterization of Thermophilic Halotolerant Aeribacillus pallidus TD1 from Tao Dam Hot Spring, Thailand
Abstract
:1. Introduction
2. Results and Discussion
3. Experimental Section
3.1. Isolation of Bacterial Strains
3.2. Pectate Lyase Assay
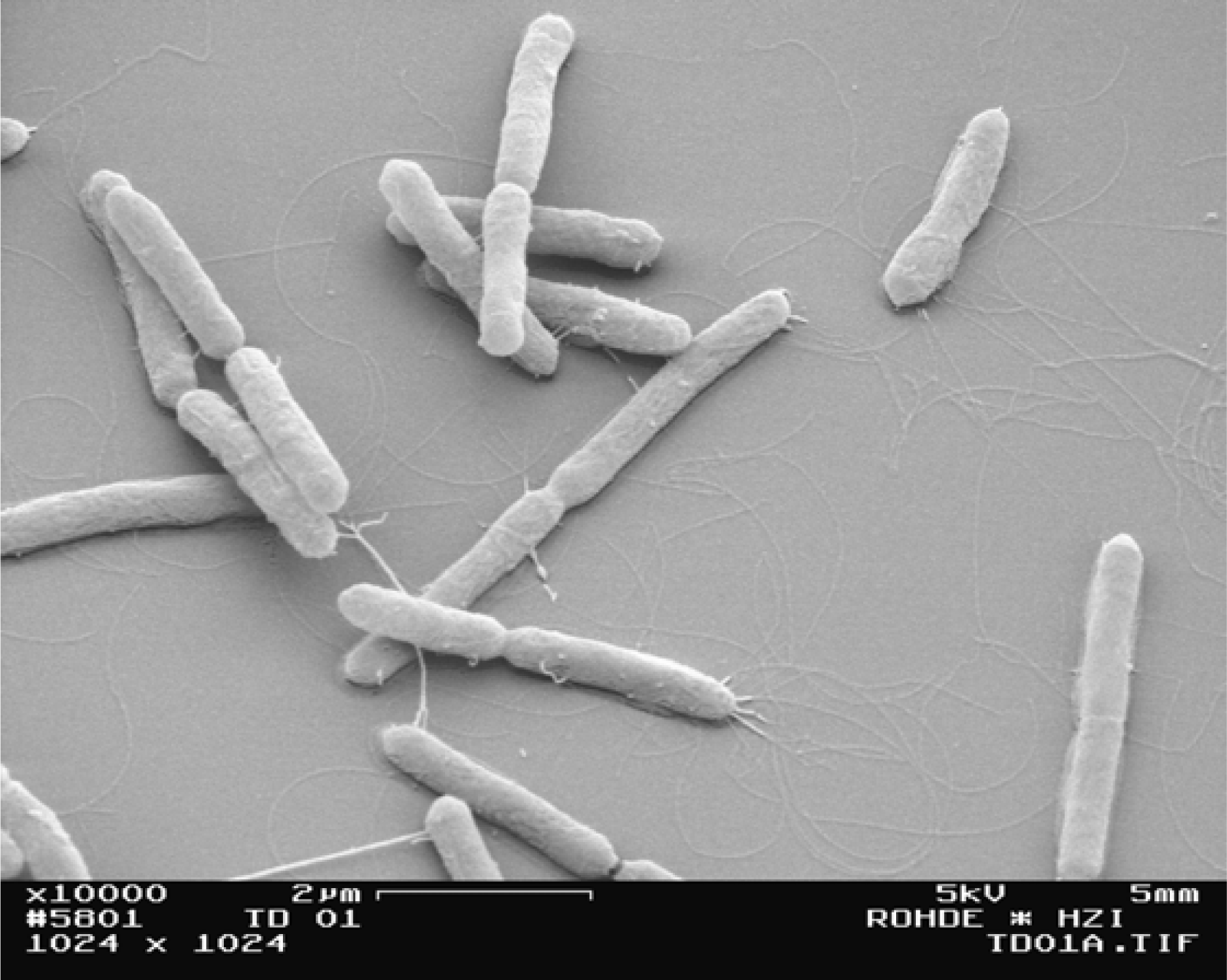
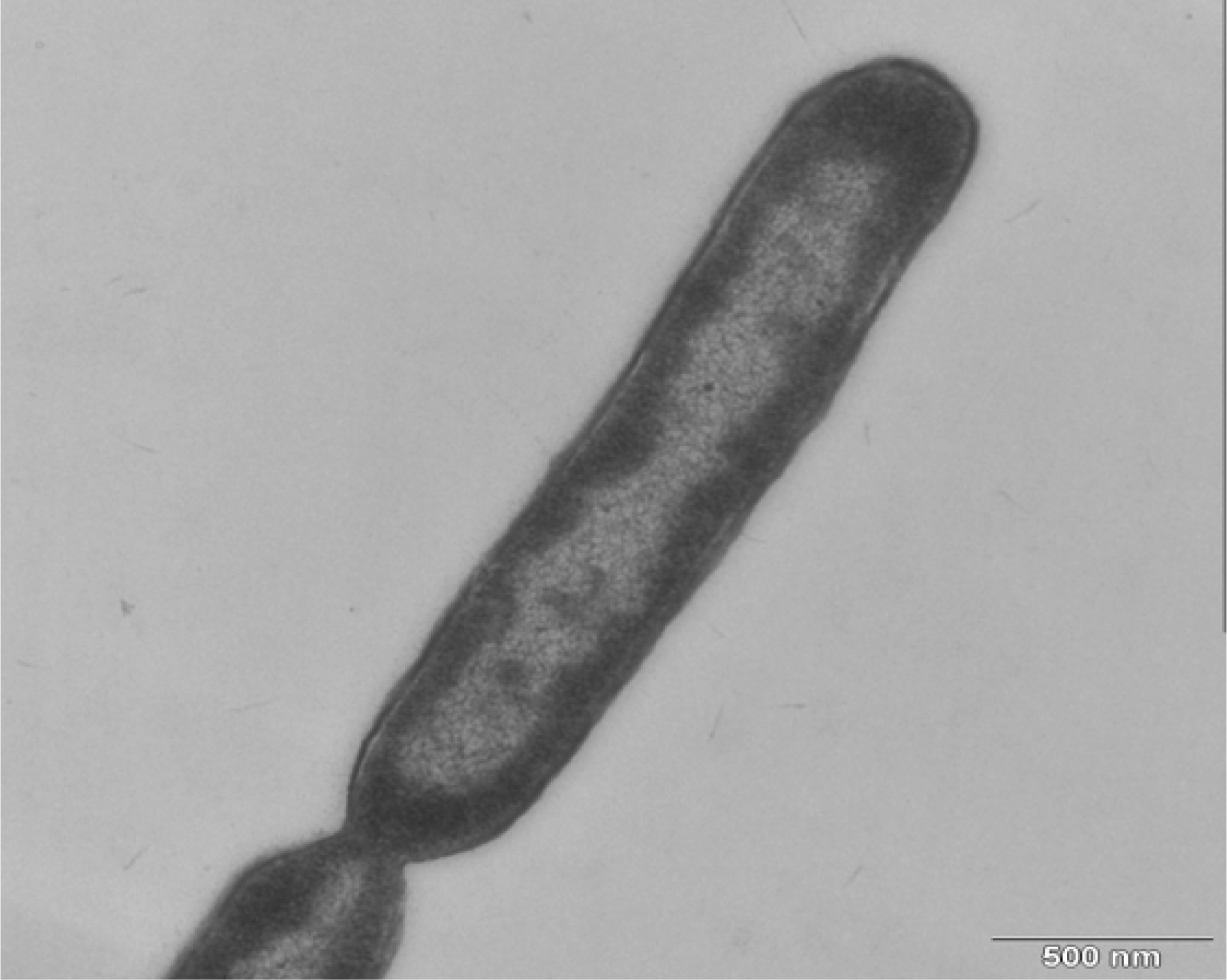
3.3. Morphology
3.4. Phenotypic Characterization
3.5. Chemotaxonomy
3.6. Molecular Systematics
4. Conclusions
Acknowledgments
References
- Turner, P; Mamo, G; Karlsson, EN. Potential and utilization of thermophiles and thermostable enzymes in biorefining. Microb. Cell Fact 2007, 6, 9. [Google Scholar]
- Niehaus, F; Bertoldo, C; Kahler, M; Antranikian, G. Extremophiles as a source of novel enzymes for industrial application. Appl. Microbiol. Biotechnol 1999, 51, 711–729. [Google Scholar]
- Carpita, NC; Gibeaut, DM. Structural models of primary cell walls in flowering plants: consistency of molecular structure with the physical properties of the walls during growth. Plant J 1993, 3, 1–30. [Google Scholar]
- Hoondal, GS; Tiwari, RP; Tewari, R; Dahiya, N; Beg, QK. Microbial alkaline pectinases and their industrial applications: A review. Appl. Microbiol. Biotechnol 2002, 59, 409–418. [Google Scholar]
- Bruhlmann, F. Purification and characterization of an extracellular pectate lyase from an Amycolata sp. Appl. Environ. Microbiol 1995, 61, 3580–3585. [Google Scholar]
- Kobayashi, T; Hatada, Y; Higaki, N; Lusterio, DD; Ozawa, T; Koike, K; Kawai, S; Ito, S. Enzymatic properties and deduced amino acid sequence of a high-alkaline pectate lyase from an alkaliphilic Bacillus isolate. Biochim. Biophys. Acta 1999, 1427, 145–154. [Google Scholar]
- Kobayashi, T; Higaki, N; Suzumatsu, A; Sawada, K; Hagihara, H; Kawai, S; Ito, S. Purification and properties of a high-molecular-weight, alkaline exopolygalacturonase from a strain of Bacillus. Enzyme Microb. Technol 2001, 29, 70–75. [Google Scholar]
- Sawada, K; Suzumatsu, A; Kobayashi, T; Ito, S. Molecular cloning and sequencing of the gene encoding an exopolygalacturonase of a Bacillus isolate and properties of its recombinant enzyme. Biochim. Biophys. Acta 2001, 1568, 162–170. [Google Scholar]
- Blanco, P; Sieiro, C; Villa, TG. Production of pectic enzymes in yeasts. FEMS Microbiol. Lett 1999, 175, 1–9. [Google Scholar]
- Guo, W; Gonzalez-Candelas, L; Kolattukudy, PE. Cloning of a novel constitutively expressed pectate lyase gene pelB from Fusarium solani f. sp. pisi (Nectria haematococca, mating type VI) and characterization of the gene product expressed in Pichia pastoris. J. Bacteriol 1995, 177, 7070–7077. [Google Scholar]
- Huertas-Gonzalez, MD; Ruiz-Roldan, MC; Garcia Maceira, FI; Roncero, MI; Di Pietro, A. Cloning and characterization of pl1 encoding an in planta-secreted pectate lyase of Fusarium oxysporum. Curr. Genet 1999, 35, 36–40. [Google Scholar]
- Ouattara, HG; Reverchon, S; Niamke, SL; Nasser, W. Biochemical properties of pectate lyases produced by three different Bacillus strains isolated from fermenting cocoa beans and characterization of their cloned genes. Appl. Environ. Microbiol 2010, 76, 5214–5220. [Google Scholar]
- Sukhumsiirchart, W; Kawanishi, S; Deesukon, W; Chansiri, K; Kawasaki, H; Sakamoto, T. Purification, characterization, and overexpression of thermophilic pectate lyase of Bacillus sp. RN1 isolated from a hot spring in Thailand. Biosci. Biotechnol. Biochem 2009, 73, 268–2673. [Google Scholar]
- Tonouchi, A; Hara, Y; Umehara, R; Sanuki, T; Fukusawa, T; Miyairi, K. Cloning of the gene encoding an endo-acting pectate lyase from Streptomyces thermocarboxydus. Biosci. Biotechnol. Biochem 2010, 74, 433–436. [Google Scholar]
- Scholz, T; Demharter, W; Hensel, R; Kandler, O. Bacillus pallidus sp. nov., a new thermophilic species from sewage. Syst. Appl. Microbiol 1987, 9, 91–96. [Google Scholar]
- Euzeby, JP. List of Bacterial Names with Standing in Nomenclature: A folder available on the Internet. Int. J. Syst. Bacteriol 1997, 47, 590–592. [Google Scholar]
- Banat, IM; Marchant, R; Rahman, TJ. Geobacillus debilis sp. nov., a novel obligately thermophilic bacterium isolated from a cool soil environment, and reassignment of Bacillus pallidus to Geobacillus pallidus comb. nov. Int. J. Syst. Evol. Microbiol 2004, 54, 2197–2201. [Google Scholar]
- Minana-Galbis, D; Pinzon, DL; Loren, JG; Manresa, A; Oliart-Ros, RM. Reclassification of Geobacillus pallidus (Scholz et al. 1988) Banat et al. 2004 as Aeribacillus pallidus gen. nov., comb. nov. Int. J. Syst. Evol. Microbiol 2010, 60, 1600–1604. [Google Scholar]
- Collmer, A; Ried, JL; Mount, MS. Assay methods for pectic enzymes. Methods Enzymol 1988, 161, 329–335. [Google Scholar]
- Spurr, AR. A low-viscosity epoxy resin embedding medium for electron microscopy. J. Ultrastruct. Res 1969, 26, 31–43. [Google Scholar]
- Lane, D. 16S/23S rRNA Sequencing. In Nucleic Acid Techniques in Bacterial Systematics; Stackebrandt, E, Goodfellow, M, Eds.; John Wiley & Sons: New York, NY, USA, 1991; pp. 115–175. [Google Scholar]
- Altschul, SF; Gish, W; Miller, W; Myers, EW; Lipman, DJ. Basic local alignment search tool. J. Mol. Biol 1990, 215, 403–410. [Google Scholar]
- Edgar, RC. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res 2004, 32, 1792–1797. [Google Scholar]
- Posada, D; Crandall, KA. MODELTEST: Testing the model of DNA substitution. Bioinformatics 1998, 14, 817–818. [Google Scholar]
- Ronquist, F; Huelsenbeck, JP. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 2003, 19, 1572–1574. [Google Scholar]
- Rambaut, A; Drummond, A. Tracer v1.4. 2007. Available online: http://beast.bio.ed.ac.uk/Tracer (accessed on 9 August 2011).

| Characteristic | A. pallidus TD1 | A. pallidus DSM 3670T |
|---|---|---|
| Cell width (μm) | 0.4 | 0.8–0.9 |
| Cell length (μm) | 2–>40 | 2–5 |
| Motility | + | + |
| Temperature range (°C) | 45–67 | 30–70 |
| DNA G+C content (mol%) | 38.9 | 39–41 |
| Producing of pectate lyase | + | − |
| Acid produced from: | ||
| Cellobiose | + | − |
| Maltose | + | d |
| Mannose | + | − |
| Sucrose | + | ND |
| Trehalose | + | d |
| Xylose | + | − |
| Arabinose | − | − |
| Ribose | + | − |
| Citrated used | + | − |
| Hydrolysis of: | ||
| Casein | − | − |
| Gelatin | − | − |
| Strach | − | +w |
| Alkane utilization | − | ND |
| Fatty Acid | A. pallidus TD1 (%) | A. pallidus DSM 3670T (%) |
|---|---|---|
| 14:0 ISO | 0.36 | 1.6 |
| 14:0 ANTEISO | – | 1.6 |
| 14:0 | 1.87 | 8.5 |
| 15:0 ISO | 16.30 | 6.2 |
| 15:0 ANTEISO | 4.50 | 4.9 |
| 15:0 | 0.33 | 1.2 |
| 16:1 w7c alcohol | 0.14 | – |
| 16:0 ISO | 11.15 | 9.3 |
| 16:1 w11c | 0.65 | – |
| 16:0 | 25.04 | 50.0 |
| ISO 17:1 w10c | 0.30 | – |
| 17:0 ISO | 17.95 | 4.0 |
| 17:0 ANTEISO | 19.74 | 6.5 |
| 17:0 | 0.19 | – |
| 18:0 ISO | 0.29 | – |
| 18:0 | 1.03 | 2.1 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yasawong, M.; Areekit, S.; Pakpitchareon, A.; Santiwatanakul, S.; Chansiri, K. Characterization of Thermophilic Halotolerant Aeribacillus pallidus TD1 from Tao Dam Hot Spring, Thailand. Int. J. Mol. Sci. 2011, 12, 5294-5303. https://doi.org/10.3390/ijms12085294
Yasawong M, Areekit S, Pakpitchareon A, Santiwatanakul S, Chansiri K. Characterization of Thermophilic Halotolerant Aeribacillus pallidus TD1 from Tao Dam Hot Spring, Thailand. International Journal of Molecular Sciences. 2011; 12(8):5294-5303. https://doi.org/10.3390/ijms12085294
Chicago/Turabian StyleYasawong, Montri, Supatra Areekit, Arda Pakpitchareon, Somchai Santiwatanakul, and Kosum Chansiri. 2011. "Characterization of Thermophilic Halotolerant Aeribacillus pallidus TD1 from Tao Dam Hot Spring, Thailand" International Journal of Molecular Sciences 12, no. 8: 5294-5303. https://doi.org/10.3390/ijms12085294




