Flavonoid Analyses and Antimicrobial Activity of Various Parts of Phaleria macrocarpa (Scheff.) Boerl Fruit
Abstract
:1. Introduction
2. Results and Discussion
2.1. Flavonoid Compounds Analyses
2.2. Antimicrobial Activity
3. Experimental Methods
3.1. Plant Materials
3.2. Extraction
3.3. Determination of Flavonoid Compounds by HPLC
3.4. Antimicrobial Activity
3.4.1. Bacteria and Fungi Cultures
3.4.2. Bacterial Susceptibility Testing
3.4.3. Fungi Susceptibility Testing
4. Conclusions
Acknowledgements
References
- Backer, C; van den Brink, R. Flora of Java (Spermatophytes Only); Noordhoff; Groningen, The Netherlands, 1965; Volume II. [Google Scholar]
- Steffan, B; Watjen, W; Michels, G; Niering, P; Wray, V; Ebel, R; Edrada, R; Kahl, R; Proksch, P. Polyphenols from plants used in traditional Indonesian medicine (Jamu): Uptake and antioxidative effects in rat H4IIE hepatoma cells. J. Pharm. Pharmacol 2005, 57, 233–240. [Google Scholar]
- Zhang, Y; Xu, X; Liu, H. Chemical constituents from Mahkota dewa. J. Asian Nat. Prod. Res 2006, 8, 119–123. [Google Scholar]
- Oshimi, S; Zaima, K; Matsuno, Y; Hirasawa, Y; Iizuka, T; Studiawan, H; Indrayanto, G; Zaini, NC; Morita, H. Studies on the constituents from the fruits of Phaleria macrocarpa. J. Nat. Med 2008, 62, 207–210. [Google Scholar]
- Hakim, R; Nawawi, A; Adnyana, I; Achmad, S; Makmur, E; Hakim, E; Syah, Y; Kitajima, M. Benzophenone glucoside from red fruit Phaleria macrocarpa and test the activity against DPPH and murine leukemia cells P-388. Bull. Soc. Nat. Prod. Chem 2004, 4, 67–70. [Google Scholar]
- Kusmaardiyani, S; Nawawi, A; Rahmi, K. Isolation of benzophenone from Phaleria macrocarpa (Scheff.) Boerl. (Thymelaeaceae) leaf. Acta Pharm. Indones 2004, 29, 150–152. [Google Scholar]
- Ao, C; Li, A; Elzaawely, AA; Xuan, TD; Tawata, S. Evaluation of antioxidant and antibacterial activities of Ficus microcarpa L. fil. extract. Food Control 2008, 19, 940–948. [Google Scholar]
- Cragg, G; Newman, D; Snader, K. Natural products in drug discovery and development. J. Nat. Prod 1997, 60, 52–60. [Google Scholar]
- Manian, R; Anusuya, N; Siddhuraju, P; Manian, S. The antioxidant activity and free radical scavenging potential of two different solvent extracts of Camellia sinensis (L.) O. Kuntz, Ficus bengalensis L. and Ficus racemosa L. Food Chem 2008, 107, 1000–1007. [Google Scholar]
- Cowan, M. Plant products as antimicrobial agents. Clin. Microbiol. Rev 1999, 12, 564. [Google Scholar]
- Cushnie, TPT; Lamb, AJ. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Agents 2005, 26, 343–356. [Google Scholar]
- Wang, H; Helliwell, K. Determination of flavonols in green and black tea leaves and green tea infusions by high-performance liquid chromatography. Food Res. Int 2001, 34, 223–227. [Google Scholar]
- Hakkinen, S; Karenlampi, S; Heinonen, I; Mykkanen, H; Torronen, A. Content of the flavonols quercetin, myricetin, and kaempferol in 25 edible berries. J. Agr. Food. Chem 1999, 47, 2274–2279. [Google Scholar]
- De Lourdes Mata Bilbao, M; Andrés-Lacueva, C; Jáuregui, O; Lamuela-Raventós, RM. Determination of flavonoids in a Citrus fruit extract by LC-DAD and LC-MS. Food Chem 2007, 101, 1742–1747. [Google Scholar]
- Kumar, B; Lakshman, K; Jayaveera, K; Krishna, N; Manjunath, M; Suresh, M. Estimation of rutin and quercetin in Amaranthus viridis Linn by HPLC. Asian J. Exp. Sci 2009, 23, 51–54. [Google Scholar]
- Crozier, A; Jensen, E; Lean, MEJ; McDonald, MS. Quantitative analysis of flavonoids by reversed-phase high-performance liquid chromatography. J. Chromatogr. A 1997, 761, 315–321. [Google Scholar]
- Othman, F. Chemical Constituents and Biological Activities of Flavonoids from Hydroponically Grown Pegaga (Centella asiatica, Linn. Urban) Extracts; Universiti Putra Malaysia: Serdang, Malaysia, 2003. [Google Scholar]
- Borris, R; Blaskó, G; Cordell, G. Ethnopharmacologic and phytochemical studies of the Thymelaeaceae. J. Ethnopharmacol 1988, 24, 41–91. [Google Scholar]
- Ayaz, F; HayIrlIoglu-Ayaz, S; Alpay-Karaoglu, S; Gruz, J; Valentová, K; Ulrichová, J; Strnad, M. Phenolic acid contents of kale (Brassica oleraceae L. var. acephala DC.) extracts and their antioxidant and antibacterial activities. Food Chem 2008, 107, 19–25. [Google Scholar]
- Rahman, M; Moon, S. Antimicrobial phenolic derivatives from Dendranthema zawadskii var. latilobum kitamura (Asteraceae). Arch. Pharm. Res 2007, 30, 1374–1379. [Google Scholar]
- Vaquero, MJR; Alberto, MR; de Nadra, MCM. Antibacterial effect of phenolic compounds from different wines. Food Control 2007, 18, 93–101. [Google Scholar]
- Teffo, L; Aderogba, M; Eloff, J. Antibacterial and antioxidant activities of four kaempferol methyl ethers isolated from Dodonaea viscosa Jacq. var. angustifolia leaf extracts. S. Afr. J. Bot 2010, 76, 25–29. [Google Scholar]
- Demetzos, C; Angelopoulou, D; Kolocouris, A; Daliani, I; Mavromoustakos, T. Structure elucidation, conformational analysis and thermal effects on membrane bilayers of an antimicrobial myricetin ether derivative. J. Heterocycl. Chem 2001, 38, 703–710. [Google Scholar]
- Li, M; Xu, Z. Quercetin in a lotus leaves extract may be responsible for antibacterial activity. Arch. Pharm. Res 2008, 31, 640–644. [Google Scholar]
- Mandalari, G; Bennett, R; Bisignano, G; Trombetta, D; Saija, A; Faulds, C; Gasson, M; Narbad, A. Antimicrobial activity of flavonoids extracted from bergamot (Citrus bergamia Risso) peel, a byproduct of the essential oil industry. J. Appl. Microbiol 2007, 103, 2056–2064. [Google Scholar]
- Boussaada, O; Chriaa, J; Nabli, R; Ammar, S; Saidana, D; Mahjoub, M; Chraeif, I; Helal, A; Mighri, Z. Antimicrobial and antioxidant activities of methanol extracts of Evax pygmaea (Asteraceae) growing wild in Tunisia. World J. Microb. Biot 2008, 24, 1289–1296. [Google Scholar]
- Quiroga, E; Sampietro, A; Vattuone, M. Screening antifungal activities of selected medicinal plants. J. Ethnopharmacol 2001, 74, 89–96. [Google Scholar]
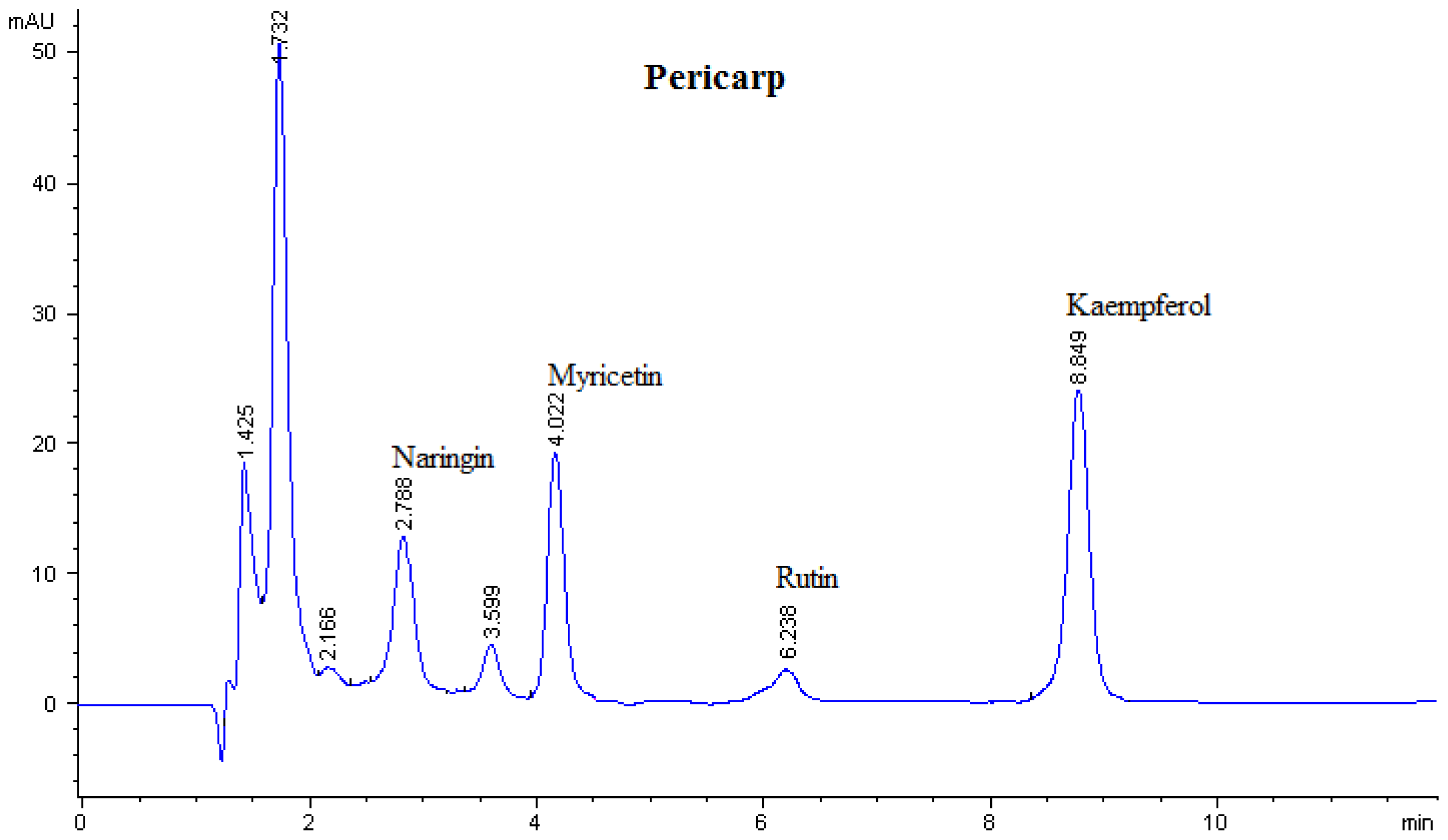
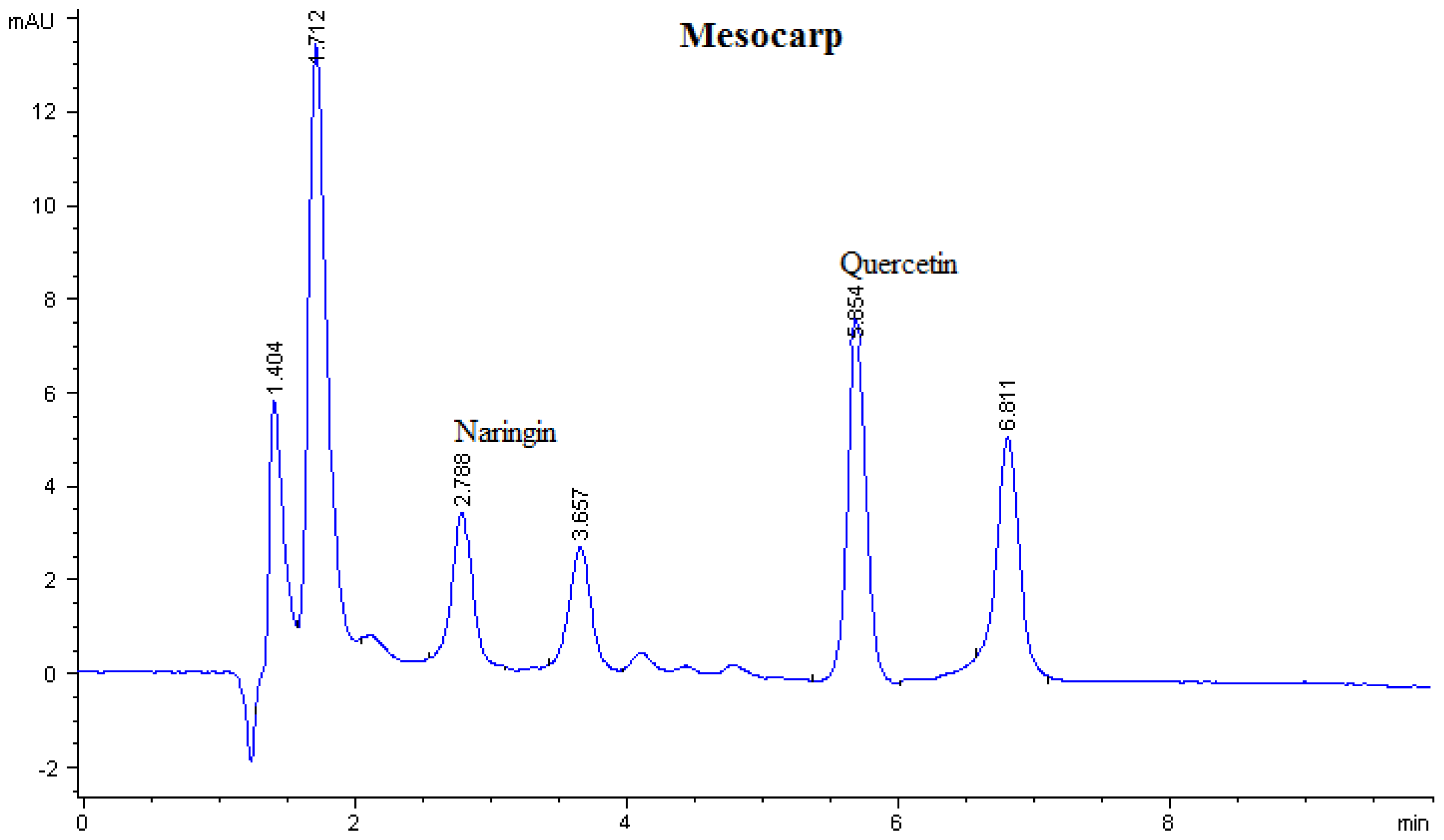
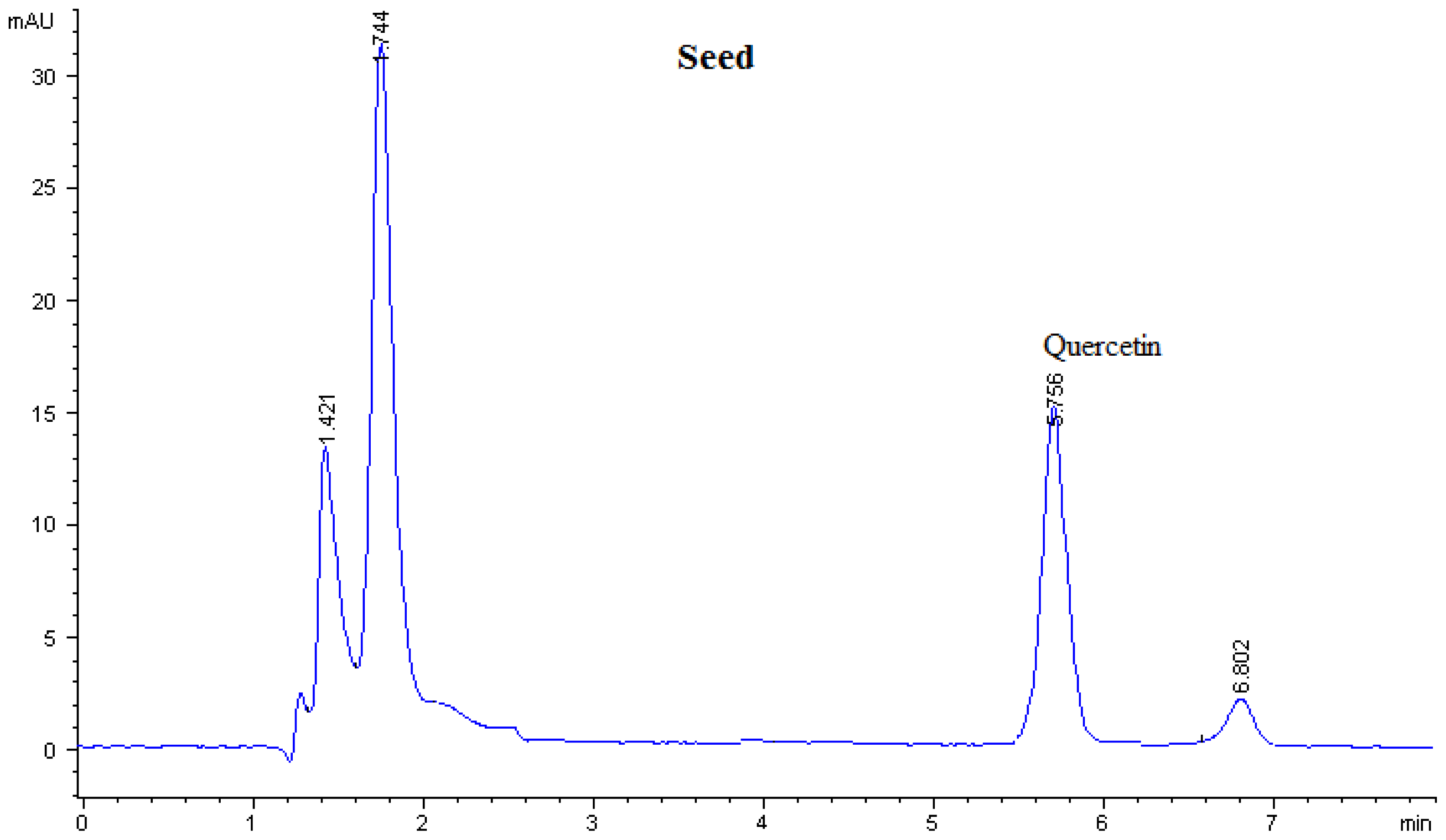
| Sample | Flavonoid contents (μg/g DW)
| ||||||
|---|---|---|---|---|---|---|---|
| Apigenin | Kaempferol | Luteolin | Myricetin | Naringin | Quercetin | Rutin | |
| Pericarp | - | 76.00 ± 0.003 | - | 59.90 ± 0.001 | 39.80 ± 0.001 | - | 17.80 ± 0.001 |
| Mesocarp | - | - | - | - | 1.90 | 31.80 ± 0.002 | - |
| Seed | - | - | - | - | - | 45.20 ± 0.003 | - |
| Microorganisms | Inhibition zone (cm)* | ||||
|---|---|---|---|---|---|
| Pericarp | Mesocarp | Seed | Kanamycin (1 μg/disc) | Amphotericin B (25 μg/well) | |
| Gram-Positive Bacteria | |||||
| B. cereus | 1.73 | 1.53 | 1.40 | 1.63 | - |
| B. subtilis | 2.33 | 2.00 | 1.83 | 1.33 | - |
| M. luteus | 1.57 | 1.47 | 1.37 | 1.35 | - |
| S. aureus | 1.53 | 1.73 | 1.40 | 1.35 | - |
| Average | 1.79 | 1.68 | 1.50 | 1.42 | - |
| Gram-Negative Bacteria | |||||
| E. aerogenes | 1.50 | 1.30 | 1.20 | 1.55 | - |
| E. coli | 2.17 | 1.47 | 1.47 | 1.73 | - |
| K. pnuomoniae | 1.23 | 1.10 | 0.97 | 1.5 | - |
| P. aeruginosa | 1.40 | 1.17 | 0.93 | 1 | - |
| Average | 1.56 | 1.26 | 1.14 | 1.45 | - |
| Fungi | |||||
| A. nige | 0 | 0 | 1.87 | - | 2.70 |
| F. oxysporum | 0 | 0 | 0 | - | 1.80 |
| G. lucidum | 0 | 0 | 0 | - | 0.40 |
| M. indicus | 0 | 0 | 0 | - | 1.20 |
| 0 | 0 | 0.47 | - | 1.53 ± 0.97 | |
© 2011 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Hendra, R.; Ahmad, S.; Sukari, A.; Shukor, M.Y.; Oskoueian, E. Flavonoid Analyses and Antimicrobial Activity of Various Parts of Phaleria macrocarpa (Scheff.) Boerl Fruit. Int. J. Mol. Sci. 2011, 12, 3422-3431. https://doi.org/10.3390/ijms12063422
Hendra R, Ahmad S, Sukari A, Shukor MY, Oskoueian E. Flavonoid Analyses and Antimicrobial Activity of Various Parts of Phaleria macrocarpa (Scheff.) Boerl Fruit. International Journal of Molecular Sciences. 2011; 12(6):3422-3431. https://doi.org/10.3390/ijms12063422
Chicago/Turabian StyleHendra, Rudi, Syahida Ahmad, Aspollah Sukari, M. Yunus Shukor, and Ehsan Oskoueian. 2011. "Flavonoid Analyses and Antimicrobial Activity of Various Parts of Phaleria macrocarpa (Scheff.) Boerl Fruit" International Journal of Molecular Sciences 12, no. 6: 3422-3431. https://doi.org/10.3390/ijms12063422




