Antimicrobial Activities of Hexane Extract and Decussatin from Stembark Extract of Ficus congensis
Abstract
:1. Introduction
2. Results and Discussion
2.1. Chemical Constituents/Isolated Compound
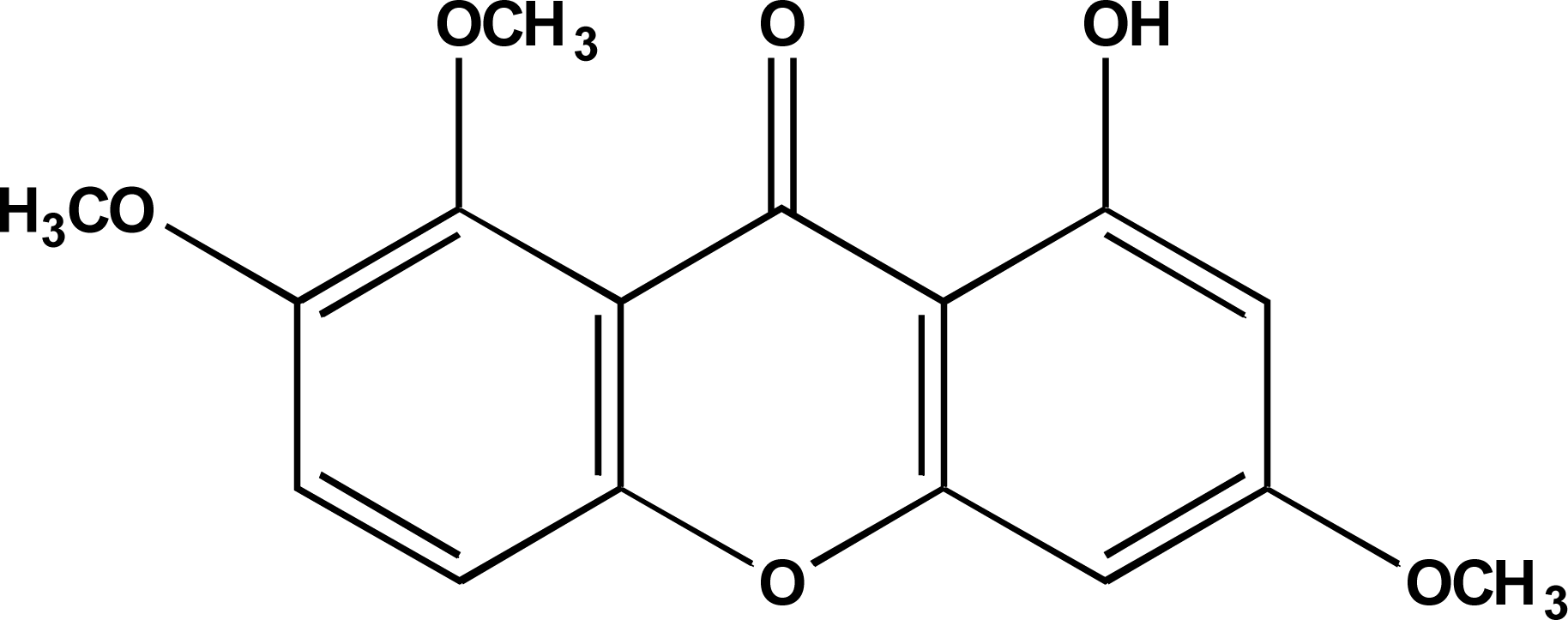
2.2. Structural and Spectra Information
Compound 1
2.3. Antimicrobial
3. Experimental Section
3.1. Collection and Preparation of Plant Material
3.2. Extraction, Isolation and Structure Elucidation
3.3. Microdilution Bioassay
3.4. Antifungal Assay
4. Conclusions
Acknowledgments
References
- Burkill, HM. The Useful Plants of West Tropical Africa, 2nd ed.; Royal Botanical Garden, Kew: London, UK, 1997; Volume 4, pp. 203–204. [Google Scholar]
- Van Noort, S; Gardiner, AJ; Tolley, KA. New records of Ficus (Moraceae) species emphasize the conservation significance of inselbergs in Mozambique. S. Afr. J. Bot 2007, 13, 642–649. [Google Scholar]
- Berg, CC. Annotated check-list of the Ficus species of the African floristic region, with special reference and a key to the taxa of Southern Africa. Kirkia 1990, 13, 253–291. [Google Scholar]
- Berg, CC. Flora Amnesiacs. Ulmaceae, Cannabaceae, Moraceae, Cecropiaceae, Urticaceae, Casuarinaceae, Salicaceae, Ceratophyllaceae. In Ficus; Launert, E, Pope, GV, Eds.; Flora Zambesiaca Managing Committee: London, UK, 1991; pp. 39–76. [Google Scholar]
- Berg, CC; Wiebes, JT. African Fig Trees and Fig Wasps; Koninklijke Nederlanse Akademie van Wetenschappen (KNAW): Amsterdam, The Netherlands, 1992; pp. 1–2. [Google Scholar]
- Burrows, J; Burrows, S. Figs of Southern and South-Central Africa; Umdaus Press: Hatfield, UK, 2003. [Google Scholar]
- Lansky, EP; Paavilainen, HM; Pawlus, AD; Newman, RA. Ficus spp. (fig): Ethnobotany and potential as anticancer and anti-inflammatory agents. J. Ethnopharmacol 2008, 119, 195–213. [Google Scholar]
- Kamatenesi-Mugisha, M; Oryem-Origa, H. Medicinal plants used to induce labour during childbirth in westen Uganda. J. Ethnopharmacol 2007, 119, 1–9. [Google Scholar]
- Ssegawa, P; Kasenene, J. Medicinal plant diversity and the uses in the Sango bay area, Southern Uganda. J. Ethnopharmacol 2007, 113, 521–540. [Google Scholar]
- Balogun, SO; Tanayen, JK; Ajayi, AM; Ibrahim, A; Ezeonwumelu, JOC; Oyewale, AA; Oloro, OJ; Goji, ADT; Kiplagat, DM; Adzu, B. Preliminary evaluation of anti-diarrheal, ulcer protective and acute toxicity of aqueous ethanolic stem bark extract of Ficus trichopoda in experimental rodents. Asian J. Med. Sci 2011, 3, 37–42. [Google Scholar]
- Ghulam, AM; Hassan, MGA. Xanthones of Centarium pulchelium. J. Phytochem 1984, 23, 1637–1638. [Google Scholar]
- Valento, P; Andrade, PB; Silva, E; Vicente, A; Santos, H; Bastos, ML; Seabra, RM. Methoxylated Xanthones in the quality control of small centaury (Centarium erythraea) flowering tops. J. Agr. Food Chem 2002, 50, 460–463. [Google Scholar]
- Okorie, D. A new Phthalide and Xanthones from Anthocleista Djalonensis and Anthocleista vogelii. J. Phytochem 1776, 15, 1799–1800. [Google Scholar]
- Peres, V; Nagem, TJ; Oliveria, FF. Tetraoxygenated naturally occurring Xanthones. J. Phytochem 2000, 55, 683–710. [Google Scholar]
- Peres, V; Nagem, TJ. Naturally occurring pentaoxygenated, hexaoxygenated and dimeric xanthones: A literature survey. Química Nova 1997, 20, 388–397. [Google Scholar]
- Eloff, JN. A sensitive and quick microplate method to determine the minimal inhibitory concentration of plant extracts for bacteria. Planta Med 1998, 64, 711–713. [Google Scholar]
- Utz, C; Shadomy, S. Antifungal activity of 5-fluorocytosine as measured by disc diffusion susceptibility testing. J. Infect. Dis 1977, 135, 970–974. [Google Scholar]
| Test strain (bacteria) | MIC (mg/mL) | ||
|---|---|---|---|
| Hexane extract | Decussatin | Neomycin | |
| E. coli | 8.00 | N.D | 0.10 |
| B. substilis | 5.00 | N.D | 1.56 |
| K. pneumonia | N.D | N.D | 1.20 |
| S. aureus | N.D | N.D | 0.39 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Alaribe, C.S.; Shode, F.; Coker, H.A.B.; Ayoola, G.; Sunday, A.; Singh, N.; Iwuanyanwu, S. Antimicrobial Activities of Hexane Extract and Decussatin from Stembark Extract of Ficus congensis. Int. J. Mol. Sci. 2011, 12, 2750-2756. https://doi.org/10.3390/ijms12042750
Alaribe CS, Shode F, Coker HAB, Ayoola G, Sunday A, Singh N, Iwuanyanwu S. Antimicrobial Activities of Hexane Extract and Decussatin from Stembark Extract of Ficus congensis. International Journal of Molecular Sciences. 2011; 12(4):2750-2756. https://doi.org/10.3390/ijms12042750
Chicago/Turabian StyleAlaribe, Chinwendum Stephenie, Francis Shode, Herbert A. B. Coker, Gloria Ayoola, Adesegun Sunday, Nisha Singh, and Silva Iwuanyanwu. 2011. "Antimicrobial Activities of Hexane Extract and Decussatin from Stembark Extract of Ficus congensis" International Journal of Molecular Sciences 12, no. 4: 2750-2756. https://doi.org/10.3390/ijms12042750




