Chitin-based Materials in Tissue Engineering: Applications in Soft Tissue and Epithelial Organ
Abstract
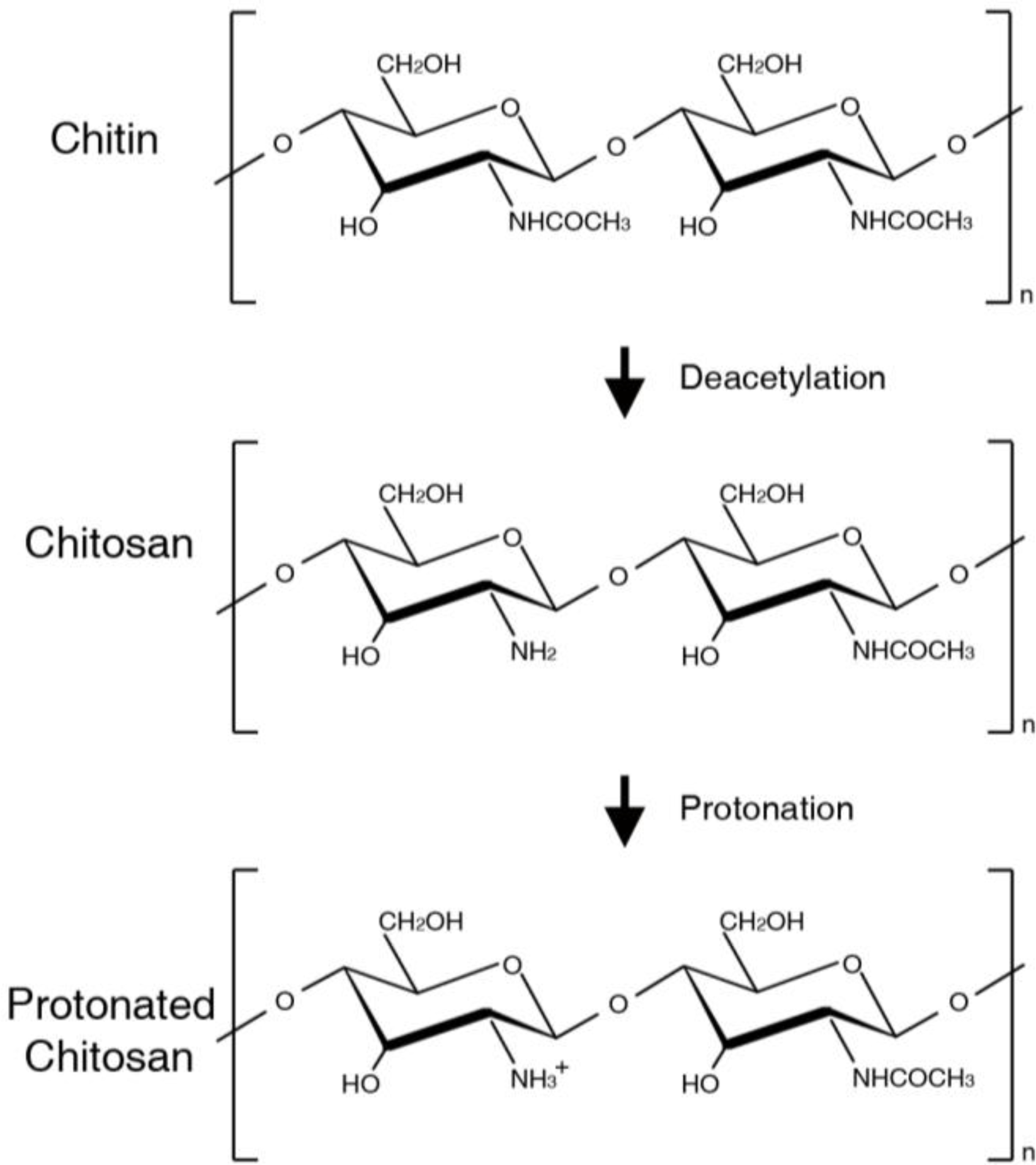
:1. Introduction
2. Scaffolds in Tissue Engineering
2.1. Chitin-based Scaffolds
2.2. Blended Chitin-based Scaffolds
2.3. Chitosan Scaffolds Loaded with Bioactive Molecules
3. Biological Modulation in Tissue Engineering
3.1. Cell Preservation
3.2. Regulation of Gene Expression
3.3. Bioactive Molecule Regulation
3.4. Extracellular Matrix Production and Deposition
4. Applications
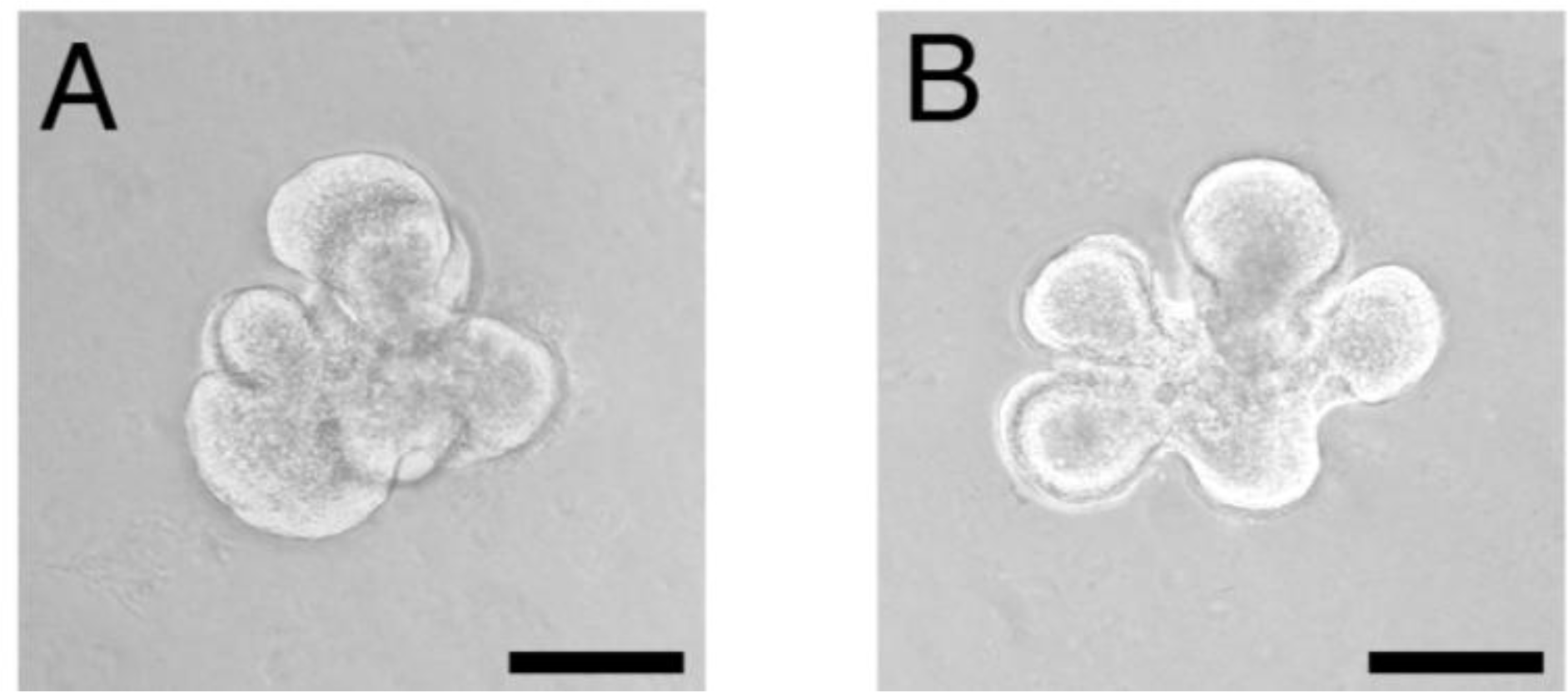
4.1. Chitin-based Materials in the Tissue Engineering of Surface-lining Layers
4.2. Chitin-based Materials in Tissue Structure Formation
4.3. Chitin-based Material in Connective Tissue Engineering
4.4. Chitin-based Material in Nerve Tissue Engineering
4.5. Chitin-based Material in Adipose Tissue Engineering
4.6. Chitin-based Material in Vascular Tissue Engineering
5. Current State, Challenge, and Future Prospects
Acknowledgments
References
- Kumar, MN; Muzzarelli, RA; Muzzarelli, C; Sashiwa, H; Domb, AJ. Chitosan chemistry and pharmaceutical perspectives. Chem. Rev 2004, 104, 6017–6084. [Google Scholar]
- Felt, O; Furrer, P; Mayer, JM; Plazonnet, B; Buri, P; Gurny, R. Topical use of chitosan in ophtalmology: Tolerance assessment and evaluation of precorneal retention. Int. J. Pharm 1999, 180, 185–193. [Google Scholar]
- Muzzarelli, RAA. Human enzymatic activities related to the therapeutic administration of chitin derivatives. Cell. Mol. Life Sci 1997, 53, 131–140. [Google Scholar]
- Kim, IY; Seo, SJ; Moon, HS; Yoo, MK; Park, IY; Kim, BC; Cho, CS. Chitosan and its derivatives for tissue engineering applications. Biotechnol. Adv 2008, 26, 1–21. [Google Scholar]
- Chung, YC; Chen, CY. Antibacterial characteristics and activity of acid-soluble chitosan. Bioresour. Technol 2008, 99, 2806–2814. [Google Scholar]
- He, P; Davis, SS; Illum, L. In vitro evaluation of the mucoadhesive properties of chitosan microspheres. Int. J. Pharm 1998, 166, 75–88. [Google Scholar]
- Calvo, P; Vila-Jato, JL; Alonso, MJ. Evaluation of cationic polymer-coated nanocapsules as ocular drug carriers. Int. J. Pharm 1997, 153, 41–50. [Google Scholar]
- Ishihara, M; Fujita, M; Obara, K; Hattori, H; Nakamura, S; Nambu, M; Kiyosawa, T; Kanatani, Y; Takase, B; Kikuchi, M; Maehara, T. Controlled releases of FGF-2 and paclitaxel from chitosan hydrogels and their subsequent effects on wound repair, angiogenesis, and tumor growth. Curr. Drug. Deliv 2006, 3, 351–358. [Google Scholar]
- Mori, T; Okumura, M; Matsuura, M; Ueno, K; Tokura, S; Okamoto, Y; Minami, S; Fujinaga, T. Effects of chitin and its derivatives on the proliferation and cytokine production of fibroblasts in vitro. Biomaterials 1997, 18, 947–951. [Google Scholar]
- Masuoka, K; Ishihara, M; Asazuma, T; Hattori, H; Matsui, T; Takase, B; Kanatani, Y; Fujita, M; Saito, Y; Yura, H; Fujikawa, K; Nemoto, K. The interaction of chitosan with fibroblast growth factor-2 and its protection from inactivation. Biomaterials 2005, 26, 3277–3284. [Google Scholar]
- Madihally, SV; Matthew, HW. Porous chitosan scaffolds for tissue engineering. Biomaterials 1999, 20, 1133–1142. [Google Scholar]
- Yang, TL; Young, TH. Chitosan cooperates with mesenchyme-derived factors in regulating salivary gland epithelial morphogenesis. J. Cell. Mol. Med 2009, 13, 2853–2863. [Google Scholar]
- Mi, FL; Shyu, SS; Peng, CK; Wu, YB; Sung, HW; Wang, PS; Huang, CC. Fabrication of chondroitin sulfate-chitosan composite artificial extracellular matrix for stabilization of fibroblast growth factor. J. Biomed. Mater. Res. A 2006, 76, 1–15. [Google Scholar]
- Prabaharan, M. Review paper: Chitosan derivatives as promising materials for controlled drug delivery. J. Biomater. Appl 2008, 23, 5–36. [Google Scholar]
- Jiang, T; Kumbar, SG; Nair, LS; Laurencin, CT. Biologically active chitosan systems for tissue engineering and regenerative medicine. Curr. Top. Med. Chem 2008, 8, 354–364. [Google Scholar]
- Langer, R; Vacanti, JP; Vacanti, CA; Atala, A; Freed, LE; Vunjak-Novakovic, G. Tissue engineering: biomedical applications. Tissue Eng 1995, 1, 151–161. [Google Scholar]
- Ladd, MR; Lee, SJ; Atala, A; Yoo, JJ. Bioreactor maintained living skin matrix. Tissue Eng. Part A 2009, 15, 861–868. [Google Scholar]
- Atala, A. Tissue engineering of artificial organs. J. Endourol 2000, 14, 49–57. [Google Scholar]
- Atala, A; Bauer, SB; Soker, S; Yoo, JJ; Retik, AB. Tissue-engineered autologous bladders for patients needing cystoplasty. Lancet 2006, 367, 1241–1246. [Google Scholar]
- Macchiarini, P; Jungebluth, P; Go, T; Asnaghi, MA; Rees, LE; Cogan, TA; Dodson, A; Martorell, J; Bellini, S; Parnigotto, PP; Dickinson, SC; Hollander, AP; Mantero, S; Conconi, MT; Birchall, MA. Clinical transplantation of a tissue-engineered airway. Lancet 2008, 372, 2023–2030. [Google Scholar]
- Langer, R; Vacanti, JP. Tissue engineering. Science 1993, 260, 920–926. [Google Scholar]
- Peter, MG. Applications and environmental aspects of chitin and chitosan. J Macromol Sci 1995, Part A. 629–640. [Google Scholar]
- Berger, J; Reist, M; Mayer, JM; Felt, O; Peppas, NA; Gurny, R. Structure and interactions in covalently and ionically crosslinked chitosan hydrogels for biomedical applications. Eur. J. Pharm. Biopharm 2004, 57, 19–34. [Google Scholar]
- Hu, Q; Li, B; Wang, M; Shen, J. Preparation and characterization of biodegradable chitosan/hydroxyapatite nanocomposite rods via in situ hybridization: A potential material as internal fixation of bone fracture. Biomaterials 2004, 25, 779–785. [Google Scholar]
- Muzzarelli, R; Baldassarre, V; Conti, F; Ferrara, P; Biagini, G; Gazzanelli, G; Vasi, V. Biological activity of chitosan: Ultrastructural study. Biomaterials 1988, 9, 247–252. [Google Scholar]
- Khor, E; Lim, LY. Implantable applications of chitin and chitosan. Biomaterials 2003, 24, 2339–2349. [Google Scholar]
- Felt, O; Carrel, A; Baehni, P; Buri, P; Gurny, R. Chitosan as tear substitute: A wetting agent endowed with antimicrobial efficacy. J. Ocul. Pharmacol 2000, 16, 261–270. [Google Scholar]
- Ayres, CE; Jha, BS; Sell, SA; Bowlin, GL; Simpson, DG. Nanotechnology in the design of soft tissue scaffolds: Innovations in structure and function. Wiley Interdiscip. Rev. Nanomed. Nanobiotech 2010, 2, 20–34. [Google Scholar]
- Di Martino, A; Sittinger, M; Risbud, MV. Chitosan: A versatile biopolymer for orthopaedic tissue-engineering. Biomaterials 2005, 26, 5983–5990. [Google Scholar]
- Risbud, MV; Sittinger, M. Tissue engineering: Advances in in vitro cartilage generation. Trends Biotech 2002, 20, 351–356. [Google Scholar]
- Oliveira, JM; Rodrigues, MT; Silva, SS; Malafaya, PB; Gomes, ME; Viegas, CA; Dias, IR; Azevedo, JT; Mano, JF; Reis, RL. Novel hydroxyapatite/chitosan bilayered scaffold for osteochondral tissue-engineering applications: Scaffold design and its performance when seeded with goat bone marrow stromal cells. Biomaterials 2006, 27, 6123–6137. [Google Scholar] [Green Version]
- Hemmrich, K; von Heimburg, D; Rendchen, R; Di Bartolo, C; Milella, E; Pallua, N. Implantation of preadipocyte-loaded hyaluronic acid-based scaffolds into nude mice to evaluate potential for soft tissue engineering. Biomaterials 2005, 26, 7025–7037. [Google Scholar]
- Ueno, H; Yamada, H; Tanaka, I; Kaba, N; Matsuura, M; Okumura, M; Kadosawa, T; Fujinaga, T. Accelerating effects of chitosan for healing at early phase of experimental open wound in dogs. Biomaterials 1999, 20, 1407–1414. [Google Scholar]
- Mattioli-Belmonte, M; Muzzarelli, B; Muzzarelli, RAA. Chitin and chitosan in wound healing and other biomedical applications. Carbohyd. Eur 1997, 19, 30–36. [Google Scholar]
- Cho, YW; Cho, YN; Chung, SH; Yoo, G; Ko, SW. Water-soluble chitin as a wound healing accelerator. Biomaterials 1999, 20, 2139–2145. [Google Scholar]
- Kojima, K; Okamoto, Y; Kojima, K; Miyatake, K; Fujise, H; Shigemasa, Y; Minami, S. Effects of chitin and chitosan on collagen synthesis in wound healing. J. Vet. Med. Sci 2004, 66, 1595–1598. [Google Scholar]
- VandeVord, PJ; Matthew, HWT; DeSilva, SP; Mayton, L; Wu, B; Wooley, PH. Evaluation of the biocompatibility of a chitosan scaffold in mice. J. Biomed. Mater. Res 2002, 59, 585–590. [Google Scholar]
- Drewa, T; Adamowicz, J; Lysik, J; Polaczek, J; Pielichowski, J. Chitosan scaffold enhances nerve regeneration within the in vitro reconstructed bladder wall: An animal study. Urol. Int 2008, 81, 330–334. [Google Scholar]
- Berger, J; Reist, M; Mayer, JM; Felt, O; Gurny, R. Structure and interactions in chitosan hydrogels formed by complexation or aggregation for biomedical applications. Eur. J. Pharm. Biopharm 2004, 57, 35–52. [Google Scholar]
- Chow, KS; Khor, E. Novel fabrication of open-pore chitin matrixes. Biomacromolecules 2000, 1, 61–67. [Google Scholar]
- Shi, C; Zhu, Y; Ran, X; Wang, M; Su, Y; Cheng, T. Therapeutic potential of chitosan and its derivatives in regenerative medicine. J. Surg. Res 2006, 133, 185–192. [Google Scholar]
- Freier, T; Montenegro, R; Shan Koh, H; Shoichet, MS. Chitin-based tubes for tissue engineering in the nervous system. Biomaterials 2005, 26, 4624–4632. [Google Scholar]
- Wang, W; Itoh, S; Matsuda, A; Ichinose, S; Shinomiya, K; Hata, Y; Tanaka, J. Influences of mechanical properties and permeability on chitosan nano/microfiber mesh tubes as a scaffold for nerve regeneration. J. Biomed. Mater. Res. A 2008, 84, 557–566. [Google Scholar]
- Zhang, L; Ao, Q; Wang, A; Lu, G; Kong, L; Gong, Y; Zhao, N; Zhang, X. A sandwich tubular scaffold derived from chitosan for blood vessel tissue engineering. J. Biomed. Mater. Res. A 2006, 77, 277–284. [Google Scholar]
- Madihally, SV; Matthew, HW. Porous chitosan scaffolds for tissue engineering. Biomaterials 1999, 20, 1133–1142. [Google Scholar]
- Ko, YG; Kawazoe, N; Tateishi, T; Chen, G. Preparation of chitosan scaffolds with a hierarchical porous structure. J. Biomed. Mater. Res. B 2010, 93, 341–350. [Google Scholar]
- Elsabee, MZ; Morsi, RE; Al-Sabagh, AM. Surface active properties of chitosan and its derivatives. Colloids Surf. B 2009, 74, 1–16. [Google Scholar]
- Nishikawa, H; Ueno, A; Nishikawa, S; Kido, J; Ohishi, M; Inoue, H; Nagata, T. Sulfated glycosaminoglycan synthesis and its regulation by transforming growth factor-beta in rat clonal dental pulp cells. J. Endod 2000, 26, 169–171. [Google Scholar]
- Bagnaninchi, PO; Yang, Y; Zghoul, N; Maffulli, N; Wang, RK; Haj, AJ. Chitosan microchannel scaffolds for tendon tissue engineering characterized using optical coherence tomography. Tissue Eng 2007, 13, 323–331. [Google Scholar]
- Ma, J; Wang, H; He, B; Chen, J. A preliminary in vitro study on the fabrication and tissue engineering applications of a novel chitosan bilayer material as a scaffold of human fetal dermal fibroblasts. Biomaterials 2001, 22, 331–336. [Google Scholar]
- Lin, SJ; Jee, SH; Hsaio, WC; Lee, SJ; Young, TH. Formation of melanocyte spheroids on the chitosan-coated surface. Biomaterials 2005, 26, 1413–1422. [Google Scholar]
- Norazril, SA; Aminuddin, BS; Norhayati, MM; Mazlyzam, AL; Fauziah, O; Ruszymah, BH. Comparison of chitosan scaffold and chitosan-collagen scaffold: A preliminary study. Med. J. Malaysia 2004, 59, 186–187. [Google Scholar]
- Huang, TW; Young, YH; Cheng, PW; Chan, YH; Young, TH. Culture of nasal epithelial cells using chitosan-based membranes. Laryngoscope 2009, 119, 2066–2070. [Google Scholar]
- Shao, HJ; Chen, CS; Lee, YT; Wang, JH; Young, TH. The phenotypic responses of human anterior cruciate ligament cells cultured on poly(epsilon-caprolactone) and chitosan. J. Biomed. Mater. Res. A 2010, 93, 1297–1305. [Google Scholar]
- Lin, SJ; Hsiao, WC; Jee, SH; Yu, HS; Tsai, TF; Lai, JY; Young, TH. Study on the effects of nylon-chitosan-blended membranes on the spheroid-forming activity of human melanocytes. Biomaterials 2006, 27, 5079–5088. [Google Scholar]
- Crompton, KE; Goud, JD; Bellamkonda, RV; Gengenbach, TR; Finkelstein, DI; Horne, MK; Forsythe, JS. Polylysine-functionalised thermoresponsive chitosan hydrogel for neural tissue engineering. Biomaterials 2007, 28, 441–449. [Google Scholar]
- Funakoshi, T; Majima, T; Iwasaki, N; Yamane, S; Masuko, T; Minami, A; Harada, K; Tamura, H; Tokura, S; Nishimura, S. Novel chitosan-based hyaluronan hybrid polymer fibers as a scaffold in ligament tissue engineering. J. Biomed. Mater. Res. A 2005, 74, 338–346. [Google Scholar]
- Zhu, C; Fan, D; Duan, Z; Xue, W; Shang, L; Chen, F; Luo, Y. Initial investigation of novel human-like collagen/chitosan scaffold for vascular tissue engineering. J. Biomed. Mater. Res. A 2009, 89, 829–840. [Google Scholar]
- Shi, H; Han, C; Mao, Z; Ma, L; Gao, C. Enhanced angiogenesis in porous collagen-chitosan scaffolds loaded with angiogenin. Tissue Eng 2008, 14, 1775–1785. [Google Scholar]
- Wu, X; Black, L; Santacana-Laffitte, G; Patrick, CW, Jr. Preparation and assessment of glutaraldehyde-crosslinked collagen-chitosan hydrogels for adipose tissue engineering. J. Biomed. Mater. Res. A 2007, 81, 59–65. [Google Scholar]
- Gobin, AS; Froude, VE; Mathur, AB. Structural and mechanical characteristics of silk fibroin and chitosan blend scaffolds for tissue regeneration. J. Biomed. Mater. Res. A 2005, 74, 465–473. [Google Scholar]
- Gobin, AS; Butler, CE; Mathur, AB. Repair and regeneration of the abdominal wall musculofascial defect using silk fibroin-chitosan blend. Tissue Eng 2006, 12, 3383–3394. [Google Scholar]
- Ma, L; Gao, CY; Mao, ZW; Zhou, J; Shen, JC; Hu, XQ; Han, CM. Collagen/chitosan porous scaffolds with improved biostability for skin tissue engineering. Biomaterials 2003, 24, 4833–4841. [Google Scholar]
- Han, CM; Zhang, LP; Sun, JZ; Shi, HF; Zhou, J; Gao, CY. Application of collagen-chitosan/fibrin glue asymmetric scaffolds in skin tissue engineering. J. Zhejiang Univ. Sci. B 2010, 11, 524–530. [Google Scholar]
- Kinikoglu, B; Auxenfans, C; Pierrillas, P; Justin, V; Breton, P; Burillon, C; Hasirci, V; Damour, O. Reconstruction of a full-thickness collagen-based human oral mucosal equivalent. Biomaterials 2009, 30, 6418–6425. [Google Scholar]
- Yang, J; Woo, SL; Yang, G; Wang, J; Cui, L; Liu, W; Cao, Y. Construction and clinical application of a human tissue-engineered epidermal membrane. Plast Reconstr. Surg 2010, 125, 901–909. [Google Scholar]
- Gafni, Y; Zilberman, Y; Ophir, Z; Abramovitch, R; Jaffe, M; Gazit, Z; Domb, A; Gazit, D. Design of a filamentous polymeric scaffold for in vivo guided angiogenesis. Tissue Eng 2006, 12, 3021–3034. [Google Scholar]
- Lee, JJ; Lee, S; Park, JC; Yang, YI; Kim, JK. Investigation on biodegradable PLGA scaffold with various pore size structure for skin tissue engineering. Curr. Appl. Phys 2007, 7, e37–e40. [Google Scholar]
- Pieper, JS; van Wachem, PB; van Luyn, MJA; Brouwer, LA; Hafmans, T; Veerkamp, JH; van Kuppevelt, TH. Attachment of glycosaminoglycans to collagenous matrices modulates the tissue response in rats. Biomaterials 2000, 21, 1689–1699. [Google Scholar]
- Ragetly, G; Griffon, DJ; Chung, YS. The effect of type II collagen coating of chitosan fibrous scaffolds on mesenchymal stem cell adhesion and chondrogenesis. Acta Biomater 2010, 6, 3988–3997. [Google Scholar]
- Ragetly, GR; Griffon, DJ; Lee, HB; Chung, YS. Effect of collagen II coating on mesenchymal stem cell adhesion on chitosan and on reacetylated chitosan fibrous scaffolds. J. Mater. Sci. Mater. Med 2010, 21, 2479–2490. [Google Scholar]
- Leipzig, ND; Xu, C; Zahir, T; Shoichet, MS. Functional immobilization of interferon-gamma induces neuronal differentiation of neural stem cells. J. Biomed. Mater. Res. A 2010, 93, 625–633. [Google Scholar]
- Akman, AC; Tigli, RS; Gumusderelioglu, M; Nohutcu, RM. bFGF-loaded HA-chitosan: A promising scaffold for periodontal tissue engineering. J. Biomed. Mater. Res. A 2010, 92, 953–962. [Google Scholar]
- Tabata, Y. Tissue regeneration based on growth factor release. Tissue Eng 2003, 9(Suppl 1), s5–15. [Google Scholar]
- Berger, J; Reist, M; Mayer, JM; Felt, O; Gurny, R. Structure and interactions in chitosan hydrogels formed by complexation or aggregation for biomedical applications. Eur. J. Pharm. Biopharm 2004, 57, 35–52. [Google Scholar]
- Ho, MH; Wang, DM; Hsieh, HJ; Liu, HC; Hsien, TY; Lai, JY; Hou, LT. Preparation and characterization of RGD-immobilized chitosan scaffolds. Biomaterials 2005, 26, 3197–3206. [Google Scholar]
- Yamada, Y; Hozumi, K; Katagiri, F; Kikkawa, Y; Nomizu, M. Biological activity of laminin peptide conjugated alginate and chitosan matrices. Biopolymers 2010, 94, 711–720. [Google Scholar]
- Kean, T; Roth, S; Thanou, M. Trimethylated chitosans as non-viral gene delivery vectors: Cytotoxicity and transfection efficiency. J. Control. Release 2005, 103, 643–653. [Google Scholar]
- Itoh, S; Matsuda, A; Kobayashi, H; Ichinose, S; Shinomiya, K; Tanaka, J. Effects of a laminin peptide (YIGSR) immobilized on crab-tendon chitosan tubes on nerve regeneration. J. Biomed. Mater. Res. B 2005, 73, 375–382. [Google Scholar]
- Mizuno, K; Yamamura, K; Yano, K; Osada, T; Saeki, S; Takimoto, N; Sakurai, T; Nimura, Y. Effect of chitosan film containing basic fibroblast growth factor on wound healing in genetically diabetic mice. J. Biomed. Mater. Res. A 2003, 64, 177–181. [Google Scholar]
- Choi, JS; Yoo, HS. Pluronic/chitosan hydrogels containing epidermal growth factor with wound-adhesive and photo-crosslinkable properties. J. Biomed. Mater. Res. A 2010, 95A, 564–573. [Google Scholar]
- Judith, R; Nithya, M; Rose, C; Mandal, AB. Application of a PDGF-containing novel gel for cutaneous wound healing. Life Sci 2010, 87, 1–8. [Google Scholar]
- Ishihara, M; Obara, K; Ishizuka, T; Fujita, M; Sato, M; Masuoka, K; Saito, Y; Yura, H; Matsui, T; Hattori, H; Kikuchi, M; Kurita, A. Controlled release of fibroblast growth factors and heparin from photocrosslinked chitosan hydrogels and subsequent effect on in vivo vascularization. J. Biomed. Mater. Res. A 2003, 64, 551–559. [Google Scholar]
- Guo, R; Xu, S; Ma, L; Huang, A; Gao, C. Enhanced angiogenesis of gene-activated dermal equivalent for treatment of full thickness incisional wounds in a porcine model. Biomaterials 2010, 31, 7308–7320. [Google Scholar]
- Zhang, Y; Cheng, X; Wang, J; Wang, Y; Shi, B; Huang, C; Yang, X; Liu, T. Novel chitosan/collagen scaffold containing transforming growth factor-beta1 DNA for periodontal tissue engineering. Biochem. Biophys. Res. Commun 2006, 344, 362–369. [Google Scholar]
- Lin, SJ; Jee, SH; Hsiao, WC; Yu, HS; Tsai, TF; Chen, JS; Hsu, CJ; Young, TH. Enhanced cell survival of melanocyte spheroids in serum starvation condition. Biomaterials 2006, 27, 1462–1469. [Google Scholar]
- Chen, YH; Wang, IJ; Young, TH. Formation of keratocyte spheroids on chitosan-coated surface can maintain keratocyte phenotypes. Tissue Eng. Part A 2009, 15, 2001–2013. [Google Scholar]
- Shao, HJ; Chen, CS; Lee, YT; Wang, JH; Young, TH. The phenotypic responses of human anterior cruciate ligament cells cultured on poly(epsilon-caprolactone) and chitosan. J. Biomed. Mater. Res. A 2010, 93, 1297–1305. [Google Scholar]
- Wang, G; Ao, Q; Gong, K; Wang, A; Zheng, L; Gong, Y; Zhang, X. The effect of topology of chitosan biomaterials on the differentiation and proliferation of neural stem cells. Acta Biomater 2010, 6, 3630–3639. [Google Scholar]
- Zhu, Y; Liu, T; Song, K; Jiang, B; Ma, X; Cui, Z. Collagen-chitosan polymer as a scaffold for the proliferation of human adipose tissue-derived stem cells. J. Mater. Sci. Mater. Med 2009, 20, 799–808. [Google Scholar]
- Thein-Han, WW; Kitiyanant, Y. Chitosan scaffolds for in vitro buffalo embryonic stem-like cell culture: an approach to tissue engineering. J. Biomed. Mater. Res. B 2007, 80, 92–101. [Google Scholar]
- Gupta, V; Davis, G; Gordon, A; Altman, AM; Reece, GP; Gascoyne, PR; Mathur, AB. Endothelial and stem cell interactions on dielectrophoretically aligned fibrous silk fibroin-chitosan scaffolds. J. Biomed. Mater. Res. A 2010, 94, 515–523. [Google Scholar]
- Lahiji, A; Sohrabi, A; Hungerford, DS; Frondoza, CG. Chitosan supports the expression of extracellular matrix proteins in human osteoblasts and chondrocytes. J. Biomed. Mater. Res 2000, 51, 586–595. [Google Scholar]
- Shen, EC; Chou, TC; Gau, CH; Tu, HP; Chen, YT; Fu, E. Releasing growth factors from activated human platelets after chitosan stimulation: a possible bio-material for platelet-rich plasma preparation. Clin. Oral Implants. Res 2006, 17, 572–578. [Google Scholar]
- Ueno, H; Nakamura, F; Murakami, M; Okumura, M; Kadosawa, T; Fujinag, T. Evaluation effects of chitosan for the extracellular matrix production by fibroblasts and the growth factors production by macrophages. Biomaterials 2001, 22, 2125–2130. [Google Scholar]
- Shao, HJ; Lee, YT; Chen, CS; Wang, JH; Young, TH. Modulation of gene expression and collagen production of anterior cruciate ligament cells through cell shape changes on polycaprolactone/chitosan blends. Biomaterials 2010, 31, 4695–4705. [Google Scholar]
- Lopes, JB; Dallan, LA; Moreira, LF; Campana Filho, SP; Gutierrez, PS; Lisboa, LA; de Oliveira, SA; Stolf, NA. Synergism between keratinocyte growth factor and carboxymethyl chitosan reduces pericardial adhesions. Ann. Thorac. Surg 2010, 90, 566–572. [Google Scholar]
- Yang, TL; Young, TH. The enhancement of submandibular gland branch formation on chitosan membranes. Biomaterials 2008, 29, 2501–2508. [Google Scholar]
- Okamoto, Y; Shibazaki, K; Minami, S; Matsuhashi, A; Tanioka, S; Shigemasa, Y. Evaluation of chitin and chitosan on open would healing in dogs. J. Vet. Med. Sci 1995, 57, 851–854. [Google Scholar]
- Kojima, K; Okamoto, Y; Miyatake, K; Fujise, H; Shigemasa, Y; Minami, S. Effects of chitin and chitosan on collagen synthesis in wound healing. J. Vet. Med. Sci 2004, 66, 1595–1598. [Google Scholar]
- Kojima, K; Okamoto, Y; Miyatake, K; Kitamura, Y; Minami, S. Collagen typing of granulation tissue induced by chitin and chitosan. Carbohyd. Polym 1998, 37, 109–113. [Google Scholar]
- Hsiao, YC; Lee, HW; Chen, YT; Young, TH; Yang, TL. The impact of compositional topography of amniotic membrane scaffold on tissue morphogenesis of salivary gland. Biomaterials 2011, (in press). [Google Scholar] [CrossRef]
- Funakoshi, T; Majima, T; Iwasaki, N; Suenaga, N; Sawaguchi, N; Shimode, K; Minami, A; Harada, K; Nishimura, S. Application of tissue engineering techniques for rotator cuff regeneration using a chitosan-based hyaluronan hybrid fiber scaffold. Am. J. Sports. Med 2005, 33, 1193–1201. [Google Scholar]
- Chen, WL; Huang, CH; Chiou, LL; Chen, TH; Huang, YY; Jiang, CC; Lee, HS; Dong, CY. Multiphoton imaging and quantitative analysis of collagen production by chondrogenic human mesenchymal stem cells cultured in chitosan scaffold. Tissue Eng 2010, 16, 913–920. [Google Scholar]
- Jayakumar, R; Prabaharan, M; Kumar, PT; Nair, SV; Tamura, H. Biomaterials based on chitin and chitosan in wound dressing applications. Biotechnol Adv 2011. [Google Scholar]
- Tashiro, T. Antibacterial and bacterium adsorbing macromolecules. Macromol. Mater. Eng 2001, 286, 63–87. [Google Scholar]
- Azad, AK; Sermsintham, N; Chandrkrachang, S; Stevens, WF. Chitosan membrane as a wound-healing dressing: Characterization and clinical application. J. Biomed. Mater. Res. B 2004, 69, 216–222. [Google Scholar]
- Kozen, BG; Kircher, SJ; Henao, J; Godinez, FS; Johnson, AS. An alternative hemostatic dressing: comparison of CELOX, HemCon, and QuikClot. Acad. Emerg. Med 2008, 15, 74–81. [Google Scholar]
- Yannas, IV; Burke, JF; Gordon, PL; Huang, C; Rubenstein, RH. Design of an artificial skin. II. Control of chemical composition. J. Biomed. Mater. Res 1980, 14, 107–132. [Google Scholar]
- Auxenfans, C; Fradette, J; Lequeux, C; Germain, L; Kinikoglu, B; Bechetoille, N; Braye, F; Auger, FA; Damour, O. Evolution of three dimensional skin equivalent models reconstructed in vitro by tissue engineering. Eur. J. Dermatol 2009, 19, 107–113. [Google Scholar]
- Yannas, IV; Burke, JF. Design of an artificial skin. I. Basic design principles. J. Biomed. Mater. Res 1980, 14, 65–81. [Google Scholar]
- Tsai, RJ; Li, LM; Chen, JK. Reconstruction of damaged corneas by transplantation of autologous limbal epithelial cells. N. Engl. J. Med 2000, 343, 86–93. [Google Scholar]
- Yeh, LK; Chen, YH; Chiu, CS; Hu, FR; Young, TH; Wang, IJ. The phenotype of bovine corneal epithelial cells on chitosan membrane. J. Biomed. Mater. Res. A 2009, 90, 18–26. [Google Scholar]
- Friedman, M; Ibrahim, H; Ramakrishnan, V. Inferior turbinate flap for repair of nasal septal perforation. Laryngoscope 2003, 113, 1425–1428. [Google Scholar]
- Davenport, EA; Nettesheim, P. Regulation of mucociliary differentiation of rat tracheal epithelial cells by type I collagen gel substratum. Am. J. Respir. Cell Mol. Biol 1996, 14, 19–26. [Google Scholar]
- Nishijkawa, A; Taira, T; Yoshizato, K. In vitro maturation of collagen fibrils modulates spreading, DNA synthesis, and collagenolysis of epidermal cells and fibroblast. Exp. Cell Res 1987, 171, 164–177. [Google Scholar]
- Taravel, MN; Domard, A. Collagen and its interactions with chitosan, III some biological and mechanical properties. Biomaterials 1996, 17, 451–455. [Google Scholar]
- Affolter, M; Zeller, R; Caussinus, E. Tissue remodelling through branching morphogenesis. Nat. Rev. Mol. Cell Biol 2009, 10, 831–842. [Google Scholar]
- Affolter, M; Bellusci, S; Itoh, N; Shilo, B; Thiery, JP; Werb, Z. Tube or not tube: Remodeling epithelial tissues by branching morphogenesis. Dev. Cell 2003, 4, 11–18. [Google Scholar]
- Yang, TL; Hsiao, YC; Young, TH. Comparison of PLGA, PCL, and chitosan in salivary gland branching morphogenesis. Biomed. Eng.-App. Bas. C 2008, 20, 287–296. [Google Scholar]
- Yang, TL; Hsiao, YC; Lin, SJ; Lee, HW; Lou, PJ; Ko, JY; Young, TH. Biomaterial mediated epithelial-mesenchymal interaction of salivary tissue under serum free condition. Biomaterials 2010, 31, 288–295. [Google Scholar]
- Yang, TL; Young, TH. The specificity of chitosan in promoting branching morphogenesis of progenitor salivary tissue. Biochem. Biophys. Res. Commun 2009, 381, 466–470. [Google Scholar]
- Umeda, Y; Miyazaki, Y; Shiinoki, H; Higashiyama, S; Nakanishi, Y; Hieda, Y. Involvement of heparin-binding EGF-like growth factor and its processing by metalloproteinases in early epithelial morphogenesis of the submandibular gland. Dev. Biol 2001, 237, 202–211. [Google Scholar]
- Grobstein, C. Inductive epitheliomesenchymal interaction in cultured organ rudiments of the mouse. Science 1953, 118, 52–55. [Google Scholar]
- Noguchi, Y; Okamoto, A; Kasama, T; Imajoh-Ohmi, S; Karatsu, T; Nogawa, H. Lysophosphatidic acid cooperates with EGF in inducing branching morphogenesis of embryonic mouse salivary epithelium. Dev. Dyn 2006, 235, 403–410. [Google Scholar]
- Ignotz, RA; Massague, J. Transforming growth-factor-beta stimulates the expression of fibronectin and collagen and their incorporation into the extracellular-matrix. J. Biol. Chem 1986, 261, 4337–4345. [Google Scholar]
- Chalfoun, CT; Wirth, GA; Evans, GR. Tissue engineered nerve constructs: Where do we stand? J. Cell. Mol. Med 2006, 10, 309–317. [Google Scholar]
- Freier, T; Montenegro, R; Shan Koh, H; Shoichet, MS. Chitin-based tubes for tissue engineering in the nervous system. Biomaterials 2005, 26, 4624–4632. [Google Scholar]
- Haipeng, G; Yinghui, Z; Jianchun, L; Yandao, G; Nanming, Z; Xiufang, Z. Studies on nerve cell affinity of chitosan-derived materials. J. Biomed. Mater. Res 2000, 52, 285–295. [Google Scholar]
- Matsuda, A; Kobayashi, H; Itoh, S; Kataoka, K; Tanaka, J. Immobilization of laminin peptide in molecularly aligned chitosan by covalent bonding. Biomaterials 2005, 26, 2273–2279. [Google Scholar]
- Mingyu, C; Kai, G; Jiamou, L; Yandao, G; Nanming, Z; Xiufang, Z. Surface modification and characterization of chitosan film blended with poly-L-lysine. J. Biomater. Appl 2004, 19, 59–75. [Google Scholar]
- Chavez-Delgado, ME; Mora-Galindo, J; Gomez-Pinedo, U; Feria-Velasco, A; Castro-Castaneda, S; Lopez-Dellamary Toral, FA; Luquin-De Anda, S; Garcia-Segura, LM; Garcia-Estrada, J. Facial nerve regeneration through progesterone-loaded chitosan prosthesis. A preliminary report. J. Biomed. Mater. Res. B 2003, 67, 702–711. [Google Scholar]
- Cheng, M; Deng, J; Yang, F; Gong, Y; Zhao, N; Zhang, X. Study on physical properties and nerve cell affinity of composite films from chitosan and gelatin solutions. Biomaterials 2003, 24, 2871–2880. [Google Scholar]
- Wang, X; Hu, W; Cao, Y; Yao, J; Wu, J; Gu, X. Dog sciatic nerve regeneration across a 30-mm defect bridged by a chitosan/PGA artificial nerve graft. Brain 2005, 128, 1897–1910. [Google Scholar]
- Fan, W; Gu, J; Hu, W; Deng, A; Ma, Y; Liu, J; Ding, F; Gu, X. Repairing a 35-mm-long median nerve defect with a chitosan/PGA artificial nerve graft in the human: A case study. Microsurgery 2008, 28, 238–242. [Google Scholar]
- Ding, F; Wu, J; Yang, Y; Hu, W; Zhu, Q; Tang, X; Liu, J; Gu, X. Use of tissue engineered nerve grafts consisting of a chitosan/poly(lactic-co-glycolic acid)-based scaffold included with bridging 50-mm dog sciatic nerve gaps. Tissue Eng Part A 2010, (in press). [Google Scholar]
- Shim, S; Ming, GL. Roles of channels and receptors in the growth cone during PNS axonal regeneration. Exp. Neurol 2010, 223, 38–44. [Google Scholar]
- Yuan, Y; Zhang, P; Yang, Y; Wang, X; Gu, X. The interaction of Schwann cells with chitosan membranes and fibers in vitro. Biomaterials 2004, 25, 4273–4278. [Google Scholar]
- Wang, W; Itoh, S; Konno, K; Kikkawa, T; Ichinose, S; Sakai, K; Ohkuma, T; Watabe, K. Effects of Schwann cell alignment along the oriented electrospun chitosan nanofibers on nerve regeneration. J. Biomed. Mater. Res. A. 2009, 91, 994–1005. [Google Scholar]
- Katz, AJ; Llull, R; Hedrick, MH; Futrell, JW. Emerging approaches to the tissue engineering of fat. Clin. Plast. Surg 1999, 26, 587–603. [Google Scholar]
- Beahm, EK; Walton, RL; Patrick, CW, Jr. Progress in adipose tissue construct development. Clin. Plast. Surg 2003, 30, 547–558. [Google Scholar]
- Wu, X; Black, L; Santacana-Laffitte, G; Patrick, CW, Jr. Preparation and assessment of glutaraldehyde-crosslinked collagen-chitosan hydrogels for adipose tissue engineering. J. Biomed. Mater. Res. A 2007, 81, 59–65. [Google Scholar]
- Laroche, G; Marois, Y; Guidoin, R; King, MW; Martin, L; How, T; Douville, Y. Polyvinylidene fluoride (PVDF) as a biomaterial: From polymeric raw material to monofilament vascular suture. J. Biomed. Mater. Res 1995, 29, 1523–1536. [Google Scholar]
- Mori, T; Irie, Y; Nishimura, SI; Tokura, S; Matsuura, M; Okumura, M; Kadosawa, T; Fujinaga, T. Endothelial cell responses to chitin and its derivatives. J. Biomed. Mater. Res 1998, 43, 469–472. [Google Scholar]
- Malafaya, PB; Santos, TC; van Griensven, M; Reis, RL. Morphology, mechanical characterization and in vivo neo-vascularization of chitosan particle aggregated scaffolds architectures. Biomaterials 2008, 29, 3914–3926. [Google Scholar]
- Chupa, JM; Foster, AM; Sumner, SR; Madihally, SV; Matthew, HW. Vascular cell responses to polysaccharide materials: In vitro and in vivo evaluations. Biomaterials 2000, 21, 2315–2322. [Google Scholar]
- Kratz, G; Arnander, C; Swedenborg, J; Back, M; Falk, C; Gouda, I; Larm, O. Heparin-chitosan complexes stimulate wound healing in human skin. Scand. J. Plast. Reconstr. Surg. Hand Surg 1997, 31, 119–123. [Google Scholar]
- Chandy, T; Rao, GH; Wilson, RF; Das, GS. Delivery of LMW heparin via surface coated chitosan/peg-alginate microspheres prevents thrombosis. Drug Deliv 2002, 9, 87–96. [Google Scholar]
- Fujita, M; Ishihara, M; Shimizu, M; Obara, K; Nakamura, S; Kanatani, Y; Morimoto, Y; Takase, B; Matsui, T; Kikuchi, M; Maehara, T. Therapeutic angiogenesis induced by controlled release of fibroblast growth factor-2 from injectable chitosan/non-anticoagulant heparin hydrogel in a rat hindlimb ischemia model. Wound Repair Regen 2007, 15, 58–65. [Google Scholar]
- Ishihara, M; Obara, K; Nakamura, S; Fujita, M; Masuoka, K; Kanatani, Y; Takase, B; Hattori, H; Morimoto, Y; Ishihara, M; Maehara, T; Kikuchi, M. Chitosan hydrogel as a drug delivery carrier to control angiogenesis. J. Artif. Organs 2006, 9, 8–16. [Google Scholar]

| Scaffold | Application | Effect | Ref. |
|---|---|---|---|
| Chitosan-hyaluronan | Ligament | Cellular adhesion | [57] |
| Cell proliferation | |||
| ECM and production | |||
| Chitosan/Collagen | Blood vessel | Cell adhesion | [58,59] |
| Cell proliferation | |||
| ECM production | |||
| Chitosan/Collagen | Adipose tissue | Vascularization induction | [60] |
| Adipose tissue formation | |||
| Chitosan/Collagen | Skin | Cell proliferation | [63] |
| Cell infiltration | |||
| Collagen-chitosan/fibrin glue | Skin | Cell growth | [64] |
| Collagen-glycosaminoglycan-chitosan | Oral mucosa | Cell differentiation | [65] |
| Cell proliferation | |||
| ECM production | |||
| Chitosan/gelatin | Skin | Cell survival | [66] |
| Chitosan/gelatin | Blood vessel | Cell growth | [44] |
| Cell migration | |||
| Chitosan/silk fibroin | Musculofascia | ECM deposition | [62] |
| Vascularization | |||
| Cellular infiltration |
| Scaffold | Factor | Application | Effect | Ref. |
|---|---|---|---|---|
| Chitosan-hydroxyapatite | FGF2 | Periodontal tissue | Cellular structure formation | [73] |
| Cell proliferation | ||||
| Mineralization | ||||
| Chitosan hydrogel | FGF2 | Wound healing | Wound healing, Angiogenesis | [8] |
| Chitosan hydrogel | FGF2 | Vascularization | Angiogenesis | [83] |
| Chitosan | FGF2 | Wound healing | Fibroblast proliferation, Vasculogenesis | [80] |
| Chitosan-Pluronic | EGF | Wound healing | Keratinocyte differentiation | [81] |
| Wound healing | ||||
| Chitosan-collagen gel | PDGF | Wound healing | Wound healing | [82] |
| Biomimetic effects | ||||
| Chemotactic effects | ||||
| collagen-chitosan/silicone | VEFG | Vascularization | Angiogenesis | [84] |
| Vasculogenesis | ||||
| Methacrylamide chitosan scaffold | IFN-γ | Nerve regeneration | Differentiation of neural progenitor cells | [72] |
| Chitosan/Collagen | TGF-β1 | Periodontal Tissue | Tissue regeneration | [85] |
| Molecule | Tissue/Organ | Effect | Ref. |
|---|---|---|---|
| FGF7 | Salivary gland | Lobular formation | [12] |
| Cell proliferation | |||
| FGF7 | Mesothelium | Synergistic effect | [97] |
| Adhesion decrease | |||
| FGF10 | Salivary gland | Ductal elongation | [12] |
| Cell proliferation | |||
| HGF | Salivary gland | Cell migration | [12] |
| Cell proliferation | |||
| Chemotaxis | |||
| FGF2 | Vasculature | Cell proliferation | [10] |
| FGF2 | Periodontal tissue | Cell proliferation | [73] |
| EGF | Skin | Cell differentiation | [81] |
| ECM component | Tissue/Organ | Effect | Ref. |
|---|---|---|---|
| Collagen, Type I | Skin | Wound healing | [101] |
| Salivary gland | Branching morphogenesis | [98] | |
| Ligament | Cell proliferation | [57] | |
| Vessel | Cell phenotype | [58] | |
| tendon | Cell ECM production | [103] | |
| Collagen, Type III | Skin | Wound healing | [101] |
| Salivary gland | Branching morphogenesis | [98] | |
| Ligament | Cell phenotypes | [54] | |
| Collagen, Type IV | Skin | Wound healing | [101] |
| Glycosaminoglycan | Vessel | Cell phenotype | [58] |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yang, T.-L. Chitin-based Materials in Tissue Engineering: Applications in Soft Tissue and Epithelial Organ. Int. J. Mol. Sci. 2011, 12, 1936-1963. https://doi.org/10.3390/ijms12031936
Yang T-L. Chitin-based Materials in Tissue Engineering: Applications in Soft Tissue and Epithelial Organ. International Journal of Molecular Sciences. 2011; 12(3):1936-1963. https://doi.org/10.3390/ijms12031936
Chicago/Turabian StyleYang, Tsung-Lin. 2011. "Chitin-based Materials in Tissue Engineering: Applications in Soft Tissue and Epithelial Organ" International Journal of Molecular Sciences 12, no. 3: 1936-1963. https://doi.org/10.3390/ijms12031936




