Identification of Receptor Tyrosine Kinase, Discoidin Domain Receptor 1 (DDR1), as a Potential Biomarker for Serous Ovarian Cancer
Abstract
:1. Introduction
2. Materials and Methods
2.1. cDNA Subtraction and Degenerate PCR
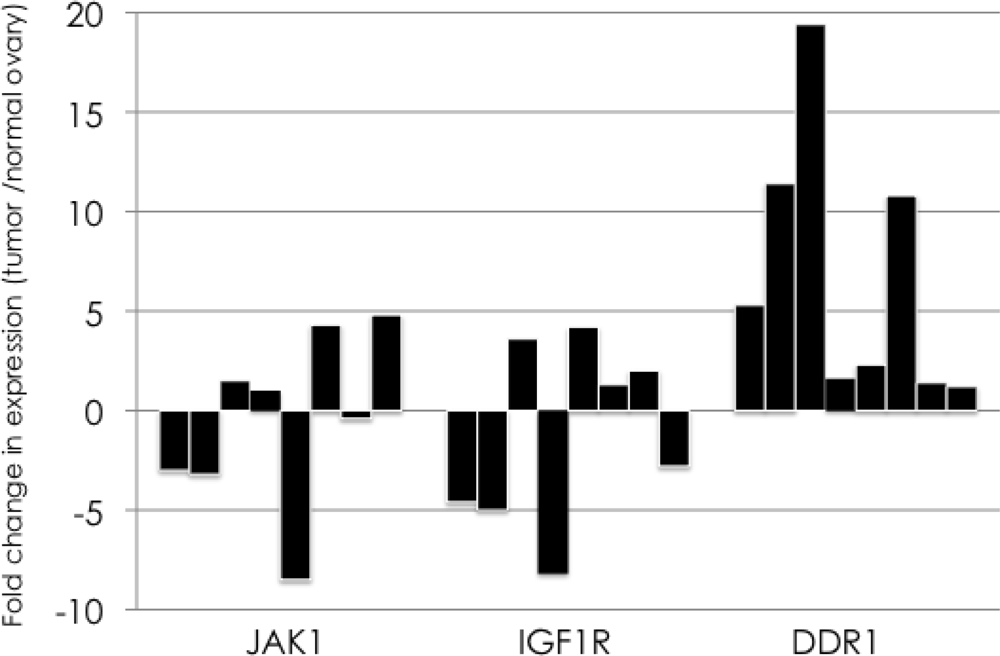
2.2. Real-Time Quantitative Reverse Transcription Polymerase Chain Reaction Assay
2.3. Immunohistochemistry
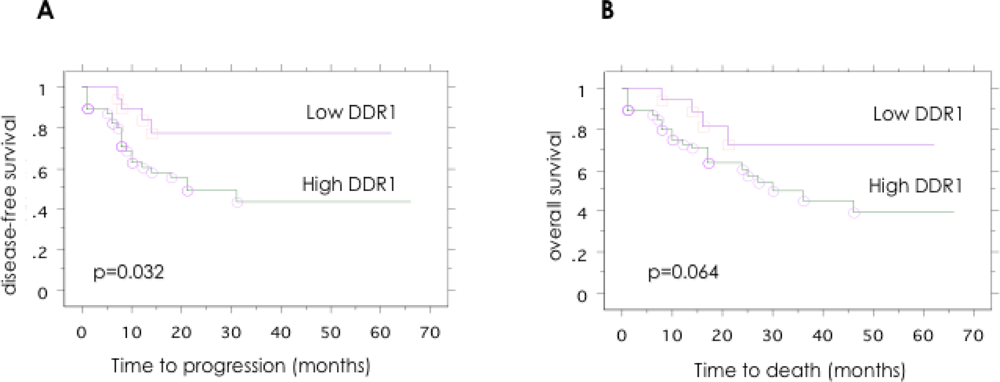
2.4. Association between the Expression of DDR1 Protein in Ovarian Cancer and the Clinical Disease Stage, Tumor Grade, and Patient Outcome
3. Results
4. Discussion
5. Conclusion
References
- Gadducci, A; Cosio, S; Tana, R; Genazzani, AR. Serum and tissue biomarkers as predictive and prognostic variables in epithelial ovarian cancer. Crit. Rev. Oncol. Hematol 2009, 69, 12–27. [Google Scholar]
- Bridges, AJ. Chemical inhibitors of protein kinases. Chem. Rev 2001, 101, 2541–2572. [Google Scholar]
- Seiden, MV; Burris, HA; Matulonis, U; Hall, JB; Armstrong, DK; Speyer, J; Weber, JD; Muggia, F. A phase II trial of EMD72000 (matuzumab), a humanized anti-EGFR monoclonal antibody, in patients with platinum-resistant ovarian and primary peritoneal malignancies. Gynecol. Oncol 2007, 104, 727–731. [Google Scholar]
- Bookman, MA; Darcy, KM; Clarke-Pearson, D; Boothby, RA; Horowitz, IR. Evaluation of monoclonal humanized anti-HER2 antibody, trastuzumab, in patients with recurrent or refractory ovarian or primary peritoneal carcinoma with overexpression of HER2: a phase II trial of the Gynecologic Oncology Group. J. Clin. Oncol 2003, 21, 283–290. [Google Scholar]
- Burger, RA; Sill, MW; Monk, BJ; Greer, BE; Sorosky, JI. Phase II trial of bevacizumab in persistent or recurrent epithelial ovarian cancer or primary peritoneal cancer: a Gynecologic Oncology Group Study. J. Clin. Oncol 2007, 25, 5165–5171. [Google Scholar]
- Hirsch, FR; Varella-Garcia, M; Cappuzzo, F. Predictive value of EGFR and HER2 overexpression in advanced non-small-cell lung cancer. Oncogene 2009, 28, S32–S37. [Google Scholar]
- Baron, AT; Wilken, JA; Haggstrom, DE; Goodrich, ST; Maihle, NJ. Clinical implementation of soluble EGFR (sEGFR) as a theragnostic serum biomarker of breast, lung and ovarian cancer. IDrugs 2009, 12, 302–328. [Google Scholar]
- Turashvili, G; Bouchal, J; Baumforth, K; Wei, W; Dziechciarkova, M; Ehrmann, J; Klein, J; Fridman, E; Skarda, J; Srovnal, J; Hajduch, M; Murray, P; Kolar, Z. Novel markers for differentiation of lobular and ductal invasive breast carcinomas by laser microdissection and microarray analysis. BMC Cancer 2007, 7, 55. [Google Scholar]
- Zerlin, M; Julius, MA; Goldfarb, M. NEP: a novel receptor-like tyrosine kinase expressed in proliferating neuroepithelia. Oncogene 1993, 8, 2731–2739. [Google Scholar]
- Sanchez, MP; Tapley, P; Saini, SS; He, B; Pulido, D; Barbacid, M. Multiple tyrosine protein kinases in rat hippocampal neurons: isolation of Ptk-3, a receptor expressed in proliferative zones of the developing brain. Proc. Natl. Acad. Sci. USA 1994, 91, 1819–1823. [Google Scholar]
- Di Marco, E; Cutuli, N; Guerra, L; Cancedda, R; De Luca, M. Molecular cloning of trkE, a novel trk-related putative tyrosine kinase receptor isolated from normal human keratinocytes and widely expressed by normal human tissues. J. Biol. Chem 1993, 268, 24290–24295. [Google Scholar]
- Bhatt, RS; Tomoda, T; Fang, Y; Hatten, ME. Discoidin domain receptor 1 functions in axon extension of cerebellar granule neurons. Genes. Dev 2000, 14, 2216–2228. [Google Scholar]
- Hou, G; Vogel, W; Bendeck, MP. The discoidin domain receptor tyrosine kinase DDR1 in arterial wound repair. J. Clin. Invest 2001, 107, 727–735. [Google Scholar]
- Vogel, WF; Aszodi, A; Alves, F; Pawson, T. Discoidin domain receptor 1 tyrosine kinase has an essential role in mammary gland development. Mol. Cell. Biol 2001, 21, 2906–2917. [Google Scholar]
- Hansen, C; Greengard, P; Nairn, AC; Andersson, T; Vogel, WF. Phosphorylation of DARPP-32 regulates breast cancer cell migration downstream of the receptor tyrosine kinase DDR1. Exp. Cell Res 2006, 312, 4011–4018. [Google Scholar]
- Jonsson, M; Andersson, T. Repression of Wnt-5a impairs DDR1 phosphorylation and modifies adhesion and migration of mammary cells. J. Cell Sci 2001, 114, 2043–2053. [Google Scholar]
- Perez, JL; Jing, SQ; Wong, TW. Identification of two isoforms of the Cak receptor kinase that are coexpressed in breast tumor cell lines. Oncogene 1996, 12, 1469–1477. [Google Scholar]
- Weiner, HL; Huang, H; Zagzag, D; Boyce, H; Lichtenbaum, R; Ziff, EB. Consistent and selective expression of the discoidin domain receptor-1 tyrosine kinase in human brain tumors. Neurosurgery 2000, 47, 1400–1409. [Google Scholar]
- Rikova, K; Guo, A; Zeng, Q; Possemato, A; Yu, J; Haack, H; Nardone, J; Lee, K; Reeves, C; Li, Y; et al. Global survey of phosphotyrosine signaling identifies oncogenic kinases in lung cancer. Cell 2007, 131, 1190–1203. [Google Scholar]
- Alves, F; Vogel, W; Mossie, K; Millauer, B; Hofler, H; Ullrich, A. Distinct structural characteristics of discoidin I subfamily receptor tyrosine kinases and complementary expression in human cancer. Oncogene 1995, 10, 609–618. [Google Scholar]
- Barker, KT; Martindale, JE; Mitchell, PJ; Kamalati, T; Page, MJ; Phippard, DJ; Dale, TC; Gusterson, BA; Crompton, MR. Expression patterns of the novel receptor-like tyrosine kinase, DDR, in human breast tumours. Oncogene 1995, 10, 569–575. [Google Scholar]
- Yang, SH; Baek, HA; Lee, HJ; Park, HS; Jang, KY; Kang, MJ; Lee, DG; Lee, YC; Moon, WS; Chung, MJ. Discoidin domain receptor 1 is associated with poor prognosis of non-small cell lung carcinomas. Oncol. Rep 2010, 24, 311–319. [Google Scholar]
- Heinzelmann-Schwarz, VA; Gardiner-Garden, M; Henshall, SM; Scurry, J; Scolyer, RA; Davies, MJ; Heinzelmann, M; Kalish, LH; Bali, A; Kench, JG; et al. Overexpression of the cell adhesion molecules DDR1, Claudin 3, and Ep-CAM in metaplastic ovarian epithelium and ovarian cancer. Clin. Cancer Res 2004, 10, 4427–4436. [Google Scholar]
- Nemoto, T; Ohashi, K; Akashi, T; Johnson, JD; Hirokawa, K. Overexpression of protein tyrosine kinases in human esophageal cancer. Pathobiology 1997, 65, 195–203. [Google Scholar]
- Yamanaka, R; Arao, T; Yajima, N; Tsuchiya, N; Homma, J; Tanaka, R; Sano, M; Oide, A; Sekijima, M; Nishio, K. Identification of expressed genes characterizing long-term survival in malignant glioma patients. Oncogene 2006, 25, 5994–6002. [Google Scholar]
- Dejmek, J; Leandersson, K; Manjer, J; Bjartell, A; Emdin, SO; Vogel, WF; Landberg, G; Andersson, T. Expression and signaling activity of Wnt-5a/discoidin domain receptor-1 and Syk plays distinct but decisive roles in breast cancer patient survival. Clin. Cancer Res 2005, 11, 520–528. [Google Scholar]
- Mihai, C; Chotani, M; Elton, TS; Agarwal, G. Mapping of DDR1 distribution and oligomerization on the cell surface by FRET microscopy. J. Mol. Biol 2009, 385, 432–445. [Google Scholar]
- Sakuma, S; Saya, H; Tada, M; Nakao, M; Fujiwara, T; Roth, JA; Sawamura, Y; Shinohe, Y; Abe, H. Receptor protein tyrosine kinase DDR is up-regulated by p53 protein. FEBS Lett 1996, 398, 165–169. [Google Scholar]
- Ongusaha, PP; Kim, JI; Fang, L; Wong, TW; Yancopoulos, GD; Aaronson, SA; Lee, SW. p53 induction and activation of DDR1 kinase counteract p53-mediated apoptosis and influence p53 regulation through a positive feedback loop. EMBO J 2003, 22, 1289–1301. [Google Scholar]
- Shintani, Y; Fukumoto, Y; Chaika, N; Svoboda, R; Wheelock, MJ; Johnson, KR. Collagen I-mediated up-regulation of N-cadherin requires cooperative signals from integrins and discoidin domain receptor 1. J. Cell Biol 2008, 180, 1277–1289. [Google Scholar]
- Su, AI; Wiltshire, T; Batalov, S; Lapp, H; Ching, KA; Block, D; Zhang, J; Soden, R; Hayakawa, M; Kreiman, G; Cooke, MP; Walker, JR; Hogenesch, JB. A gene atlas of the mouse and human protein-encoding transcriptomes. Proc. Natl. Acad. Sci. USA 2004, 101, 6062–6067. [Google Scholar]
- Posadas, EM; Liel, MS; Kwitkowski, V; Minasian, L; Godwin, AK; Hussain, MM; Espina, V; Wood, BJ; Steinberg, SM; Kohn, EC. A phase II and pharmacodynamic study of gefitinib in patients with refractory or recurrent epithelial ovarian cancer. Cancer 2007, 109, 1323–1330. [Google Scholar]
- Matulonis, UA; Berlin, S; Ivy, P; Tyburski, K; Krasner, C; Zarwan, C; Berkenblit, A; Campos, S; Horowitz, N; Cannistra, SA; et al. Cediranib, an oral inhibitor of vascular endothelial growth factor receptor kinases, is an active drug in recurrent epithelial ovarian, fallopian tube, and peritoneal cancer. J. Clin. Oncol 2009, 27, 5601–5606. [Google Scholar]
- Bantscheff, M; Eberhard, D; Abraham, Y; Bastuck, S; Boesche, M; Hobson, S; Mathieson, T; Perrin, J; Raida, M; Rau, C; et al. Quantitative chemical proteomics reveals mechanisms of action of clinical ABL kinase inhibitors. Nat. Biotechnol 2007, 25, 1035–1044. [Google Scholar]
- Rix, U; Hantschel, O; Durnberger, G; Remsing Rix, LL; Planyavsky, M; Fernbach, NV; Kaupe, I; Bennett, KL; Valent, P; Colinge, J; Kocher, T; Superti-Furga, G. Chemical proteomic profiles of the BCR-ABL inhibitors imatinib, nilotinib, and dasatinib reveal novel kinase and nonkinase targets. Blood 2007, 110, 4055–4063. [Google Scholar]

| DDR1 positive | DDR1 negative | |||
|---|---|---|---|---|
| G1 | 12 | 12 |  | P = 0.015 |
| G2 | 21 | 5 | ||
| G3 | 13 | 4 | ||
| Stage | Cases No. | DDR1 positive | Percent of DDR1 positive | |
|---|---|---|---|---|
| I | 22 | 11 |  | p = 0.006 |
| II | 6 | 3 | ||
| III | 35 | 29 | ||
| IV | 4 | 3 | ||
| total | 67 | 46 | ||
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Quan, J.; Yahata, T.; Adachi, S.; Yoshihara, K.; Tanaka, K. Identification of Receptor Tyrosine Kinase, Discoidin Domain Receptor 1 (DDR1), as a Potential Biomarker for Serous Ovarian Cancer. Int. J. Mol. Sci. 2011, 12, 971-982. https://doi.org/10.3390/ijms12020971
Quan J, Yahata T, Adachi S, Yoshihara K, Tanaka K. Identification of Receptor Tyrosine Kinase, Discoidin Domain Receptor 1 (DDR1), as a Potential Biomarker for Serous Ovarian Cancer. International Journal of Molecular Sciences. 2011; 12(2):971-982. https://doi.org/10.3390/ijms12020971
Chicago/Turabian StyleQuan, Jinhua, Tetsuro Yahata, Sosuke Adachi, Kosuke Yoshihara, and Kenichi Tanaka. 2011. "Identification of Receptor Tyrosine Kinase, Discoidin Domain Receptor 1 (DDR1), as a Potential Biomarker for Serous Ovarian Cancer" International Journal of Molecular Sciences 12, no. 2: 971-982. https://doi.org/10.3390/ijms12020971




