Development of a Chitosan-Based Biofoam: Application to the Processing of a Porous Ceramic Material
Abstract
:1. Introduction
2. Experimental Methods
2.1. Material
2.2. Preparation of the Foam Samples
2.3. Testing Methods
3. Physical Characterization Results
3.1. Effect of Chitosan Concentration on the Microstructure
3.2. Effect of Chitosan Concentration on the Density
3.3. Effect of Chitosan Concentration on Thermal Conductivity
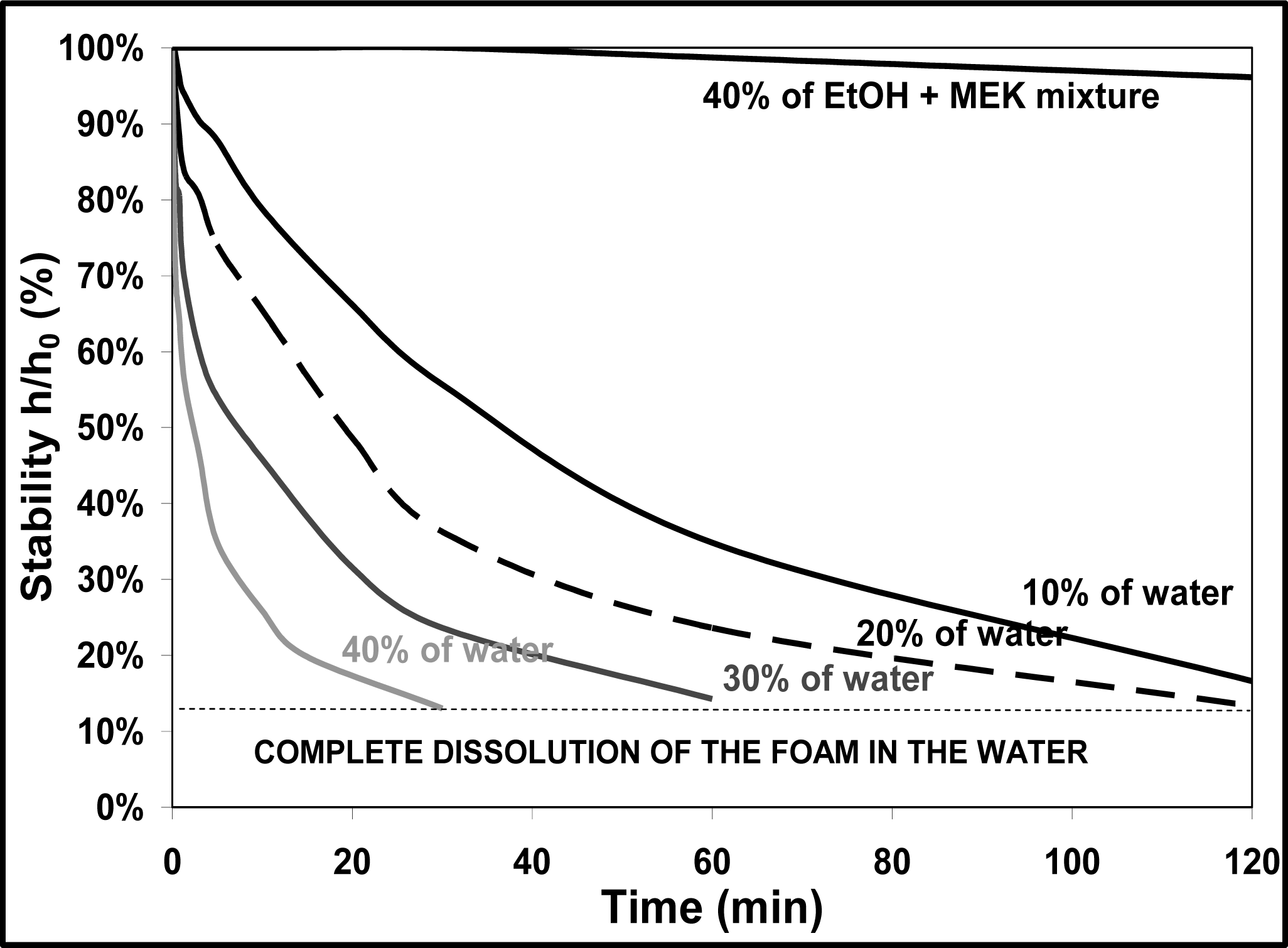
3.4. Influence of a Wet Media
4. Application: Elaboration of a Porous Ceramic Material
4.1. Principles of Replica Technique
4.2. Preparation of the Ceramic Slurry
4.3. Infiltration of the Foam
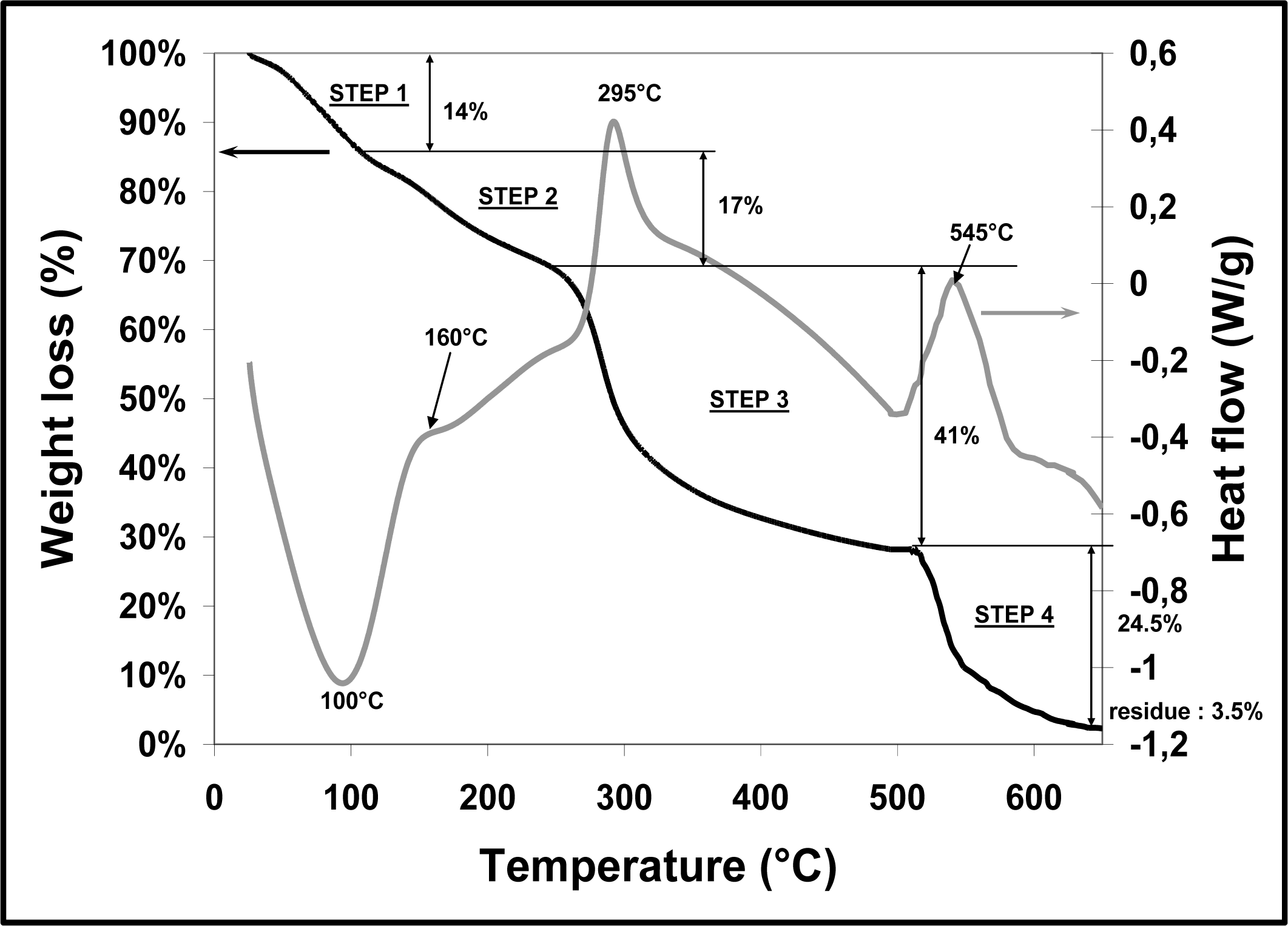
4.4. Pyrolytic Decomposition of the Chitosan Foam
4.5. Sintering of the Green Ceramic Body
4. Conclusion
References
- Resins report. In Modern Plastics; Mc-Graw Hill Co: New York, NY, USA, 1999; pp. 49–80.
- Wang, W; Flores, RA; Huang, CT. Physical properties of two biological cushioning materials from wheat and cornstarches. Cereal Chem 1995, 72(1), 38–41. [Google Scholar]
- Lin, Y; Huff, HE; Parsons, MH; Iannotti, E; Hsieh, F. Mechanical properties of extruded high amylase starch for loose-fill packaging material. Lebensm-Wiss u-Technol 1995, 28(2), 163–168. [Google Scholar]
- Bhatnagar, S; Hanna, MA. Amylose-lipid complex formation during single screw extrusion of various cornstarches. Cereal Chem 1994, 71(6), 582–587. [Google Scholar]
- Bhatnagar, S; Hanna, MA. Physical, mechanical and thermal properties of starch-based plastic foams. Trans ASAE 1995, 38(2), 567–571. [Google Scholar]
- Bhatnagar, S; Hanna, MA. Effect of talc on properties of cornstarch extrudates. Starch:Staerke 1996, 48(3), 94–101. [Google Scholar]
- Lay, G; Rehm, J; Stepto, RF; Thoma, M; Sachetto, J; Lentz, DJ; Silbiger, J. Polymer compositions containing destructible starch. U.S. Patent No. 5,095,054. 1992. [Google Scholar]
- Fang, Q; Hanna, MA. Preparation and characterization of biodegradable copolyester-starch based foams. Bioresour Technol 2001, 78(2), 115–122. [Google Scholar]
- Behling, A. Keep it cool with soybeans: Soy-based foam shows potential as insulation. Soybean Digest 1998, 66(2), 2. [Google Scholar]
- Guo, A; Javni, I; Petrovic, Z. Rigid polyurethane foams based on soybean oil. J Appl Polymer Sci 2000, 77(2), 467–473. [Google Scholar]
- Chain, KS; Gan, LH. Development of a rigid polyurethane foam from palm oil. J Appl Polymer Sci 1998, 68(3), 509–515. [Google Scholar]
- Baser, SA; Khakhar, DV. Castor oil-glycerol blends as polyols for rigid polyurethane. Foams. Cell. Polymer 1993, 12, 390–401. [Google Scholar]
- Muzzarelli, R. Chitosan. In Natural Chelating Polymers; Muzzarelli, R, Ed.; Pergamon Press: Oxford, UK, 1973; pp. 144–176. [Google Scholar]
- Peter, MG. Applications and environmental aspects of chitin and chitosan. J. Macromol. Sci 1995, A32, 629–640. [Google Scholar]
- Salam, A; Pawlak, JJ; Venditti, RA; El-Tahlawy, K. Synthesis and characterization of starch citrate-chitosan foam with superior water and saline absorbance properties. Biomacromolecules 2010, 11, 1453–1459. [Google Scholar]
- He, P; Davis, SS; Illum, L. In vitro evaluation of the mucoadhesive properties of chitosan microspheres. Int. J. Pharm 1998, 166, 75–88. [Google Scholar]
- Muzzarelli, RA. Colorimetric determination of chitosan. Anal. Biochem 1998, 260, 255–257. [Google Scholar]
- Davies, DH; Hayes, ER. Determination of the degree of acetylation of chitin and chitosan. Meth. Enzymol 1988, 161, 442–446. [Google Scholar]
- Hladik, J. Métrologie des propriétés thermophysiques des; Masson: Issy les Moulineaux Cedex, France, 1990. [Google Scholar]
- Colombo, P. Conventional and novel processing methods for cellular ceramics. Phil Trans R Soc 2006, A1(5), 109–124. [Google Scholar]
- Liu, J; Mioa, X. Porous alumina ceramics prepared by slurry infiltration of expanded polystyrene beads. J. Mater. Sci 2005, 40, 6145–6150. [Google Scholar] [Green Version]
- Boch, P; Chartier, T. Ceramic processing techniques: The case of tape casting. Ceram. For. Int 1989, 4, 55–67. [Google Scholar]
- Seal, A; Chattopadhyay, D; Das Sharma, A; Sen, A; Maiti, HS. Influence of ambient temperature on the rheological properties of alumina tape casting slurry. J. Europ. Ceramic Soc 2004, 24, 2275–2283. [Google Scholar]
- Jingxiana, Z; Donglianga, J; Weisenselb, L; Greilb, P. Binary solvent mixture for tape casting of TiO2 sheets. J. Europ. Ceramic Soc 2004, 24, 147–155. [Google Scholar]
- Zawadzki, J; Kaczmarek, H. Thermal treatment of chitosan in various conditions. Carbohyd. Polym 2010, 80, 394–400. [Google Scholar]



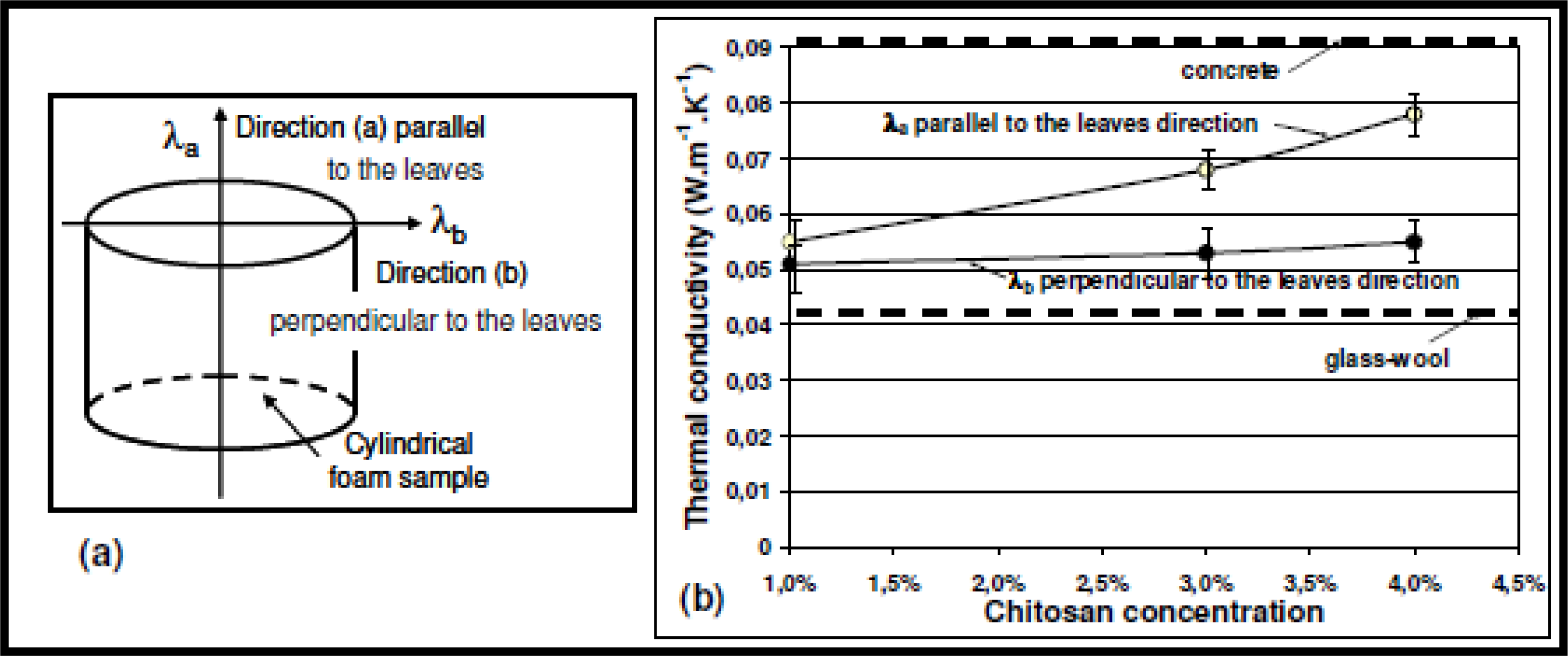

| Chitosan concentration | 1% | 3% | 4% |
| Apparent density ρapp (g·cm−3) | 0.043 | 0.081 | 0.088 |
| Overall porosity rate π | 96% | 93% | 92% |
| Thermal conductivity λa measured in direction [a] (W·m−1·K−1) | 0.055 | 0.068 | 0.078 |
| Thermal conductivity λb measured in direction [b] (W·m−1·K−1) | 0.051 | 0.052 | 0.054 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Mathias, J.-D.; Tessier-Doyen, N.; Michaud, P. Development of a Chitosan-Based Biofoam: Application to the Processing of a Porous Ceramic Material. Int. J. Mol. Sci. 2011, 12, 1175-1186. https://doi.org/10.3390/ijms12021175
Mathias J-D, Tessier-Doyen N, Michaud P. Development of a Chitosan-Based Biofoam: Application to the Processing of a Porous Ceramic Material. International Journal of Molecular Sciences. 2011; 12(2):1175-1186. https://doi.org/10.3390/ijms12021175
Chicago/Turabian StyleMathias, Jean-Denis, Nicolas Tessier-Doyen, and Philippe Michaud. 2011. "Development of a Chitosan-Based Biofoam: Application to the Processing of a Porous Ceramic Material" International Journal of Molecular Sciences 12, no. 2: 1175-1186. https://doi.org/10.3390/ijms12021175
APA StyleMathias, J.-D., Tessier-Doyen, N., & Michaud, P. (2011). Development of a Chitosan-Based Biofoam: Application to the Processing of a Porous Ceramic Material. International Journal of Molecular Sciences, 12(2), 1175-1186. https://doi.org/10.3390/ijms12021175





