The Relationships Between Biological Activities and Structure of Flavan-3-Ols
Abstract
:1. Introduction
2. Results and Discussion
2.1. The Antiproliferative Effect of Flavan-3-Ols and GA
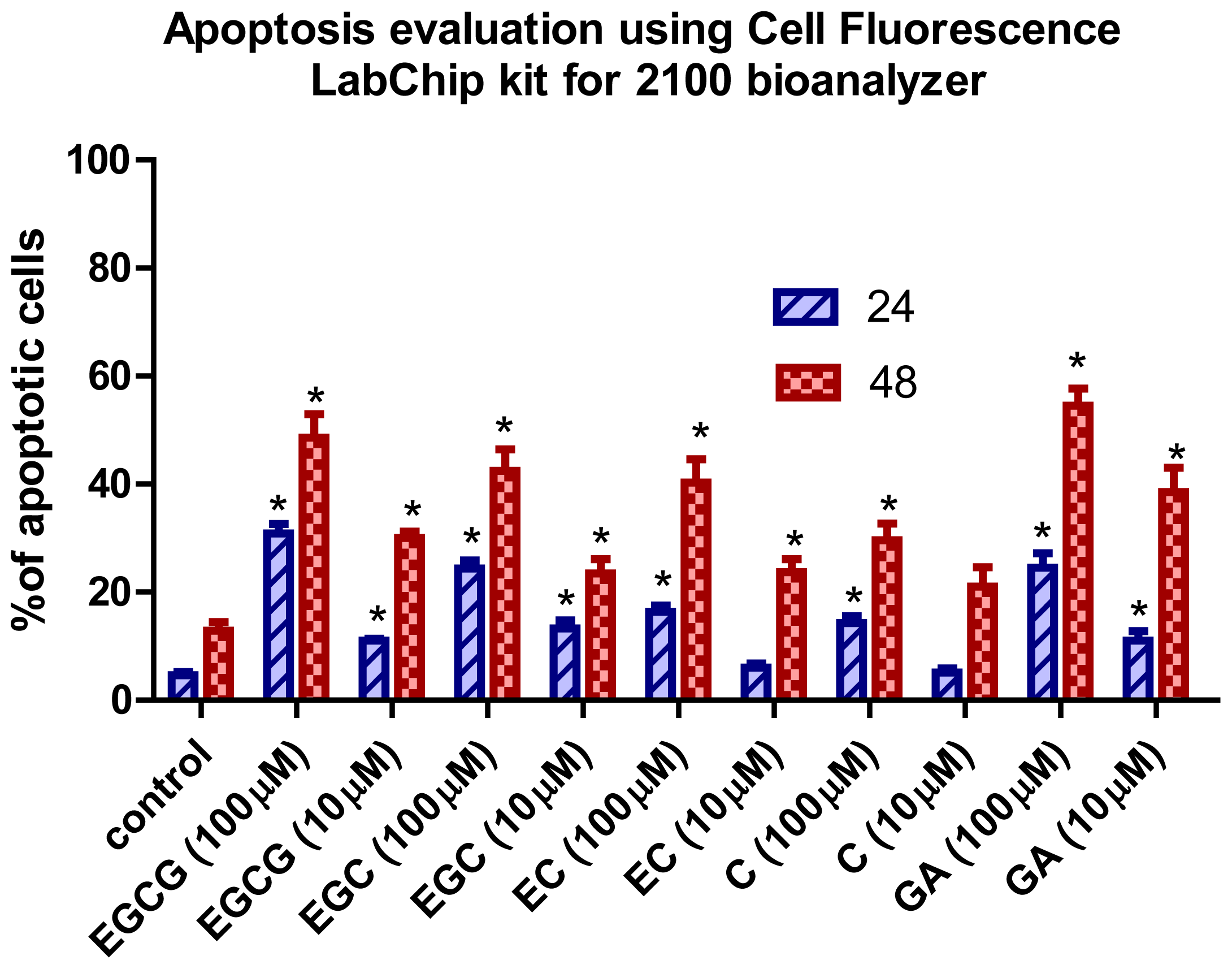
2.2. Apoptotic Effects of Flavan-3-Ols and GA
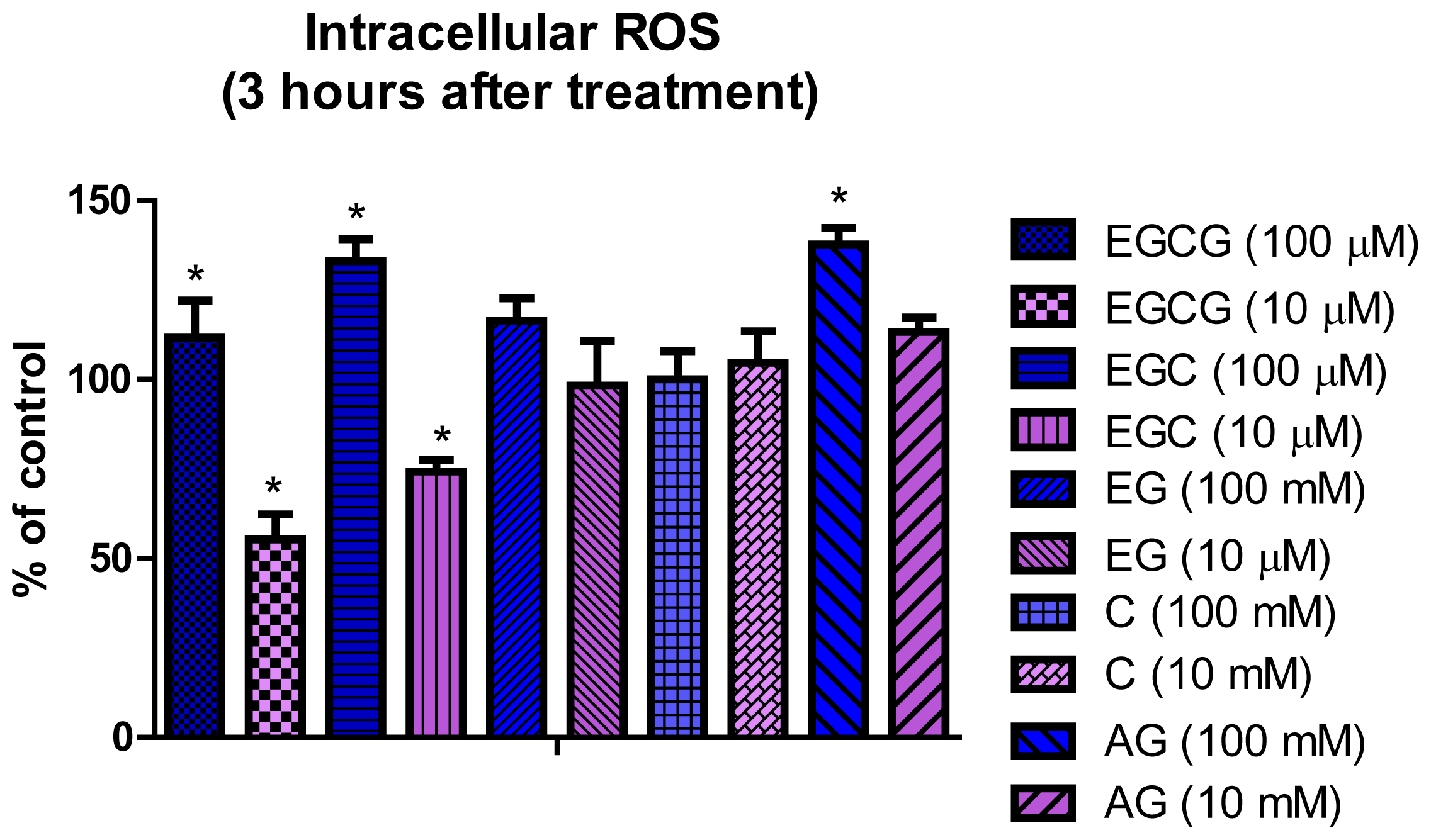
2.3. Detection of Intracellular ROS
3. Experimental Section
3.1. Cell Culture
3.2. Cell Treatment
3.3. MTT Assay
3.4. Detection of Intracellular ROS
3.5. On-Chip Flow Cytometry
4. Conclusions
Acknowledgments
References
- Henning, S.; Choo, J.; Heber, D. Nongallated compared with gallated flavan-3-ols in green and black tea are more bioavailable. J. Nutr 2008, 138, 1529S–1534S. [Google Scholar]
- Velayutham, P.; Babu, A.; Liu, D. Green tea catechins and cardiovascular health: An update. Curr. Med. Chem 2008, 15, 1840–1850. [Google Scholar]
- Thangapazham, R.L.; Passi, N.; Maheshwari, R.K. Green tea polyphenol and epigallocatechin gallate induce apoptosis and inhibit invasion in human breast cancer cells. Cancer Biol. Ther 2007, 6, 1938–1943. [Google Scholar]
- Kampa, M.; Alexaki, V.-I.; Notas, G. Antiproliferative and apoptotic effects of selective phenolic acids on T47D human breast cancer cells: Potential mechanisms of action breast. Cancer Res 2004, 6, R63–R74. [Google Scholar]
- Chen, C.; Shen, G.; Hebbar, V.; Hu, R.; Owuor, E.D.; Kong, A.N. Epigallocatechin-3-gallate-induced stress signals in HT-29 human colon adenocarcinoma cells. Carcinogenesis 2003, 24, 1369–1378. [Google Scholar]
- Chen, Z.P.; Schell, J.B.; Ho, C.T.; Chen, K.Y. Green tea epigallocatechin gallate shows a pronounced growth inhibitory effect on cancerous cells but not on their normal counterparts. Cancer Lett 1998, 129, 173–179. [Google Scholar]
- Tang, Y.Q.; Jaganath, I.B.; Sekaran, S.D. Phyllanthus SPP. induces selective growth inhibition of PC-3 and MeWo human cancer cells through modulation of cell cycle and induction of apoptosis. PLoS One 2009, 5, e12644:1–e12644:11. [Google Scholar]
- Kavanagh, K.T.; Hafer, L.J.; Kim, D.W.; Mann, K.K.; Sherr, D.H.; Rogers, A.E.; Sonenshein, G.E. Green tea extracts decrease carcinogen-induced mammary tumor burden in rats and rate of breast cancer cell proliferation in culture. J. Cell. Biochem 2001, 82, 387–398. [Google Scholar]
- Muzolf-Panek, M.; Gliszczyńska-Świglo, A.; de Haan, L.; Aarts, J.M.M.J.G.; Vervoort, J.; Tyrakowska, B.; Rietjens, I.M.C.M. Prooxidant activity of catechins induces EpRE-mediated gene expression. Chem. Res. Toxicol. 2008, 21, 2352–2360. [Google Scholar]
- Saeki, K.; Hayakawa, S.; Isemura, M.; Miyase, T. Importance of a pyrogallol-type structure in catechin compounds for apoptosis-inducing activity. Phytochemistry 2000, 53, 391–394. [Google Scholar]
- Isemura, M.; Saeki, K.; Kimura, T.; Hayakawa, S.; Minami, T.; Sazuka, M. Tea catechins and related polyphenols as anti-cancer agents. Biofactors 2000, 13, 81–85. [Google Scholar]
- Horie, N.; Hirabayashi, N.; Takahashi, Y.; Miyauchi, Y.; Taguchi, H.; Takeishi, K. Synergistic effect of green tea catechins on cell growth and apoptosis induction in gastric carcinoma cells. Biol. Pharm. Bull 2005, 28, 574–579. [Google Scholar]
- Sakagami, H.; Satoh, K.; Hatano, T.; Yoshida, T.; Okuda, T. Possible role of radical intensity and oxidation potential for gallic acid-induced apoptosis. Anticancer Res 1997, 17, 377–380. [Google Scholar]
- Yang, C.S.; Kim, S.; Yang, G.Y.; Lee, M.J. Inhibition of carcinogenesis by tea: Bioavailability of tea polyphenols and mechanisms of actions. Proc. Soc. Exp. Biol. Med 1999, 220, 213–217. [Google Scholar]
- Aldini, G.; Yeum, K.J.; Carini, M.; Krinsky, N.I.; Russell, R. (−)-Epigallocatechin-3-gallate prevents oxidative damage in both the aqueous and lipid compartments of human plasma. Biochem. Biophys. Res. Commun. 2003, 302, 409–414. [Google Scholar]
- Chedea, V.S.; Braicu, C.; Socaciu, C. Antioxidant/prooxidant activity of a polyphenolic grape seed extract. Food Chem 2010, 121, 132–139. [Google Scholar]
- Peng, I.-W.; Kuo, S.-M. Flavonoid structure affects the inhibition of lipid peroxidation in Caco-2 intestinal cells at physiological concentrations. J. Nutr 2003, 133, 2184–2187. [Google Scholar]
- Lotito, S.B.; Fraga, C.G. Catechins delay lipid oxidation and alpha-tocopherol and beta-carotene depletion following ascorbate depletion in human plasma. Proc. Soc. Exp. Biol. Med 2000, 225, 32–38. [Google Scholar]
- Guo, Q.; Zhao, B.; Shen, S.; Hou, J.; Hu, J.; Xin, W. ESR study on the structure–antioxidant activity relationship of tea catechins and their epimers. Biochim. Biophys. Acta 1999, 1427, 13–23. [Google Scholar]
- Chobot, V.; Huber, C.; Trettenhahn, G.; Hadacek, F. (±)-Catechin: Chemical weapon, antioxidant, or stress regulator. J. Chem. Ecol. 2009, 35, 980–996. [Google Scholar]
- Nanjo, F.; Mori, M.; Goto, K.; Hara, Z. Radical scavenging activity of tea catechins and their related compounds. Biosci. Biotechnol. Biochem 1999, 6, 1621–1623. [Google Scholar]
- Heim, K.E.; Tagliaferro, A.R.; Bobilya, D.J. Flavonoid antioxidants: Chemistry, metabolism and structure-activity relationships. J. Nutr. Biochem 2002, 13, 572–584. [Google Scholar]
- Dwibedy, P.; Dey, G.R.; Naik, D.B.; Kishore, K.; Moorthy, P.N. Pulse radiolysis studies on redox reactions of gallic acid: One electron oxidation of gallic acid by gallic acid–OH adductPhys. Chem. Chem. Phys 1999, 1, 1915–1918. [Google Scholar]
- Weisburg, J.H.; Weissman, D.B.; Sedaghat, T.; Babich, H. In vitro cytotoxicity of epigallocatechin gallate and tea extracts to cancerous and normal cells from the human oral cavity. Basic Clin. Pharmacol. Toxicol 2004, 95, 191–200. [Google Scholar]
- Azam, S.; Hadi, N.; Khan, N.U.; Hadi, S.M. Prooxidant property of green tea polyphenols epicatechin and epigallocatechin-3-gallate: Implications for anticancer properties. Toxicol. In Vitro 2004, 18, 555–561. [Google Scholar]
- Azam, S.; Hadi, N.; Khan, N.U.; Hadi, S.M. Prooxidant property of green tea polyphenols epicatechin and epigallocatechin-3-gallate: Implications for anticancer properties. Toxicol. In Vitro 2004, 18, 555–561. [Google Scholar]
- Khan, N.; Afaq, F.; Saleem, M.; Ahmad, N.; Mukhtar, H. Targeting multiple signaling pathways by green tea polyphenol (−)-epigallocatechin-3-gallate. Cancer Res 2006, 66, 2500–2505. [Google Scholar]
- Chen, L.-J.; Yang, X.-G.; Shen, S.-R.; Wang, Y.-F. Mechanism of scavenging reactive oxygen radicals of tea catechins. J. Zhejiang Univ 2002, 28, 563–574. [Google Scholar]
- Braicu, C.; Rugina, D.; Chedea, V.S.; Balacescu, L.; Brie, I.; Soritau, O.; Socaciu, C.; Irimie, A. Protective action of different natural flavan-3-ols against aflatoxin B1-related cytotoxicity. J. Food Biochem 2010, 34, 595–610. [Google Scholar]


| Time (h) | Compound | IC50 (μM) | Slope | R2 |
|---|---|---|---|---|
| 24 | EGCG | 131.6 | −1.086 | 0.9522 |
| ECG | 155.1 | −0.7287 | 0.9601 | |
| EC | 180.4 | −0.7303 | 0.7820 | |
| C | 326.8 | −0.4809 | 0.6794 | |
| GA | 211.1 | −0.7720 | 0.9016 | |
| 48 | EGCG | 15.8 | −0.3760 | 0.6944 |
| EGC | 43.49 | −0.7227 | 0.5283 | |
| EC | 89.59 | −1.220 | 0.9251 | |
| C | 250.0 | −1.233 | 0.9324 | |
| GA | 40.14 | −1.002 | 0.9424 | |
| 72 | EGCG | 17.75 | −0.9903 | 0.9509 |
| ECG | 32.56 | −0.7985 | 0.9446 | |
| EC | 67.45 | −0.5975 | 0.8716 | |
| C | 233 | −1.205 | 0.9136 | |
| GA | 31.67 | −1.270 | 0.9394 |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Braicu, C.; Pilecki, V.; Balacescu, O.; Irimie, A.; Berindan Neagoe, I. The Relationships Between Biological Activities and Structure of Flavan-3-Ols. Int. J. Mol. Sci. 2011, 12, 9342-9353. https://doi.org/10.3390/ijms12129342
Braicu C, Pilecki V, Balacescu O, Irimie A, Berindan Neagoe I. The Relationships Between Biological Activities and Structure of Flavan-3-Ols. International Journal of Molecular Sciences. 2011; 12(12):9342-9353. https://doi.org/10.3390/ijms12129342
Chicago/Turabian StyleBraicu, Cornelia, Valentina Pilecki, Ovidiu Balacescu, Alexandru Irimie, and Ioana Berindan Neagoe. 2011. "The Relationships Between Biological Activities and Structure of Flavan-3-Ols" International Journal of Molecular Sciences 12, no. 12: 9342-9353. https://doi.org/10.3390/ijms12129342







