Melanogenesis Inhibitory Activity of Two Generic Drugs: Cinnarizine and Trazodone in Mouse B16 Melanoma Cells
Abstract
:1. Introduction
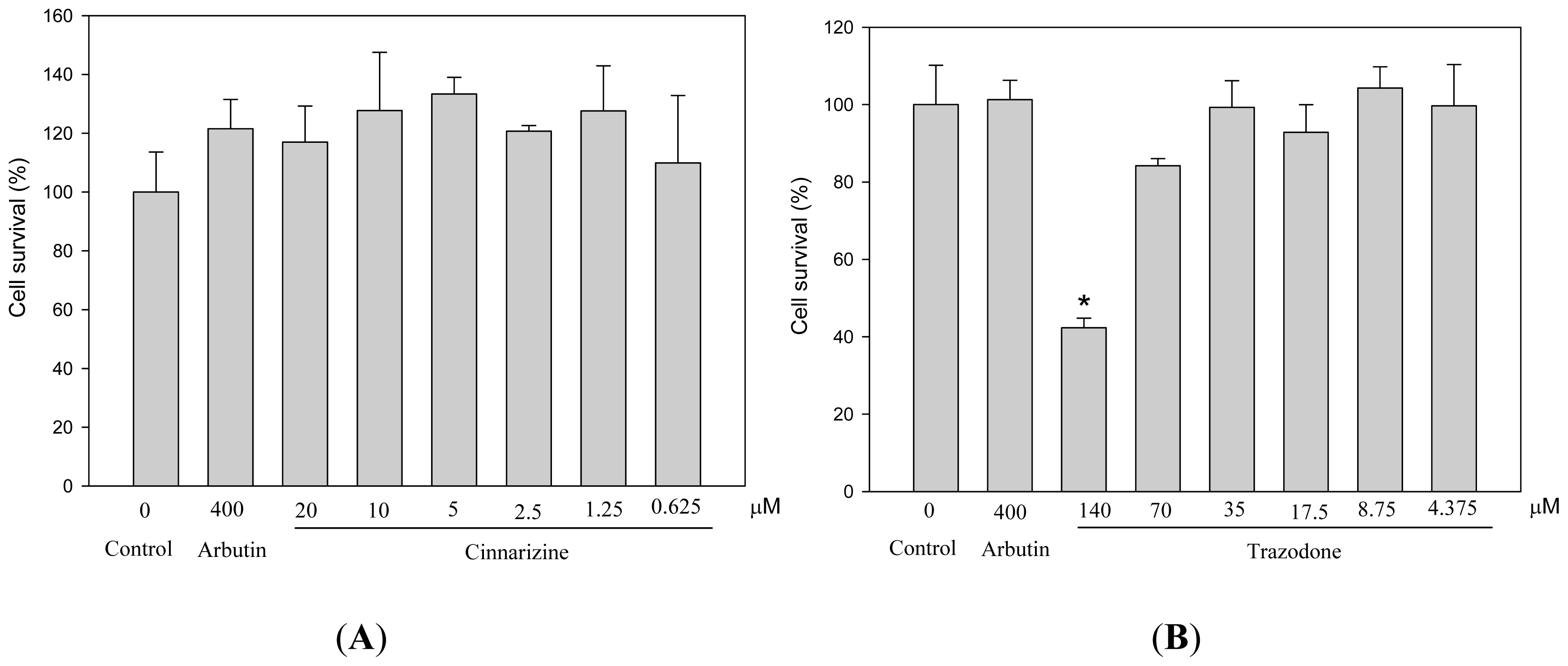
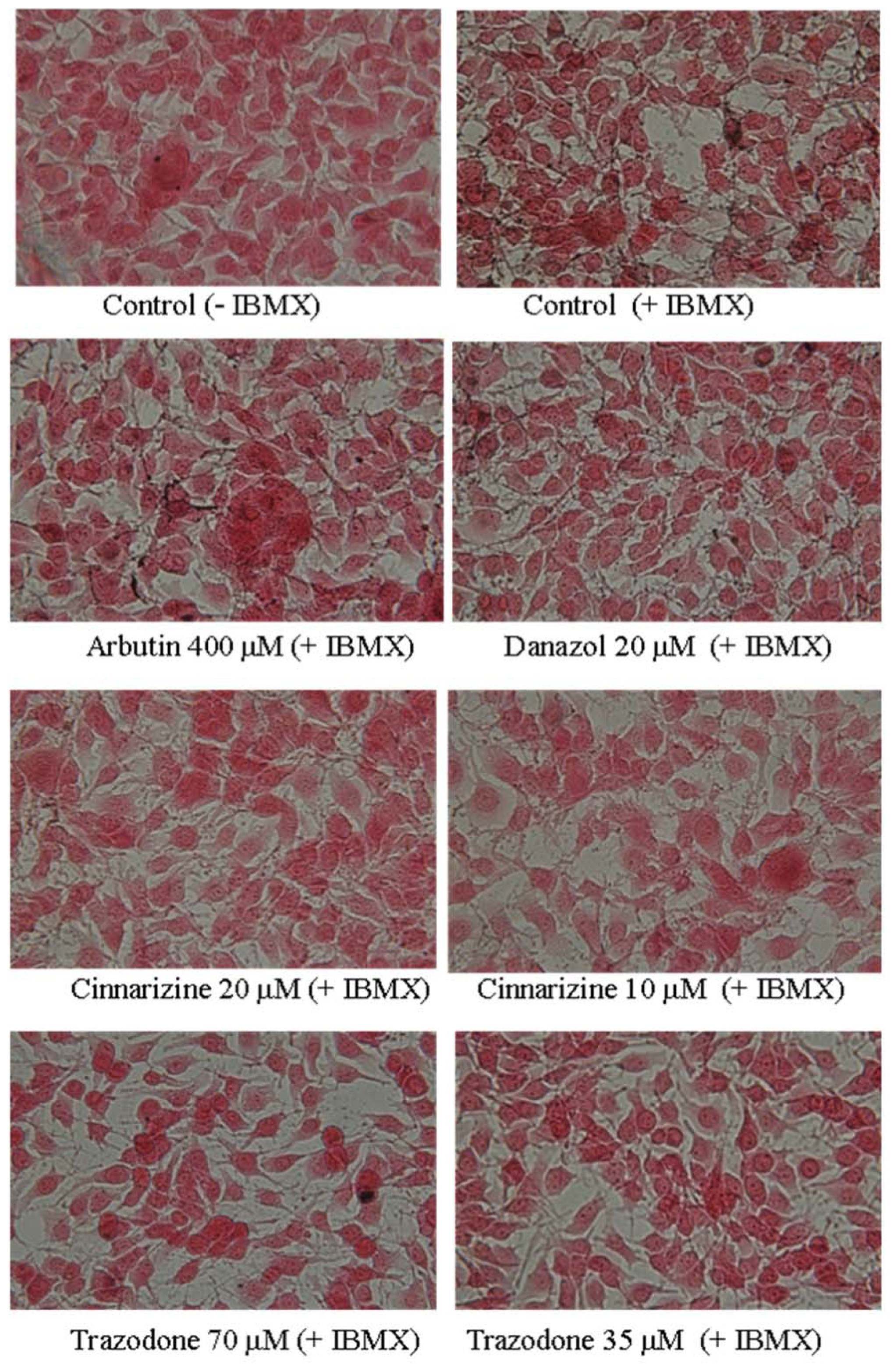
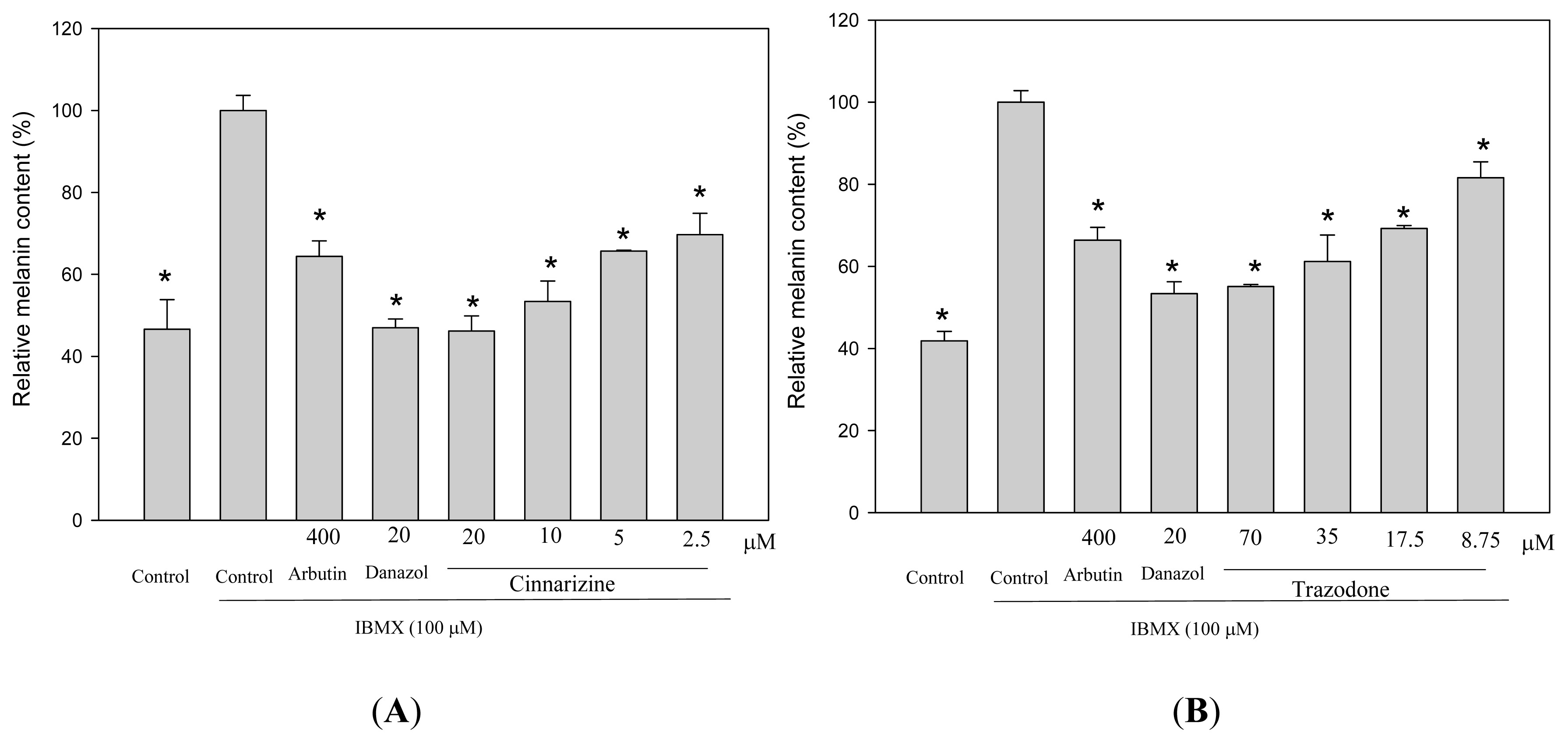
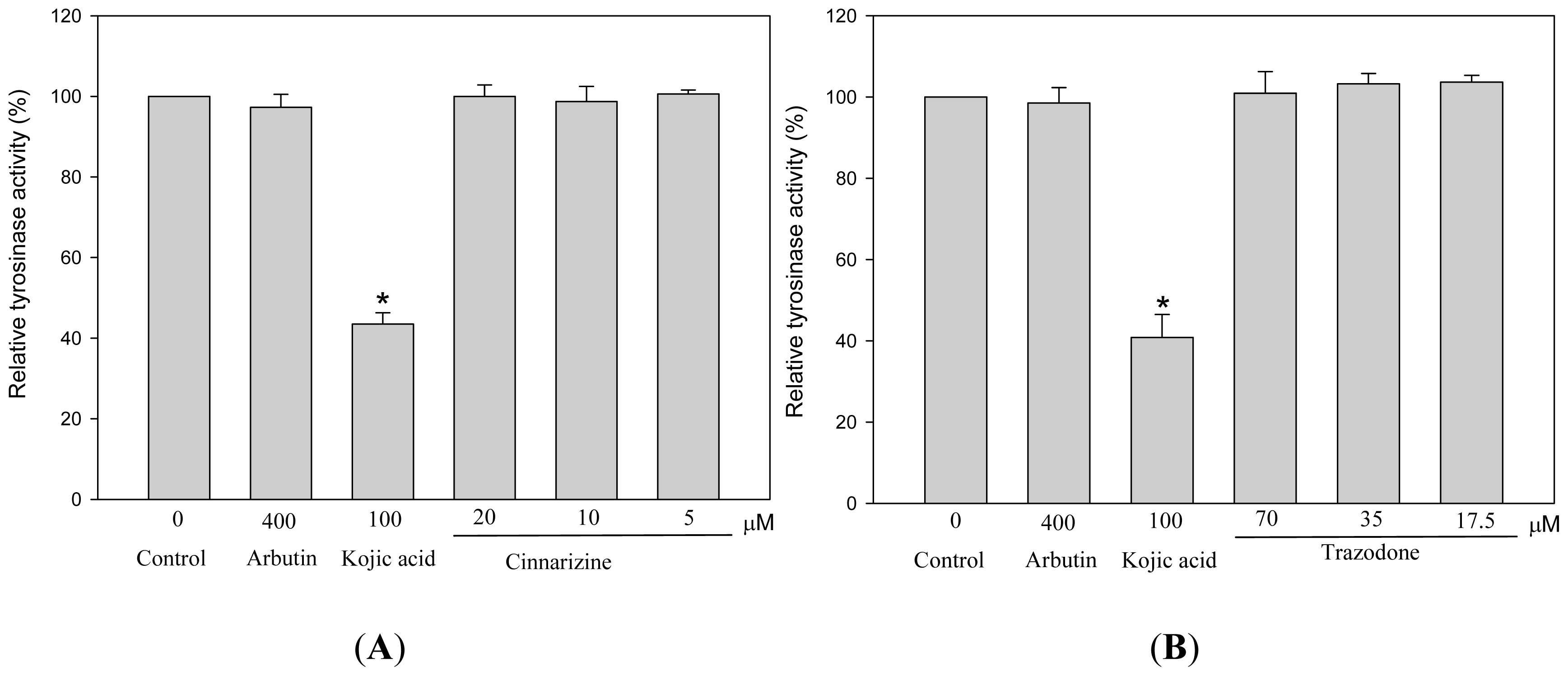
2. Results and Discussion
3. Experimental Section
3.1. Materials
3.2. Cell Cultures and Drug Treatments
3.3. Measurements of Cell Viability
3.4. Fontana-Masson Stain
3.5. Determination of Melanin Content
3.6. Measurements of Tyrosinase Activity
3.7. Statistical Analysis
4. Conclusions
Acknowledgments
- Conflict of InterestThe author declares no conflict of interest.
References
- Chang, T.S. An updated review on tyrosinase inhibitors. Int. J. Mol. Sci 2009, 10, 2400–2475. [Google Scholar]
- Sato, K.; Toriyama, M. The inhibitory effect of non-steroidal anti-inflammatory drugs (NSAIDs) on the monophenolase and diphenolase activities of mushroom tyrosinase. Int. J. Mol. Sci 2011, 12, 3998–4008. [Google Scholar]
- Lin, C.C.; Yang, C.H.; Chang, N.F.; Wu, P.S.; Chen, Y.S.; Lee, S.M.; Chen, C.W. Study on the stability of deoxyarbutin in an anhydrous emulsion systemy. Int. J. Mol. Sci 2011, 12, 5946–5954. [Google Scholar]
- Ding, H.Y.; Chang, T.S.; Shen, H.C.; Tai, S.S.K. Murine tyrosinase inhibitors from Cynanchum bungei and evaluation of in vitro and in vivo depigmenting activity. Exp. Dermatol 2011, 20, 720–724. [Google Scholar]
- Lin, C.C.; Ding, H.Y.; Tsai, P.C.; Wu, J.Y.; Lu, Y.H.; Chang, T.S. In vitro and in vivo melanogenesis inhibition by biochanin A from Trifolium pretense. Biosci. Biotechnol. Biochem 2011, 75, 914–918. [Google Scholar]
- Tai, S.S.K.; Lin, C.G.; Wu, M.H.; Chang, T.S. Evaluation of depigmenting activity by 8-hydroxydaidzein in mouse B16 melanoma cells and human volunteers. Int. J. Mol. Sci 2009, 10, 4257–4266. [Google Scholar]
- Lin, V.H.; Ding, H.Y.; Kuo, S.Y.; Chin, L.W.; Wu, J.Y.; Chang, T.S. Evaluation of in vitro and in vivo depigmenting activity of raspberry ketone from Rheum officinale. Int. J. Mol. Sci 2011, 12, 4819–4835. [Google Scholar]
- Sato, K.; Takahashi, H.; Iraha, R.; Toriyama, M. Down-regulation of tyrosinase expression by acetylsalicylic acid in murine B16 melanoma. Biol. Pharm. Bull 2008, 31, 33–37. [Google Scholar]
- Mun, Y.J.; Lee, S.W.; Jeong, H.W.; Lee, K.G.; Kim, J.H.; Woo, W.H. Inhibitory effect of miconazole on melanogenesis. Biol. Pharm. Bull 2004, 27, 806–809. [Google Scholar]
- Chang, T.S.; Lin, J.J. Inhibitory effect of danazol on melanogenesis in mouse B16 melanoma cells. Arch. Pharm. Res 2010, 33, 1959–1965. [Google Scholar]
- Chang, T.S.; Chen, C.T. Inhibitory effect of homochlorcyclizine on melanogenesis in α-melanocyte stimulating hormone-stimulated mouse B16 melanoma cells. Arch. Pharm. Res 2011, in press. [Google Scholar]
- Imokawa, G. Autocrine and paracrine regulation of melanocytes in human skin and in pigmentary disorders. Pigment Cell Res 2004, 17, 96–110. [Google Scholar]
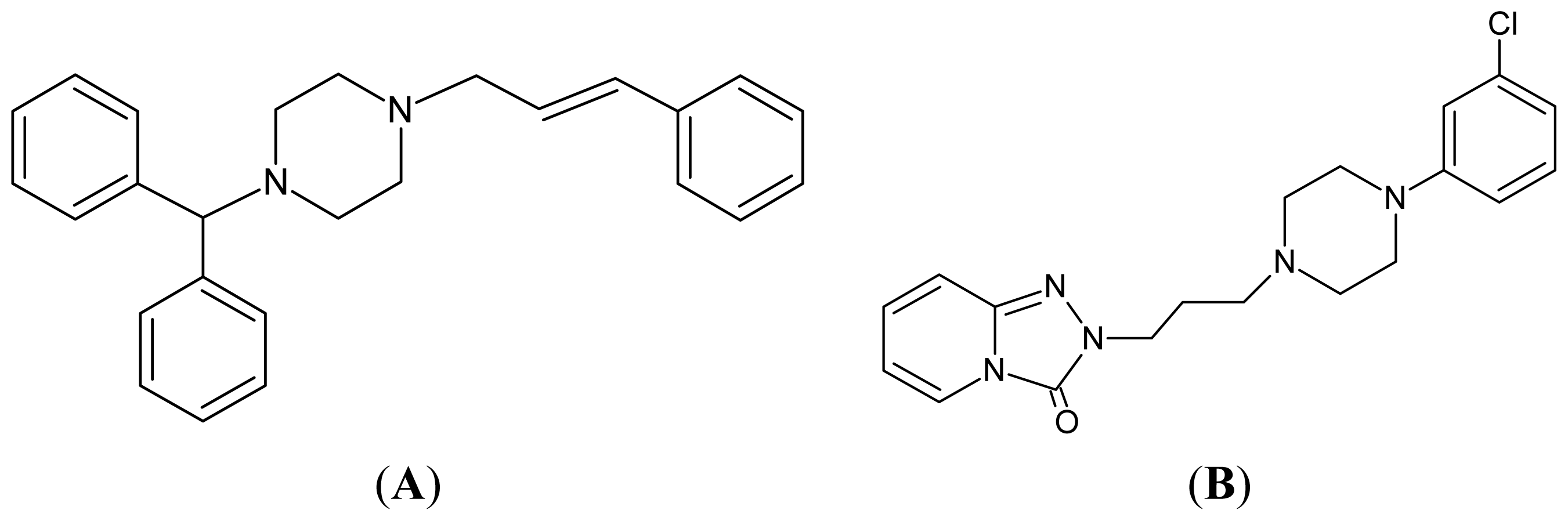
| The generic drugs screened in this study | ||||
|---|---|---|---|---|
| Acarbose | Colchicine | Fexofenadine | Meloxicam | Pertiazem |
| Acemetacin | Cortisone | Fluconazole | Mepenzolate | Phenytoin |
| Acetaminophen | Coumadin | Fludiazepam | Mephenoxalone | Pioglitazone |
| Aldioxa | Cyprohetadine | Flunarizine | Mequitazine | Piracetam |
| Allopurinol | Dacoton | Fluoxetine | Metformin | Polaramine |
| Alusa | Danazol | Formoterol | Methimazole | Ranitidine |
| Ambroxol | Dexamethasone | Furosemide | Methocarbamol | Rifater |
| Amlodipine | Dexchlorpheniramine | Gemfibrozil | Methotrexate | Ritampin |
| Amoxicillin | Diclofenac potassium | Ginkgoflavon | Metoclopramide | Rosiglitazone |
| Apresoline | Dicloxacillin | Glibenclamide | Metronidazole | Selegiline |
| Aspirin | Dihydroergotoxine | Glimepiride | Mexiletine | Sennoside |
| Atenolol | Dimethicone | Glipizide | Miconazole | Silymarin |
| Baclofen | Diovan | Glucosamine | Minocycline | Simvastatin |
| Benzbromarone | Diphenidol | Homochlorcyclizine | Mosapride | Songora |
| Benzonatate | Dipyridamole | Hydralazine | Nalidixic acid | Souriree |
| Bethanechol | Ditiazem | Hydroxychloroquine | Naproxen | Spironolaotone |
| Biorix | Domperidol | Hydroxyzine | Narcaricin | Strocain |
| Bisacodyl | Doxazosin | Hyoscyamine | Neomycin | Sulfinpyrazone |
| Bismuth | Doxycycline | Ibuprofen | Nicardipine | Sulindac |
| Bisoprolol | Doxymycin | Imdur | Nicergoline | Tamoxifen |
| Bromhexine | Elistin | Indapamide | Nicorandil | Terazosin |
| Captopril | Enalapril | Indomethanoin | Nifedipine | Theophyllin |
| Carvedilol | Entacapone | Iodopropylidene | Nifuroxazide | Thyroxine |
| Cefadroxil | Enzdase | Isoniazid | Nimodipine | Trazodone |
| Celecoxib | Eprazine | Isosorbide | Nitroxoline | Trihexyphenidy |
| Cephalexin | Ergonovine | Ketoconazole | Nystatin | Trimethoprim |
| Cetirizine | Erythromycin | Ketorolac | Ofloxacin | Tsurupinate |
| Cimetidine | Escitalopram | Levamisole | Oxatomide | Uliden |
| Cinnarizine | Ethambutol | Loperamide | Pantoprazole | Ultracet |
| Ciprofloxacin | Famotidine | Loratadine | Pecolin | Valproic acid |
| Clindamycin | Felodipine | Mapirocin | Pentoxifylline | Verapamil |
| Clonazepam | Fenoterol | Mebendazole | Peptidin | Warfarin |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chang, T.-S.; Lin, V.C.-H. Melanogenesis Inhibitory Activity of Two Generic Drugs: Cinnarizine and Trazodone in Mouse B16 Melanoma Cells. Int. J. Mol. Sci. 2011, 12, 8787-8796. https://doi.org/10.3390/ijms12128787
Chang T-S, Lin VC-H. Melanogenesis Inhibitory Activity of Two Generic Drugs: Cinnarizine and Trazodone in Mouse B16 Melanoma Cells. International Journal of Molecular Sciences. 2011; 12(12):8787-8796. https://doi.org/10.3390/ijms12128787
Chicago/Turabian StyleChang, Te-Sheng, and Victor Chia-Hsiang Lin. 2011. "Melanogenesis Inhibitory Activity of Two Generic Drugs: Cinnarizine and Trazodone in Mouse B16 Melanoma Cells" International Journal of Molecular Sciences 12, no. 12: 8787-8796. https://doi.org/10.3390/ijms12128787




