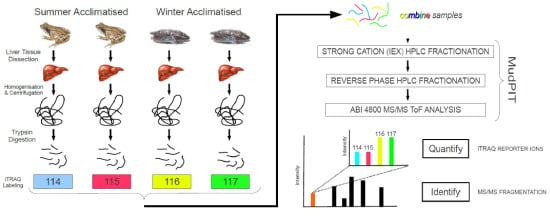
Seasonal Variation in the Hepatoproteome of the Dehydration- and Freeze-Tolerant Wood Frog, Rana sylvatica
Abstract
:1. Introduction
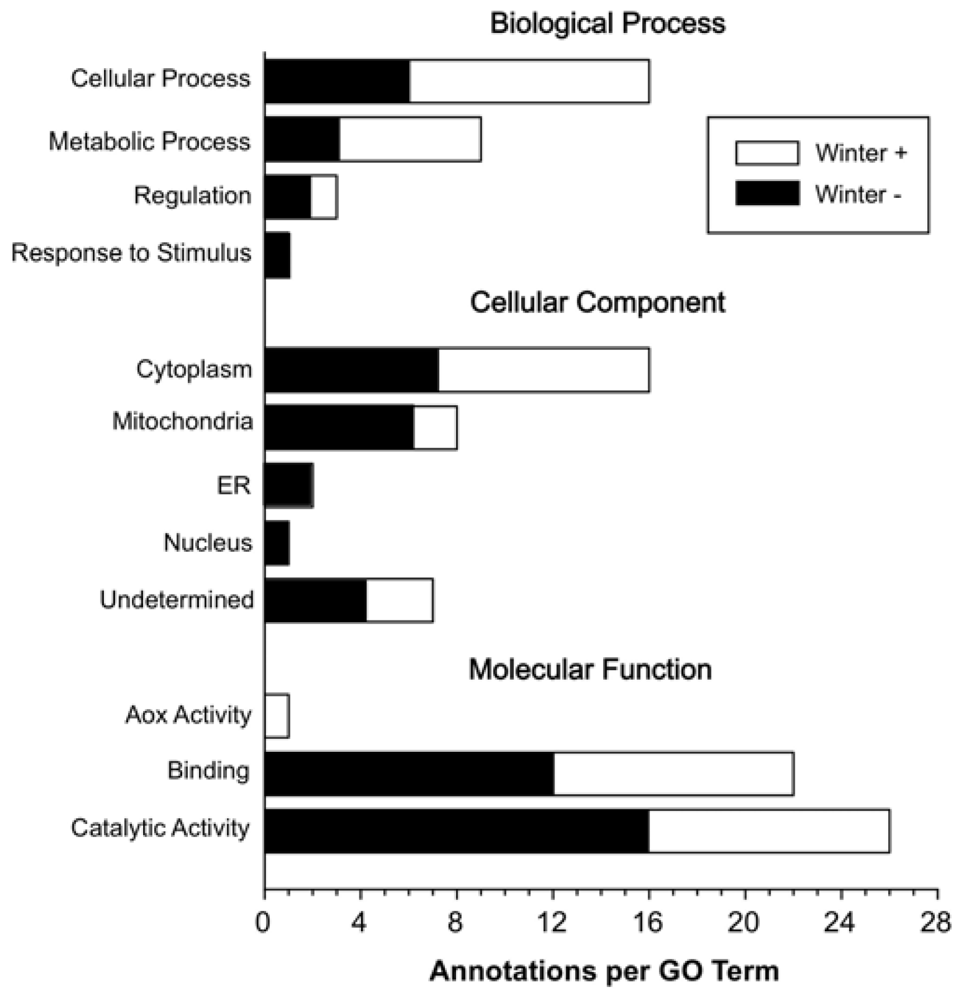
2. Results and Discussion
3. Experimental Section
Acknowledgments
References
- Costanzo, J.P.; Lee, R.E. Cryoprotection by urea in a terrestrially-hibernating frog. J. Exp. Biol 2005, 208, 4079–4089. [Google Scholar]
- Andrews, P.C. Personal communication, University of Michigan: Ann Arbor, MI, USA, 2010.
- Peregrín-Alvarez, J.M.; Sanford, C.; Parkinson, J. The conservation and evolutionary modularity of metabolism. Genome Biol 2009, 10, R63:1–R63:17. [Google Scholar]
- Bhatia, V.N.; Perlman, D.H.; Costello, C.E.; McComb, M.E. Software tool for researching annotations of proteins: Open-source protein annotation software with data visualization. Anal. Chem 2009, 81, 9819–9823. [Google Scholar]
- Storey, K.B.; Storey, J.M. Tribute to P. L. Lutz: Putting life on ‘pause’—molecular regulation of hypometabolism. J. Exp. Biol 2007, 210, 1700–1714. [Google Scholar]
- Wu, S.; DeCroos, A.; Storey, K.B. Cold acclimation-induced up-regulation of the ribosomal protein L7 gene in the freeze tolerant wood frog, Rana sylvatica. Gene 2008, 424, 48–55. [Google Scholar]
- Carrasco, M.A.; Tan, J.C.; Duman, J.G. A cross-species compendium of proteins/gene products related to cold stress identified by bioinformatic approaches. J. Insect Physiol 2011, 57, 1127–1135. [Google Scholar]
- Storey, K.B. Stress-Induced Gene Expression in Freeze Tolerant and Anoxia Tolerant Vertebrates. In Environmental Stress and Gene Regulation; Storey, K.B., Ed.; BIOS Scientific Publishers: Oxford, UK, 1999; pp. 1–23. [Google Scholar]
- Gülow, K.; Bienert, D.; Haas, I.G. BiP is feed-back regulated by control of protein translation efficiency. J. Cell Sci 2002, 115, 2443–2452. [Google Scholar]
- Chuang, M.H.; Wu, M.-S.; Lo, W.-L.; Lin, J.-T.; Wong, C.-H.; Chiou, S.-H. The antioxidant protein alkylhydroperoxide reductase of Helicobacter pylori switches from a peroxide reductase to a molecular chaperone function. Proc. Natl. Acad. Sci. USA 2006, 103, 2552–2557. [Google Scholar]
- Colell, A.; Green, D.R.; Ricci, J.-E. Novel roles for GAPDH in cell death and carcinogenesis. Cell Death Differ 2009, 16, 1573–1581. [Google Scholar] [Green Version]
- Pinder, A.W.; Storey, K.B.; Ultsch, G.R. Estivation and Hibernation. In Environmental Physiology of the Amphibians; Feder, M.E., Burggren, W.W., Eds.; The University of Chicago Press: Chicago, IL, USA, 1992; pp. 250–274. [Google Scholar]
- Costanzo, J.P.; et al. Miami University: Oxford, OH, USA, Unpublished work; 2011.
- Schiller, T.M.; Costanzo, J.P.; Lee, R.E. Urea production capacity in the wood frog (Rana sylvatica) varies with season and experimentally induced hyperuremia. J. Exp. Zool. A Ecol. Genet. Physiol 2008, 309, 484–493. [Google Scholar]
- Muir, T.J.; Costanzo, J.P.; Lee, R.E. Osmotic and metabolic responses to dehydration and urea-loading in a dormant, terrestrially-hibernating frog. J. Comp. Physiol. B 2007, 177, 917–926. [Google Scholar]
- Nagano, H.; Itoh, H.; Shukuya, R. Seasonal variation in hepatic phosphoenolpyruvate carboxykinase activity of bullfrog, Rana catesbeiana. Comp. Biochem. Physiol. B 1975, 51, 255–256. [Google Scholar]
- Storey, K.B. Glycolysis and the regulation of cryoprotectant synthesis in liver of the freeze tolerant wood frog. J. Comp. Physiol. B 1987, 157, 373–380. [Google Scholar]
- Pasanen, S.; Koskela, P. Seasonal and age variation in the metabolism of the common frog, Rana temporaria L. in Northern Finland. Comp. Biochem. Physiol. A 1974, 47, 635–654. [Google Scholar]
- Scapin, S.; Di Guiseppe, G. Seasonal variations of glycogen synthase and phosphorylase activities in the liver of the frog Rana esculenta. Comp. Biochem. Physiol. B 1994, 107, 189–195. [Google Scholar]
- Swanson, D.L.; Graves, B.M.; Koster, K.L. Freezing tolerance/intolerance and cryoprotectant synthesis in terrestrially overwintering anurans in the Great Plains, USA. J. Comp. Physiol. B 1996, 166, 110–119. [Google Scholar]
- Ackrell, B.A.C.; Kearney, E.B.; Mayr, M. Role of oxalacetate in the regulation of mammalian succinate dehydrogenase. J. Biol. Chem 1974, 249, 2021–2027. [Google Scholar]
- Muller, F.L.; Liu, Y.; Abdul-Ghani, M.A.; Lustgarten, M.S.; Bhattacharya, A.; Jang, Y.C.; van Remmen, H. High rates of superoxide production in skeletal-muscle mitochondria respiring on both complex I- and complex II-linked substrates. Biochem. J 2008, 409, 491–499. [Google Scholar]
- Joanisse, D.R.; Storey, K.B. Oxidative damage and antioxidants in Rana sylvatica, the freeze-tolerant wood frog. Am. J. Physiol 1996, 271, R545–R553. [Google Scholar]
- Ziekske, L.R. A perspective on the use of iTRAQ™ reagent technology for protein complex and profiling studies. J. Exp. Bot 2006, 57, 1501–1508. [Google Scholar]
- Yates, J.R.; Ruse, C.I.; Nakorchevsky, A. Proteomics by mass spectrometry: Approaches, advances, and applications. Annu. Rev. Biomed. Eng 2009, 11, 49–79. [Google Scholar]
- Shilov, I.V.; Seymour, S.L.; Patel, A.A.; Loboda, A.; Tang, W.H.; Keating, S.P.; Hunter, C.L.; Nuwaysir, L.M.; Schaeffer, D.A. The Paragon Algorithm, a next generation search engine that uses sequence temperature values and feature probabilities to identify peptides from tandem mass spectra. Mol. Cell. Proteomics 2007, 6, 1638–1655. [Google Scholar]
| GI | Protein | AR 1 | Species | Function |
|---|---|---|---|---|
| 129730 | protein disulfide isomerase precursor | 2.34 | Oryctolagus cuniculus | protein folding |
| 9755362 | acetaldehyde dehydrogenase | 2.28 | Mus musculus | alcohol metabolism |
| 85719973 | peptidylprolyl isomerase B | 2.22 | Ictalurus punctatus | protein folding |
| 57087309 | mRNA transport regulator 3 | 1.74 | Canis familiaris | protein synthesis |
| 47498070 | aspartate aminotransferase 1 | 1.68 | Xenopus tropicalis | amino acid metabolism |
| 146415164 | RNA recognition motif (hypothetical) | 1.65 | Pichia guilliermondii | protein synthesis |
| 5921957 | carbamoyl-phosphate synthase I | 1.53 | Rana catesbeiana | urea metabolism |
| 148226795 | binding immunoglobulin protein (BiP) | 1.51 | Xenopus laevis | protein folding/stress |
| 121594245 | protein DUF891 | 1.51 | Acidovorax sp. | unknown |
| 47210694 | glutamate dehydrogenase 1 | 1.49 | Tetraodon nigroviridis | nitrogen metabolism |
| 45383354 | histidine ammonia-lyase | 1.38 | Gallus gallus | amino acid metabolism |
| 124266729 | acetyl-CoA C-acetyltransferase 1 | 1.34 | Methylibium petroleiphilum | protein/lipid metabolism |
| 58332740 | aconitase 2 | 1.33 | Xenopus tropicalis | TCA cycle |
| 148230238 | homogentisate 1,2-dioxygenase | 1.32 | Xenopus laevis | amino acid metabolism |
| 126632707 | long-chain acyl-CoA dehydrogenase | 1.26 | Danio rerio | fatty acid metabolism |
| 50417404 | alanine-glyoxylate aminotransferase | 1.24 | Xenopus laevis | amino acid metabolism |
| 56377788 | elongation factor 1-α | 1.22 | Pelodiscus sinensis | protein synthesis |
| 89886140 | phosphoenolpyruvate carboxykinase 1 | 1.21 | Xenopus tropicalis | gluconeogenesis |
| GI | Protein | AR 1 | Species | Function |
|---|---|---|---|---|
| 147901600 | glycogen phosphorylase | 2.04 | Xenopus laevis | glycogen catabolism |
| 145545139 | protein kinase C | 1.72 | Paramecium tetraurelia | signal transduction |
| 148227386 | pyruvate carboxylase, gene 1 | 1.67 | Xenopus laevis | gluconeogenesis |
| 134254218 | pyruvate carboxylase, gene 2 | 1.51 | Xenopus tropicalis | gluconeogenesis |
| 118083730 | 1,4-α-glucan branching enzyme | 1.42 | Gallus gallus | glycogen synthesis |
| 63146078 | heat shock protein 70 (hsp70) | 1.34 | Oxyuranus scutellatus | stress response |
| 2196882 | heat shock cognate protein 70 (hsc70) | 1.32 | Pleurodeles waltl | protein folding |
| 148225037 | pyruvate kinase type M2 | 1.32 | Xenopus laevis | glycolysis |
| 147902026 | peroxiredoxin 6 | 1.30 | Xenopus laevis | antioxidation |
| 91084329 | protein phosphatase-1 α | 1.29 | Tribolium castaneum | glycogen metabolism |
| 148234835 | β-ureidopropionase | 1.27 | Xenopus laevis | amino acid metabolism |
| 54020777 | hydroxyacyl-CoA dehydrogenase | 1.24 | Xenopus tropicalis | fatty acid metabolism |
| 148224534 | Arp2/3, subunit 2 | 1.24 | Xenopus laevis | actin polymerization |
| 148223127 | glyceraldehyde-3-phosphate dehydrogenase | 1.21 | Xenopus laevis | glycolysis; non-metabolic processes |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Kiss, A.J.; Muir, T.J.; Lee, Jr, R.E.; Costanzo, J.P. Seasonal Variation in the Hepatoproteome of the Dehydration- and Freeze-Tolerant Wood Frog, Rana sylvatica. Int. J. Mol. Sci. 2011, 12, 8406-8414. https://doi.org/10.3390/ijms12128406
Kiss AJ, Muir TJ, Lee, Jr RE, Costanzo JP. Seasonal Variation in the Hepatoproteome of the Dehydration- and Freeze-Tolerant Wood Frog, Rana sylvatica. International Journal of Molecular Sciences. 2011; 12(12):8406-8414. https://doi.org/10.3390/ijms12128406
Chicago/Turabian StyleKiss, Andor J., Timothy J. Muir, Richard E. Lee, Jr, and Jon P. Costanzo. 2011. "Seasonal Variation in the Hepatoproteome of the Dehydration- and Freeze-Tolerant Wood Frog, Rana sylvatica" International Journal of Molecular Sciences 12, no. 12: 8406-8414. https://doi.org/10.3390/ijms12128406