1. Introduction
Rice is a staple cereal and widely consumed in the world. Recent attempts have been focused on the physiological functions of rice to prevent the life style-related diseases [
1–
4], in which the association of rice protein (RP) consumption with modulation of plasma cholesterol level has been demonstrated in some studies [
5–
9]. However, evidence as to how the hypocholesterolemic mechanism exerted by rice protein works is lacking.
As for isolation of rice protein, alkaline treatment and α-amylase degradation are two major industrial processing [
5,
10]. Alkaline treatment is a common process for rice protein extraction, by which the digestibility of rice protein is suggested to be effectively improved rather than by α-amylase degradation [
5,
11,
12]. Morita
et al. indicated that the true digestibility of rice protein extracted by alkaline treatment was higher than that treated by α-amylase, and showed that rice protein isolates produced by the two different methods (alkaline treatment and α-amylase degradation) could lower serum cholesterol concentration in Sprague-Dawley rats compared with casein [
5]. Taken together, these findings provided evidence that there may be a link of extraction method and protein digestibility which may be connected to the physiological function of rice protein.
Some studies have suggested that the biological utilization of a protein is primarily dependent on its digestibility by gastric, pancreatic and intestinal peptidases [
13–
15], further providing the insight that the digestibility may play a major role in the modulation of physiological function of dietary protein. In addition, the most frequently suggested mechanism responsible for the cholesterol-lowering effect of plant protein is the interference with enterohepatic circulation of cholesterol, leading to an inhibition of intestinal cholesterol absorption and an increase in fecal steroid excretion [
16]. Taking advantage of this view, some studies suggest that the low digestibility of dietary protein is a factor involved with the inhibition of intestinal cholesterol absorption and the disturbance of enterohepatic circulation to lead to the cholesterol-lowering action [
17,
18]. Thus, the digestibility of rice protein may be a major factor to influence cholesterol metabolism.
Therefore, in the present study, two extraction methods for preparation of rice protein, namely, an alkaline treatment and heat-stable α-amylase degradation, were developed to evaluate and compare the physiological function of rice protein. The key question addressed are: (1) whether the digestibility is responsible for the hypocholesterolemic action of rice protein and (2) how the digestibility of rice protein possesses a vital function in improving cholesterol metabolism in growing rats fed cholesterol-free diets. In addition, the present work also focused on the effect of rice protein on triglyceride metabolism.
2. Experimental Section
2.1. Protein Sources
Casein (CAS) (Gansu Hualing Industrial Group, Gansu, China) and rice proteins extracted from
Oryza sativa L. cv.
Longjing 26 (Rice Research Institute of Heilongjiang Academy of Agricultural Sciences, Jiamusi, China) were used as the dietary protein sources. Two methods were conducted for preparation of rice proteins, a classical extraction method with alkaline followed by precipitation with acidic solution (RP-A) [
5], and a method for rice protein isolation by starch degradation using a heat-stable α-amylase (Sigma, St. Louis, MO, USA) (RP-E) [
5].
The chemical composition of dietary proteins was analyzed, according to the previous study [
6]. Amino acid analysis of these proteins was performed using a Biochrom 30 amino acid analyzer (Biochrom, Holliston, MA, USA) according to Yang
et al. [
6].
2.2. In Vitro Digestion Study
The lipid fraction of protein samples (CAS, RP-A, RP-E) was removed by chloroform-methanol (2:1, v/v) extraction [
19], and the
in vitro digestion of CAS, RP-A and RP-E with pepsin and pancreatin was performed according to previous study [
17].
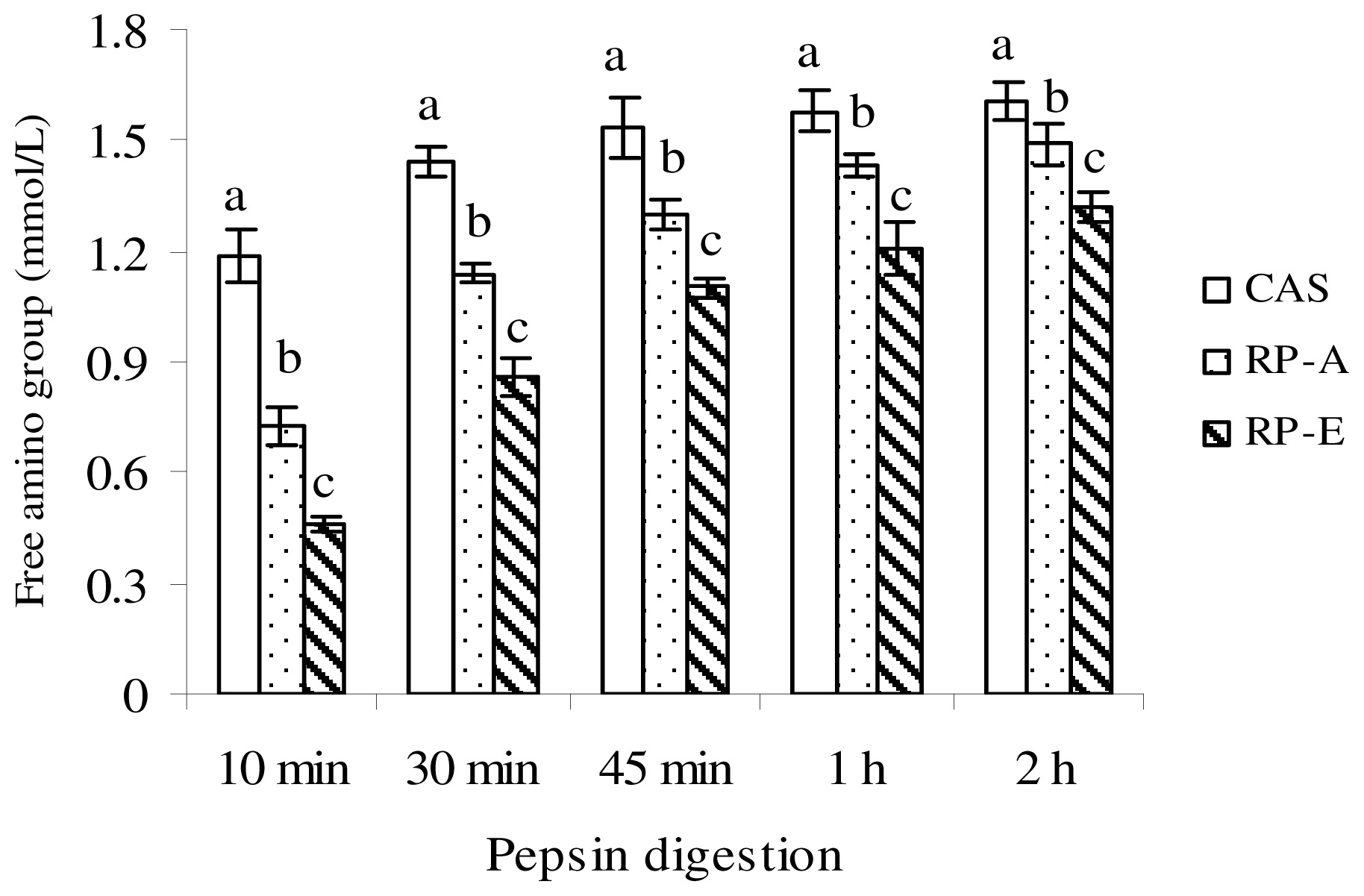
Of the defatted sample (CAS, RP-A, RP-E), protein solution (5% w/v, in distilled water) was adjusted to pH 2.0 with dilute HCl, and incubated at 37 °C with 5 mg of porcine pepsin (Sigma, St. Louis, MO, USA). During the pepsin digestion, the digest was sampled at intervals of 10 min, 30 min, 45 min, 1 h and 2 h. After deproteinization by 30% trichloroacetic acid, free amino group, which was reacted with 2,4,6-trinitrobenzenesulfonic acid at 37 °C for 2 h, was measured at 420 nm to evaluate the pepsin digestion.
The
in vitro protein digestibility was conducted according to the method of Mohamed
et al. and Ali
et al. [
13,
20], with slight modification. After 2 h of pepsin digestion, the peptic digest was adjusted to pH 8.5 with NaHCO
3, and then treated with 15 mg porcine pancreatin (Sigma, St. Louis, MO, USA). The mixture was incubated at 37 °C for 24 h. At appropriate intervals (4, 6, 8 and 24 h), samples were taken and treated with 30% trichloroacetic acid and centrifuged at 12,000×
g for 5 min at room temperature. After centrifugation, the acid-soluble fraction was estimated according to the method of Lowry
et al. [
21].
In vitro protein digestibility was calculated as:
In vitro protein digestibility (%) = Protein in the supernatant/Protein in the sample × 100.
2.3. Animals and Diets
The present experiments followed the same as the previous study [
6]. 7-week-old male Wistar rats were purchased from Animal Center of Harbin Medical University (SCXK20020002, Harbin, China) and individually housed in metabolic cages in a room maintained at 22 ± 2 °C under a 12 h light-dark cycle (07:00–19:00 for light). Rats were allowed free access to commercial pellets (Animal Center of Harbin Medical University, Harbin, China) for 3 days. After acclimatization, rats were randomly divided into three groups of similar body weight. Each group consisted of six rats.
All animals were fed
ad libitum with experimental diets according to the formula recommended by American Institute of Nutrition [
22]. For 3 weeks, growing rats were fed cholesterol-free diets with dietary protein level of 20% (as crude protein) of CAS, RP-A and RP-E, respectively. Diets were completed to 100% with starch. The composition of experimental diets is shown in
Table 1.
2.4. Samples Collection
During the feeding period, food consumption and body weight were recorded daily in the morning before replenishing the diet. Feces were collected for the final 3 days of the experimental period and dried to a constant weight and ground to a fine powder for fecal steroids determination according to Yang
et al. [
6].
At the end of the feeding period, the rats were deprived for 18 h and then sacrificed. Blood was withdrawn from abdominal vein into a heparinized syringe under anesthesia with sodium pentobarbital (50 mg/kg body weight), immediately cooled on ice and separated by centrifugation at 12,000× g for 5 min. The plasma obtained was frozen at −20 °C until analysis. After blood collection, the liver, perirenal fat and epididymal fat were excised immediately, rinsed in saline and weighed after blotted on a filter paper. The whole liver was cut into three portions and quickly freeze-clamped in liquid nitrogen and stored at −80 °C until analysis.
2.5 Plasma Lipid Analysis
Plasma concentrations of total cholesterol (TC), low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C) and triglyceride (TG) were measured colorimetrically with commercial kits (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Plasma very-low-density lipoprotein cholesterol (VLDL-C) was calculated as: VLDL-C = TC − HDL-C − LDL-C.
2.6. Liver Lipid Analysis
The lipids in the liver were extracted and purified according to the method of Folch
et al. [
19], and were analyzed as described by Yang
et al. [
6]. Samples of liver were extracted with chloroform/methanol (2:1, v/v). Total- and free-cholesterol and triglyceride were measured with a commercial kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Total lipids were determined gravimetrically. The concentration of esterified cholesterol was calculated as: esterified cholesterol = total cholesterol − free cholesterol. Cholesterol esterification ratio was described as: cholesterol esterification ratio (%) = (total cholesterol − free cholesterol) × 100/total cholesterol.
2.7. Determination of Fecal Excretion
Fecal bile acid and neutral sterol concentrations were measured as described in detail previously [
6]. Total bile acids were determined by 3α-hydroxysteroid dehydrogenase (Sigma, St. Louis, MO, USA) with sodium taurocholate as the standard. Cholesterol and coprostanol were identified by their retention time and mass-spectrum standard material by GCMS (GCMS QP5050A, Shimadzu, Kyoto, Japan), and the other sterols were identified by mass-spectrum and calculated by coprostanol’s standard curve. Apparent cholesterol absorption was calculated as: apparent cholesterol absorption = cholesterol intake − neutral sterol excretion. After lipid extraction by chloroform-methanol (2:1, v/v) [
19], fecal triglyceride in the extracted lipid was assayed with a commercial kit (Nanjing Jiancheng Bioengineering Institute, Nanjing, China). Fecal nitrogen content was determined by Kjeldahl method [
23]. Apparent digestibility of protein was calculated as: Apparent protein digestibility (%) = (Protein intake − Fecal protein) × 100/Protein intake.
2.8. Statistical Analysis
Data are presented as means ± SEM. Differences between groups were examined for statistical significance using the one-way analysis of variance (ANOVA), and then determined with the least significant difference test. The criterion for significance was P < 0.05.
4. Discussion
We examined the cholesterol-lowering potential of rice protein extracted by different method, suggesting that the hypocholesterolemic action of RP was associated with lower protein digestibility and higher fecal excretion of neutral sterols. Our findings clearly indicate that the digestibility of rice protein is a major factor to influence cholesterol metabolism through the inhibition of cholesterol absorption.
Conversion of cholesterol to bile acids is the principal regulated pathway whereby cholesterol is eliminated from the body, primarily via the enterohepatic circulation [
7,
8]. In the present study, our data suggested that the fecal excretion of bile acids can be affected by the digestibility of RP. Results indicated that RP-E significantly stimulated fecal excretion of bile acids (
P < 0.05), whereas fecal bile acids excretion of RP-A was comparable to CAS (
P > 0.05). These findings support the previous studies that alkaline treatment may improve the digestibility of rice protein [
11,
12], suggesting that RP-A appears to have less influence on binding bile acids in the intestinal tract due to its higher digestibility, in line with our previous findings [
6]. Nevertheless, of interest was the finding that RP-A caused the cholesterol-lowering effect in growing rats under the present experimental condition, despite the fecal excretion of bile acids was not significantly stimulated by RP-A feeding. The precise mechanism by which RP affects cholesterol metabolism is not fully established, but the higher extent to which RP-E and RP-A enhanced excretions of neutral sterols in fecal output should be taken into account. Clearly, the finding observed in this study suggests that hypocholesterolemic action induced by RP cannot be explained by the result of a simple diversion of bile acids toward fecal excretion, whereas the inhibition of intestinal cholesterol absorption, which was induced by the lower digestibility, might be one of possible mechanisms exerted by rice protein due to its lower digestibility.
In this study, we provided the
in vivo and
in vitro evidences for the lower digestibility of RP as compared with CAS. These results confirmed the finding that the digestibility of RP-E extracted by α-amylase was significantly lower than RP-A by alkaline treatment, further supporting the view that the digestibility of RP could be affected by the processing of extraction [
5,
11,
12]. More importantly, to support our hypothesis that lower digestibility of RP might produce the cholesterol-lowering action, we found a significant positive correlation between the protein digestibility and the concentration of plasma cholesterol (
r = 0.7357,
P < 0.01), as well as liver cholesterol accumulation (
r = 0.9025,
P < 0.01) (
Table 7). These data clearly indicated that there was a close link of lower protein digestibility and cholesterol-lowering action exerted by RP.
The most frequently suggested mechanism responsible for the cholesterol-lowering effect of dietary protein is to inhibit the intestinal cholesterol absorption and an increase in fecal steroid excretion [
5,
16–
18,
24]. In the present study, our results confirm and expand this view, indicating that higher fecal excretions of neutral sterols and nitrogen were closely associated with lower cholesterol concentration in plasma and liver (
Table 7). Results showed a significant negative correlation between fecal neutral sterols and plasma cholesterol concentration (
r = −0.7451,
P < 0.01), as well as liver cholesterol concentration (
r = −0.8629,
P < 0.01). Furthermore, with the increasing proportion of nitrogen in feces, the absorption of cholesterol was more efficaciously depressed in rats, indicating a significant positive correlation between the protein digestibility and the apparent cholesterol absorption (
r = 0.8662,
P < 0.01). Thus, with the lower digestibility, RP posses a cholesterol-lowering action through enhancing fecal excretion of neutral sterols, further suggesting the digestibility of rice protein is a major factor to influence cholesterol metabolism through the inhibition of cholesterol absorption.
Here, it must be noted that the hypocholesterolemic effect of RP-E was not more effective than that of RP-A. Also, we did not observe a difference of liver cholesterol-lowering effect between RP-E and RP-A. Thus, the question arises why the stronger inhibition of cholesterol absorption induced by RP-E with lower digestibility did not lead to lower cholesterol level in rats fed RP-E than those fed RP-A. The precise mechanism by which RP affects cholesterol metabolism is not fully understood, but the mechanism responsible for the effect on the removal of non-HDL-C from circulation by hepatic uptake should be taken into account. Our data obtained from plasma HDL-C and non-HDL-C indicated that RP-E and RP-A produced the similar ratio of non-HDL-C to HDL-C in the present study (RP-E, 0.42; RP-A, 0.45), which may result in the similar transfer of peripheral free cholesterol to the liver by a mechanism known as “reverse cholesterol transfer” promoted by HDL-C. Thus, the findings observed in this study suggest that, except for the inhibition of cholesterol absorption induced by lower digestibility of RP, other mechanisms, such as the stimulation of the uptake of lipoprotein cholesterol by the liver may also come into play to fully explain the hypocholesterolemic response to RP. Clearly, the observed effect of RP on cholesterol metabolism remains to be clarified in further studies.
Cholesterol absorption has been also suggested to be affected by amino acid composition. Vahouny
et al. reported that the cholesterol-lowering effect of soy protein was due mainly to the ratio of arginine (Arg) to lysine (Lys), which was involved with the inhibition of intestinal lipid absorption [
25]. Results shown by Vahouny
et al. indicated that the addition of Arg to a CAS diet to increase the Arg/Lys ratio resulted in a slower rate of lipid absorption, and the addition of Lys to the soy protein diet to decrease the Arg/Lys ratio caused a faster rate of lipid absorption. In addition, some studies have led to better understanding of the higher ratio of Arg/Lys can result in an elevation in hepatic 7α-hydroxylase activity, which is a rate-limiting enzyme for conversion of cholesterol to bile acids [
26]. Thus, whatever the mechanism of action, the effect of Arg/Lys ratio on cholesterol metabolism is important. Considering this view, the highest Arg/Lys ratio in RP-E (3.15), the higher in RP-A (2.56) and the lowest in CAS (0.44) might explain the different levels in plasma cholesterol and fecal output of sterols in the present study. In light of these findings, the higher ratio of Arg/Lys in RP, which may depress the cholesterol absorption to result in hypocholesterolemia, should be also emphasized in the present study. Thus, in respect to the influence on digestibility, the difference of amino acid composition induced by the different extraction method may represent a principal mechanism for inhibition of cholesterol absorption to induce the cholesterol-lowering action of RP.
Additionally, some studies have suggested that other non-protein contents, e.g., carbohydrate, dietary fiber, unsaturated fatty acids, also could induce the cholesterol-lowering action [
27]. However, it does not seem very likely that the other remaining non-protein components, e.g., dietary fiber, carbohydrate, in RP may contribute to the cholesterol-lowering effects in this study because their concentrations are extremely low compared to the concentrations normally used for the induction of hypolipidemia. This may explain the phenomenon that RP-E containing more dietary fiber could not produce more effective cholesterol-lowering effect than RP-A. Clearly, additional studies are required to confirm this view.