1. Introduction
Lipases, which have large biocatalytic potential in both aqueous and non-aqueous media, find their widespread applications in biomedical sciences and chemical synthesis. It has been proved that most of the lipases from microorganisms have multi-faceted properties and play an important role in ever-growing modern biotechnology [
1,
2]. Lipases have a special catalytic mechanism which is so-called interfacial activation. Lipases may exist in two different conformations, an open and active conformation and a closed and inactive one. In an aqueous homogeneous media, these forms are in equilibrium shifted towards the closed form. In the presence of hydrophobic surfaces (e.g., a drop of oil), the lipase is adsorbed, shifting the equilibrium towards the open conformation one [
3]. However, the two open lipase molecules have a trend to give biomolecular aggregated by interacting with each other via the large hydrophobic surface surrounding the active center of lipases [
4,
5], which may alter the properties of the lipases and reduce their activity. In contrast, the mechanism “interfacial activation” of lipases could be a new tool for purification, immobilization and modulation of lipases properties [
6,
7].
Lipases from Candida species such as
Candida rugosa,
Candida Antarctica have been intensively studied and widely used in many applications for producing valuable esters [
8,
9]. Although ten lipases (Lip1-Lip10) have been found in yeast
Candida albicans (
C. albicans) [
10], the characterization and potential application of most of the lipases has not been reported yet. Until now, only lipase 4 and lipase 5 from
C. albicans have been biochemically characterized [
11,
12]. Lipase 4 has been produced in
Saccharomyces cerevisiae and revealed a very efficient acyltransfer activity [
11]. And lipase 5 was active at low temperature and behaved as a cold-active lipase [
12].
C. albicans obeys a special protein translation system which is different from that in the universal host.
C. albicans encodes a unique seryl-tRNA (CAG) that should decode the leucine codon CUG as serine [
13]. Besides
C. albicans, several species of yeast in the
Candida genus, such as
parapsilosus,
zeylanoides,
ragosa, and
melibiosica, also use CUG as a codon for serine instead of leucine [
14]. The methylotrophic yeast
P. pastoris is considered as a valuable host for the production of various proteins of interest, such as enzyme and vaccine [
15]. However, functional productions of lipase from
Candida genus in
P. pastoris are unfeasible due to the non-universal codon CUG [
12,
16]. To overcome this problem, mutagenesis of the unusual CUG serine codon into a universal one by site-directed mutagenesis needs to be conducted before expression in the universal host.
One of the most important applications of lipase is to produce fatty acids or special glycerides from oils [
17,
18]. For example, full hydrolysis of fish oils to produce poly-unsaturated fatty acids has attracted much attention recently [
19]. However, partial hydrolysis of triglycerides also leads to production of valuable glycerides [
20]. Another application of lipase is used as an additive in detergents, but the activity of lipase might be inhibited by other components in detergents such as proteases and surfactants. Immobilization of lipase on a suitable support provides its reuse and protects it from protease attack and surfactants inhibition. Immobilization can help to enable the application of enzymes in different solvents, at extremes of pH and temperature [
21]. The substrate-specificity, enantioselectivity and reactivity of lipase can also be modified [
21].
An effective production of lipases is an essential step in determining their biochemical properties, catalytic behavior and sequence-structure-function relationships. However, it is difficult to isolate the individual isoenzymes from
C. albicans directly because of their high similarities in physical properties. A feasible approach is to produce a lipase by the recombinant DNA technology combined with manipulating culture conditions in a heterologous expression system. A number of statistical experimental designs, such as central composite design (CCD), with response surface methodology (RSM) have been employed for optimizing enzyme production from microorganisms [
22–
26]. In contrast to “one-at-a-time” technique, the response surface methodology (RSM), which includes factorial design and regression analysis, helps in evaluating the effective factors and building models to study interaction and select optimum conditions of variables for a desirable response [
27].
In the present work, our attempt was to produce lipase 10 from C. albicans (CaLIP10) and determine its biochemical properties. The two non-universal serine codons (CTG) in the gene were mutated into universal TCT serine codons by overlap extension PCR. The screened CaLIP10 with high productivity was constitutively expressed in Pichia pastoris (P. pastoris), and its culture process was optimized by response surface methodology. The recombinant CaLIP10 was purified and characterized by monitoring the effect of pH, temperature, ions, detergents, water-miscible solvents on lipase activity and studying the hydrolysis of coconut oil.
3. Experimental Section
3.1. Materials
All chemicals used in the study were analytical pure grade. The substrates p-nitrophenol caprylate (p-NPc) were purchased from Sigma-Aldrich (Germany). Other reagents were purchased from Takara (Japan) or Omega (USA).
3.3. Cloning and Mutagenesis of CalIP10
C. albicans genomic DNA was prepared using a fungal genome extraction kit (Omega) according to the manufacturer’s instruction. The mature protein-coding sequence without the signal peptide of
C. albicans lipase 10 (namely
lip10) was amplified by polymerase chain reaction (PCR) from genomic DNA. Specific primers 10F/10R (
Table 1) were used. The PCR product was first purified and digested by restriction enzymes, then ligated with predigested pBluescript SK vector, and was termed pBluescript-
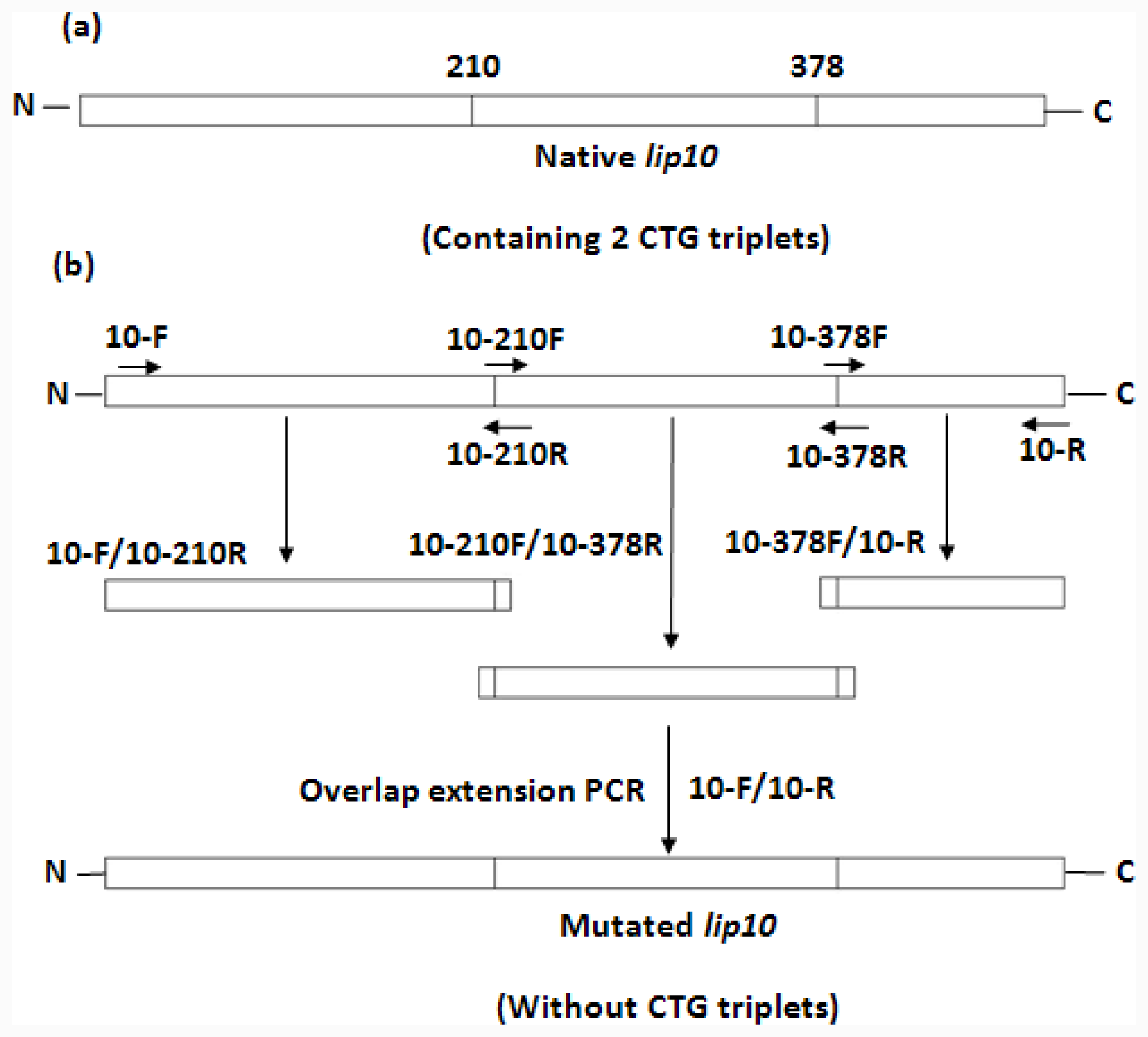
lip10. The plasmid construct was used as the template for the mutagenesis to convert 2 non-universal CTG-serine codons (residue number 210 and 378) in CaLIP10 into universal TCT-serine codons. The simultaneous multiple mutagenesis was carried out by the overlap extension PCR [
37]. The product of the mutagenesis was ligated into the pBluescript SK vector, and was termed pBluescript-
lip10-
dm. The sequence of mutated
lip10 containing the mentioned replacements was confirmed by the full-length DNA sequencing. The scheme for mutation is shown in
Figure 1 and the mutagenic primers are shown in
Table 1. Subcloning of
lip10 and
lip10-dm into the expression vector
pGAPZαA were performed to generate pGAPZαA-
lip10 and pGAPZαA-
lip10-
dm.
3.4. Screening Recombinant P. pastoris with High Yield of CaLIP10
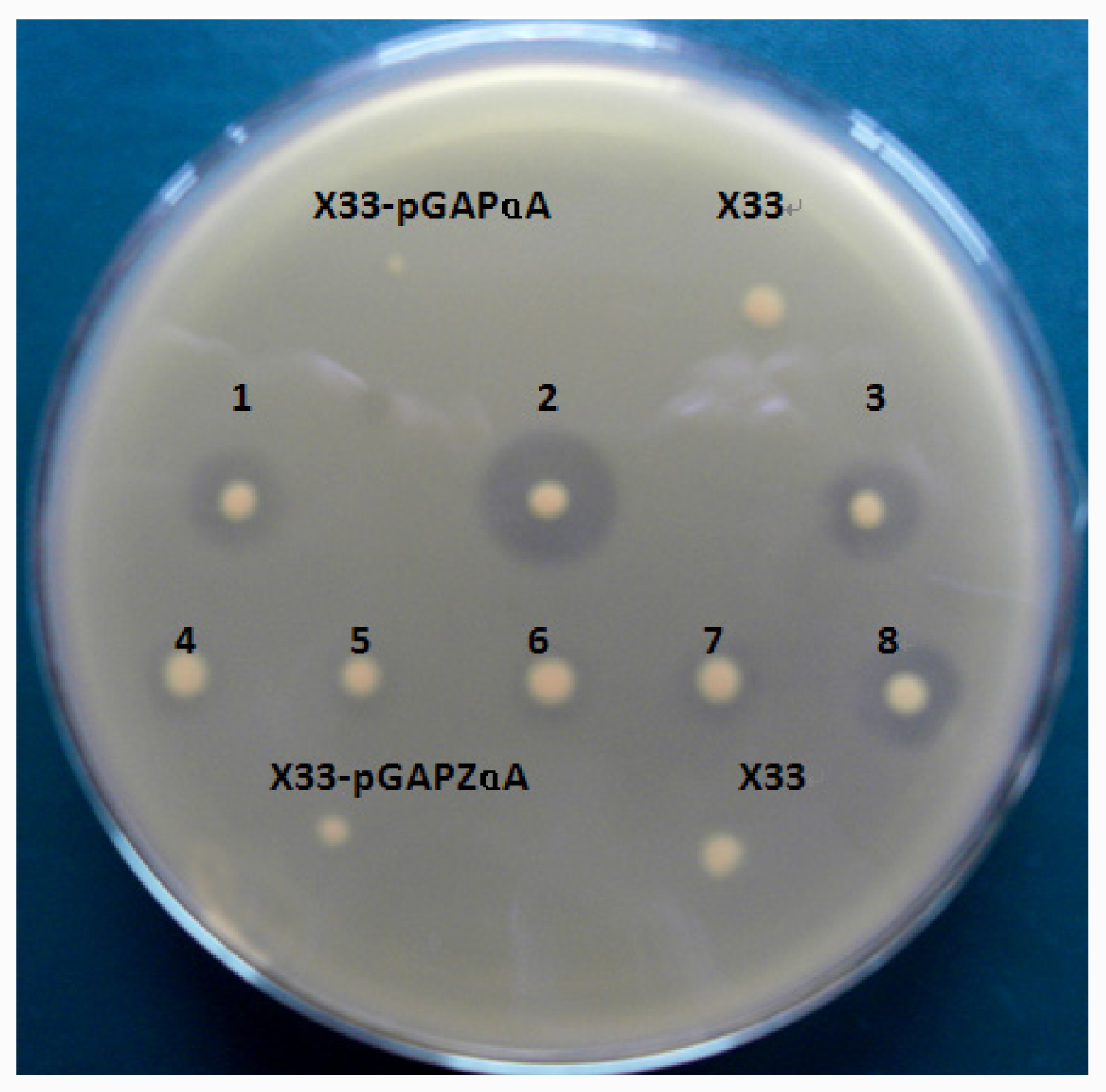
The plasmid pGAPZαA-lip10 and pGAPZαA-lip10-dm were linearized with AvrII (NEB, USA) and transformed into P. pastoris X33 by electroporation using Gene Pulser (Bio-Rad) apparatus. Transformants were screened on YPD plates (containing 2% agar and 100 μg/mL Zeocin) to isolate Zeocin-resistant clones. Individual colonies were pitched and patched on 1% tributyrin-emulsion YPD plates to identify lipase-secreting transformants which would form a clear zone on the opaque tributyrin emulsion. P. pastoris transformed with pGAPZαA was used as a negative control. Strains with high yield of CaLIP10 were maintained at −80 °C in 50% glycerol (v/v).
3.5. Optimizing the Production of CaLIP10 by Central Composite Design
3.5.1. Culture of the Recombinant P. pastoris
Seed medium consisting of YPD was inoculated and incubated in a rotary shaker (30 °C, 250 rpm, 24 h) and then 5 mL was used to inoculate 50 mL of production medium (YPD) in a 300 mL glass flask. This culture was grown under the different conditions to optimize the culture process.
3.5.2. Experimental Design
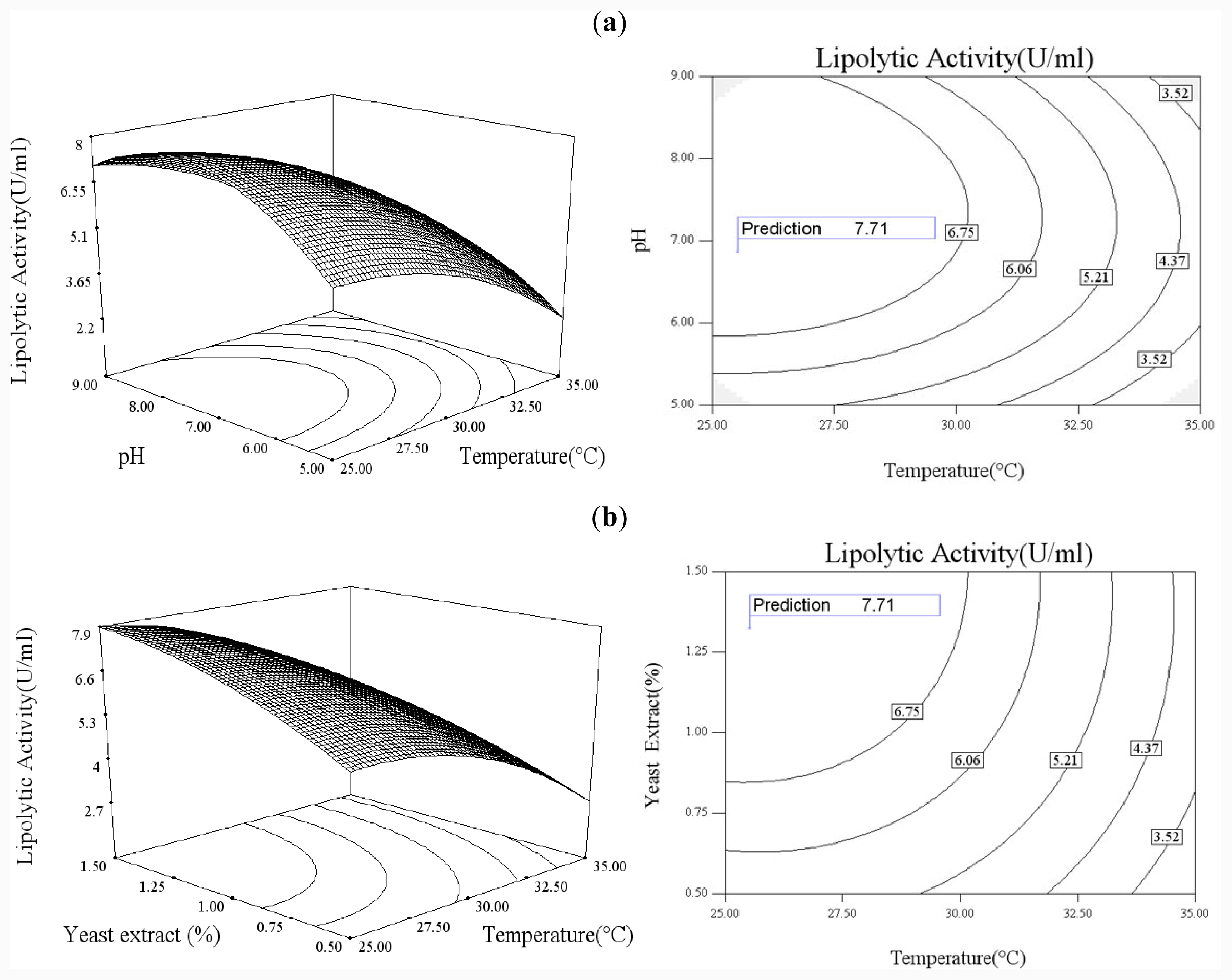
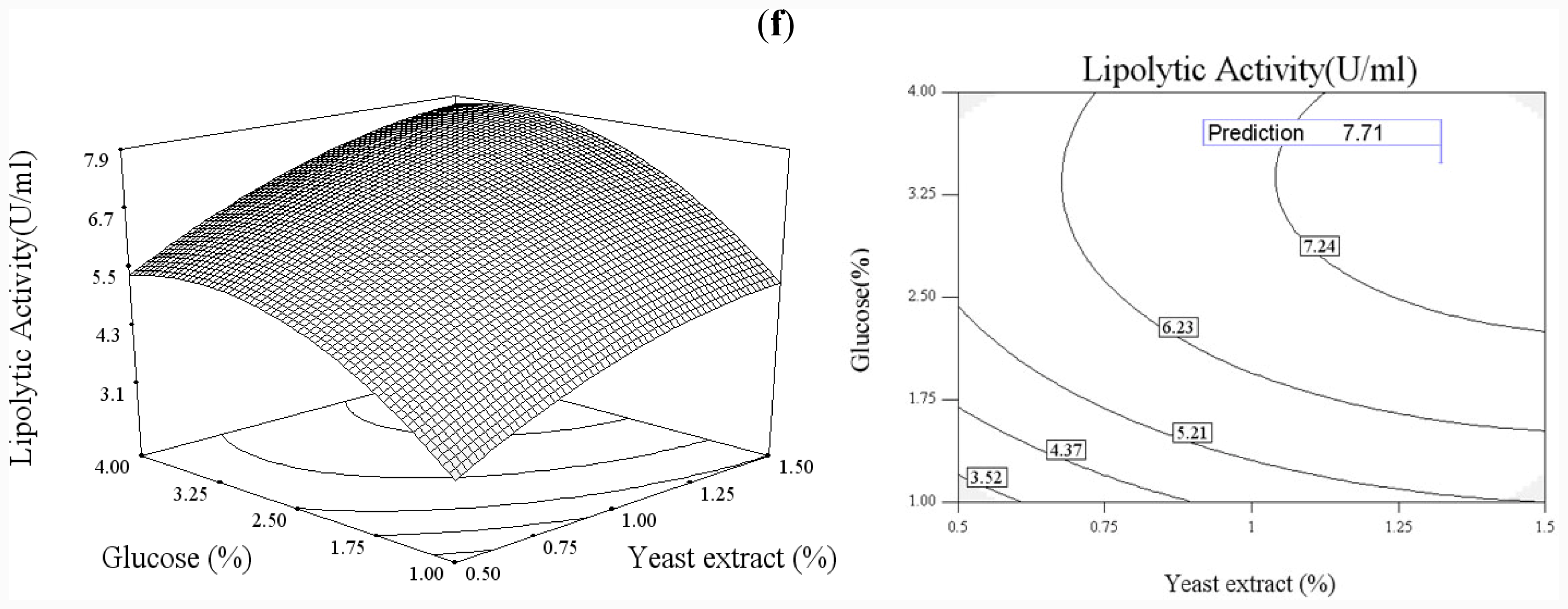
In order to explore the effect of culture medium (glucose, yeast extract) and growth condition (pH and temperature), a statistical approach using the Box–Wilson central composite design (CCD) was conducted [
38]. In this study, the four independence variables were studied at five levels (−2, −1, 0, 1, 2) and a 4-factor-5-level CCD that consisted of 30-run experiments (
Table 2) generated by Design Expert 7.0 (Stat-Ease Inc., USA) was used. The lipolytic activity of lipase was taken as responses.
The CCD experimental results were fitted with a second-order polynomial equation of
Equation 2 [
39]:
where Y is response (lipolytic activity); βk0, βi, βkii, and βkij are constant coefficients and xi the uncoded independent variable. The analysis of variance (ANOVA) was performed to evaluate significance of the model and coefficients.
3.6. Purification of Recombinant CaLIP10
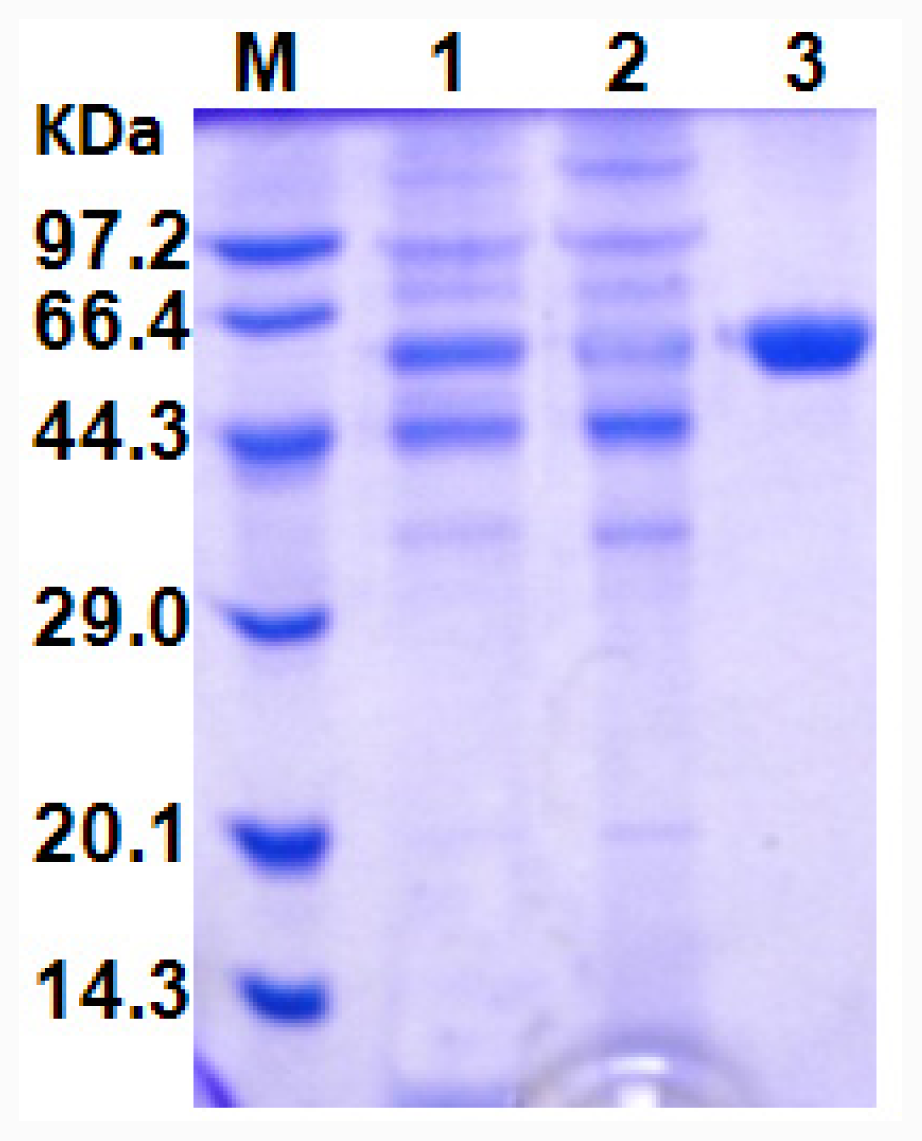
All procedures were performed at 4 °C. The culture broth was centrifuged (12,000 × g, 4 min) to remove the cells. The crude enzyme solution was adjusted to pH 5.5 using HCl, and applied onto a CM SepharoseTM Fast Flow column (CM FF, 1.6 cm × 28 cm, GE Healthcare) equilibrated with 20 mM sodium acetate buffer (pH 5.5). The column was washed with 20 mM sodium acetate buffer (pH 5.5) and then bound protein was eluted with a gradient of the same buffer containing 0–1.0 M NaCl at a flow rate of 3 mL/min. The active fractions (50 mL) were pooled, concentrated and 10 mL loaded onto the Sephadex G25 column (2.5 cm × 20 cm) equilibrated with 100 mM sodium phosphate buffer (pH 7.0). The column was then washed with the same buffer and the lipase-active fractions pooled and stored at 4 °C.
The molecular mass of the purified recombinant lipase was determined in denaturing conditions by SDS-PAGE with 5% polyacrylamide-stacking gel and a 12% polyacrylamide-resolving gel. The total protein in the samples was quantified with a BCA Protein Assay Kit (Sangon, Shanghai, China) using bovine serum albumin as standard according to the manufacturer’s instruction.
3.7. Lipase Activity Assay
The lipase activity was determined spectrophotometrically using p-nitrophenol caprylate as substrate. The reaction mixture consisted of 50 mM Tris-HCl buffer (pH 8.0), 2 mM p-NPc and an appropriate amount of the lipase and the amount of released p-nitrophenol quantified after 5 min reaction time by its absorbance at 405 nm. One unit of activity was defined as the amount of enzyme needed to release 1 μmol of p-nitrophenol per minute under the assay conditions.
3.8. Characterization of CaLIP10
3.8.1. Effect of Temperature on CaLIP10 Activity and Stability
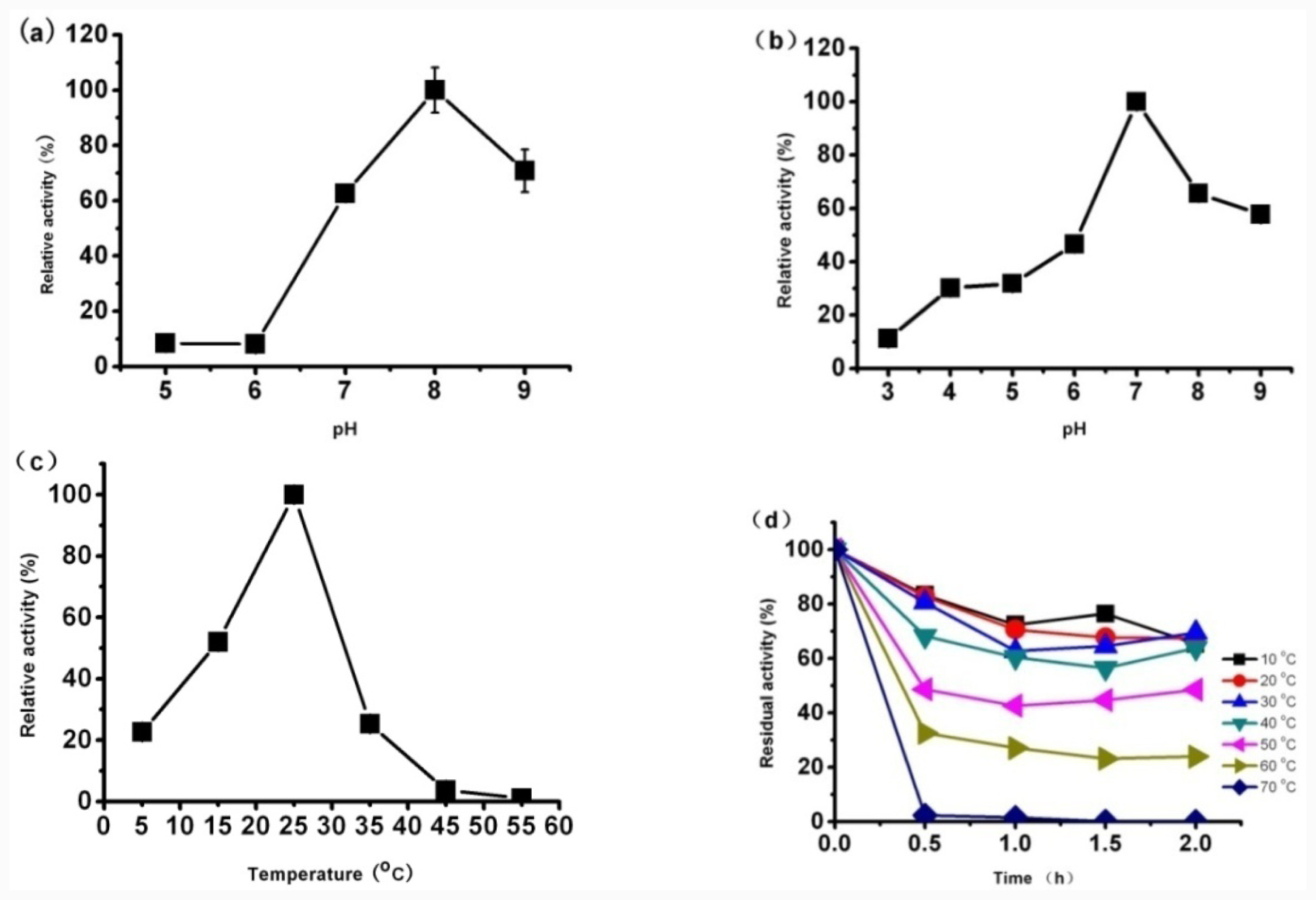
Effect of temperature on the lipase activity was determined by measuring the hydrolytic activity at different temperatures in 50 mM Tris-HCl buffer (pH 8.0). The temperature was set as 5 °C, 15 °C, 25 °C, 35 °C, 45 °C and 55 °C. The activity was expressed as percent relative activity in relation to the temperature optimum, which was considered as 100%.
The thermostability was investigated by incubating the lipase at the different temperatures for 2 h and measuring its activity every 30 min. The activity was expressed as percent relative activity with respect to maximum activity, which was considered as 100%.
3.8.2. Effect of pH on the CaLIP10 Activity and Stability
The optimum pH was investigated by measuring the hydrolytic activity of lipase at 25 °C at different pH between 3.0 and 9.0 by using the following buffers: 100 mM sodium acetate buffer (pH 3.0–5.0), 100 mM sodium phosphate buffer (pH 6.0, pH 7.0), 50 mM Tris-HCl buffer (pH 8.0) and 50 mM Gly-NaOH buffer (pH 9.0). The activity was expressed as percent relative activity with respect to maximum activity, which was considered as 100%.
To analyze pH stability of the lipase, the purified CaLIP10 was pre-incubated (4 °C, 20 h) in buffers with each of the different pH value and the residual activity was measured The activity was expressed as percent relative activity with respect to maximum activity, which was considered as 100%.
3.8.4. Hydrolysis of Coconut Oil
5 g of coconut oil and 0.5 g of distilled water were added into a 50 mL Florence flask, and the reaction was preceded under magnetic stirring (200 rpm) at 25 °C with an addition of 100 U of CalIP10 per gram of coconut oil. About 0.1 g of the reaction mixture was withdrawn periodically and centrifuged at 10,000 rpm for 5 min. The upper layer was transferred into another tube, and anhydrous sodium sulfate was added to remove the residual water. And then 1 mL of n-hexane and 2-propanol (15:1 v/v) was added to the mixture and swirled thoroughly. The supernatant was collected for HPLC analysis by centrifugation at 10,000 rpm for 1 min.
3.8.5. Analysis of Hydrolytic Products by HPLC and GC
After 48 h of hydrolysis of coconut oil, 100 mg of reaction mixture were withdrawn into a separatory funnel, and then hexane (10 mL) and phenolphthalein solution was added. The reaction mixture was neutralized with KOH 0.5 N hydroehanolic solution (30% ethanol). The lower phase was acidified (pH 1.0) with 6 N HCl. The liberated free fatty acids were then extracted with 50 mL of hexane. The hexane layer was dried over anhydrous sodium sulfate, and the solvent was removed with a rotary evaporator at 40 °C. Free fatty acids were methylated and analyzed by GC [
40]. The dacylglycerols in the reaction mixture were analyzed by HPLC [
41].
4. Conclusions
Functional expression of CaLIP10 in P. pastoris and indicated biochemical properties of CaLIP10 were performed. 210 and 378-CUG-Ser codons play an impact role in the lipase activity, and about 2-fold activity increased when they were mutated into universal TCT serine codons. The lipolytic activity of CaLIP10 was further increased by 64% at optimal conditions under initial pH of culture at 6.86, temperature at 25.53 °C, 3.48% of glucose and 1.32% of yeast extract.
The optimum conditions for lipolytic activity were 25 °C and pH 8. Ions, such as Fe3+ at a high concentration (5 mM) reduced lipolytic activity of CaLIP10 dramatically. The hydrophilicity of non-ionic surfactants at low concentration (1%) might help to increase the stability and activity of CaLIP10 to some degree. CaLIP10 exhibited a good stability in non-ionic surfactants and retained more than 73% of maximal activity after being treated with ethanol. CaLIP10 could effectively hydrolyze coconut oil, but exhibited no obvious preference to the fatty acids with different carbon lengths, and diacylglycerol was accumulated in the reaction products, suggesting that CaLIP10 is a potential lipase for application in the oil industry.