N-Acetylcysteine Reduces Markers of Differentiation in 3T3-L1 Adipocytes
Abstract
:1. Introduction
2. Results and Discussion
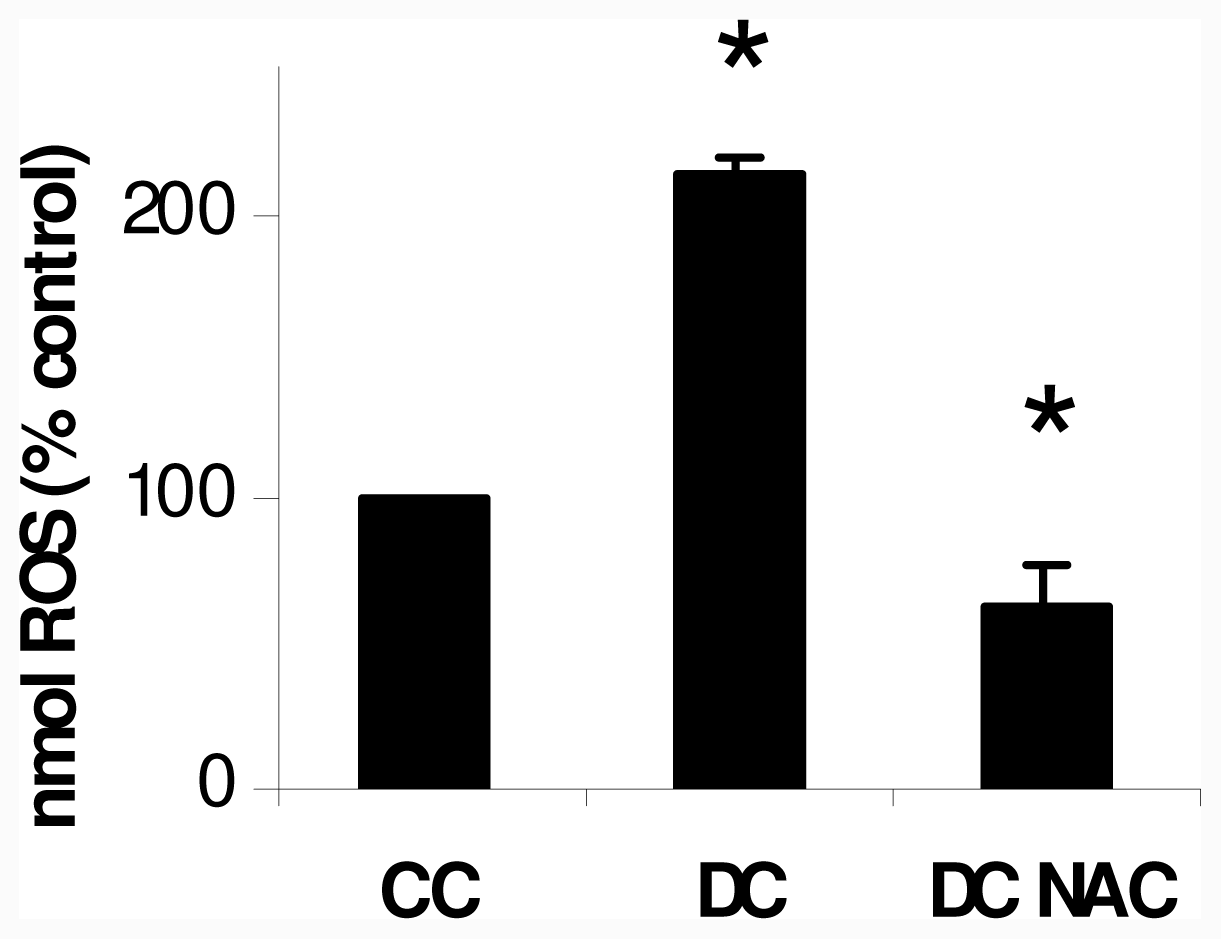
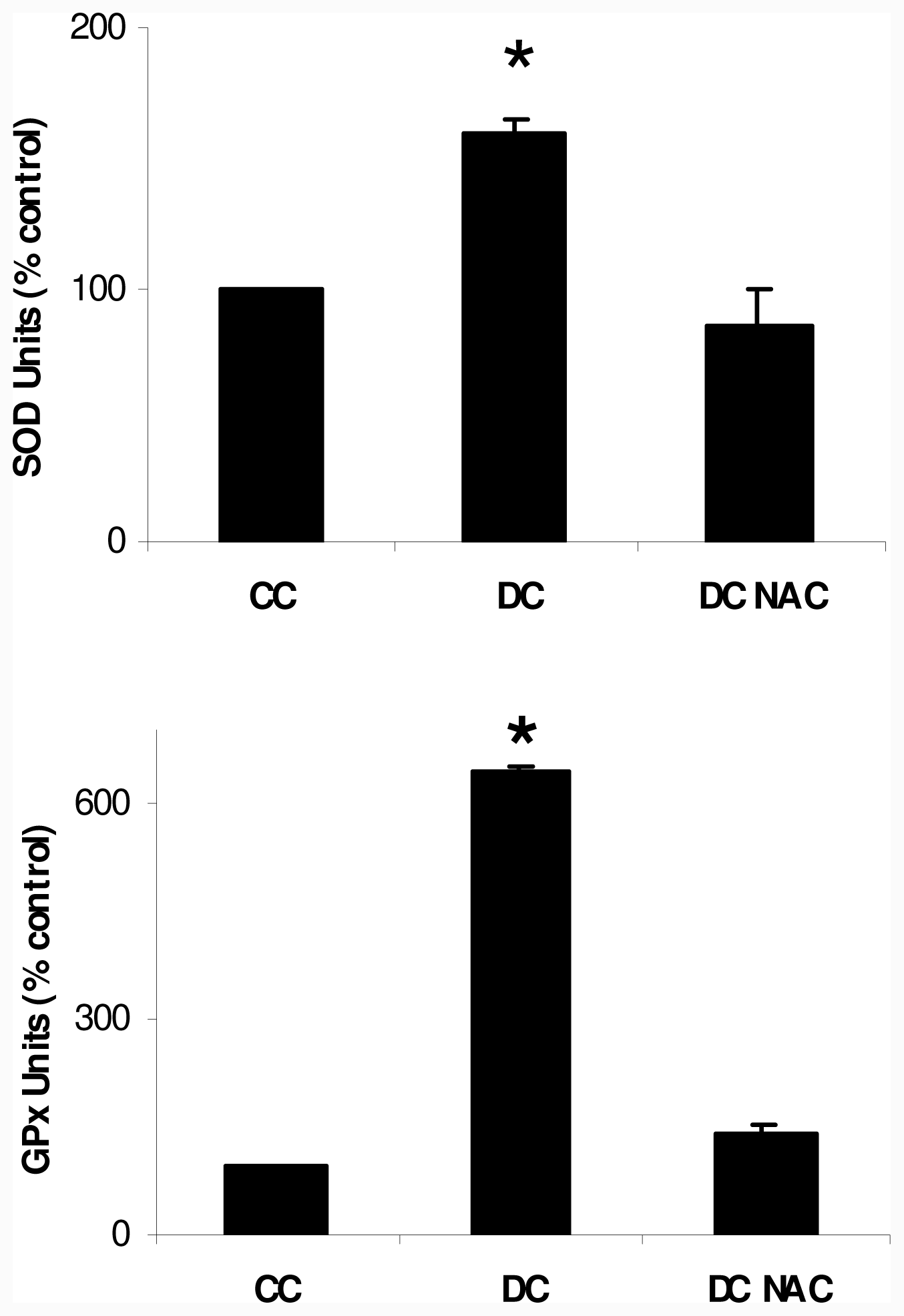
2.1. NAC Effect on Oxidative Markers
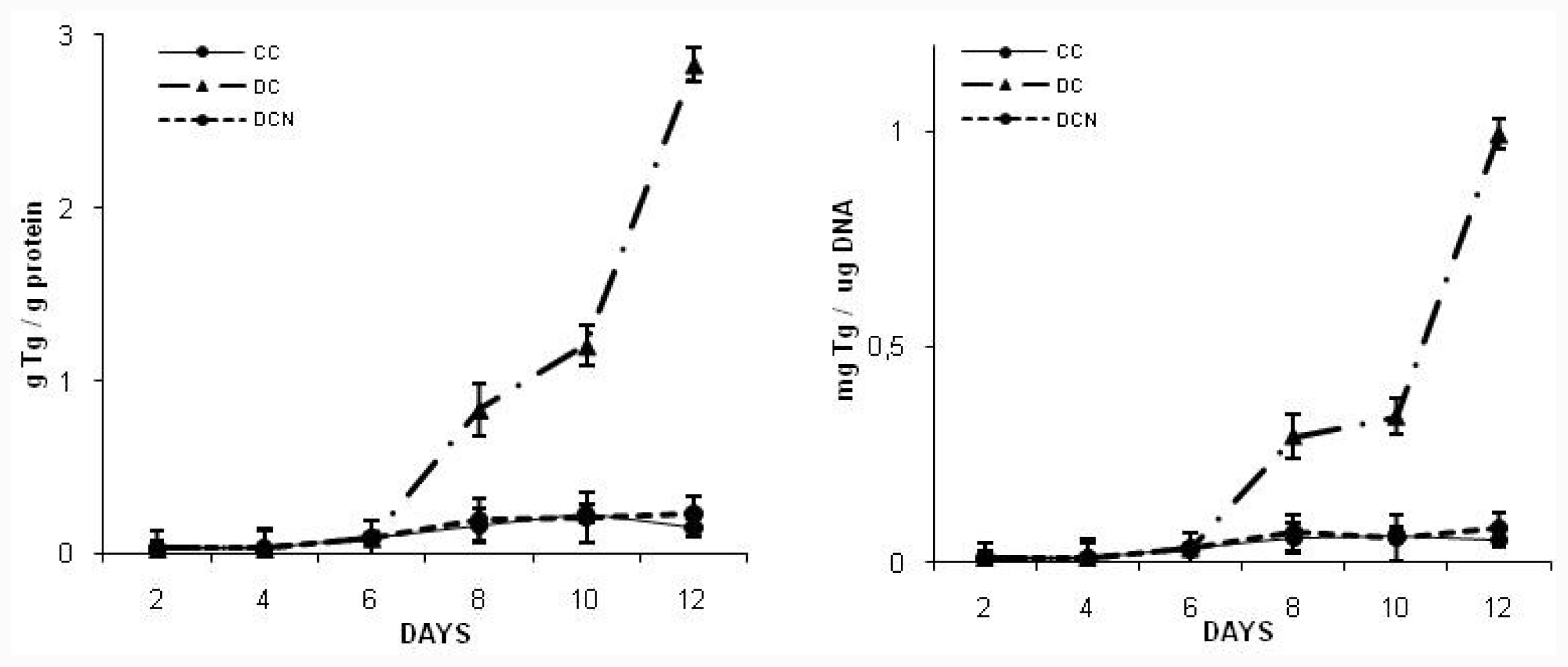
2.2. NAC Effect on Triglyceride and GSH Content
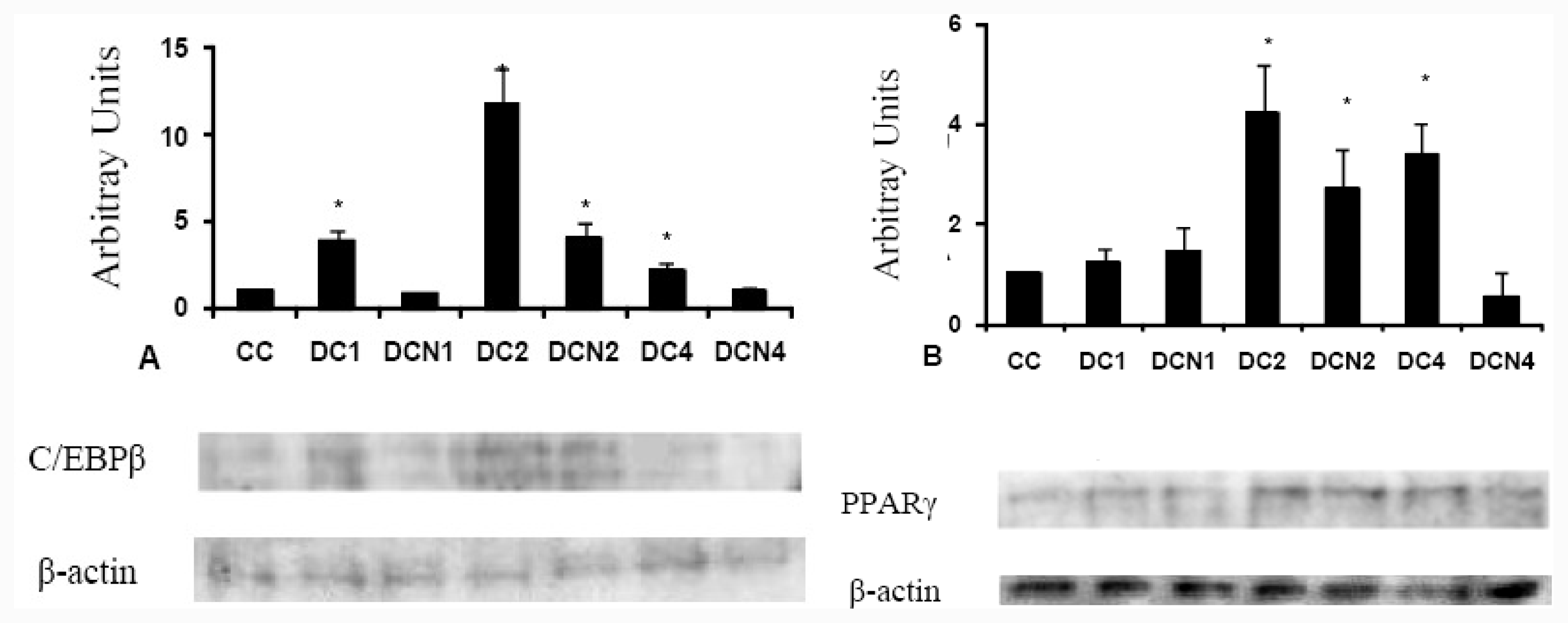
2.3. NAC Effect on Adipocyte Differentiation Markers
3. Materials and Methods
3.1. Cell Cultures and Drugs
3.2. Differentiation of 3T3-L1 Preadipocytes into Adipocytes
3.3. Determination of Triglyceride
3.4. Glutathione Determination
3.5. Determination of Protein and DNA
3.6. Determination of Superoxide Dismutase
3.7. Determination of Glutathione Peroxidase
3.8. Measurement of Intracellular ROS Generation
3.9. Western Blot Analysis
3.10. Akt Assays
3.11. Statistics
4. Conclusions
Acknowledgments
Abbreviations
| GSH | Glutathione |
| NAC | N-acetylcysteine |
| C/EBP β | CCAAT/enhancer binding protein beta |
| PPAR γ | peroxisomal proliferator activated receptor gamma |
References
- Frost, SC; Lane, MD. Evidence for the involvement of vicinal sulfhydryl groups in insulin-activated hexose transport by 3T3-L1 adipocytes. J. Biol. Chem 1985, 260, 2646–2652. [Google Scholar]
- Mac Dougald, O; Lane, MD. Transcriptional regulation of gene expression during adipocyte differentiation. Ann. Rev. Biochem 1995, 64, 345–373. [Google Scholar]
- Furukawa, S; Fujita, T; Shimabukuro, M; Iwaki, M; Yamada, Y; Nakajima, Y; Nakayama, O; Makishima, M; Matsuda, M; Shimomura, I. Increased oxidative stress in obesity and its impact on metabolic syndrome. J. Clin. Invest 2004, 114, 1752–1761. [Google Scholar]
- Bustamante, J; Guerra, LN; Bredeston, J; Mordoh, J; Boveris, A. Melanin content and hydroperoxide metabolism in human melanoma cells. Exp. Cell. Res 1991, 196, 172–176. [Google Scholar]
- Guerra, LN; Moiguer, S; Karner, M; de Molina, MC; Sreider, CM; Burdman, JA. Antioxidants in the treatment of Graves disease. IUBMB Life 2001, 51, 105–109. [Google Scholar]
- Guerra, LN; Rios de Molina, MC; Miler, EA; Moiguer, S; Karner, M; Burdman, JA. Antioxidants and methimazole in the treatments of Graves’ disease: Effect on urinary malondialdehyde levels. Clin. Chim. Acta 2005, 352, 115–120. [Google Scholar]
- Jaswal, S; Metha, HC; Sood, AK; Kaur, J. Antioxidant status in rheumatoid arthritis and role of antioxidant therapy. Clin. Chim. Acta 2003, 338, 123–129. [Google Scholar]
- Browniee, M. Biochemistry and molecular cell biology of diabetic complications. Nature 2001, 414, 813–820. [Google Scholar]
- Adam-Vizi, V; Chinopoulos, C. Bioenergetics and the formation of mitochondrial reactive oxygen species. Trends Pharmacol. Sci 2006, 27, 639–645. [Google Scholar]
- Kuehl, FA; Egan, RN. Prostaglandins, arachidonic acid and inflammation. Science 1980, 210, 978–984. [Google Scholar]
- Hille, R; Nishino, T. Xantine oxidase and xantine dehydrogenase. FASEB J 1995, 9, 995–1003. [Google Scholar]
- Dahlgren, C; Karlsson, A. Respiratory burst in human neuthrophils. J. Immunol. Methods 1999, 232, 3–14. [Google Scholar]
- Gottlieb, RA. Cytochrome p450: Major player in reperfusion injury. Arch. Biochem. Biophys 2003, 420, 262–267. [Google Scholar]
- Scrader, M; Fahimi, HD. Peroxisomes and oxidative stress. Biochim. Biophys. Acta 2006, 1763, 1755–1766. [Google Scholar]
- Shonfeld, P; Wojtczak, L. Fatty acids as modulators of the cellular production of reactive oxygen species. Free Radic. Biol. Med 2008, 45, 231–241. [Google Scholar]
- Druge, W. Free radicals in physiological control of cell function. Physiol. Rev 2002, 82, 47–95. [Google Scholar]
- Stone, JR; Yang, SP. Hydrogen peroxide: A signaling messenger. Antioxid. Redox Signal 2006, 8, 243–270. [Google Scholar]
- Mahadev, K; Wu, X; Zilbering, A; Zhu, L; Todd, J; Lawrence, R. Hydrogen peroxide generated during cellular insulin stimulation is integral to activation of the distal insulin signaling cascade in 3T3-L1 adipocytes. J. Biol. Chem 2001, 276, 48662–48669. [Google Scholar]
- Lee, H; Jeong, Y; Choi, H; Ko, E; Kim, J. Reactive oxygen species facilitate adipocyte differentiation by accelerating mitotic clonal expansion. J. Biol. Chem 2009, 284, 10601–10609. [Google Scholar]
- Kim, JR; Ryu, HH; Chung, HJ; Lee, JH; Kim, SW; Kwun, WH; Baek, SH; Kim, JH. Association of anti-obesity activity of N-acteylcysteine with metalothionein-II down-regulation. Exp. Mol. Med 2006, 30, 162–172. [Google Scholar]
- Blouet, C; Miriotti, F; Azzout-Marniche, D; Mathe, V; Mikogami, T; Torne, D; Huncau, JF. Dietary cysteine alleviates sucrose-induced oxidative stress and insulin resistance. Free Radic. Biol. Med 2007, 42, 1096–1097. [Google Scholar]
- Lin, Y; Berg, AH; Iyengar, P; Lam, T; Giacca, A; Combs, TP; Rajala, MW; Du, X; Rollman, B; Li, W; et al. The hyperglycemia-induced in inflammatory response in adipocytes. J. Biol. Chem 2005, 280, 4617–4626. [Google Scholar]
- Alberici, L; Oliveira, H; Palm, B; Mantello, C; Augusto, A; Zecchin, K; Gurgeira, S; Kowlstowski, A; Vercesi, A. Mitochondrial ATP-sensitive K+ channels as redox signals to liver mitochondria in response to hypertriglyceridemia. Free Radic. Biol. Med 2009, 47, 1422–1427. [Google Scholar]
- McClung, J; Roneker, C; Mu, W; Lisk, D; Langlais, P; Liu, F; Lei, XG. Development of insulin resistant and obesity in mice overexpressing cellular glutathione peroxidase. Proc. Natl. Acad. Sci. USA 2004, 101, 8852–8857. [Google Scholar]
- Miler, EA; Rios, MC; Dominguez, G; Guerra, LN. Thyroid hormone effect in human hepatocytes. Red. Rep 2008, 13, 185–191. [Google Scholar]
- Cotgreave, IA. N-acetylcysteine: Pharmacological considerations and experimental and clinical applications. Adv. Pharmacol 1997, 38, 205–207. [Google Scholar]
- Araki, S; Dobashi, K; Kugo, K; Yamamoto, Y; Asayama, K; Shirahata, A. N-acetylcysteine attenuates TNF-alpha induced changes in secretion of interleukin-6, plaminogen activator inhibitor-1 and adiponectin from 3T3-L1 adipocytes. Life Sci 2006, 79, 2405–2412. [Google Scholar]
- Kobayashi, H; Matsuda, M; Fukuhara, A; Komuro, R; Shimomura, I. Dysregulated glutathione metabolism links to impaired insulin action in adipocytes. Am. J. Physiol. Endocrinol. Metab 2009, 296, 1326–1334. [Google Scholar]
- Hallenborg, P; Jorgensen, C; Petersen, R; Feddersen, S; Araujo, P; Markt, P; Langer, T; Furstenberger, G; Krieg, P; Koppen, A; et al. Epidermis-type lipoxigenase 3 regulates adipocyte differentiation and peroxisome proliferator activated receptor activity. Mol Cell Biol 2010, 30, 4077–4091. [Google Scholar]
- Durlinton, GJ; Ross, SE; Mac Dougald, OA. The role of C/EBP genes in adipocyte diffentiation. J. Biol. Chem 1998, 272, 30057–30060. [Google Scholar]
- Tang, Q; Otto, T; Lane, D. CCAAT/enhancer-binding protein β is required for mitotic clonal expansion during adipogenesis. Proc. Natl. Acad. Sci. USA 2003, 100, 850–855. [Google Scholar]
- Carriere, A; Carmona, MC; Fernandez, Y; Rigoulet, M; Wengeri, RH; Penicaud, L; Casteilla, L. Mitochondrial reactive oxygen species control the transcription factor CHOP-10/GADD 153 and adipocyte differentiation. J. Biol. Chem 2004, 39, 40462–40469. [Google Scholar]
- Tang, Q; Lane, MD. Role of C/EBP β homologous protein (CHOP-10) in the programmed activation of CCAAT/enhancer binding protein beta during adipogenesis. Proc. Natl. Acad. Sci. USA 2000, 97, 12446–12450. [Google Scholar]
- Rubin, GL; Zhao, Y; Kalus, AM; Simpson, ER. Peroxisome proliferator-activated receptor gamma ligands inhibit estrogen biosynthesis in human breast adipose tissue: Possible implications for breast cancer therapy. Cancer Res 2000, 60, 1604–1608. [Google Scholar]
- Park, BH; Qiang, L; Farmer, SR. Phosphorylation of C/EBP β at a consensus extracelluar signal-regulated kinase/glycogen synthase kinase 3 site is required for the induction of adiponectin gene expression during the differentiation of mouse fibroblast into adipocytes. Mol. Cell Biol 2004, 24, 8671–8679. [Google Scholar]
- Gou, W; Xie, W; Han, J. Modulation of adipocytes lipogenesis by octanoate: Involvement of reactive oxygen species. Nutr. Metab 2006, 3, 30–37. [Google Scholar]
- Fukuoka, H; Iida, K; Nishizawa, H; Imanaka, M; Takeno, R; Iguchi, G; Takahashi, M; Okimura, Y; Kaji, H; Chihara, K; et al. IGF-1 stimulates reactive oxygen species (ROS) production and inhibits insulin-dependent glucose uptake via ROS in 3T3L-1 adipocytes. Growth Horm. IGF Res 2010, 20, 212–219. [Google Scholar]
- Lin, D; Yin, M. Effects on cysteine-containing compounds on biosynthesis of triacylgycerol and cholesterol and anti-oxidative protection in liver from mice consuming a high-fat diet. Br. J. Nutr 2008, 99, 37–41. [Google Scholar]
- Awasthi, YC; Ansari, GAS; Anasthi, S. Regulation of 4-hydroxynonenal mediated signaling by glutathione S-transferases. Methods Enzymol 2005, 401, 379–407. [Google Scholar]
- Uchida, K. 4-hydroxy-2-nonenal: A product and mediator of oxidative stress. Prog. Lipid Res 2003, 42, 13–343. [Google Scholar]
- Pizzimenti, S; Laurora, S; Briatore, F; Ferreti, C; Dianzani, MU; Barrera, G. Sinergistic effect of 4-hydroxinonenal and PPAR ligands in controlling human leukemic cell growth and differentiation. Free Radic. Biol. Med 2002, 32, 233–245. [Google Scholar]
- Muzio, G; Trombetta, A; Maggiora, M; Martinasso, G; Vasiliou, V; Lassen, N; Canuto, RA. Arachidonic and suppressor growth human lung tumor A 549 cells through down regulation of ALDH 3 A1 expression. Free Radic. Biol. Med 2006, 40, 1929–1938. [Google Scholar]
- Ristow, M; Zarse, K; Oberbach, A; Kloting, N; Birringer, M; Kiehntopt, M; Stumvoll, M; Kahn, R; Bluher, M. Antioxidants prevents health promoting effects of physical exercise in humans. Proc. Natl. Acad. Sci. USA 2009, 106, 8665–8670. [Google Scholar]
- Hsu, CL; Yen, GC. Effect of flavonoids and phenolic acid on the inhibition of adipogenesis in 3T3-L1 adipocytes. J. Agric. Food Chem 2007, 17, 8404–8410. [Google Scholar]
- Park, HS; Kim, SH; Kim, YS; Ryu, SY; Hwang, JT; Yang, HJ; Kim, GH; Kwon, DY; Kim, MS. Luteolin inhibits adipogenic differentiation by regulating PPAR gamma activation. Biofactors 2009, 35, 373–379. [Google Scholar]
- Uto-Kondo, H; Ohmori, R; Kiyose, C; Kishimoto, Y; Saito, H; Igarashi, O; Kondo, K. Tocotrienol suppresses adipocyte differentiation and AKT phosphorylation in 3T3-L1 preadipocytes. J. Nutr 2009, 139, 51–57. [Google Scholar]
- Julianelli, VL; Guerra, LN; Calvo, JC. Cell-cell communication between mouse mammary epithelial cells and 3T3-L1 preadipocytes: Effect on triglyceride accumulations and cell proliferarion. Biocell 2007, 31, 237–245. [Google Scholar]
- Baker, MA; Cerniglia, GJ; Zaman, A. Microtiter plate assay for measurement of glutathione and glutathione disulfide in large number of biological samples. Anal. Biochem 1990, 190, 360–365. [Google Scholar]
- Bradford, MM. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem 1976, 72, 248–254. [Google Scholar]
- Kaufman, PB; Wu, W; Kim, D; Cseke, LJ. Handbook of Molecular and Cellular Methods in Biology and Medicine, 1st ed; CRC Press: Boca Raton, FL USA, 1995; pp. 2–25. [Google Scholar]
- McCord, JM; Fridovich, I. Superoxide dismutase. An enzymatic function for erythrocuprein. J. Biol. Chem 1969, 224, 6049–6055. [Google Scholar]
- Arthur, JR; Boyne, R. Superoxide dismutase and glutathione peroxidase activities in neutrophiles from selenium deficient and copper deficient cattle. Life Sci 1985, 36, 1569–1575. [Google Scholar]
- Paglia, DE; Valentine, WN. Studies on the quantitative and qualitative characterization of erythrocyte glutathione peroxidase. J. Lab. Clin. Med 1967, 70, 158–169. [Google Scholar]
- Analytical Software, Statistix 8; McGraw-Hill/Irwin: Tallahases, FL, USA, 2003.


© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Calzadilla, P.; Sapochnik, D.; Cosentino, S.; Diz, V.; Dicelio, L.; Calvo, J.C.; Guerra, L.N. N-Acetylcysteine Reduces Markers of Differentiation in 3T3-L1 Adipocytes. Int. J. Mol. Sci. 2011, 12, 6936-6951. https://doi.org/10.3390/ijms12106936
Calzadilla P, Sapochnik D, Cosentino S, Diz V, Dicelio L, Calvo JC, Guerra LN. N-Acetylcysteine Reduces Markers of Differentiation in 3T3-L1 Adipocytes. International Journal of Molecular Sciences. 2011; 12(10):6936-6951. https://doi.org/10.3390/ijms12106936
Chicago/Turabian StyleCalzadilla, Pablo, Daiana Sapochnik, Soledad Cosentino, Virginia Diz, Lelia Dicelio, Juan Carlos Calvo, and Liliana N. Guerra. 2011. "N-Acetylcysteine Reduces Markers of Differentiation in 3T3-L1 Adipocytes" International Journal of Molecular Sciences 12, no. 10: 6936-6951. https://doi.org/10.3390/ijms12106936



