Evaluation of Antioxidant Compounds and Total Sugar Content in a Nectarine [Prunus persica (L.) Batsch] Progeny
Abstract
:1. Introduction
2. Results and Discussion
2.1. Agronomical and Basic Fruit Quality Traits
2.2. Correlations between Agronomical and Basic Biochemical Traits
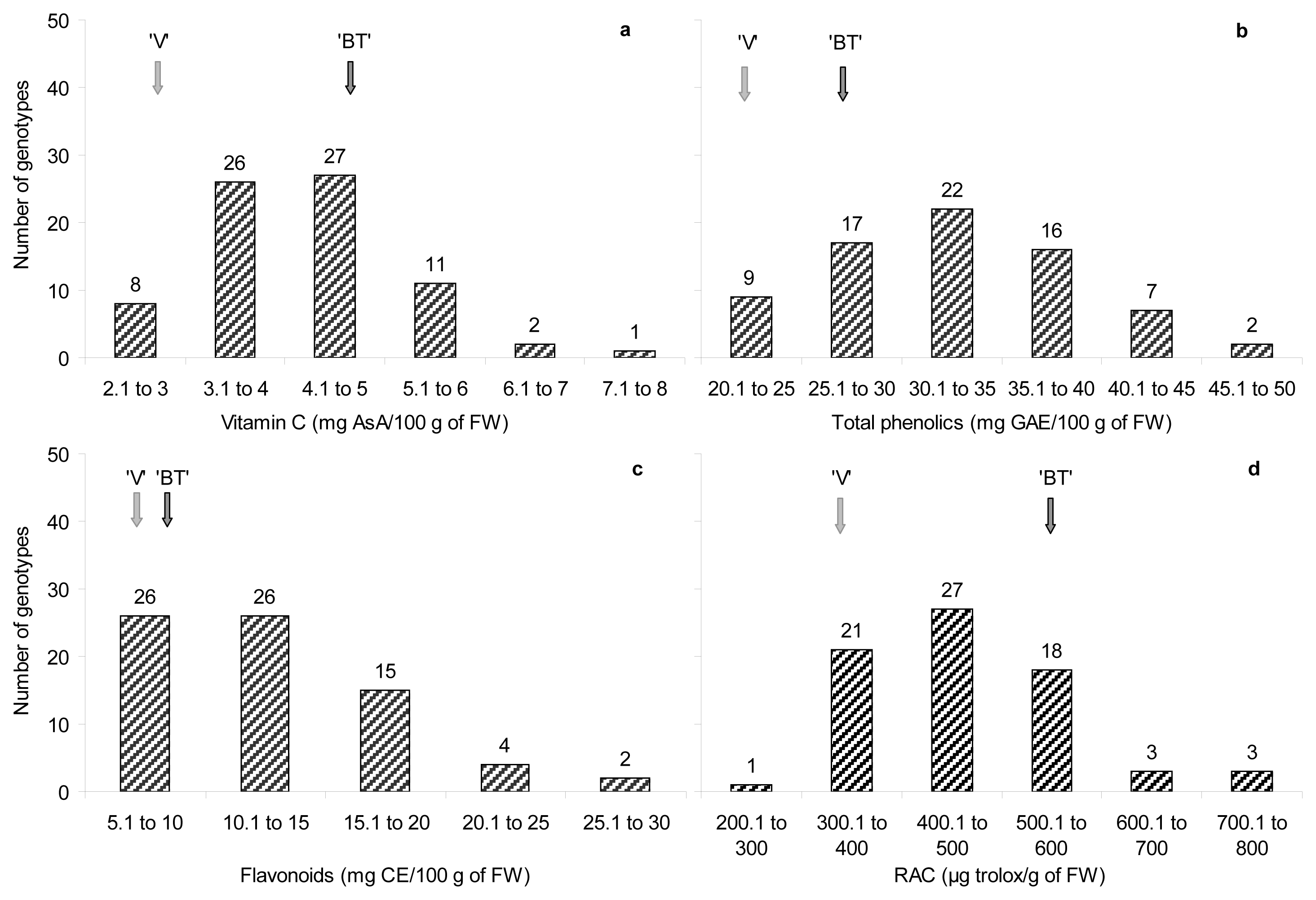
2.3. Antioxidant Compounds Content
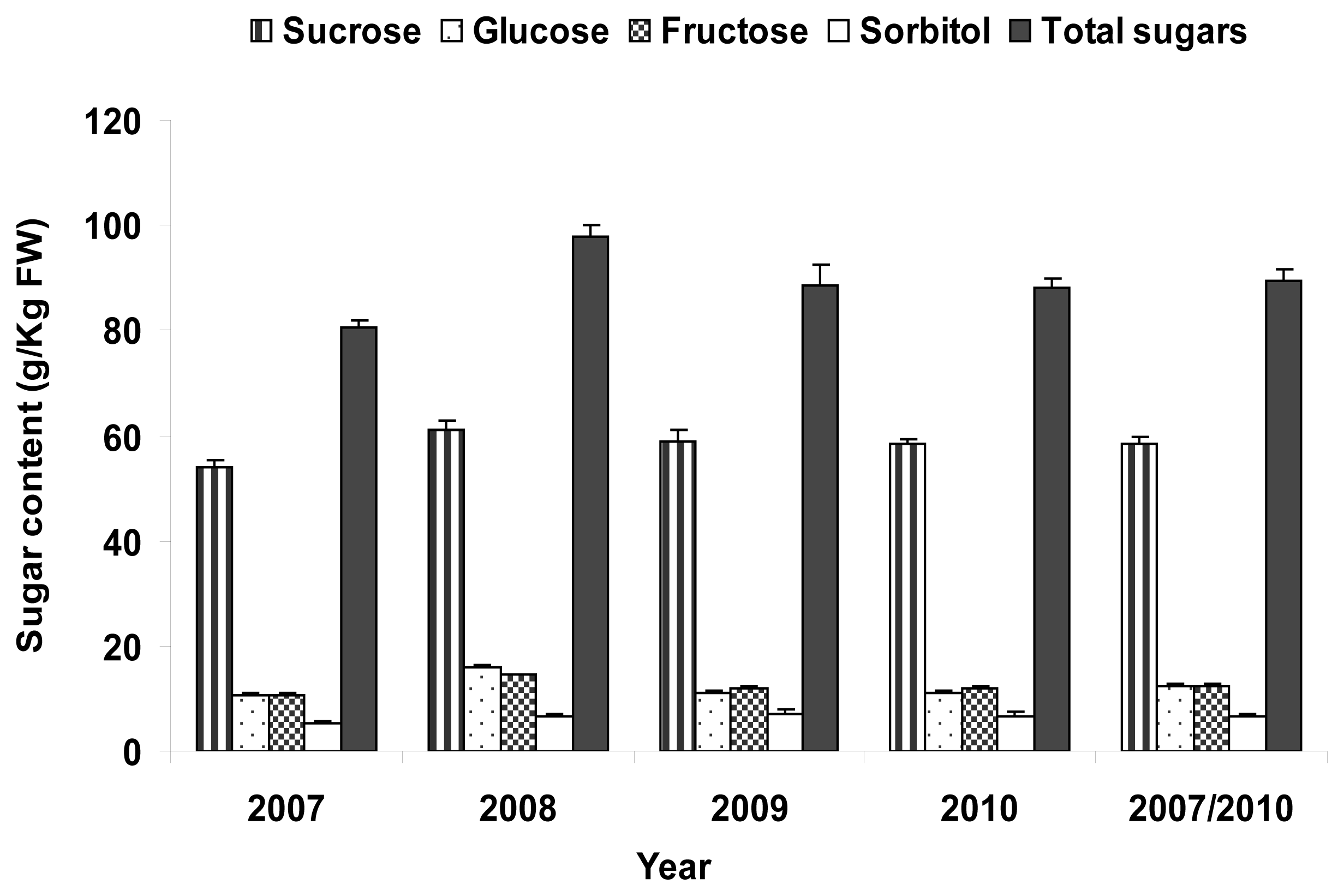
2.4. Total Sugar Content
2.5. Correlations between Phytochemicals Traits
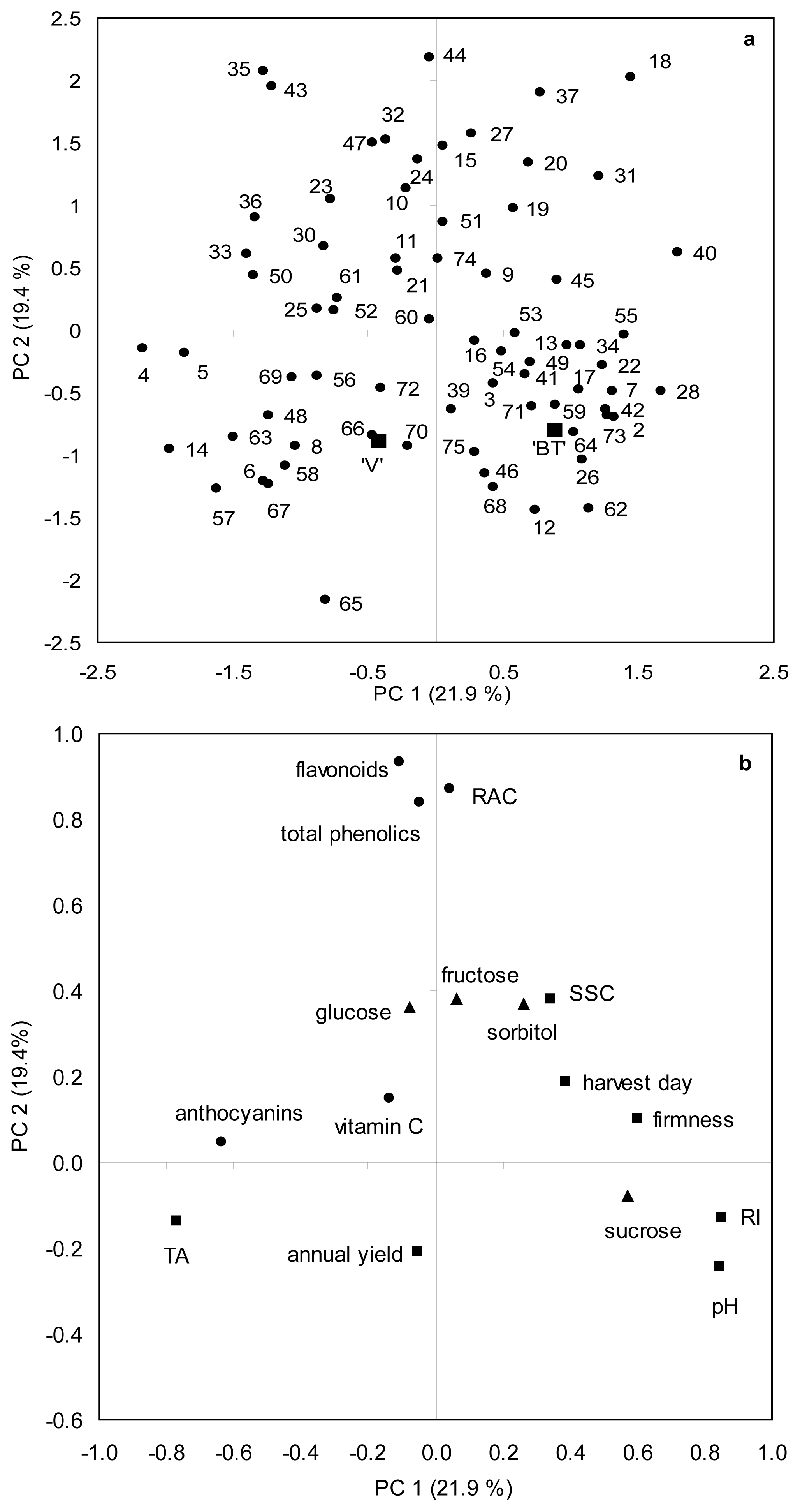
2.6. Principal Component Analysis for Agronomical and Biochemical Traits
3. Experimental Section
3.1. Plant Material
3.2. Agronomical and Basic Fruit Quality Parameters
3.3. Phytochemical Extraction
3.4. Antioxidant Compounds Analysis
3.5. Total Sugars Analysis
3.6. Statistical Analysis
4. Conclusions
Acknowledgments
References
- FAOSTAT. 2011. Available online: http://www.faostat.fao.org accessed on 10 October 2011.
- Wolfe, KL; Kang, X; He, X; Dong, M; Zhang, Q; Liu, RH. Cellular antioxidant activity of common fruits. J. Agric. Food Chem 2008, 56, 8418–8426. [Google Scholar]
- Hegedús, A; Engel, R; Abrankó, L; Balogh, E; Blázovics, A; Hermán, R; Halász, J; Ercisli, S; Pedryc, A; Stefanovits-Banyai, E. Antioxidant and antiradical capacities in apricot (Prunus armeniaca L.) fruits: Variations from genotypes, years, and analytical methods. J. Food Sci 2010, 75, C722–C730. [Google Scholar]
- Müller, L; Gnoyke, S; Popken, AM; Böhm, V. Antioxidant capacity and related parameters of different fruit formulations. Food Sci. Technol 2010, 43, 992–999. [Google Scholar]
- Tsantili, E; Shin, Y; Nock, JF; Watkins, CB. Antioxidant concentrations during chilling injury development in peaches. Postharvest Biol. Technol 2010, 57, 27–34. [Google Scholar]
- Orazem, P; Stampar, F; Hudina, M. Quality analysis of “Redhaven” peach fruit grafted on 11 rootstocks of different genetic origin in a replant soil. Food Chem 2011, 124, 1691–1698. [Google Scholar]
- Bravo, I. Polyphenols: Chemistry, dietary source, metabolism, and nutritional significance. Nutr. Rev 1998, 56, 317–333. [Google Scholar]
- Slimestad, R; Vangdal, E; Brede, C. Analysis of phenolic compounds in six norwegian plum cultivars (Prunus domestica L.). J. Agric. Food Chem 2009, 57, 11370–11375. [Google Scholar]
- Koh, E; Wimalasiri, KMS; Chassy, AW; Mitchell, AE. Content of ascorbic acid, quercetin, kaempferol and total phenolics in commercial broccoli. J. Food Comp. Anal 2009, 22, 637–643. [Google Scholar]
- Cantín, C; Moreno, MA; Gogorcena, Y. Evaluation of the antioxidant capacity, phenolic compounds, and vitamin C content of different peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. J. Agric. Food Chem 2009, 57, 4586–4592. [Google Scholar]
- Rossato, SB; Haas, C; Raseira, MC; Moreira, JC; Zuanazzi, JA. Antioxidant potential of peels and fleshes of peaches from different cultivars. J. Med. Food 2009, 12, 1119–1126. [Google Scholar]
- Crisosto, CH; Crisosto, GM. Relationship between ripe soluble solids concentration (RSSC) and consumer acceptance of high and low acid melting flesh peach and nectarine [Prunus persica (L.) Batsch] cultivars. Postharvest Biol. Technol 2005, 38, 239–246. [Google Scholar]
- Génard, M; Lescourret, F; Gómez, L; Habib, R. Changes in fruit sugar concentrations in response to assimilate supply, metabolism and dilution: A modeling approach applied to peach fruit (Prunus persica). Tree Physiol 2003, 23, 373–385. [Google Scholar]
- Cantín, C; Gogorcena, Y; Moreno, MA. Analysis of phenotypic variation of sugar profile in different peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. J. Sci. Food Agric 2009, 89, 1909–1917. [Google Scholar]
- Kulp, K; Lorenz, K; Stone, M. Functionality of carbohydrate ingredients in bakery products. Food Technol 1991, 45, 136–142. [Google Scholar]
- Dirlewanger, E; Moing, A; Rothan, C; Svanella, L; Pronier, V; Guye, A; Plomion, C; Monet, R. Mapping QTLs controlling fruit quality in peach [P. persica (L.) Batsch]. Theor. Appl. Genet 1999, 98, 18–31. [Google Scholar]
- Cantín, C; Gogorcena, Y; Moreno, MA. Phenotypic diversity and relationships of fruit quality traits in peach and nectarine [Prunus persica (L.) Batsch] breeding progenies. Euphytica 2010, 171, 211–226. [Google Scholar]
- Milatović, D; Nikolić, D; Đurović, D. Variability, heritability and correlations of some factors affecting productivity in peach. Hort. Sci 2010, 37, 79–87. [Google Scholar]
- Valero, C; Crisosto, CH; Slaughter, D. Relationship between nondestructive firmness measurements and commercially important ripening fruit stages for peaches, nectarines and plums. Postharvest Biol. Technol 2007, 44, 248–253. [Google Scholar] [Green Version]
- Kader, AA. Fruit maturity, ripening, and quality relationships. Acta Hort 1999, 485, 203–208. [Google Scholar]
- Quilot, B; Génard, M; Kervella, J; Lescourret, F. Analysis of genotypic variation in fruit flesh total sugar content via an ecophysiological model applied to peach. Theor. Appl. Genet 2004, 109, 440–449. [Google Scholar]
- Monet, R. Transmission Génétique du Caractère “Fruit Doux” Chez le Pêcher. Incidence sur la Sélection Pour la Qualité. In Eucarpia Fruit Section, Tree Fruit Breeding; INRA: Angers, France, 1979; pp. 273–276. [Google Scholar]
- Hilaire, C. The Peach Industry in France: State of Art, Research and Development. Proceeding of the First Mediterranean Peach Symposium, Agrigento, Italy, 10 September, 2003.
- Bassi, D; Selli, R. Evaluation of fruit quality in peach and apricot. Adv. Hort. Sci 1990, 4, 107–112. [Google Scholar]
- Crisosto, CH; Garner, D; Andris, HL; Day, KR. Controlled delayed cooling extends peach market life. Hort. Technol 2004, 14, 99–104. [Google Scholar]
- Crisosto, CH; Slaughter, D; Garner, D; Boyd, J. Stone fruit critical bruising. J. Am. Pomol. Soc 2001, 55, 76–81. [Google Scholar]
- Tavarini, S; Degl’Innocenti, E; Remorini, D; Massai, R; Guidi, L. Preliminary characterisation of peach cultivars for their antioxidant capacity. J. Food Sci. Technol 2008, 43, 810–815. [Google Scholar]
- Gil, M; Tomás-Barberán, AT; Hess-Pierce, B; Kader, AA. Antioxidant capacities, phenolic compounds, carotenoids and vitamin C content of nectarine and plum cultivars from California. J. Agric. Food Chem 2002, 50, 4976–4982. [Google Scholar]
- Vauzour, D; Vafeiadou, K; Rodriguez-Mateos, A; Rendeiro, C; Spencer, J. The neuroprotective potential of flavonoids: A multiplicity of effects. Genes Nutr 2008, 3, 115–126. [Google Scholar]
- Giorgi, M; Capocasa, F; Scalzo, J; Murri, G; Battino, M; Mezzetti, B. The rootstock effects on plant adaptability, production, fruit quality, and nutrition in the peach (cv. “Suncrest”). Sci. Hort 2005, 107, 36–42. [Google Scholar]
- Tulipani, S; Marzban, G; Hernd, A; Laimer, M; Mezzetti, B; Battino, M. Influence of environmental and genetic factors on health-related compounds in strawberry. Food Chem 2011, 124, 906–913. [Google Scholar]
- Robertson, JA; Meredith, FI; Scorza, R. Characteristics of fruit from high- and low-quality peach cultivars. HortScience 1988, 23, 1032–1034. [Google Scholar]
- Colaric, M; Veberic, R; Stampar, F; Hudina, M. Evaluation of peach and nectarine fruit quality and correlations between sensory and chemical attributes. J. Sci. Food Agric 2005, 85, 2611–2616. [Google Scholar]
- Wu, BH; Quilot, B; Génard, M; Kervella, J; Li, SH. Changes in sugar and organic acid concentrations during fruit maturation in peaches, P. davidiana and hybrids as analyzed by principal component analysis. Sci. Hort 2005, 103, 429–439. [Google Scholar]
- Pirie, A; Mullins, MG. Interrelationships of sugars, anthocyanins, total phenols and dry weight in the skin of grape berries during ripening. Am. J. Enol. Vitic 1977, 28, 204–209. [Google Scholar]
- Tabart, J; Kevers, C; Pincemail, J; Defraigne, JO; Dommes, J. Evaluation of spectrophotometric methods for antioxidant compound measurement in relation to total antioxidant capacity in beverages. Food Chem 2010, 120, 607–614. [Google Scholar]
- Escarpa, A; González, MC. Approach to the content of total extractable phenolic compounds from different food samples by comparison of chromatographic and spectrophotometric methods. Ana. Chim. Acta 2001, 427, 119–127. [Google Scholar]
- Jiménez, S; Ollat, N; Deborde, C; Maucourt, M; Rellán-Álvarez, R; Moreno, MA; Gogorcena, Y. Metabolic response in roots of Prunus rootstocks submitted to iron chlorosis. J. Plant Physiol 2011, 168, 415–423. [Google Scholar]
| Progenitors | Progeny a | ||||
|---|---|---|---|---|---|
| Traits | “Venus” | “Big Top” | Min | Max | Mean ± SE |
| Fruit weight | 178.0 ± 58.3 | 204.0 ± 39.3 | 109.2 | 261.8 | 190.2 ± 3.8 |
| Firmness | 36.1 ± 0.1 | 49.2 ± 6.9 | 24.2 | 50.7 | 39.2 ± 0.6 |
| SSC | 13.9 ± 0.1 | 14.4 ± 0.1 | 11.2 | 17.5 | 13.9 ± 0.2 |
| pH | 3.4 ± 0.1 | 3.9 ± 0.1 | 3.2 | 4.0 | 3.6 ± 0.1 |
| TA | 0.7 ± 0.1 | 0.4 ± 0.1 | 0.5 | 1.1 | 0.7 ± 0.1 |
| RI | 20.3 ± 0.3 | 35.2 ± 0.3 | 13.8 | 35.8 | 23.8 ± 0.7 |
| Progenitors | Progeny | ||||
|---|---|---|---|---|---|
| Compounds | “Venus” | “Big Top” | Min | Max | Mean ± SE |
| Vitamin C | 3.0 ± 0.7 | 4.9 ± 0.7 | 2.1 | 7.2 | 4.0 ± 0.1 |
| Total phenolics | 22.1 ± 8.0 | 26.4 ± 9.8 | 22.5 | 49.2 | 32.6 ± 0.7 |
| Flavonoids | 7.6 ± 3.8 | 7.8 ± 4.6 | 5.9 | 33.8 | 12.5 ± 0.6 |
| Anthocyanins | 2.1 ± 0.1 | 5.9 ± 2.2 | 1.2 | 9.5 | 3.2 ± 0.2 |
| RAC | 386.1 ± 18.5 | 521.4 ± 47.4 | 292.4 | 835.8 | 464.2 ± 12.5 |
| Compounds | 2007 | 2008 | 2009 | 2010 | Mean ± SE |
|---|---|---|---|---|---|
| Vitamin C | 2.8 ± 0.1 | 3.9 ± 0.2 | 6.3 ± 0.2 | 3.2 ± 0.2 | 4.0 ± 0.1 |
| Total phenolics | 36.9 ± 1.7 | 44.2 ± 0.7 | 21.2 ± 0.6 | 23.3 ± 0.8 | 32.6 ± 0.7 |
| Flavonoids | 12.9 ± 1.0 | 21.7 ± 1.2 | 6.5 ± 0.4 | 8.1 ± 0.6 | 12.5 ± 0.6 |
| Anthocyanins | 2.2 ± 0.2 | 1.7 ± 0.1 | 4.0 ± 0.5 | 4.6 ± 0.4 | 3.2 ± 0.2 |
| RAC | 380.6 ± 14 | 617.0 ± 23 | 322.6 ± 11 | 444.8 ± 10 | 464.2 ± 12.5 |
| Progenitors | Progeny | ||||
|---|---|---|---|---|---|
| Sugar content | “Venus” | “Big Top” | Min | Max | Mean ± SE |
| Sucrose | 41.0 ± 5.7 | 85.1 ± 17.0 | 40.7 | 102.3 | 58.4 ± 1.2 |
| Glucose | 10.0 ± 0.4 | 8.9 ± 0.8 | 8.3 | 23.4 | 12.2 ± 0.3 |
| Fructose | 13.4 ± 0.5 | 10.9 ± 1.7 | 8.9 | 19.1 | 12.4 ± 0.2 |
| Sorbitol | 8.6 ± 3.8 | 6.5 ± 1.7 | 1.7 | 19.5 | 6.6 ± 0.5 |
| Sucrose/glucose | 4.0 ± 0.4 | 9.5 ± 2.1 | 3.2 | 7.6 | 4.9 ± 0.1 |
| Glucose/fructose | 0.7 ± 0.1 | 0.8 ± 0.1 | 0.8 | 1.2 | 0.9 ± 0.1 |
| % Sorbitol | 5.6 ± 1.9 | 2.9 ± 0.4 | 1.1 | 8.7 | 3.5 ± 0.2 |
| Total sugars | 73.0 ± 9.6 | 111.5 ± 14.1 | 67.4 | 138.9 | 89.7 ± 1.6 |
| Traits | Flavonoids | Total phenolics | RAC | SSC | Total Sugars |
|---|---|---|---|---|---|
| Vitamin C | 0.420 ** | 0.374 ** | 0.455 ** | 0.579 ** | 0.350 ** |
| Flavonoids | 0.807 ** | 0.851 ** | 0.482 ** | ns | |
| Total phenolics | 0.738 ** | 0.524 ** | 0.398 ** | ||
| RAC | 0.597 ** | 0.384 ** |
© 2011 by the authors; licensee MDPI, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Abidi, W.; Jiménez, S.; Moreno, M.Á.; Gogorcena, Y. Evaluation of Antioxidant Compounds and Total Sugar Content in a Nectarine [Prunus persica (L.) Batsch] Progeny. Int. J. Mol. Sci. 2011, 12, 6919-6935. https://doi.org/10.3390/ijms12106919
Abidi W, Jiménez S, Moreno MÁ, Gogorcena Y. Evaluation of Antioxidant Compounds and Total Sugar Content in a Nectarine [Prunus persica (L.) Batsch] Progeny. International Journal of Molecular Sciences. 2011; 12(10):6919-6935. https://doi.org/10.3390/ijms12106919
Chicago/Turabian StyleAbidi, Walid, Sergio Jiménez, María Ángeles Moreno, and Yolanda Gogorcena. 2011. "Evaluation of Antioxidant Compounds and Total Sugar Content in a Nectarine [Prunus persica (L.) Batsch] Progeny" International Journal of Molecular Sciences 12, no. 10: 6919-6935. https://doi.org/10.3390/ijms12106919






