A Single Amino Acid Substitution Changes Antigenicity of ORF2-Encoded Proteins of Hepatitis E Virus
Abstract
:1. Introduction
2. Results
2.1. Preparation of Mabs against p166Bur, p166Pak and p166Mor
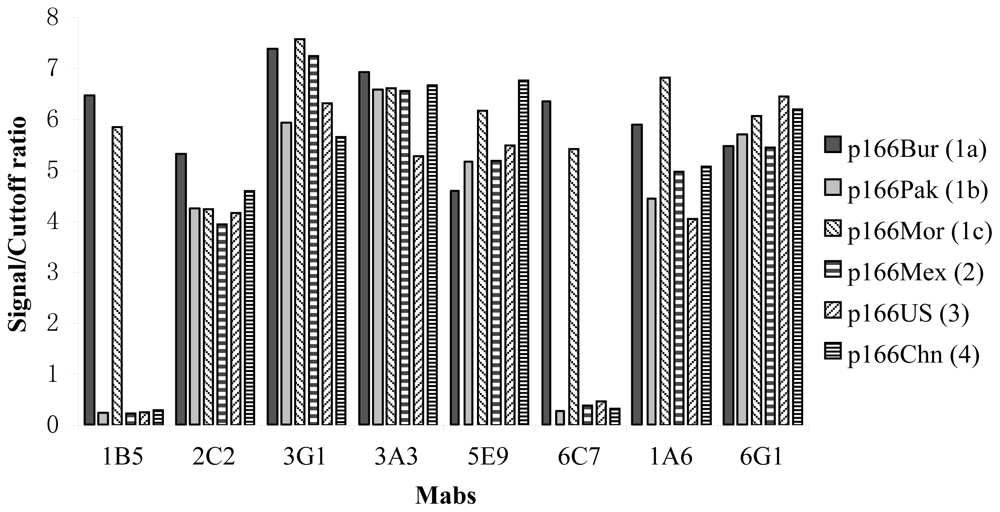
2.2. Difference in Immunoreactivity of the Mabs
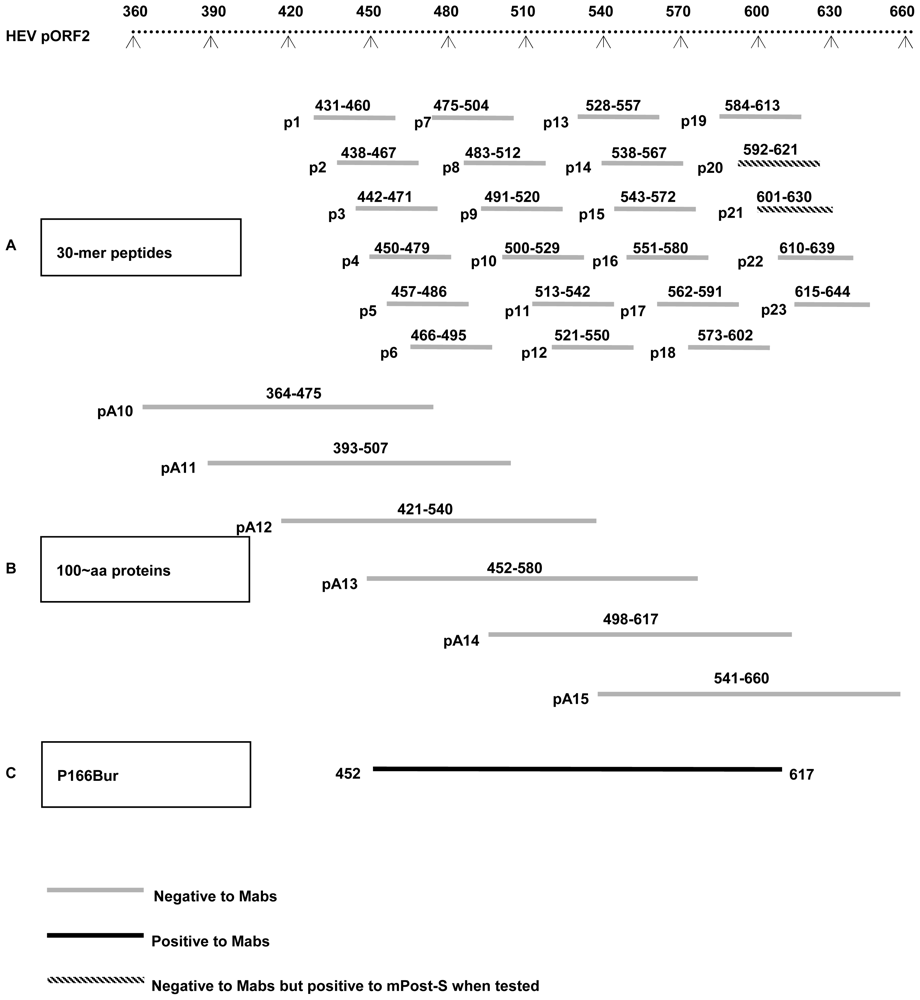
2.3. Epitopes Recognized by the Mabs Are Conformation-Dependent
2.4. Position 606 Is Critical for the Epitope Recognized by Mab 1B5 and 6C7
3. Discussion
4. Materials and Methods
4.1. Recombinant Proteins and Synthetic Peptides
4.2. Generation of Mabs
4.3. ELISA
4.4. Western Blotting
Acknowledgements
References
- Dalton, HR; Bendall, R; Ijaz, S; Banks, M. Hepatitis E: An emerging infection in developed countries. Lancet Infect. Dis 2008, 8, 698–709. [Google Scholar]
- Kamar, N; Selves, J; Mansuy, JM; Ouezzani, L; Peron, JM; Guitard, J; Cointault, O; Esposito, L; Abravanel, F; Danjoux, M; Durand, D; Vinel, JP; Izopet, J; Rostaing, L. Hepatitis E virus and chronic hepatitis in organ-transplant recipients. N. Eng. J. Med 2008, 358, 811–817. [Google Scholar]
- Pan, JS; Zhang, K; Zhou, JY; Wu, C; Zhuang, H; Zhou, YH. Application of truncated immunodominant polypeptide from hepatitis E virus (HEV) ORF2 in an assay to exclude nonspecific binding in detecting anti-HEV immunoglobulin M. J. Clin. Microbiol 2010, 48, 779–784. [Google Scholar]
- Emerson, SU; Anderson, D; Arankalle, A; Meng, XJ; Purdy, M; Schlauder, GG; Tsarev, SA. Fauquet, CM, Mayo, MA, Maniloff, J, Desselberger, U, Ball, LA, Eds.; Hepevirus. In Virus Taxonomy: Eighth Report of the International Committee on Taxonomy of Viruses; Elsevier Academic Press: London, UK, 2004; pp. 851–855. [Google Scholar]
- Okamoto, H. Genetic variability and evolution of hepatitis E virus. Virus Res 2007, 127, 216–228. [Google Scholar]
- Lu, L; Li, CH; Hagedorn, CH. Phylogenetic analysis of global hepatitis E virus sequences: Genetic diversity, subtypes and zoonosis. Rev. Med. Virol 2006, 16, 5–36. [Google Scholar]
- Zhai, L; Dai, X; Meng, J. Hepatitis E virus genotyping based on full-length genome and partial genomic regions. Virus Res 2006, 120, 57–69. [Google Scholar]
- Reyes, GR; Purdy, MA; Kim, JP; Luk, KC; Young, LM; Fry, KE; Bradley, DW. Isolation of a cDNA from the virus responsible for enterically transmitted non-A, non-B hepatitis. Science 1990, 247, 1335–1339. [Google Scholar]
- Huang, CC; Nguyen, D; Fernandez, J; Yun, KY; Fry, KE; Bradley, DW; Tam, AW; Reyes, GR. Molecular-cloning and sequencing of the Mexico isolate of hepetitis-E virus (HEV). Virology 1992, 191, 550–558. [Google Scholar]
- Schlauder, GG; Dawson, GJ; Erker, JC; Kwo, PY; Knigge, MF; Smalley, DL; Rosenblatt, JE; Desai, SM; Mushahwar, IK. The sequence and phylogenetic analysis of a novel hepatitis E virus isolated from a patient with acute hepatitis reported in the United States. J. Gen. Virol 1998, 79, 447–456. [Google Scholar]
- Wang, YC; Zhang, HY; Ling, R; Li, HM; Harrison, TJ. The complete sequence of hepatitis E virus genotype 4 reveals an alternative strategy for translation of open reading frames 2 and 3. J. Gen. Virol 2000, 81, 1675–1686. [Google Scholar]
- Engle, RE; Yu, C; Emerson, SU; Meng, XJ; Purcell, RH. Hepatitis E virus (HEV) capsid antigens derived from viruses of human and swine origin are equally efficient for detecting anti- HEV by enzyme immunoassay. J. Clin. Microbiol 2002, 40, 4576–4580. [Google Scholar]
- Zhou, YH; Purcell, RH; Emerson, SU. An ELISA for putative neutralizing antibodies to hepatitis E virus detects antibodies to genotypes 1, 2, 3, and 4. Vaccine 2004, 22, 2578–2585. [Google Scholar]
- Arankalle, VA; Lole, KS; Deshmukh, TM; Chobe, LP; Gandhe, SS. Evaluation of human (genotype 1) and swine (genotype 4)-ORF2-based ELISAs for anti-HEV IgM and IgG detection in an endemic country and search for type 4 human HEV infections. J. Viral. Hepat 2007, 14, 435–445. [Google Scholar]
- Peralta, B; Casas, M; de Deus, N; Martin, M; Ortuno, A; Perez-Martin, E; Pina, S; Mateu, E. Anti-HEV antibodies in domestic animal species and rodents from Spain using a genotype 3-based ELISA. Vet. Microbiol 2009, 137, 66–73. [Google Scholar]
- Wang, YC; Zhang, HY; Li, Z; Gu, WJ; Lan, HY; Hao, W; Ling, R; Li, HM; Harrison, TJ. Detection of sporadic cases of hepatitis e virus (HEV) infection in china using immunoassays based on recombinant open reading frame 2 and 3 polypeptides from HEV genotype 4. J. Clin. Microbiol 2001, 39, 4370–4379. [Google Scholar]
- Herremans, M; Duizer, E; Jusic, E; Koopmans, MPG. Detection of hepatitis E virus-specific immunoglobulin A in patients infected with hepatitis E virus genotype 1 or 3. Clin. Vaccine Immunol 2007, 14, 276–280. [Google Scholar]
- Bendall, R; Ellis, V; Ijaz, S; Thurairajah, P; Dalton, HR. Serological response to hepatitis E virus genotype 3 infection: IgG quantitation, avidity, and IgM response. J. Med. Virol 2008, 80, 95–101. [Google Scholar]
- Jiménez de Oya, N; Galindo, I; Gironés, O; Duizer, E; Escribano, J; Saiz, J. Serological immunoassay for detection of hepatitis E virus on the basis of genotype 3 open reading frame 2 recombinant proteins produced in Trichoplusia ni larvae. J. Clin. Microbiol 2009, 47, 3276–3282. [Google Scholar]
- Meng, JH; Dai, X; Chang, JC; Lopareva, E; Pillot, J; Fields, HA; Khudyakov, YE. Identification and characterization of the neutralization epitope(s) of the hepatitis E virus. Virology 2001, 288, 203–211. [Google Scholar]
- Deng, L; Meng, J-h; Zhao, Y; Zhang, H-m; Dai, X. Hepatitis E virus of different genotypes contains multiple-type antigenic epitopes. Acta Microbiol. Sinica 2006, 46, 120–126. [Google Scholar]
- Zhao, Y; Liang, JH; Meng, JH; Zhang, LF; Zhang, HM; Ding, F; Ji, XH. Comparison of B ell antigenic epitopes between hepatitis E virus genotype I, II and genotype III, IV by using monoclonal antibodies. Chin. J. Immunol 2004, 20, 591–595. [Google Scholar]
- Schofield, D; Purcell, R; Nguyen, H; Emerson, S. Monoclonal antibodies that neutralize HEV recognize an antigenic site at the carboxyterminus of an ORF2 protein vaccine. Vaccine 2003, 22, 257–267. [Google Scholar]
- Meng, JH; Cong, ME; Dai, X; Pillot, J; Purdy, MA; Fields, HA; Khudyakov, YE. Primary structure of open reading frame 2 and 3 of the hepatitis E virus isolated from Morocco. J. Med. Virol 1999, 57, 126–133. [Google Scholar]
- Meng, XJ; Purcell, RH; Halbur, PG; Lehman, JR; Webb, DM; Tsareva, TS; Haynes, JS; Thacker, BJ; Emerson, SU. A novel virus in swine is closely related to the human hepatitis E virus. Proc. Natl. Acad. Sci. USA 1997, 94, 9860–9865. [Google Scholar]
- Wang, YC; Zhang, HY; Xia, NS; Peng, G; Lan, HY; Zhuang, H; Zhu, YH; Li, SW; Tian, KG; Gu, WJ; Lin, JX; Wu, X; Li, HM; Harrison, TJ. Prevalence, isolation, and partial sequence analysis of hepatitis E virus from domestic animals in China. J. Med. Virol 2002, 67, 516–521. [Google Scholar]
- Ji, YL; Zhu, YH; Liang, JR; Wei, XF; Yang, X; Wang, L; Li, LJ; Chang, YB; Tang, RL; Zhuang, H. Swine hepatitis E virus in rural southern China: Genetic characterization and experimental infection in rhesus monkeys (Macaca mulatta). J. Gastroenterol 2008, 43, 565–570. [Google Scholar]
- Kase, JA; Correa, MT; Luna, C; Sobsey, MD. Isolation, detection and characterization of swine hepatitis E virus from herds in Costa Rica. Int. J. Environ. Health Res 2008, 18, 165–176. [Google Scholar]
- Xia, HY; Liu, LH; Linde, AM; Belak, S; Norder, H; Widen, F. Molecular characterization and phylogenetic analysis of the complete genome of a hepatitis E virus from European swine. Virus Gene 2008, 37, 39–48. [Google Scholar]
- Li, TC; Chijiwa, K; Sera, N; Ishibashi, T; Etoh, Y; Shinohara, Y; Kurata, Y; Ishida, M; Sakamoto, S; Takeda, N; Miyamura, T. Hepatitis E virus transmission from wild boar meat. Emerg. Infect. Dis 2005, 11, 1958–1960. [Google Scholar]
- Nishizawa, T; Takahashi, M; Endo, K; Fujiwara, S; Sakuma, N; Kawazuma, F; Sakamoto, H; Sato, Y; Bando, M; Okamoto, H. Analysis of the full-length genome of hepatitis E virus isolates obtained from wild boars in Japan. J. Gen. Virol 2005, 86, 3321–3326. [Google Scholar]
- Martelli, F; Caprioli, A; Zengarini, M; Marata, A; Fiegna, C; Di Bartolo, I; Ruggeri, FM; Delogu, M; Ostanello, F. Detection of Hepatitis E virus (HEV) in a demographic managed wild boar (Sus scrofa scrofa) population in Italy. Vet. Microbiol 2008, 126, 74–81. [Google Scholar]
- Kaci, S; Nockler, K; Johne, R. Detection of hepatitis E virus in archived German wild boar serum samples. Vet. Microbiol 2008, 128, 380–385. [Google Scholar]
- Takahashi, K; Kitajima, N; Abe, N; Mishiro, S. Complete or near-complete nucleotide sequences of hepatitis E virus genome recovered from a wild boar, a deer, and four patients who ate the deer. Virology 2004, 330, 501–505. [Google Scholar]
- Zhao, CY; Ma, ZR; Harrison, TJ; Feng, RF; Zhang, CT; Qiao, ZL; Fan, JP; Ma, HX; Li, MS; Song, AJ; Wang, YC. A novel genotype of hepatitis E virus prevalent among farmed rabbits in China. J. Med. Virol 2009, 81, 1371–1379. [Google Scholar]
- Tei, S; Kitajima, N; Takahashi, K; Mishiro, S. Zoonotic transmission of hepatitis E virus from deer to human beings. Lancet 2003, 362, 371–373. [Google Scholar]
- Meng, JH; Pillot, J; Dai, X; Fields, HA; Khudyakov, YE. Neutralization of different geographic strains of the hepatitis E virus with anti-hepatitis E virus-positive serum samples obtained from different sources. Virology 1998, 249, 316–324. [Google Scholar]
- Zhou, YH; Chen, ZC; Purcell, RH; Emerson, SU. Positive reactions on Western blots do not necessarily indicate the epitopes on antigens are continuous. Immunol. Cell Biol 2007, 85, 73–78. [Google Scholar]
- Yamashita, T; Mori, Y; Miyazaki, N; Cheng, RH; Yoshimura, M; Unno, H; Shima, R; Moriishi, K; Tsukihara, T; Li, TC; Takeda, N; Miyamura, T; Matsuura, Y. Biological and immunological characteristics of hepatitis E virus-like particles based on the crystal structure. Proc. Natl. Acad. Sci. USA 2009, 106, 12986–12991. [Google Scholar]
- He, J. Molecular detection and sequence analysis of a new hepatitis E virus isolate from Pakistan. J. Viral Hepat 2006, 13, 840–844. [Google Scholar]
- Shen, Q; Zhang, W; Cao, X; Mou, J; Cui, L; Hua, X. Cloning of full genome sequence of hepatitis E virus of Shanghai swine isolate using RACE method. Virology 2007, 4, 98. [Google Scholar]
- Glaser, L; Stevens, J; Zamarin, D; Wilson, IA; Garcia-Sastre, A; Tumpey, TM; Basler, CF; Taubenberger, JK; Palese, P. A single amino acid substitution in 1918 influenza virus hemagglutinin changes receptor binding specificity. Virology 2005, 79, 11533–11536. [Google Scholar]
- Shiomi, H; Urasawa, T; Urasawa, S; Kobayashi, N; Abe, S; Taniguchi, K. Isolation and characterisation of poliovirus mutants resistant to heating at 50 degrees C for 30 min. J. Med. Virol 2004, 74, 484–491. [Google Scholar]
- Zhang, H; Dai, X; Shan, X; Meng, J. Characterization of antigenic epitopes of the ORF2 protein from hepatitis E virus genotype 4. Virus Res 2009, 142, 140–143. [Google Scholar]
- Khudyakov, YE; Lopareva, EN; Jue, DL; Crews, TK; Thyagarajan, SP; Fields, HA. Antigenic domains of the open reading frame 2-encoded protein of hepatitis E virus. J. Clin. Microbiol 1999, 37, 2863–2871. [Google Scholar]
- Adler, B; Faine, S. A pomona serogroup-specific, agglutinating antigen in leptospira, identified by monoclonal-antibodies. Pathology 1983, 15, 247–250. [Google Scholar]
- Obriadina, A; Meng, JH; Lopareva, EN; Spelbring, J; Burkov, A; Krawczynski, K; Khudyakov, YE; Fields, HA. Antigenic properties of recombinant proteins of hepatitis E virus. In Proceedings of 10th International Symposium on Viral Hepatitis and Liver Disease; International Medical Press: London, UK, 2002; pp. 119–123. [Google Scholar]
| HEV genotype | Mutations associated with HEV genotypes/subtypes within the p166 region* | ||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| 483 | 490 | 492 | 497 | 500 | 509 | 511 | 517 | 527 | 537 | 569 | 571 | 580 | 587 | 590 | 593 | 599 | 606 | 609 | 614 | ||
| 1a | S | G | V | S | L | A | A | T | S | F | L | V | A | S | A | V | A | A | L | L | |
| 1 | 1b | S | G | V | S | L | A | A | T | S | F | L | V | A | S | A | V | A | A/V | L | M |
| 1c | S | G | V | S | L | A | A | T | S | F | L | V | A | S | A | V | A | A | L | L | |
| 2 | S | G | V | S | L | A | A | S | P | F | I | I | A | R | A | V | A | A | L | F | |
| 3 | T | N | M | T | F/L | A | A | S | T | Y | I | I/V | A | S | A | T | G | A/V | V | A/V | |
| 4 | T | N | M | T | F | G | S | S | T/M | F/Y | I | I | C | N | S | V | G | A | A/V | V/A | |
| Proteins | Primer sequence |
|---|---|
| pA10 (aa 364–475) | Forward 5′-CCCGGATCCATCGGCCGCGGGATAGCCCTCAC-3′ Reverse 5′-CCCCTCGAGTCAGAGAGAGAGCCAAAGCACATC-3′ |
| pA11 (aa 393–507) | Forward 5′-CCCGGATCCGGCCAGCTGTTCTACTCCCGTCC-3′ Reverse 5′-CCCCTCGAGTCACGCGCCGGTCGCAACATTAACC-3′ |
| pA12 (aa 421–540) | Forward 5′-CCCGGATCCCAGGATAAGGGTATTGCAATCC-3′ Reverse 5′-CCCCTCGAGTCACGGCAGGACAAAGAAGGTCTTC-3′ |
| pA13 (aa 452–580) | Forward 5′-CCCGGATCCCCGACGCCTTCTCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGCGACCCGGTGCCCGGCGGCA-3′ |
| pA14 (aa 498–617) | Forward 5′-CCCGGATCCACCTTGGTTAATGTTGCGACC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCAAGGTATCCTCAA-3′ |
| pA15 (aa 541–660) | Forward 5′-CCCGGATCCCTCCGCGGTAAGCTCTCTTTC-3′ Reverse 5′-CCCCTCGAGCTACAACTCCCGAGTTTTACC-3′ |
| p166Bur (aa 452–617) | Forward 5′-CCCGGATCCCCGACGCCTTCTCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCAAGGTATCCTCAA-3′ |
| p166Bur/A155V | Mutant primer 5′-CTCTGTGCTAGCATTGCTTGAGGATAC-3′ Forward 5′-CCCGGATCCCCGACGCCTTCTCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCAAGGTATCCTCAA-3′ |
| p166Bur/L163M | Forward 5′-CCCGGATCCCCGACGCCTTCTCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCATGGTATCCTCAA-3′ |
| p166Pak (aa 452–617) | Forward 5′-CCCGGATCCCCGACACCTTCCCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCATGGTATCCTCAA-3′ |
| p166Pak/V155A | Mutant primer 5′-CTCTGCGCTAGCATTGCTTGAGGATAC-3′ Forward 5′-CCCGGATCCCCGACACCTTCCCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCATGGTATCCTCAA-3′ |
| p166Pak/M163L | Forward 5′-CCCGGATCCCCGACACCTTCCCCAGCCCCATC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAGTCCAAGGTATCCTCAA-3′ |
| p166Mor (aa 452–617) | Forward 5′-CCCGGATCCCCGACACCTTCCCCAGCCCCGTC-3′ Reverse 5′-CCCCTCGAGCCCACCCCGTCGCCTGCGCCATC-3′ |
| p166Mex (aa 452–617) | Forward 5′-CCCGGATCCCCCACCCCGTCGCCTGCGCCATC-3′ Reverse 5′-CCCCTCGAGTCACGGATAATCAAAAGTATCCTCCA-3′ |
| p166US (aa 452–617) | Forward 5′-CCCGGATCCCCTACCCCGTCACCTGCCCCCTC-3′ Reverse 5′-CCCCTCGAGTCAAGGATAATCAACAGTATCCTCGA-3′ |
| p166Chn (aa 452–617) | Forward 5′-CCCGGATCCCCTACCCCCTCTCCTGCTCCCTC-3′ Reverse 5′-CCCCTCGAGTCAAGGGTAATCAACAGTGTCCTCCA-3′ |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Liang, J.-H.; Dai, X.; Dong, C.; Meng, J.-H. A Single Amino Acid Substitution Changes Antigenicity of ORF2-Encoded Proteins of Hepatitis E Virus. Int. J. Mol. Sci. 2010, 11, 2962-2975. https://doi.org/10.3390/ijms11082962
Liang J-H, Dai X, Dong C, Meng J-H. A Single Amino Acid Substitution Changes Antigenicity of ORF2-Encoded Proteins of Hepatitis E Virus. International Journal of Molecular Sciences. 2010; 11(8):2962-2975. https://doi.org/10.3390/ijms11082962
Chicago/Turabian StyleLiang, Jiu-Hong, Xing Dai, Chen Dong, and Ji-Hong Meng. 2010. "A Single Amino Acid Substitution Changes Antigenicity of ORF2-Encoded Proteins of Hepatitis E Virus" International Journal of Molecular Sciences 11, no. 8: 2962-2975. https://doi.org/10.3390/ijms11082962




