Flavanols and Anthocyanins in Cardiovascular Health: A Review of Current Evidence
Abstract
:1. Introduction
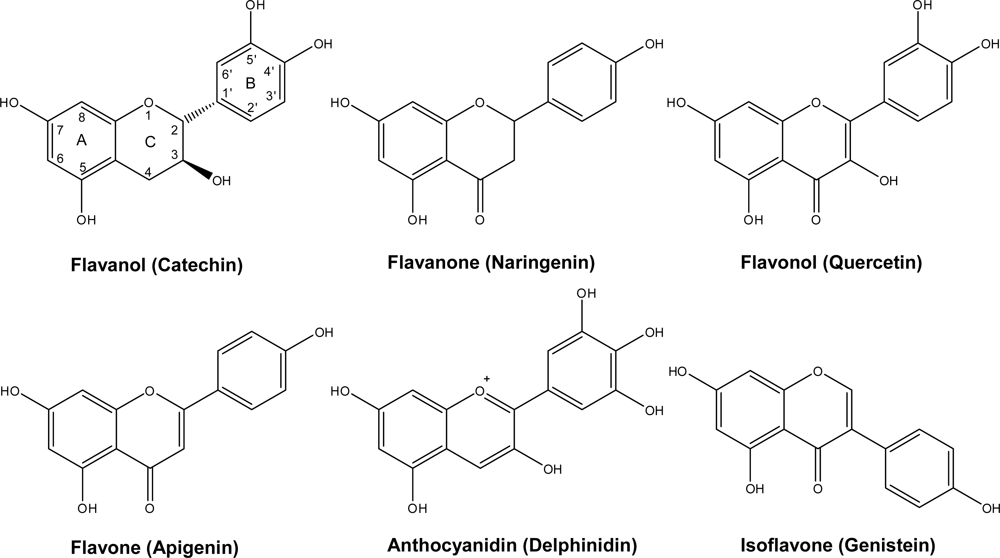
2. Chemistry and Distribution
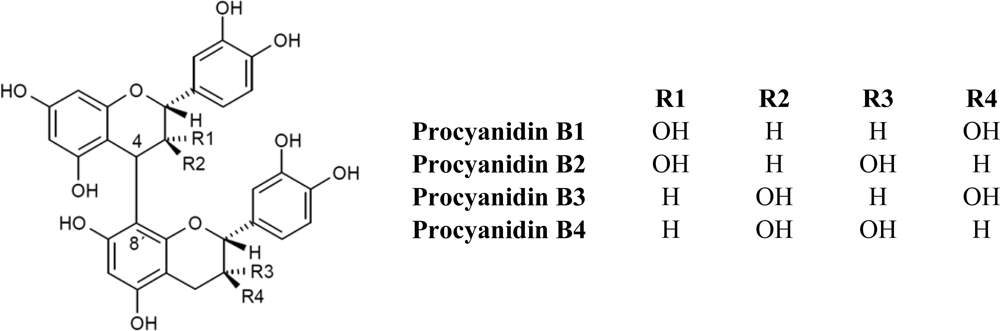
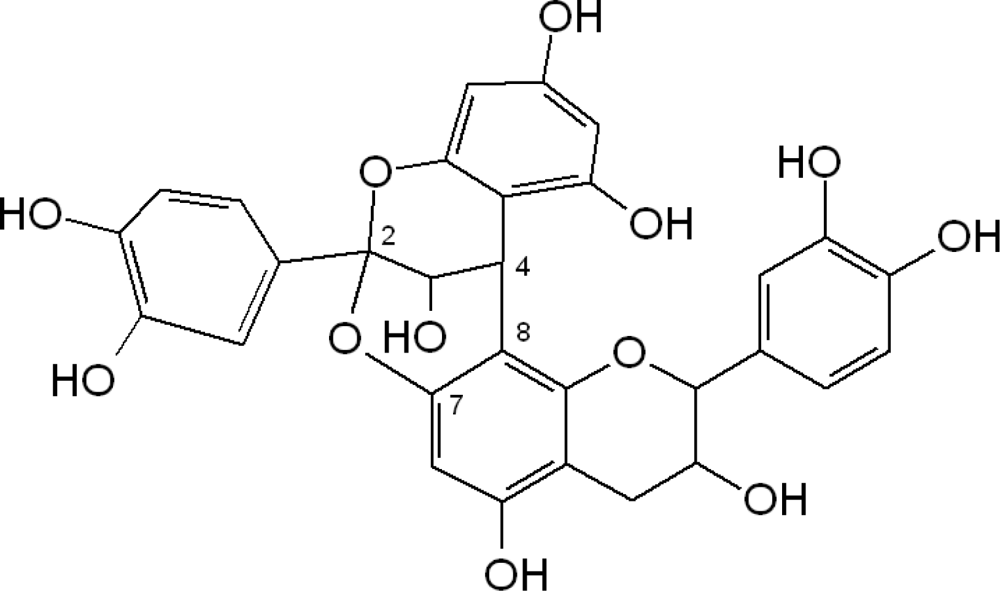
2.1. Flavanols: Monomeric Catechins and Proanthocyanidins
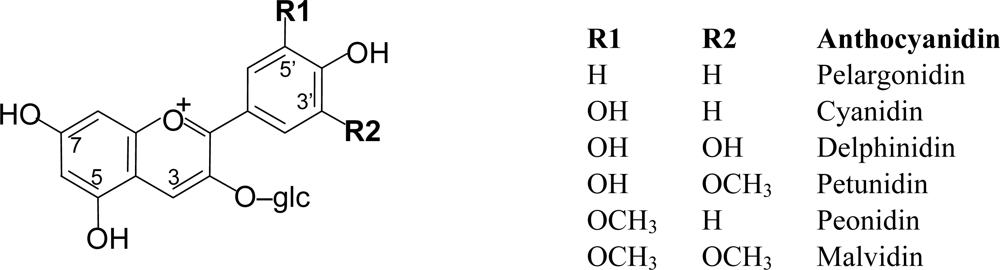
2.2. Anthocyanins
3. Cardiovascular Effects
3.1. Flavanols: Monomeric Catechins And (Oligomeric) Proanthocyanidins
3.1.1. Catechins
3.1.2. Oligomeric Procyanidins
3.2. Anthocyanins
4. Conclusions
Acknowledgments
References
- Harborne, JB; Baxter, H. The Handbook of Natural Flavonoids; Wiley: West Sussex, UK, 1999; Volume 2. [Google Scholar]
- Harborne, JB. The Flavonoids: Advances in Research since 1986; Chapman and Hall: London, UK, 1993. [Google Scholar]
- U.S. Department of Agriculture (USDA). USDA Database for the Flavonoid Content of Selected Foods—2003. 2003. Available at: http://www.nal.usda.gov/fnic/foodcomp/Data/Flav/flav.html (Accessed on 17 September 2009).
- Williams, RJ; Spencer, JP; Rice-Evans, C. Flavonoids: Antioxidants or signalling molecules? Free Radic. Biol. Med 2004, 36, 838–849. [Google Scholar]
- World Health Organization (W.H.O.). Cardiovascular diseases (CVDs) Fact sheet Nº 317. September 2009. Available at: http://www.who.int/mediacentre/factsheets/fs317/en/index.html (Accessed on 12 April 2010).
- Cassidy, A; de Pascual-Teresa, S; Rimbach, G. Molecular mechanisms by which dietary isoflavones potentially prevent atherosclerosis. Expert Rev. Mol. Med 2003, 5, 1–15. [Google Scholar]
- Boots, AW; Haenen, GR; Bast, A. Health effects of quercetin: from antioxidant to nutraceutical. Eur. J. Pharm 2008, 585, 325–337. [Google Scholar]
- Crozier, A; Jaganath, IB; Clifford, MN. Dietary phenolics: chemistry, bioavailability and effects on health. Nat. Product Rep 2009, 26, 10001–1043. [Google Scholar]
- Modrianský, M; Gabrielová, E. Uncouple my heart: the benefits of inefficiency. J. Bioenerg. Biomembr 2009, 41, 133–136. [Google Scholar]
- Lou, HX; Yamazaki, Y; Sasaki, T; Uchida, M; Tanaka, H; Oka, S. A-type proanthocyanidins from peanut skins. Phytochemistry 1999, 51, 297–308. [Google Scholar]
- Monagas, M; Garrido, I; Lebron-Aguilar, R; Bartolome, B; Gomez-Cordoves, C. Almond (Prunus dulcis (Mill.) D.A. Webb) skins as a potential source of bioactive polyphenols. J. Agric. Food Chem 2007, 55, 8498–8507. [Google Scholar]
- Maatta-Riihinen, KR; Kahkonen, MP; Torronen, AR; Heinonen, IM. Catechins and procyanidins in berries of Vaccinium species and their antioxidant activity. J. Agric. Food Chem 2005, 53, 8485–8491. [Google Scholar]
- de Pascual-Teresa, S; Santos-Buelga, C; Rivas-Gonzalo, JC. Quantitative analysis of flavan-3-ols in Spanish foodstuffs and beverages. J. Agric. Food Chem 2000, 48, 5331–5337. [Google Scholar]
- Arts, IC; van de Putte, B; Hollman, PC. Catechin contents of foods commonly consumed in The Netherlands. 1. Fruits, vegetables, staple foods, and processed foods. J. Agric. Food Chem 2000, 48, 1746–1751. [Google Scholar]
- Heinonen, IM; Meyer, AS; Frankel, EN. Antioxidant activity of berry phenolics on human low-density lipoprotein and liposome oxidation. J. Agric. Food Chem 2000, 48, 1746–1751. [Google Scholar]
- Veberic, R; Colaric, M; Stampar, F. Phenolic acids and flavonoids of fig fruit (Ficus carica L.) in the northern Mediterranean region. Food Chem 2008, 106, 153–157. [Google Scholar]
- Tsanova-Savova, S; Ribarova, F; Gerova, M. (+)-Catechin and (−)-epicatechin in Bulgarian fruits. J. Food Compos. Anal 2005, 18, 691–698. [Google Scholar]
- Suzukia, T; Someyaa, S; Hua, F; Tanokura, M. Comparative study of catechin compositions in five Japanese persimmons (Diospyros kaki). Food Chem 2005, 93, 149–152. [Google Scholar]
- Pallauf, K; Rivas-Gonzalo, JC; del Castillo, MD; Cano, MP; de Pascual-Teresa, S. Characterization of the antioxidant composition of strawberry tree (Arbutus unedo L.) fruits. J. Food Compos. Anal 2008, 21, 273–281. [Google Scholar]
- Suarez-Valles, B; Santamaria-Victorero, J; Mangas-Alonso, JJ; Blanco-Gomis, D. High performance Liquid Chromatography of the neutral phenolic compounds of low molecular weight in apple juice. J. Agric. Food Chem 1994, 42, 2732–2736. [Google Scholar]
- Siriwoharn, T; Wrolstad, RE; Finn, CE; Pereira, CB. Influence of cultivar, maturity, and sampling on blackberry (Rubus L. Hybrids) anthocyanins, polyphenolics, and antioxidant properties. J. Agric. Food Chem 2004, 52, 8021–8030. [Google Scholar]
- Amiot, MJ; Tacchini, M; Aubert, SY; Oleszek, W. Influence of cultivar, maturity stage, and storage-conditions on phenolic composition and enzymatic browning of pear fruits. J. Agric. Food Chem 1995, 43, 1132–1137. [Google Scholar]
- Bourzeix, M; Weyland, D; Heredia, N; Desfeux, C. Étude des catéchines et des procyanidols de la grape de raisin du vin et d’ autres derivés de la vigne. Bull OIV 1986, 669–670, 1179–1254. [Google Scholar]
- Ricardo da Silva, JM; Rosec, JP; Bourzeix, M; Heredia, N. Separation and quantitative determination of grape and wine procyanidins by high performance reversed phase liquid chromatography. J. Agric. Food Chem 1990, 53, 85–92. [Google Scholar]
- Mendez, CDV; Forster, MP; Rodriguez-Delgado, MA; Rodriguez-Rodriguez, EM; Romero, CD. Content of free phenolic compounds in bananas from Tenerife (Canary Islands) and Ecuador. Eur. Food Res. Technol 2003, 217, 287–290. [Google Scholar]
- de Pascual-Teresa, S; Rivas-Gonzalo, JC; Santos-Buelga, C. Catechins intake in the Spanish diet. In COST 916 - Bioactive Compounds in Plant Foods Health Effects and Perspectives for the Food Industry; Amadò, R, Abt, B, Bravo, L, Goni, I, Saura-Calixto, F, Eds.; Office for Official Publications of the European Communities: Tenerife, Spain, 2002; pp. 317–318. [Google Scholar]
- Arts, IC; Hollman, PC; Feskens, EJ; Bueno de Mesquita, HB; Kromhout, D. Catechin intake and associated dietary and lifestyle factors in a representative sample of Dutch men and women. Eur. J. Clin. Nutr 2001, 55, 76–81. [Google Scholar]
- Manach, C; Williamson, G; Morand, C; Scalbert, A; Rémésy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr 2005, 81, 230S–242S. [Google Scholar]
- Stalmach, A; Troufflard, S; Serafini, M; Crozier, A. Absorption, metabolism and excretion of Choladi green tea flavan-3-ols by humans. Mol. Nutr. Food Res 2009, 53, S44–S53. [Google Scholar]
- van Amelsvoort, JM; van Hof, KH; Mathot, JN; Mulder, TP; Wiersma, A; Tijburg, LB. Plasma concentrations of individual tea catechins after a single oral dose in humans. Xenobiotica 2001, 31, 891–901. [Google Scholar]
- Holt, RR; Lazarus, SA; Sullards, MC; Zhu, QY; Schramm, DD; Hammerstone, JF; Fraga, CG; Schmitz, HH; Keen, CL. Procyanidin dimer B2 [epicatechin-(4β-8)-epicatechin] in human plasma after the consumption of a flavanol-rich cocoa. Am. J. Clin. Nutr 2002, 76, 798–804. [Google Scholar]
- Sano, A; Yamakoshi, J; Tokutake, S; Tobe, K; Kubota, Y; Kikuchi, M. Procyanidin B1 is detected in human serum after intake of proanthocyanidin-rich grape seed extract. Biosci. Biotechnol. Biochem 2003, 67, 1140–1143. [Google Scholar]
- Natsume, M; Osakabe, N; Oyama, M; Sasaki, M; Baba, S; Nakamura, Y; Osawa, T; Terao, J. Structures of (−)-epicatechin glucuronide identified from plasma and urine after oral ingestion of (−)-epicatechin: differences between human and rat. J. Free Radic. Biol. Med 2003, 34, 840–849. [Google Scholar]
- Gu, L; Kelm, MA; Hammerstone, JF; Beecher, G; Holden, J; Haytowitz, D; Gebhardt, S; Prior, RL. Concentrations of proanthocyanidins in common foods and estimations of normal consumption. J. Nutr 2004, 134, 613–617. [Google Scholar]
- Holt, RR; Lazarus, SA; Sullards, MC; Zhu, QY; Schramm, DD; Hammerstone, JF; Fraga, CG; Schmitz, HH; Keen, CL. Procyanidin dimer B2 [epicatechin-(4beta-8)-epicatechin] in human plasma after the consumption of a flavanol-rich cocoa. Am. J. Clin. Nutr 2002, 76, 798–804. [Google Scholar]
- Yamakoshi, J; Sano, A; Tokutake, S; Saito, M; Kikuchi, M; Kubota, Y; Kawachi, Y; Otsuka, F. Oral intake of proanthocyanidin-rich extract from grape seed improves Chloasma. Phytother. Res 2004, 18, 895–899. [Google Scholar]
- Urpi-Sarda, M; Monagas, M; Khana, N; Lamuela-Raventos, RM; Santos-Buelga, C; Sacanell, E; Castell, M; Permanyer, J; Andres-Lacueva, C. Epicatechin, procyanidins, and phenolic microbial metabolites after cocoa intake in humans and rats. Anal. Bioanal. Chem 2009, 394, 1545–1556. [Google Scholar]
- Shoji, T; Masumoto, S; Moriichi, N; Akiyama, H; Kanda, T; Ohtake, Y; Goda, Y. Apple procyanidin oligomers absorption in rats after oral administration: analysis of procyanidins in plasma using the porter method and high-performance liquid chromatography/tandem mass spectrometry. J. Agric. Food Chem 2006, 54, 884–892. [Google Scholar]
- Appeldoorn, MM; Vincken, JP; Gruppen, H; Hollman, PCH. Procyanidin Dimers A1, A2, and B2 Are Absorbed without Conjugation or Methylation from the Small Intestine of Rats. J. Nutr 2009, 139, 1469–1473. [Google Scholar]
- Li, C; Lee, MJ; Sheng, S; Meng, X; Prabhu, S; Winnik, B; Huang, B; Chung, JY; Yan, S; Ho, CT; Yang, CS. Structural identification of two metabolites of catechins and their kinetics in human urine and blood after tea ingestion. Chem. Res. Toxicol 2000, 13, 177–184. [Google Scholar]
- Déprez, S; Brezillon, C; Rabot, S; Philippe, C; Mila, I; Lapierre, C; Scalbert, A. Polymeric proanthocyanidins are catabolized by human colonic microflora into low-molecular-weight phenolic acids. J. Nutr 2000, 130, 2733–2738. [Google Scholar]
- Rios, LY; Gonthier, MP; Rémésy, C; Mila, I; Lapierre, C; Lazarus, SA; Williamson, G; Scalbert, A. Chocolate intake increases urinary excretion of polyphenol-derived phenolic acids in healthy human subjects. Am. J. Clin. Nutr 2003, 77, 912–918. [Google Scholar]
- Rechner, AR; Smith, MA; Kuhnle, G; Gibson, GR; Debnam, ES; Srai, SKS; Moore, KP; Rice-Evans, CA. Colonic metabolism of dietary polyphenol: influence of structure on microbial fermentation products. J. Free Radic. Biol. Med 2004, 36, 212–225. [Google Scholar]
- Meselhy, MR; Nakamura, N; Hattori, M. Biotransformation of (−)-epicatechin-3-O-gallate by human intestinal bacteria. Chem. Pharm. Bull 1997, 45, 888–893. [Google Scholar]
- Gonthier, MP; Cheynier, V; Donovan, JL; Manach, C; Morand, C; Mila, I; Lapierre, C; Rémésy, C; Scalbert, A. Microbial aromatic acid metabolites formed in the gut account for a major fraction of the polyphenols excreted in urine of rats fed red wine polyphenols. J. Nutr 2003, 133, 461–467. [Google Scholar]
- Groenewoud, G; Hundt, HKL. The microbial metabolism of condensed (+)-catechins bt rat-cecal microflora. Xenobiotica 1986, 16, 99–107. [Google Scholar]
- van’t Slot, G; Humpf, HU. Degradation and metabolism of catechin, epigallocatehin-3-gallate (EGCG), and related compounds by the intestinal microbiota in the pig cecum model. J. Agric. Food Chem 2009, 57, 8041–8048. [Google Scholar]
- de Pascual-Teresa, S; Sanchez-Ballesta, MT. Anthocyanins: from plant to health. Phytochem. Rev 2008, 7, 281–299. [Google Scholar]
- Clifford, MN. Anthocyanins-nature, occurrence and dietary burden. J. Sci. Food Agric 2000, 80, 1063–1072. [Google Scholar]
- Koponen, JM; Happonen, AM; Mattila, PH; Torronen, AR. Contents of anthocyanins and ellagitannins in selected foods consumed in Finland. J. Agric. Food Chem 2007, 55, 1612–1619. [Google Scholar]
- Wojdylo, A; Oszmianski, J; Laskowski, P. Polyphenolic compounds and antioxidant activity of new and old apple varieties. J. Agric. Food Chem 2008, 56, 6520–6530. [Google Scholar]
- Mazza, G; Miniati, E. Anthocyanins in Fruits, Vegetables, and Grains; CRC Press: Boca Raton, FL, USA, 1993. [Google Scholar]
- Macz-Pop, GA; Rivas-Gonzalo, JC; Perez-Alonso, J; Gonzalez-Paramas, AM. Natural occurrence of free anthocyanin aglycones in beans (Phaseolus vulgaris L.). Food Chem 2006, 94, 448–456. [Google Scholar]
- Timberlake, CF. The biological properties of anthocyanin compounds. NATCOL Quart. Bull 1988, 1, 4–15. [Google Scholar]
- Wu, XL; Beecher, GR; Holden, JM; Haytowitz, DB; Gebhardt, SE; Prior, RL. Concentrations of anthocyanins in common foods in the United States and estimation of normal consumption. J. Agric. Food Chem 2006, 54, 4069–4075. [Google Scholar]
- Ryu, SN; Park, SZ; Ho, CT. High performance liquid chromatographic determination of anthocyanin pigments in some varieties of black rice. J. Food Drug Anal 1998, 6, 729–736. [Google Scholar]
- Torre, LC; Barritt, BH. Quantitative evaluation of Rubus fruit anthocyanin pigments. J. Food Sci 1977, 42, 488–490. [Google Scholar]
- Wang, SY; Lin, HS. Antioxidant activity in fruits and leaves of blackberry, raspberry, and strawberry varies with cultivar and developmental stage. J. Agric. Food Chem 2000, 48, 140–146. [Google Scholar]
- Prior, RL; Cao, G; Martin, A; Sofic, E; McEwen, J; O’Brien, C; Lischner, N; Ehlenfeldt, M; Kalt, W; Krewer, G; Mainland, CM. Antioxidant Capacity As Influenced by Total Phenolic and Anthocyanin Content, Maturity, and Variety of Vaccinium Species. J. Agric. Food Chem 1998, 46, 2686–2693. [Google Scholar]
- Wang, SY; Chen, CT; Sciarappa, W; Wang, CY; Camp, MJ. Fruit quality, antioxidant capacity, and flavonoid content of organically and conventionally grown blueberries. J. Agric. Food Chem 2008, 56, 5788–5794. [Google Scholar]
- Gao, L; Mazza, G. Characterization, quantification and distribution of anthocyanins and colourless phenolics in sweet cherry. J. Agric. Food Chem 1995, 43, 343–346. [Google Scholar]
- Kaack, K; Austed, T. Interaction of vitamin C and flavonoids in elderberry (Sambucus nigra L.) during juice processing. Plant Food. Hum. Nutr 1998, 52, 187–198. [Google Scholar]
- Kosar, M; Altintas, A; Kirimer, N; Baser, KHC. Determination of the free radical scavenging activity of Lycium extracts. Chem. Nat. Compd 2003, 39, 531–535. [Google Scholar]
- Pantelidis, GE; Vasilakakis, M; Manganaris, GA; Diamantidis, G. Antioxidant capacity, phenol, anthocyanin and ascorbic acid contents in raspberries, blackberries, red currants, gooseberries and cornelian cherries. Food Chem 2007, 102, 777–783. [Google Scholar]
- Alighourchi, H; Barzegar, M; Abbasi, S. Anthocyanins characterization of 15 Iranian pomegranate (Punica granatum L.) varieties and their variation after cold storage and pasteurization. Eur. Food Res. Technol 2008, 227, 881–887. [Google Scholar]
- Cevallos-Casals, BA; Cisneros-Zevallos, L. Stoichiometric and kinetic studies of phenolic antioxidants from Andean purple corn and red-fleshed sweetpotato. J. Agric. Food Chem 2003, 51, 3313–3319. [Google Scholar]
- Lamikanra, O. Anthocyanins of Vitis rotundifolia hybrid grapes. Food Chem 1989, 33, 225–237. [Google Scholar]
- Ferreres, F; Gil, MI; Tomas-Barberan, FA. Anthocyanins and flavonoids from shredded red onion and changes during storage in perforated films. Food Res. Int 1996, 29, 389–395. [Google Scholar]
- Giusti, MM; Wrolstad, RE. Characterization of red radish anthocyanins. J. Food Sci 1996, 61, 322–326. [Google Scholar]
- Teissedre, PL; Landrault, T. Wine phenolics: contribution to dietary intake and bioavailability. Food Res. Int 2000, 33, 461–467. [Google Scholar]
- Frankel, EN; Waterhouse, AL; Teissedre, PL. Principal phenolic phytochemicals in selected California wines and their antioxidant activity in inhibiting oxidation of human low-density lipoproteins. J. Agric. Food Chem 1995, 43, 890–894. [Google Scholar]
- Sanchez-Moreno, C; Cao, GH; Ou, BX; Prior, RL. Anthocyanin and proanthocyanidin content in selected white and red wines. Oxygen radical absorbance capacity comparison with nontraditional wines obtained from highbush blueberry. J. Agric. Food Chem 2003, 51, 4889–4896. [Google Scholar]
- Lopes-da-Silva, F; de Pascual-Teresa, S; Rivas-Gonzalo, J; Santos-Buelga, C. Identification of anthocyanin pigments in strawberry (cv Camarosa) by LC using DAD and ESI-MS detection. Eur. Food Res. Technol 2002, 214, 248–253. [Google Scholar]
- Tomás-Barberán, FA; Gil-Izquierdo, A; Moreno, DA. Bioavailability and metabolism of phenolic compounds and glucosinolates. In Designing Functional Foods: Understanding, Measuring and Controlling Food Structure Breakdown and Nutrient Absorption for the Development of Health-Promoting Foods Part 3 Digestion and Adsorption of Food Components; McClemments, DJ, Decker, EA, Eds.; Woodhead Publishing Ltd/CRC Press: Boca Raton, FL, USA, 2009; Chapter 19, 194–221. [Google Scholar]
- Escribano-Bailon, MT; Santos-Buelga, C; Rivas-Gonzalo, JC. Anthocyanins in cereals. J. Chromatogr. A 2004, 1054, 129–141. [Google Scholar]
- Kuhnau, J. The flavonoids. A class of semi-essential food components: Their role in human nutrition. World Rev. Nutr. Dietet 1976, 24, 117–191. [Google Scholar]
- Chun, OK; Chung, SJ; Song, WO. Estimated dietary flavonoid intake and major food sources of U.S. adults. J. Nutr 2007, 137, 1244–1252. [Google Scholar]
- Lapidot, T; Harel, S; Grani, R; Kannaer, J. Bioavailability of red wine anthocyanins as detected in human urine. J. Agric. Food Chem 1998, 46, 4297–4302. [Google Scholar]
- Felgines, C; Talavera, S; Gonthier, MP; Texier, O; Scalbert, A; Lamaison, JL; Rémésy, C. Strawberry anthocyanins are recovered in urine as glucuro- and sulfoconjugates in humans. J. Nutr 2003, 133, 1296–1301. [Google Scholar]
- Galvano, F; La Fauci, L; Lazzarino, G; Gloginao, V; Triieni, A; Ciappellano, S; Battistini, NC; Tavazzi, B; Galvano, G. Cyanidins: metabolism and biological properties. J. Nutr. Biochem 2004, 15, 2–11. [Google Scholar]
- Galvano, F; La Fauci, L; Vitaglione, P; Fogliano, V; Vanella, L; Felgines, C. Bioavailability, antioxidant and biological properties of the natuarl free-radical scavengers cyanidin and related glycosides. Ann. Ist. Sup. Sanitá 2007, 43, 382–393. [Google Scholar]
- Felgines, C; Talavera, S; Texier, O; Gil-Izquierdo, A; Lamaison, JL; Rémésy, C. Blackberry anthocyanins are mainly recovered in urine as methylated and glucuronidated conjugates in humans. J. Agric. Food Chem 2005, 53, 7721–7727. [Google Scholar]
- Felgines, C; Texier, O; Besson, C; Lyan, B; Lamaison, JL; Scalbert, A. Strawberry pelargonidin glycosides are excreted in urine as intact glycosides and glucuronidated pelargonidin derivatives in rats. Br. J. Nutr 2007, 98, 1126–1131. [Google Scholar]
- Talavera, S; Felgines, C; Texier, O; Besson, C; Lamaison, JL; Rémésy, C. Anthocyanins are efficiently absorbed from the stomach in anesthetized rats. J. Nutr 2003, 133, 4178–4182. [Google Scholar]
- Kay, CD; Kroon, PA; Cassidy, A. The bioactivity of dietary anthocyanins is likely to be mediated by their degradation products. Mol. Nutr. Food Res 2009, 53, S92–S101. [Google Scholar]
- Tian, Q; Giusti, MM; Stoner, GD; Schwartz, SJ. Urinary excretion of black raspberry (Rubus occidentalis) anthocyanins and their metabolites. J. Agric. Food Chem 2006, 54, 1467–1472. [Google Scholar]
- Bitsch, R; Netzel, M; Sonntag, S; Strass, G; Frank, T; Bitsch, I. Urinary excretion of cyanidin clucosides and glucuronides in healthy humans after elderberry juice ingestion. J. Biomed. Biotechnol 2004, 4, 343–345. [Google Scholar]
- Wu, X; Cao, G; Prior, RL. Absorption and metabolism of anthocyanins in elderly women after consumption of elderberry or blueberry. J. Nutr 2002, 132, 1865–1871. [Google Scholar]
- Talavera, S; Felgines, C; Texier, O; Besson, C; Gil-Izquierdo, A; Lamaison, JL; Rémésy, C. Anthocyanin metabolism in rats and their distribution to digestive area, kidney, and brain. J. Agric. Food Chem 2005, 23, 3902–3908. [Google Scholar]
- Andrés-Lacueva, C; Shukitt-Hale, B; Galli, RL; Jauregui, O; Lamuela-Raventós, RM; Joseph, JA. Anthocyanins in aged blueberry-fed rats are found centrally and may enhance memory. Nutr. Neurosci 2005, 8, 111–120. [Google Scholar]
- Passamonti, S; Vrhovsek, U; Vanzo, A; Mattivi, F. Fast access of some grape pigments to the brain. J. Agric. Food Chem 2005, 53, 7029–7034. [Google Scholar]
- Keppler, K; Humpf, HU. Metabolism of anthocyanis and their phenolic degradation products by the intestinal microflora. Bioorg. Med. Chem 2005, 13, 5195–5205. [Google Scholar]
- Aura, AM; Martin-Lopez, P; O’Leary, KA; Williamson, G; Oksman-Caldentey, KM; Poutanen, K; Santos-Buelga, C. In vitro metabolism of anthocyanins by human gut microflora. Eur. J. Nutr 2005, 44, 133–142. [Google Scholar]
- Selma, MV; Espin, JC; Tomas-Barberan, FA. Interaction between phenolics and gut microbiota: role in human health. J. Agric. Food Chem 2009, 57, 6485–6501. [Google Scholar]
- Avila, M; Hidalgo, M; Sanchez-Moreno, C; Pelaez, C; Requena, T; de Pascual-Teresa, S. Bioconversion of anthocyanin glycosides by Bifidobacteria and Lactobacillus. Food Res. Int 2009, 42, 1453–1461. [Google Scholar]
- McGhie, TK; Walton, MC. The bioavailability and absorption of anthocyanins: towards a better understanding. Mol. Nutr. Food Res 2007, 51, 702–713. [Google Scholar]
- Demrow, HS; Slane, PR; Folts, JD. Administration of wine and grape juice inhibits in vivo platelet activity and thrombosis in stenosed canine coronary arteries. Circulation 1995, 91, 1182–1188. [Google Scholar]
- Yamakuchi, M; Bao, C; Ferlito, M; Lowenstein, CJ. Epigallocatechin gallate inhibits endothelial exocytosis. Biol. Chem 2008, 389, 935–941. [Google Scholar]
- Lin, YL; Lin, JK. (−)-Epigallocatechin-3-gallate blocks the induction of nitric oxide synthase by down-regulating lipopolysaccharide-induced activity of transcription factor nuclear factor-kappaB. Mol. Pharmacol 1997, 52, 465–472. [Google Scholar]
- Danesi, F; Di Nunzio, M; Boschetti, E; Bordoni, A. Green tea extract selectively activates peroxisome proliferator-activated receptor beta/delta in cultured cardiomyocytes. Br. J. Nutr 2009, 101, 1736–1739. [Google Scholar]
- Hsu, SP; Wu, MS; Yang, CC; Huang, KC; Liou, SY; Hsu, SM; Chien, CT. Chronic green tea extract supplementation reduces hemodialysis-enhanced production of hydrogen peroxide and hypochlorous acid, atherosclerotic factors, and proinflammatory cytokines. Am. J. Clin. Nutr 2007, 86, 539–547. [Google Scholar]
- Fisher, ND; Hughes, M; Gerhard-Herman, M; Hollenberg, NK. Flavanol-rich cocoa induces nitric-oxide-dependent vasodilation in healthy humans. J. Hypertens 2003, 21, 2281–2286. [Google Scholar]
- Lorenz, M; Hellige, N; Rieder, P; Kinkel, HT; Trimpert, C; Staudt, A; Felix, SB; Baumann, G; Stangl, K; Stangl, V. Positive inotropic effects of epigallocatechin-3-gallate (EGCG) involve activation of Na+/H+ and Na+/Ca2+ exchangers. Eur. J. Heart Fail 2008, 10, 439–445. [Google Scholar]
- Sheng, R; Gu, ZL; Xie, ML; Zhou, WX; Guo, CY. EGCG inhibits cardiomyocyte apoptosis in pressure overload-induced cardiac hypertrophy and protects cardiomyocytes from oxidative stress in rats. Acta Pharmacol. Sinica 2007, 28, 191–201. [Google Scholar]
- Rimbach, G; Melchin, M; Moehring, J; Wagner, AE. Polyphenols from cocoa and vascular health-a critical review. Int. J. Mol. Sci 2009, 10, 4290–4309. [Google Scholar]
- Matsuyama, T; Tanaka, Y; Kamimaki, I; Nagao, T; Tokimitsu, I. Catechin safely improved higher levels of fatness, blood pressure, and cholesterol in children. Obesity (Silver Spring Md.) 2008, 16, 1338–1348. [Google Scholar]
- Brown, AL; Lane, J; Coverly, J; Stocks, J; Jackson, S; Stephen, A; Bluck, L; Coward, A; Hendrickx, H. Effects of dietary supplementation with the green tea polyphenol epigallocatechin-3-gallate on insulin resistance and associated metabolic risk factors: randomized controlled trial. Br. J. Nutr 2009, 101, 886–894. [Google Scholar]
- Velayutham, P; Babu, A; Liu, D. Green Tea Catechins and Cardiovascular Health: An Update. Curr Med Chem 2008, 15, 1840–1850. [Google Scholar]
- Tinahones, FJ; Rubio, MA; Garrido-Sánchez, L; Ruiz, C; Gordillo, E; Cabrerizo, L; Cardona, F. Green tea reduces LDL oxidability and improves vascular function. J. Am. Coll. Nutr 2008, 27, 209–213. [Google Scholar]
- Mursu, J; Voutilainen, S; Nurmi, T; Tuomainen, TP; Kurl, S; Salonen, JT. Flavonoid intake and the risk of ischaemic stroke and CVD mortality in middle-aged Finish men: the Kuopio Ischaemic Heat Disease Risk Factor Study. Br. J. Nutr 2008, 100, 890–895. [Google Scholar]
- Hooper, L; Kroon, PA; Rimm, EB; Cohn, JS; Harvey, I; Le Cornu, KA; Ryder, JJ; Hall, WL; Cassidy, A. Flavonoids, flavonoid-rich foods, and cardiovascular risk: A meta-analysis of randomized controlled trials. Am. J. Clin. Nutr 2008, 88, 38–50. [Google Scholar]
- Frank, J; George, TW; Lodge, JK; Rodriguez-Mateos, AM; Spencer, JP; Minihane, AM; Rimbach, G. Daily consumption of an aqueous green tea extract supplement does not impair liver function or alter cardiovascular disease risk biomarkers in healthy men. J. Nutr 2009, 139, 58–62. [Google Scholar]
- Rein, D; Paglieroni, TG; Wun, T; Pearson, DA; Schmitz, HH; Gosselin, R; Keen, CL. Cocoa inhibits platelet activation and function. Am. J. Clin. Nutr 2000, 72, 30–35. [Google Scholar]
- Vitseva, O; Varghese, S; Chakrabarti, S; Folts, JD; Freedman, JE. Grape seed and skin extracts inhibit platelet function and release of reactive oxygen intermediates. J. Cardiovasc. Pharmacol 2005, 46, 445–451. [Google Scholar]
- Garcia-Conesa, MT; Tribolo, S; Guyot, S; Tomas-Barberan, FA; Kroon, PA. Oligomeric procyanidins inhibit cell migration and modulate the expression of migration and proliferation associated genes in human umbilical vascular endothelial cells. Mol. Nutr. Food Res 2009, 53, 266–276. [Google Scholar]
- DalBo, S; Goulart, S; Horst, H; Pizzolatti, MG; Ribeiro-do-Valle, RM. Activation of endothelial nitric oxide synthase by proanthocyanidin-rich fraction from Croton celtidifolius (Euphorbiaceae): involvement of extracellular calcium influx in rat thoracic aorta. J. Pharmacol. Sci 2008, 107, 181–189. [Google Scholar]
- Terra, X; Montagut, G; Bustos, M; Llopiz, N; Ardèvol, A; Bladé, C; Fernández-Larrea, J; Pujadas, G; Salvadó, J; Arola, L; Blay, M. Grape-seed procyanidins prevent low-grade inflammation by modulating cytokine expression in rats fed a high-fat diet. J. Nutr. Biochem 2009, 20, 210–218. [Google Scholar]
- Del Bas, JM; Fernández-Larrea, J; Blay, M; Ardèvol, A; Salvado, MJ; Arola, L; Blade, C. Grape seed procyanidins improve atherosclerotic risk index and induce liver CYP7A1 and SHP expression in healthy rats. FASEB J 2005, 19, 479–481. [Google Scholar]
- Del Bas, JM; Ricketts, ML; Baiges, I; Quesada, H; Ardevol, A; Salvado, MJ; Pujadas, G; Blay, M; Arola, L; Blade, C; Moore, DD; Fernandez-Larrea, J. Dietary procyanidins lower triglyceride levels signaling through the nuclear receptor small heterodimer partner. Mol. Nutr. Food Res 2008, 52, 1172–1181. [Google Scholar]
- Baba, S; Natsume, M; Yasuda, A; Nakamura, Y; Tamura, T; Osakabe, N; Kanegae, M; Kondo, K. Plasma LDL and HDL cholesterol and oxidized LDL concentrations are altered in normo- and hypercholesterolemic humans after intake of different levels of cocoa powder. J. Nutr 2007, 137, 1436–1441. [Google Scholar]
- Monagas, M; Khan, N; Andres-Lacueva, C; Casas, R; Urpi-Sarda, M; Llorach, R; Lamuela-Raventos, RM; Estruch, R. Effect of cocoa powder on the modulation of inflammatory biomarkers in patients at high risk of cardiovascular disease. Am. J. Clin. Nutr 2009, 90, 1144–1150. [Google Scholar]
- Kenny, TP; Keen, CL; Schmitz, HH; Gershwin, ME. Immune effects of cocoa procyanidin oligomers on peripheral blood mononuclear cells. Exp. Biol. Med 2007, 232, 293–300. [Google Scholar]
- Du, Y; Lou, H. Catechin and proanthocyanidin B4 from grape seeds prevent doxorubicin-induced toxicity in cardiomyocytes. Eur. J. Pharmacol 2008, 591, 96–101. [Google Scholar]
- Rechner, AR; Kroner, C. Anthocyanins and colonic metabolites of dietary polyphenols inhibit platelet function. Thromb. Res 2005, 116, 327–334. [Google Scholar]
- Garcia-Alonso, M; Minihane, AM; Rimbach, G; Rivas-Gonzalo, JC; de Pascual-Teresa, S. Red wine anthocyanins are rapidly absorbed in humans and affect monocyte chemoattractant protein 1 levels and antioxidant capacity of plasma. J. Nutr. Biochem 2009, 20, 521–529. [Google Scholar]
- Oak, MH; Bedoui, JE; Madeira, SV; Chalupsky, K; Schini-Kerth, VB. Delphinidin and cyanidin inhibit PDGF(AB)-induced VEGF release in vascular smooth muscle cells by preventing activation of p38 MAPK and JNK. Br. J. Pharmacol 2006, 149, 283–290. [Google Scholar]
- Bell, DR; Gochenaur, K. Direct vasoactive and vasoprotective properties of anthocyanin-rich extracts. J. Appl. Physiol 2006, 100, 1164–1170. [Google Scholar]
- Toufektsian, MC; de Lorgeril, M; Nagy, N; Salen, P; Donati, MB; Giordano, L; Mock, HP; Peterek, S; Matros, A; Petroni, K; Pilu, R; Rotilio, D; Tonelli, C; de Leiris, J; Boucher, F; Martin, C. Chronic dietary intake of plant-derived anthocyanins protects the rat heart against ischemia-reperfusion injury. J. Nutr 2008, 138, 747–752. [Google Scholar]
- Palfi, A; Bartha, E; Copf, L; Mark, L; Gallyas, F, Jr; Veres, B; Kalman, E; Pajor, L; Toth, K; Ohmacht, R; Sumegi, B. Alcohol-free red wine inhibits isoproterenol-induced cardiac remodeling in rats by the regulation of Akt1 and protein kinase C alpha/beta II. J. Nutr. Biochem 2009, 20, 418–425. [Google Scholar]
- Xia, M; Ling, W; Zhu, H; Wang, Q; Ma, J; Hou, M; Tang, Z; Li, L; Ye, Q. Anthocyanin prevents CD40-activated proinflammatory signaling in endothelial cells by regulating cholesterol distribution. Arterioscler. Thromb. Vasc. Biol 2007, 27, 519–524. [Google Scholar]
- Hämäläinen, M; Nieminen, R; Vuorela, P; Heinonen, M; Moilanen, E. Anti-inflammatory effects of flavonoids: genistein, kaempferol, quercetin, and daidzein inhibit STAT-1 and NF-kappaB activations, whereas flavone, isorhamnetin, naringenin, and pelargonidin inhibit only NF-kappaB activation along with their inhibitory effect on iNOS expression and NO production in activated macrophages. Mediat Inflamm 2007, 2007. [Google Scholar]
- Pergola, C; Rossi, A; Dugo, P; Cuzzocrea, S; Sautebin, L. Inhibition of nitric oxide biosynthesis by anthocyanin fraction of blackberry extract. Nitric Oxide 2006, 15, 30–39. [Google Scholar] [Green Version]
- Scarabelli, TM; Mariotto, S; Abdel-Azeim, S; Shoji, K; Darra, E; Stephanou, A; Chen-Scarabelli, C; Marechal, JD; Knight, R; Ciampa, A; Saravolatz, L; de Prati, AC; Yuan, Z; Cavalieri, E; Menegazzi, M; Latchman, D; Pizza, C; Perahia, D; Suzuki, H. Targeting STAT1 by myricetin and delphinidin provides efficient protection of the heart from ischemia/reperfusion-induced injury. FEBS Lett 2009, 583, 531–541. [Google Scholar]
- Curtis, PJ; Kroon, PA; Hollands, WJ; Walls, R; Jenkins, G; Kay, CD; Cassidy, A. Cardiovascular disease risk biomarkers and liver and kidney function are not altered in postmenopausal women after ingesting an elderberry extract rich in anthocyanins for 12 weeks. J. Nutr 2009, 139, 2266–2271. [Google Scholar]
| Fruit | Flavanols (mg/100 g fresh weight) | References |
|---|---|---|
| Apple | 0.1–45 | [13,14,17,20] |
| Apricot | 0.3–11 | [13,14] |
| Avocado | 0.1–0.6 | [13,14] |
| Banana | 0.1–10.3 | [13,25] |
| Black currant | 1.2 | [14] |
| Blackberry | 3.3–23.8 | [13–15,17] |
| Blueberry | 1–7 | [13–15] |
| Cherry | 6.3–23 | [13–15,17] |
| Custard apple | 18–25 | [13] |
| Fig | 0.1–4.8 | [13,16,17] |
| Grape | 0.1–20 | [13–15,23,24] |
| Kiwi | 0.3–0.8 | [13,14] |
| Loquat fruit | 2.5–2.9 | [13] |
| Mango | 1.7 | [13] |
| Peach | 2–17 | [13,14] |
| Pear | 0.4–12 | [13,14,22] |
| Persimmon | 0.4–1.7 | [13,18] |
| Plum | 3.7–79 | [13,14,17] |
| Pomegranate | 0.8–1.2 | [13] |
| Quince | 3–7 | [13,14] |
| Raspberry | 2–48 | [13–15,17] |
| Red currant | 2–7 | [13,14] |
| Strawberry | 2–6 | [13–15,17] |
| Strawberry tree fruit | 10–29 | [13,19] |
| Food | Content | Unit * | References |
|---|---|---|---|
| Apple | 0.0–60.0 | mg per 100 g f.w. | [50,51] |
| Bilberry | 300–698 | mg per 100 g f.w. | [50,52] |
| Black bean | 24.1–44.5 | mg per 100 g f.w. | [53] |
| Black currant | 130–476 | mg per 100 g f.w. | [54,55] |
| Black olives | 42–228 | mg per 100 g f.w. | [52] |
| Black rice | 10–493 | mg per 100 g f.w. | [56] |
| Blackberry | 82.5–325.9 | mg per 100 g f.w. | [57,58] |
| Blueberry | 61.8–299.6 | mg per 100 g f.w. | [59,60] |
| Bog whortleberry | 154 | mg per 100 g f.w. | [50] |
| Cherry | 2–450 | mg per 100 g f.w. | [52,61] |
| Chokeberry | 410–1480 | mg per 100 g f.w. | [50,55] |
| Cranberry | 67–140 | mg per 100 g f.w. | [50,55] |
| Crowberry | 360 | mg per 100 g f.w. | [59] |
| Eggplant | 8–85 | mg per 100 g f.w. | [50,55] |
| Elderberry | 664–1816 | mg per 100 g f.w. | [62] |
| Goji | 49.4 | mg per 100 g f.w. | [63] |
| Gooseberry | 2.0–43.3 | mg per 100 g f.w. | [55,64] |
| Grapefruit | 5.9 | mg per 100 g f.w. | [50] |
| Lettuce | 2.5–5.2 | mg per 100 g f.w. | [50,55] |
| Nectarine | 2.4 | mg per 100 g f.w. | [50] |
| Peach | 4.2 | mg per 100 g f.w. | [50] |
| Pear | 5–10 | mg per 100 g f.w. | [52] |
| Plum | 2–25 | mg per 100 g f.w. | [55] |
| Pomegranate, unprocessed juice | 15–252 | mg per L | [65] |
| Purple corn | 1642 | mg per 100 g f.w. | [66] |
| Raspberry | 20–687 | mg per 100 g f.w. | [55,58] |
| Red cabbage | 322 | mg per 100 g f.w. | [55] |
| Red currant | 22 | mg per 100 g f.w. | [50] |
| Red grape | 30–750 | mg per 100 g f.w. | [67] |
| Red onion, processed | 23.3–48.5 | mg per 100 g f.w. | [55,68] |
| Red radish | 100–154 | mg per 100 g f.w. | [55,69] |
| Red wine | 16.4–35 | mg per 100 mL | [70–72] |
| Rhubarb | 4–200 | mg per 100 g f.w. | [50] |
| Rowanberry | 14 | mg per 100 g f.w. | [50] |
| Sakatoon berry | 234 | mg per 100 g f.w. | [50] |
| Strawberry | 19–55 | mg per 100 g f.w. | [73] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
De Pascual-Teresa, S.; Moreno, D.A.; García-Viguera, C. Flavanols and Anthocyanins in Cardiovascular Health: A Review of Current Evidence. Int. J. Mol. Sci. 2010, 11, 1679-1703. https://doi.org/10.3390/ijms11041679
De Pascual-Teresa S, Moreno DA, García-Viguera C. Flavanols and Anthocyanins in Cardiovascular Health: A Review of Current Evidence. International Journal of Molecular Sciences. 2010; 11(4):1679-1703. https://doi.org/10.3390/ijms11041679
Chicago/Turabian StyleDe Pascual-Teresa, Sonia, Diego A. Moreno, and Cristina García-Viguera. 2010. "Flavanols and Anthocyanins in Cardiovascular Health: A Review of Current Evidence" International Journal of Molecular Sciences 11, no. 4: 1679-1703. https://doi.org/10.3390/ijms11041679
APA StyleDe Pascual-Teresa, S., Moreno, D. A., & García-Viguera, C. (2010). Flavanols and Anthocyanins in Cardiovascular Health: A Review of Current Evidence. International Journal of Molecular Sciences, 11(4), 1679-1703. https://doi.org/10.3390/ijms11041679







