Bioavailability of the Polyphenols: Status and Controversies
Abstract
:1. Introduction
2. Distribution and Food Content
3. Main Factors Affecting the Bioavailability of the Polyphenols
3.1. External Factors
3.2. Food Processing Related Factors
3.3. Food Related Factors
3.4. Interaction with Other Compounds
3.5. Chemical Structure
3.6. Host Related Factors
4. Limits and Difficulties of the Studies on the Bioavailability of the Polyphenols
5. Conclusions
- The potential biological activity of the metabolites of many dietary polyphenols needs to be better investigated. In fact, the identification and the quantification of metabolites currently represents an important and growing field of research.
- Strategies to improve the bioavailability of the polyphenols need to be developed. Moreover it is necessary to determine whether these methods translate into increased biological activity.
- Whereas in vitro studies shed light on the mechanisms of action of individual dietary polyphenols, these findings need to be supported by in vivo experiments. The health benefits of dietary polyphenols must be demonstrated in appropriate animal models of disease and in humans at appropriate doses.
References and Notes
- Hertog, MG; Kromhout, D; Aravanis, C; Blackburn, H; Buzina, R; Fidanza, F; Giampaoli, S; Jansen, A; Menotti, A; Nedeljkovic, S; Pekkarinen, M; Simic, BS; Toshima, H; Feskens, EJM; Hollman, PCH; Katan, MB. Flavonoid intake and long-term risk of coronary heart disease and cancer in the seven countries study. Arch Int. Med 1995, 155, 381–386. [Google Scholar]
- Arts, IC; Hollman, PC. Polyphenols and disease risk in epidemiologic studies. Am. J. Clin. Nutr 2005, 81, 317S–325S. [Google Scholar]
- Hirvonen, T; Virtamo, J; Korhonen, P; Albanes, D; Pietinen, P. Flavonol and flavone intake and the risk of cancer in male smokers (Finland). Cancer Causes Control 2001, 12, 789–796. [Google Scholar]
- Lambert, JD; Hong, J; Yang, GY; Liao, J; Yang, CS. Inhibition of carcinogenesis by polyphenols: Evidence from laboratory investigations. Am. J. Clin Nutr 2005, 81, 284S–291S. [Google Scholar]
- Williamson, G; Manach, C. Bioavailability and bioefficacy of polyphenols in humans. II. Review of 93 intervention studies. Am. J. Clin Nutr 2005, 81, 243S–255S. [Google Scholar]
- Williams, C. Healthy eating: Clarifying advice about fruit and vegetables. Bmj 1995, 310, 1453–1455. [Google Scholar]
- Dietary Guidelines for Americans 2005; US Department of Agriculture (USDA): Washington, DC, USA, 2005.
- Masella, R; Di Benedetto, R; Vari, R; Filesi, C; Giovannini, C. Novel mechanisms of natural antioxidant compounds in biological systems: Involvement of glutathione and glutathione-related enzymes. J. Nutr. Biochem 2005, 16, 577–586. [Google Scholar]
- Halliwell, B; Rafter, J; Jenner, A. Health promotion by flavonoids, tocopherols, tocotrienols, and other phenols: Direct or indirect effects? Antioxidant or not? Am. J. Clin Nutr 2005, 81, 268S–276S. [Google Scholar]
- Romier, B; Schneider, YJ; Larondelle, Y; During, A. Dietary polyphenols can modulate the intestinal inflammatory response. Nutr. Rev 2009, 67, 363–378. [Google Scholar]
- D'Archivio, M; Santangelo, C; Scazzocchio, B; Vari, R; Filesi, C; Masella, R; Giovannini, C. Modulatory effects of polyphenols on apoptosis induction: Relevance for cancer prevention. Int. J. Mol. Sci 2008, 9, 213–228. [Google Scholar]
- Noratto, G; Porter, W; Byrne, D; Cisneros-Zevallos, L. Identifying peach and plum polyphenols with chemopreventive potential against estrogen-independent breast cancer cells. J. Agric. Food Chem 2009, 57, 5219–5126. [Google Scholar]
- Leifert, WR; Abeywardena, MY. Grape seed and red wine polyphenol extracts inhibit cellular cholesterol uptake, cell proliferation, and 5-lipoxygenase activity. Nutr. Res 2008, 28, 842–850. [Google Scholar]
- Naasani, I; Oh-Hashi, F; Oh-Hara, T; Feng, WY; Johnston, J; Chan, K; Tsuruo, T. Blocking telomerase by dietary polyphenols is a major mechanism for limiting the growth of human cancer cells in vitro and in vivo. Cancer Res 2003, 63, 824–830. [Google Scholar]
- Hussain, T; Gupta, S; Adhami, VM; Mukhtar, H. Green tea constituent epigallocatechin-3-gallate selectively inhibits COX-2 without affecting COX-1 expression in human prostate carcinoma cells. Int. J. Cancer 2005, 113, 660–669. [Google Scholar]
- O'Leary, KA; de Pascual-Tereasa, S; Needs, PW; Bao, YP; O'Brien, NM; Williamson, G. Effect of flavonoids and vitamin E on cyclooxygenase-2 (COX-2) transcription. Mutat. Res 2004, 551, 245–254. [Google Scholar]
- Sadik, CD; Sies, H; Schewe, T. Inhibition of 15-lipoxygenases by flavonoids: Structure-activity relations and mode of action. Biochem. Pharmacol 2003, 65, 773–781. [Google Scholar]
- Schewe, T; Sadik, C; Klotz, LO; Yoshimoto, T; Kuhn, H; Sies, H. Polyphenols of cocoa: Inhibition of mammalian 15-lipoxygenase. Biol. Chem 2001, 382, 168716–96. [Google Scholar]
- de la Puerta, R; Ruiz Gutierrez, V; Hoult, JR. Inhibition of leukocyte 5-lipoxygenase by phenolics from virgin olive oil. Biochem. Pharmacol 1999, 57, 445–49. [Google Scholar]
- Kong, AN; Yu, R; Chen, C; Mandlekar, S; Primiano, T. Signal transduction events elicited by natural products: Role of MAPK and caspase pathways in homeostatic response and induction of apoptosis. Arch Pharm. Res 2000, 23, 1–16. [Google Scholar]
- Spencer, JP; Rice-Evans, C; Williams, RJ. Modulation of pro-survival Akt/protein kinase B and ERK1/2 signaling cascades by quercetin and its in vivo metabolites underlie their action on neuronal viability. J. Biol. Chem 2003, 278, 34783–34793. [Google Scholar]
- Wiseman, S; Mulder, T; Rietveld, A. Tea flavonoids: Bioavailability in vivo and effects on cell signaling pathways in vitro. Antioxid. Redox Signal 2001, 3, 1009–1021. [Google Scholar]
- Rosenblat, M; Aviram, M. Paraoxonases role in the prevention of cardiovascular diseases. Biofactors 2009, 35, 98–104. [Google Scholar]
- Masella, R; Vari, R; D'Archivio, M; Di Benedetto, R; Matarrese, P; Malorni, W; Scazzocchio, B; Giovannini, C. Extra virgin olive oil biophenols inhibit cell-mediated oxidation of LDL by increasing the mRNA transcription of glutathione-related enzymes. J .Nutr 2004, 134, 785–791. [Google Scholar]
- Monasterio, A; Urdaci, MC; Pinchuk, IV; Lopez-Moratalla, N; Martinez-Irujo, JJ. Flavonoids induce apoptosis in human leukemia U937 cells through caspase- and caspase-calpain-dependent pathways. Nutr. Cancer 2004, 50, 90–100. [Google Scholar]
- Way, TD; Kao, MC; Lin, JK. Degradation of HER2/neu by apigenin induces apoptosis through cytochrome c release and caspase-3 activation in HER2/neu-overexpressing breast cancer cells. FEBS Lett 2005, 579, 145–52. [Google Scholar]
- Fischer, PM; Lane, DP. Inhibitors of cyclin-dependent kinases as anti-cancer therapeutics. Curr. Med. Chem 2000, 7, 1213–1245. [Google Scholar]
- Murphy, KJ; Chronopoulos, AK; Singh, I; Francis, MA; Moriarty, H; Pike, MJ; Turner, AH; Mann, NJ; Sinclair, AJ. Dietary flavanols and procyanidin oligomers from cocoa (Theobroma cacao) inhibit platelet function. Am. J. Clin. Nutr 2003, 77, 1466–1473. [Google Scholar]
- Carluccio, MA; Siculella, L; Ancora, MA; Massaro, M; Scoditti, E; Storelli, C; Visioli, F; Distante, A; De Caterina, R. Olive oil and red wine antioxidant polyphenols inhibit endothelial activation: Antiatherogenic properties of Mediterranean diet phytochemicals. Arterioscler Thromb Vasc Biol 2003, 23, 622–629. [Google Scholar]
- Porrini, M; Riso, P. Factors influencing the bioavailability of antioxidants in foods: A critical appraisal. Nutr. Metab. Cardiovasc Dis 2008, 18, 647–650. [Google Scholar]
- Crozier, A; Jaganath, IB; Clifford, MN. Dietary phenolics: Chemistry, bioavailability and effects on health. Nat. Prod. Rep 2009, 26, 1001–1043. [Google Scholar]
- Cheynier, V. Polyphenols in foods are more complex than often thought. Am. J. Clin. Nutr 2005, 81, 223S–229S. [Google Scholar]
- Manach, C; Scalbert, A; Morand, C; Remesy, C; Jimenez, L. Polyphenols: Food sources and bioavailability. Am. J. Clin. Nutr 2004, 79, 727–747. [Google Scholar]
- Sanoner, P; Guyot, S; Marnet, N; Molle, D; Drilleau, JP. Polyphenol profiles of French cider apple varieties (Malus domestica sp.). J. Agric. Food Chem 1999, 47, 4847–4853. [Google Scholar]
- Guyot, S; Marnet, N; Sanoner, P; Drilleau, JF. Variability of the polyphenolic composition of cider apple (Malus domestica) fruits and juices. J .Agric. Food Chem 2003, 51, 6240–6247. [Google Scholar]
- Forester, SC; Waterhouse, AL. Metabolites are key to understanding health effects of wine polyphenolics. J. Nutr 2009, 139, 1824S–1831S. [Google Scholar]
- Waterhouse, AL. Wine Phenolics. Ann. N. Y. Acad. Sci 2002, 957, 21–36. [Google Scholar]
- Vitaglione, P; Ottanelli, B; Milani, S; Morisco, F; Caporaso, N; Fogliano, V. Dietary trans-resveratrol bioavailability and effect on CCl4-induced liver lipid peroxidation. J. Gastroenterol. Hepatol 2009, 24, 618–622. [Google Scholar]
- Brown, L; Kroon, PA; Das, DK; Das, S; Tosaki, A; Chan, V; Singer, MV; Feick, P. The biological responses to resveratrol and other polyphenols from alcoholic beverages. Alcohol Clin. Exp. Res 2009, 33, 1513–1523. [Google Scholar]
- Bhat, KPL; Kosmeder, JW, II; Pezzuto, JM. Biological effects of resveratrol. Antioxid. Redox Signal 2001, 3, 1041–1064. [Google Scholar]
- Hung, LM; Chen, JK; Huang, SS; Lee, RS; Su, MJ. Cardioprotective effect of resveratrol, a natural antioxidant derived from grapes. Cardiovasc Res 2000, 47, 549–555. [Google Scholar]
- Godichaud, S; Krisa, S; Couronne, B; Dubuisson, L; Merillon, JM; Desmouliere, A; Rosenbaum, J. Deactivation of cultured human liver myofibroblasts by trans-resveratrol, a grapevine-derived polyphenol. Hepatology 2000, 31, 922–931. [Google Scholar]
- Lamuela-Raventós, RM; Waterhouse, AL. Occurrence of resveratrol in selected California wines by a new HPLC method. J. Agric. Food Chem 1993, 41, 521–523. [Google Scholar]
- Chu, Q; O’Dwyer, M; Zeece, MG. Direct analysis of resveratrol in wine by Micel capillary electrophoresis. J. Agric. Food Chem 1998, 46, 509–513. [Google Scholar]
- Cicerale, S; Conlan, XA; Sinclair, AJ; Keast, RS. Chemistry and health of olive oil phenolics. Crit. Rev. Food Sci. Nutr 2009, 49, 218–236. [Google Scholar]
- Tripoli, E; Giammanco, M; Tabacchi, G; Di Majo, D; Giammanco, S; La Guardia, M. The phenolic compounds of olive oil: Structure, biological activity and beneficial effects on human health. Nutr. Res. Rev 2005, 18, 98–112. [Google Scholar]
- Baccouri, B; Temime, SB; Taamalli, W; Daoud, D; M’Sallem, M; Zarrouk, M. Analytical characteristics of virgin olive oil from two new varieties obtained by controlled crossing on meski variety. J. Food Lip 2007, 14, 19–34. [Google Scholar]
- Carrasco-Pancorbo, A; Cerretani, L; Bendini, A; Segura-Carretero, A; Gallina-Toschi, T; Fernandez-Gutierrez, A. Analytical determination of polyphenols in olive oils. J. Sep. Sci 2005, 28, 837–858. [Google Scholar]
- Scalbert, A; Williamson, G. Dietary intake and bioavailability of polyphenols. J. Nutr 2000, 130, 2073S–85S. [Google Scholar]
- Femia, AP; Caderni, G; Ianni, M; Salvadori, M; Schijlen, E; Collins, G; Bovy, A; Dolara, P. Effect of diets fortified with tomatoes or onions with variable quercetin-glycoside content on azoxymethane-induced aberrant crypt foci in the colon of rats. Eur. J. Nutr 2003, 42, 346–352. [Google Scholar]
- Gomez-Rico, A; Salvador, MD; La Greca, M; Fregapane, G. Phenolic and volatile compounds of extra virgin olive oil (Olea europaea L. Cv. Cornicabra) with regard to fruit ripening and irrigation management. J. Agric. Food Chem 2006, 54, 7130–7136. [Google Scholar]
- Macheix, JJ; Fleuriet, A; Billot, J. Fruit Phenolics; CRC Press: Boca Raton, FL, USA, 1990; pp. 101–126. [Google Scholar]
- Bonoli, M; Bendini, A; Cerretani, L; Lercker, G; Toschi, TG. Qualitative and semiquantitative analysis of phenolic compounds in extra virgin olive oils as a function of the ripening degree of olive fruits by different analytical techniques. J. Agric. Food Chem 2004, 52, 7026–7032. [Google Scholar]
- Jemai, H; Bouaziz, M; Sayadi, S. Phenolic composition, sugar contents and antioxidant activity of Tunisian sweet olive cultivar with regard to fruit ripening. J. Agric. Food Chem 2009, 57, 2961–2968. [Google Scholar]
- Bouaziz, M; Chamkha, M; Sayadi, S. Comparative study on phenolic content and antioxidant activity during maturation of the olive cultivar Chemlali from Tunisia. J. Agric. Food Chem 2004, 52, 5476–5481. [Google Scholar]
- Xu, B; Chang, SK. Total phenolic, phenolic acid, anthocyanin, flavan-3-ol, and flavonol profiles and antioxidant properties of pinto and Black Beans (Phaseolus vulgaris L.) as affected by thermal processing. J. Agric. Food Chem 2009, 57, 4754–4764. [Google Scholar]
- Xu, B; Chang, SK. Phytochemical profiles and health-promoting effects of cool-season food legumes as influenced by thermal processing. J. Agric. Food Chem 2009, 57, 10718–10731. [Google Scholar]
- Rocha-Guzman, NE; Gonzlez-Laredo, RF; Ibarra-Prez, FJ; Nava-Bermen, CA; Gallegos-Infante, JA. Effect of pressure cooking on the antioxidant activity of extracts from three common bean (Phaseolus vulgaris L.) cultivars. Food Chem 2007, 100, 31–35. [Google Scholar]
- Khatun, M; Eguchi, S; Yamaguchi, T; Takamura, H; Matoba, T. Effect of the thermal treatment on radical-scavenging activity of some spices. Food Sci. Technol. Res 2006, 12, 178–185. [Google Scholar]
- Carrasco-Pancorbo, A; Cerretani, L; Bendini, A; Segura-Carretero, A; Lercker, G; Fernandez-Gutierrez, A. Evaluation of the influence of thermal oxidation on the phenolic composition and on the antioxidant activity of extra-virgin olive oils. J. Agric. Food Chem 2007, 55, 4771–4780. [Google Scholar]
- Ferracane, R; Pellegrini, N; Visconti, A; Graziani, G; Chiavaro, E; Miglio, C; Fogliano, V. Effects of different cooking methods on antioxidant profile, antioxidant capacity, and physical characteristics of artichoke. J. Agric. Food Chem 2008, 56, 8601–8608. [Google Scholar]
- Miglio, C; Chiavaro, E; Visconti, A; Fogliano, V; Pellegrini, N. Effects of different cooking methods on nutritional and physicochemical characteristics of selected vegetables. J. Agric. Food Chem 2008, 56, 139–147. [Google Scholar]
- van der Sluis, AA; Dekker, M; de Jager, A; Jongen, WM. Activity and concentration of polyphenolic antioxidants in apple: Effect of cultivar, harvest year, and storage conditions. J. Agric. Food Chem 2001, 49, 3606–3613. [Google Scholar]
- DuPont, MS; Mondin, Z; Williamson, G; Price, KR. Effect of variety, processing, and storage on the flavonoid glycoside content and composition of lettuce and endive. J. Agric. Food Chem 2000, 48, 3957–3964. [Google Scholar]
- Baiano, A; Gambacorta, G; Terracone, C; Previtali, MA; Lamacchia, C; La Notte, E. Changes in phenolic content and antioxidant activity of Italian extra-virgin olive oils during storage. J. Food. Sci 2009, 74, C177–183. [Google Scholar]
- Brenes, M; Garcia, A; Dobarganes, MC; Velasco, J; Romero, C. Influence of thermal treatments simulating cooking processes on the polyphenol content in virgin olive oil. J. Agric. Food Chem 2002, 50, 5962–5967. [Google Scholar]
- Xu, X; Li, W; Lu, Z; Beta, T; Hydamaka, AW. Phenolic content, composition, antioxidant activity, and their changes during domestic cooking of potatoes. J. Agric. Food Chem 2009, 57, 10231–10238. [Google Scholar]
- Gomez-Alonso, S; Fregapane, G; Salvador, MD; Gordon, MH. Changes in phenolic composition and antioxidant activity of virgin olive oil during frying. J. Agric. Food Chem 2003, 51, 667–672. [Google Scholar]
- Andrikopoulos, NK; Dedoussis, GV; Falirea, A; Kalogeropoulos, N; Hatzinikola, HS. Deterioration of natural antioxidant species of vegetable edible oils during the domestic deep-frying and pan-frying of potatoes. Int. J. Food Sci. Nutr 2002, 53, 351–363. [Google Scholar]
- Bugianesi, R; Salucci, M; Leonardi, C; Ferracane, R; Catasta, G; Azzini, E; Maiani, G. Effect of domestic cooking on human bioavailability of naringenin, chlorogenic acid, lycopene and beta-carotene in cherry tomatoes. Eur. J. Nutr 2004, 43, 360–366. [Google Scholar]
- Gliszczynska-Swiglo, A; Ciska, E; Pawlak-Lemanska, K; Chmielewski, J; Borkowski, T; Tyrakowska, B. Changes in the content of health-promoting compounds and antioxidant activity of broccoli after domestic processing. Food Addit. Contam 2006, 23, 1088–1098. [Google Scholar]
- Gliszczynska-Swiglo, A; Tyrakowska, B. Quality of commercial apple juices evaluated on the basis of the polyphenol content and the TEAC antioxidant activity. J. Food Sci 2003, 68, 1844–1849. [Google Scholar]
- Mullen, W; Stewart, AJ; Lean, ME; Gardner, P; Duthie, GG; Crozier, A. Effect of freezing and storage on the phenolics, ellagitannins, flavonoids, and antioxidant capacity of red raspberries. J. Agric. Food Chem 2002, 50, 5197–5201. [Google Scholar]
- Vallejo, F; Tomas-Barberan, F; Garcia-Viguera, C. Health-promoting compounds in broccoli as influenced by refrigerated transport and retail sale period. J. Agric. Food Chem 2003, 51, 3029–3034. [Google Scholar]
- Zafrilla, P; Morillas, J; Mulero, J; Cayuela, JM; Martinez-Cacha, A; Pardo, F; Lopez Nicolas, JM. Changes during storage in conventional and ecological wine: Phenolic content and antioxidant activity. J. Agric. Food Chem 2003, 51, 4694–4700. [Google Scholar]
- Napolitano, A; Cascone, A; Graziani, G; Ferracane, R; Scalfi, L; Di Vaio, C; Ritieni, A; Fogliano, V. Influence of variety and storage on the polyphenol composition of apple flesh. J. Agric. Food Chem 2004, 52, 6526–6531. [Google Scholar]
- Klaiber, RG; Baur, S; Koblo, A; Carle, R. Influence of washing treatment and storage atmosphere on phenylalanine ammonia-lyase activity and phenolic acid content of minimally processed carrot sticks. J. Agric. Food Chem 2005, 53, 1065–1072. [Google Scholar]
- Gutierrez, F; Fernandez, JL. Determinant parameters and components in the storage of virgin olive oil. Prediction of storage time beyond which the oil is no longer of “extra” quality. J. Agric. Food Chem 2002, 50, 571–577. [Google Scholar]
- Okogeri, O; Tasioula-Margari, M. Changes occurring in phenolic compounds and alpha-tocopherol of virgin olive oil during storage. J. Agric. Food Chem 2002, 50, 1077–1080. [Google Scholar]
- Lavelli, V; Fregapane, G; Salvador, MD. Effect of storage on secoiridoid and tocopherol contents and antioxidant activity of monovarietal extra virgin olive oils. J. Agric. Food Chem 2006, 54, 3002–3007. [Google Scholar]
- Cinquanta, L; Esti, M; La Notte, E. Evolution of phenolic compounds in virgin olive oil during storage. J. Am. Oil Chem. Soc 1997, 74, 1259–1264. [Google Scholar]
- Porrini, M; Riso, P; Testolin, G. Absorption of lycopene from single or daily portions of raw and processed tomato. Br. J. Nutr 1998, 80, 353–361. [Google Scholar]
- Azuma, K; Ippoushi, K; Ito, H; Higashio, H; Terao, J. Combination of lipids and emulsifiers enhances the absorption of orally administered quercetin in rats. J. Agric. Food Chem 2002, 50, 1706–1712. [Google Scholar]
- Lesser, S; Cermak, R; Wolffram, S. Bioavailability of quercetin in pigs is influenced by the dietary fat content. J. Nutr 2004, 134, 1508–1511. [Google Scholar]
- Visioli, F; Galli, C; Grande, S; Colonnelli, K; Patelli, C; Galli, G; Caruso, D. Hydroxytyrosol excretion differs between rats and humans and depends on the vehicle of administration. J. Nutr 2003, 133, 2612–2615. [Google Scholar]
- Donovan, JL; Bell, JR; Kasim-Karakas, S; German, JB; Walzem, RL; Hansen, RJ; Waterhouse, AL. Catechin is present as metabolites in human plasma after consumption of red wine. J. Nutr 1999, 129, 1662–1668. [Google Scholar]
- Donovan, JL; Kasim-Karakas, S; German, JB; Waterhouse, AL. Urinary excretion of catechin metabolites by human subjects after red wine consumption. Br. J. Nutr 2002, 87, 31–37. [Google Scholar]
- Roura, E; Andres-Lacueva, C; Estruch, R; Lourdes Mata Bilbao, M; Izquierdo-Pulido, M; Lamuela-Raventos, RM. The effects of milk as a food matrix for polyphenols on the excretion profile of cocoa (−)-epicatechin metabolites in healthy human subjects. Br. J. Nutr 2008, 100, 846–851. [Google Scholar]
- Serafini, M; Bugianesi, R; Maiani, G; Valtuena, S; De Santis, S; Crozier, A. Plasma antioxidants from chocolate. Nature 2003, 424, 1013. [Google Scholar]
- Schroeter, H; Holt, RR; Orozco, TJ; Schmitz, HH; Keen, CL. Nutrition: Milk and absorption of dietary flavanols. Nature 2003, 426, 787–788. [Google Scholar]
- Schramm, DD; Karim, M; Schrader, HR; Holt, RR; Kirkpatrick, NJ; Polagruto, JA; Ensunsa, JL; Schmitz, HH; Keen, CL. Food effects on the absorption and pharmacokinetics of cocoa flavanols. Life Sci 2003, 73, 857–869. [Google Scholar]
- van het Hof, KH; Kivits, GA; Weststrate, JA; Tijburg, LB. Bioavailability of catechins from tea: The effect of milk. Eur. J. Clin. Nutr 1998, 52, 356–359. [Google Scholar]
- Roura, E; Andres-Lacueva, C; Estruch, R; Mata-Bilbao, ML; Izquierdo-Pulido, M; Waterhouse, AL; Lamuela-Raventos, RM. Milk does not affect the bioavailability of cocoa powder flavonoid in healthy human. Ann. Nutr. Metab 2007, 51, 493–498. [Google Scholar]
- Meng, X; Maliakal, P; Lu, H; Lee, MJ; Yang, CS. Urinary and plasma levels of resveratrol and quercetin in humans, mice, and rats after ingestion of pure compounds and grape juice. J. Agric. Food Chem 2004, 52, 935–942. [Google Scholar]
- Goldberg, DM; Yan, J; Soleas, GJ. Absorption of three wine-related polyphenols in three different matrices by healthy subjects. Clin. Biochem 2003, 36, 79–87. [Google Scholar]
- Ortega, N; Reguant, J; Romero, MP; Macia, A; Motilva, MJ. Effect of fat content on the digestibility and bioaccessibility of cocoa polyphenol by an in vitro digestion model. J. Agric. Food Chem 2009, 57, 5743–5749. [Google Scholar]
- Rao, SS; Lu, C; Schulze-Delrieu, K. Duodenum as a immediate brake to gastric outflow: A videofluoroscopic and manometric assessment. Gastroenterology 1996, 110, 740–747. [Google Scholar]
- Mullen, W; Edwards, CA; Serafini, M; Crozier, A. Bioavailability of pelargonidin-3-O-glucoside and its metabolites in humans following the ingestion of strawberries with and without cream. J. Agric. Food Chem 2008, 56, 713–719. [Google Scholar]
- Mullen, W; Archeveque, MA; Edwards, CA; Matsumoto, H; Crozier, A. Bioavailability and metabolism of orange juice flavanones in humans: Impact of a full-fat yogurt. J. Agric. Food Chem 2008, 56, 11157–11164. [Google Scholar]
- Miro-Casas, E; Covas, MI; Fito, M; Farre-Albadalejo, M; Marrugat, J; de la Torre, R. Tyrosol and hydroxytyrosol are absorbed from moderate and sustained doses of virgin olive oil in humans. Eur. J. Clin. Nutr 2003, 57, 186–190. [Google Scholar]
- Visioli, F; Galli, C; Bornet, F; Mattei, A; Patelli, R; Galli, G; Caruso, D. Olive oil phenolics are dose-dependently absorbed in humans. FEBS Lett 2000, 468, 159–160. [Google Scholar]
- Tuck, KL; Freeman, MP; Hayball, PJ; Stretch, GL; Stupans, I. The in vivo fate of hydroxytyrosol and tyrosol, antioxidant phenolic constituents of olive oil, after intravenous and oral dosing of labeled compounds to rats. J. Nutr 2001, 131, 1993–1996. [Google Scholar]
- Tamura, M; Nakagawa, H; Tsushida, T; Hirayama, K; Itoh, K. Effect of pectin enhancement on plasma quercetin and fecal flora in rutin-supplemented mice. J. Food. Sci 2007, 72, S648–S651. [Google Scholar]
- Perez-Jimenez, J; Serrano, J; Tabernero, M; Arranz, S; Diaz-Rubio, ME; Garcia-Diz, L; Goni, I; Saura-Calixto, F. Bioavailability of phenolic antioxidants associated with dietary fiber: Plasma antioxidant capacity after acute and long-term intake in humans. Plant Foods Hum. Nutr 2009, 64, 102–107. [Google Scholar]
- Sazuka, M; Itoi, T; Suzuki, Y; Odani, S; Koide, T; Isemura, M. Evidence for the interaction between (−)-epigallocatechin gallate and human plasma proteins fibronectin, fibrinogen, and histidine-rich glycoprotein. Biosci. Biotechnol. Biochem 1996, 60, 1317–1319. [Google Scholar]
- Cicerale, S; Conlan, XA; Barnett, NW; Sinclair, AJ; Keast, RS. Influence of heat on biological activity and concentration of oleocanthal--a natural anti-inflammatory agent in virgin olive oil. J. Agric. Food Chem 2009, 57, 1326–1330. [Google Scholar]
- Nurmi, T; Mursu, J; Heinonen, M; Nurmi, A; Hiltunen, R; Voutilainen, S. Metabolism of berry anthocyanins to phenolic acids in humans. J. Agric. Food Chem 2009, 57, 2274–2281. [Google Scholar]
- Passamonti, S; Vrhovsek, U; Mattivi, F. The interaction of anthocyanins with bilitranslocase. Biochem. Biophys. Res. Commun 2002, 296, 631–636. [Google Scholar]
- Wu, X; Cao, G; Prior, RL. Absorption and metabolism of anthocyanins in elderly women after consumption of elderberry or blueberry. J. Nutr 2002, 132, 1865–1871. [Google Scholar]
- Semenza, G. Mammalian Ectoenzymes; Kenny, AJ, Turner, AJ, Eds.; Elsevier: Amsterdam, Holand, 1987; pp. 256–287. [Google Scholar]
- Wilkinson, AP; Gee, JM; Dupont, MS; Needs, PW; Mellon, FA; Williamson, G; Johnson, IT. Hydrolysis by lactase phlorizin hydrolase is the first step in the uptake of daidzein glucosides by rat small intestine in vitro. Xenobiotica 2003, 33, 255–264. [Google Scholar]
- Day, AJ; Canada, FJ; Diaz, JC; Kroon, PA; McLauchlan, R; Faulds, CB; Plumb, GW; Morgan, MR; Williamson, G. Dietary flavonoid and isoflavone glycosides are hydrolysed by the lactase site of lactase phlorizin hydrolase. FEBS Lett 2000, 468, 166–170. [Google Scholar]
- Gee, JM; DuPont, MS; Day, AJ; Plumb, GW; Williamson, G; Johnson, IT. Intestinal transport of quercetin glycosides in rats involves both deglycosylation and interaction with the hexose transport pathway. J. Nutr 2000, 130, 2765–2771. [Google Scholar]
- Aura, AM; Martin-Lopez, P; O'Leary, KA; Williamson, G; Oksman-Caldentey, KM; Poutanen, K; Santos-Buelga, C. In vitro metabolism of anthocyanins by human gut microflora. Eur. J. Nutr 2005, 44, 133–142. [Google Scholar]
- Kuhnau, J. The flavonoids. A class of semi-essential food components: Their role in human nutrition. World Rev. Nutr. Diet 1976, 24, 117–191. [Google Scholar]
- Setchell, KD; Brown, NM; Lydeking-Olsen, E. The clinical importance of the metabolite equol-a clue to the effectiveness of soy and its isoflavones. J. Nutr 2002, 132, 3577–3584. [Google Scholar]
- Munoz, Y; Garrido, A; Valladares, L. Equol is more active than soy isoflavone itself to compete for binding to thromboxane A(2) receptor in human platelets. Thromb Res 2009, 123, 740–744. [Google Scholar]
- Lu, LJ; Anderson, KE. Sex and long-term soy diets affect the metabolism and excretion of soy isoflavones in humans. Am. J. Clin. Nutr 1998, 68, 1500S–1504S. [Google Scholar]
- Morton, MS; Arisaka, O; Miyake, N; Morgan, LD; Evans, BA. Phytoestrogen concentrations in serum from Japanese men and women over forty years of age. J. Nutr 2002, 132, 3168–3171. [Google Scholar]
- Setchell, KD; Clerici, C; Lephart, ED; Cole, SJ; Heenan, C; Castellani, D; Wolfe, BE; Nechemias-Zimmer, L; Brown, NM; Lund, TD; Handa, RJ; Heubi, JE. S-equol, a potent ligand for estrogen receptor beta, is the exclusive enantiomeric form of the soy isoflavone metabolite produced by human intestinal bacterial flora. Am. J. Clin. Nutr 2005, 81, 1072–1079. [Google Scholar]
- Decroos, K; Vanhemmens, S; Cattoir, S; Boon, N; Verstraete, W. Isolation and characterisation of an equol-producing mixed microbial culture from a human faecal sample and its activity under gastrointestinal conditions. Arch Microbiol 2005, 183, 45–55. [Google Scholar]
- Tamura, M; Tsushida, T; Shinohara, K. Isolation of an isoflavone-metabolizing, Clostridium-like bacterium, strain TM-40, from human faeces. Anaerobe 2007, 13, 32–35. [Google Scholar]
- Wang, XL; Kim, HJ; Kang, SI; Kim, SI; Hur, HG. Production of phytoestrogen S-equol from daidzein in mixed culture of two anaerobic bacteria. Arch Microbiol 2007, 187, 155–160. [Google Scholar]
- Yee, S; Burdock, GA; Kurata, Y; Enomoto, Y; Narumi, K; Hamada, S; Itoh, T; Shimomura, Y; Ueno, T. Acute and subchronic toxicity and genotoxicity of SE5-OH, an equol-rich product produced by Lactococcus garvieae. Food Chem. Toxicol 2008, 46, 2713–2720. [Google Scholar]
- Felgines, C; Talavera, S; Texier, O; Gil-Izquierdo, A; Lamaison, JL; Remesy, C. Blackberry anthocyanins are mainly recovered from urine as methylated and glucuronidated conjugates in humans. J. Agric. Food Chem 2005, 53, 7721–7727. [Google Scholar]
- Day, AJ; Bao, Y; Morgan, MR; Williamson, G. Conjugation position of quercetin glucuronides and effect on biological activity. Free Radic. Biol. Med 2000, 29, 1234–1243. [Google Scholar]
- Piskula, MK; Terao, J. Accumulation of (−)-epicatechin metabolites in rat plasma after oral administration and distribution of conjugation enzymes in rat tissues. J. Nutr 1998, 128, 1172–1178. [Google Scholar]
- Tilgmann, C; Ulmanen, I. Purification methods of mammalian catechol-O-methyltransferases. J Chromatogr B: Biomed Appl 1996, 684, 147–161. [Google Scholar]
- Falany, CN. Enzymology of human cytosolic sulfotransferases. Faseb. J 1997, 11, 206–216. [Google Scholar]
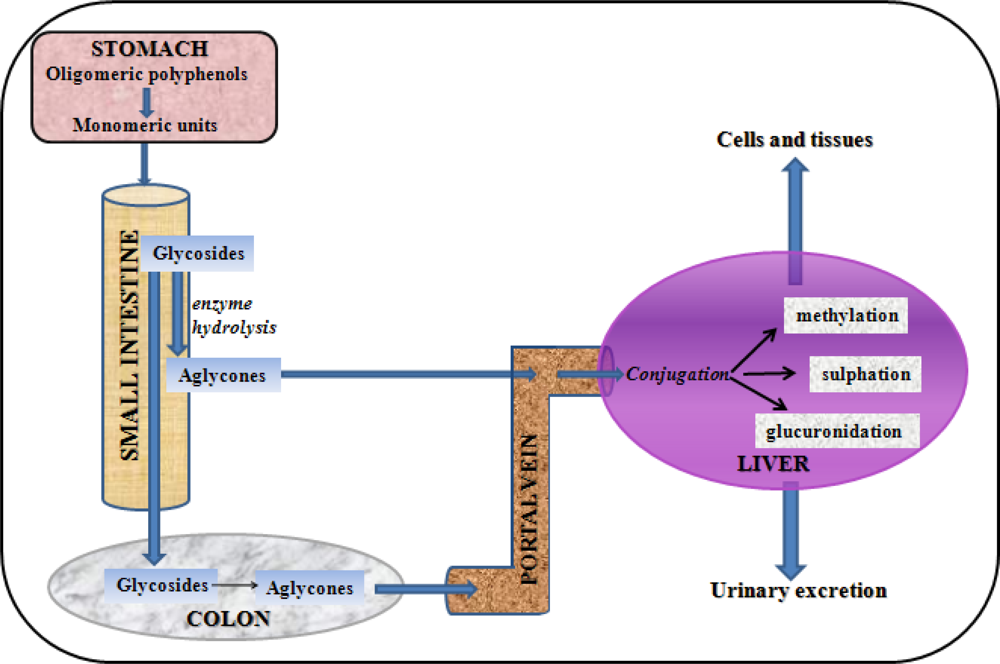
- Spencer, JP; Chowrimootoo, G; Choudhury, R; Debnam, ES; Srai, SK; Rice-Evans, C. The small intestine can both absorb and glucuronidate luminal flavonoids. FEBS Lett 1999, 458, 224–230. [Google Scholar]
- Crespy, V; Morand, C; Besson, C; Manach, C; Demigne, C; Remesy, C. Comparison of the intestinal absorption of quercetin, phloretin and their glucosides in rats. J. Nutr 2001, 131, 2109–2114. [Google Scholar]
- Boersma, MG; van der Woude, H; Bogaards, J; Boeren, S; Vervoort, J; Cnubben, NH; van Iersel, ML; van Bladeren, PJ; Rietjens, IM. Regioselectivity of phase II metabolism of luteolin and quercetin by UDP-glucuronosyl transferases. Chem. Res. Toxicol 2002, 15, 662–670. [Google Scholar]
- Lee, MJ; Maliakal, P; Chen, L; Meng, X; Bondoc, FY; Prabhu, S; Lambert, G; Mohr, S; Yang, CS. Pharmacokinetics of tea catechins after ingestion of green tea and (−)-epigallocatechin-3-gallate by humans: Formation of different metabolites and individual variability. Cancer Epidemiol. Biomarkers Prev 2002, 11, 1025–1032. [Google Scholar]
- Mullen, W; Edwards, CA; Crozier, A. Absorption, excretion and metabolite profiling of methyl-, glucuronyl-, glucosyl- and sulpho-conjugates of quercetin in human plasma and urine after ingestion of onions. Br. J. Nutr 2006, 96, 107–116. [Google Scholar]
- Zhang, Y; Song, TT; Cunnick, JE; Murphy, PA; Hendrich, S. Daidzein and genistein glucuronides in vitro are weakly estrogenic and activate human natural killer cells at nutritionally relevant concentrations. J. Nutr 1999, 129, 399–405. [Google Scholar]
- Wen, X; Walle, T. Methylated flavonoids have greatly improved intestinal absorption and metabolic stability. Drug Metab. Dispos 2006, 34, 1786–1792. [Google Scholar]
- Wen, X; Walle, T. Methylation protects dietary flavonoids from rapid hepatic metabolism. Xenobiotica 2006, 36, 387–397. [Google Scholar]
- Landis-Piwowar, KR; Milacic, V; Dou, QP. Relationship between the methylation status of dietary flavonoids and their growth-inhibitory and apoptosis-inducing activities in human cancer cells. J. Cell Biochem 2008, 105, 514–523. [Google Scholar]
- Del Rio, D; Costa, LG; Lean, ME; Crozier, A. Polyphenols and health: What compounds are involved? Nutr. Metab. Cardiovasc. Dis 2010, 20, 1–6. [Google Scholar]
- Monteiro, M; Farah, A; Perrone, D; Trugo, LC; Donangelo, C. Chlorogenic acid compounds from coffee are differentially absorbed and metabolized in humans. J. Nutr 2007, 137, 2196–2201. [Google Scholar]
- Seeram, NP; Zhang, Y; McKeever, R; Henning, SM; Lee, RP; Suchard, MA; Li, Z; Chen, S; Thames, G; Zerlin, A; Nguyen, M; Wang, D; Dreher, M; Heber, D. Pomegranate juice and extracts provide similar levels of plasma and urinary ellagitannin metabolites in human subjects. J. Med. Food 2008, 11, 390–394. [Google Scholar]
- Manach, C; Williamson, G; Morand, C; Scalbert, A; Remesy, C. Bioavailability and bioefficacy of polyphenols in humans. I. Review of 97 bioavailability studies. Am. J. Clin. Nutr 2005, 81, 230S–242S. [Google Scholar]
- Zaripheh, S; Erdman, JW, Jr. The biodistribution of a single oral dose of [14C]-lycopene in rats prefed either a control or lycopene-enriched diet. J. Nutr 2005, 135, 2212–2218. [Google Scholar]
- Gladine, C; Rock, E; Morand, C; Bauchart, D; Durand, D. Bioavailability and antioxidant capacity of plant extracts rich in polyphenols, given as a single acute dose, in sheep made highly susceptible to lipoperoxidation. Br. J. Nutr 2007, 98, 691–701. [Google Scholar]
- Khan, SA; Priyamvada, S; Farooq, N; Khan, S; Khan, MW; Yusufi, AN. Protective effect of green tea extract on gentamicin-induced nephrotoxicity and oxidative damage in rat kidney. Pharmacol. Res 2009, 59, 254–262. [Google Scholar]
- Sato, S; Mukai, Y; Yamate, J; Kato, J; Kurasaki, M; Hatai, A; Sagai, M. Effect of polyphenol-containing azuki bean (Vigna angularis) extract on blood pressure elevation and macrophage infiltration in the heart and kidney of spontaneously hypertensive rats. Clin. Exp. Pharmacol. Physiol 2008, 35, 43–49. [Google Scholar]
- Seeram, NP; Aronson, WJ; Zhang, Y; Henning, SM; Moro, A; Lee, RP; Sartippour, M; Harris, DM; Rettig, M; Suchard, MA; Pantuck, AJ; Belldegrun, A; Heber, D. Pomegranate ellagitannin-derived metabolites inhibit prostate cancer growth and localize to the mouse prostate gland. J. Agric. Food Chem 2007, 55, 7732–7737. [Google Scholar]
- de Boer, VC; Dihal, AA; van der Woude, H; Arts, IC; Wolffram, S; Alink, GM; Rietjens, IM; Keijer, J; Hollman, PC. Tissue distribution of quercetin in rats and pigs. J. Nutr 2005, 135, 1718–1725. [Google Scholar]
- Day, AJ; Mellon, F; Barron, D; Sarrazin, G; Morgan, MR; Williamson, G. Human metabolism of dietary flavonoids: Identification of plasma metabolites of quercetin. Free Radic. Res 2001, 35, 941–952. [Google Scholar]
- Hong, SJ; Kim, SI; Kwon, SM; Lee, JR; Chung, BC. Comparative study of concentration of isoflavones and lignans in plasma and prostatic tissues of normal control and benign prostatic hyperplasia. Yonsei Med. J 2002, 43, 236–241. [Google Scholar]
- Henning, SM; Aronson, W; Niu, Y; Conde, F; Lee, NH; Seeram, NP; Lee, RP; Lu, J; Harris, DM; Moro, A; Hong, J; Pak-Shan, L; Barnard, RJ; Ziaee, HG; Csathy, G; Go, VL; Wang, H; Heber, D. Tea polyphenols and theaflavins are present in prostate tissue of humans and mice after green and black tea consumption. J. Nutr 2006, 136, 1839–1843. [Google Scholar]
- Veeriah, S; Balavenkatraman, KK; Bohmer, F; Kahle, K; Glei, M; Richling, E; Scheppach, W; Pool-Zobel, BL. Intervention with cloudy apple juice results in altered biological activities of ileostomy samples collected from individual volunteers. Eur. J. Nutr 2008, 47, 226–234. [Google Scholar]
- Paetau, I; Rao, D; Wiley, ER; Brown, ED; Clevidence, BA. Carotenoids in human buccal mucosa cells after 4 wk of supplementation with tomato juice or lycopene supplements. Am. J. Clin. Nutr 1999, 70, 490–494. [Google Scholar]
- Maubach, J; Bracke, ME; Heyerick, A; Depypere, HT; Serreyn, RF; Mareel, MM; De Keukeleire, D. Quantitation of soy-derived phytoestrogens in human breast tissue and biological fluids by high-performance liquid chromatography. J. Chromatogr. B: Anal. Technol. Biomed. Life Sci 2003, 784, 137–144. [Google Scholar]
- Gu, L; Laly, M; Chang, HC; Prior, RL; Fang, N; Ronis, MJ; Badger, TM. Isoflavone conjugates are underestimated in tissues using enzymatic hydrolysis. J. Agric. Food Chem 2005, 53, 6858–6863. [Google Scholar]
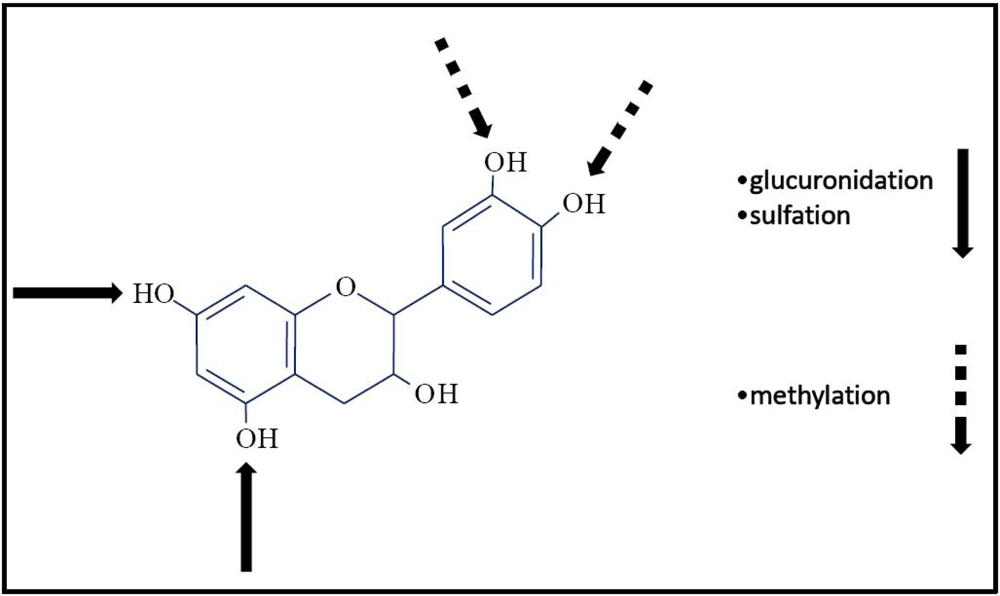
| External factors | Environmental factors (i.e., sun exposure, degree of ripeness); food availability |
| Food processing related factors | Thermal treatments; homogenization; liophylization; cooking and methods of culinary preparation; storage |
| Food related factors | Food matrix; presence of positive or negative effectors of absorption (i.e., fat, fiber) |
| Interaction with other compounds | Bonds with proteins (i.e., albumin) or with polyphenols with similar mechanism of absorption |
| Polyphenols related factors | Chemical structure; concentration in food; amount introduced |
| Host related factors | Intestinal factors (i.e., enzyme activity; intestinal transit time; colonic microflora). Systemic factors (i.e., gender and age; disorders and/or pathologies; genetics; physiological condition) |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
D’Archivio, M.; Filesi, C.; Varì, R.; Scazzocchio, B.; Masella, R. Bioavailability of the Polyphenols: Status and Controversies. Int. J. Mol. Sci. 2010, 11, 1321-1342. https://doi.org/10.3390/ijms11041321
D’Archivio M, Filesi C, Varì R, Scazzocchio B, Masella R. Bioavailability of the Polyphenols: Status and Controversies. International Journal of Molecular Sciences. 2010; 11(4):1321-1342. https://doi.org/10.3390/ijms11041321
Chicago/Turabian StyleD’Archivio, Massimo, Carmelina Filesi, Rosaria Varì, Beatrice Scazzocchio, and Roberta Masella. 2010. "Bioavailability of the Polyphenols: Status and Controversies" International Journal of Molecular Sciences 11, no. 4: 1321-1342. https://doi.org/10.3390/ijms11041321
APA StyleD’Archivio, M., Filesi, C., Varì, R., Scazzocchio, B., & Masella, R. (2010). Bioavailability of the Polyphenols: Status and Controversies. International Journal of Molecular Sciences, 11(4), 1321-1342. https://doi.org/10.3390/ijms11041321




