Structural Features and Biological Properties of Ellagitannins in Some Plant Families of the Order Myrtales
Abstract
:1. Introduction
2. Monomeric Ellagitannins
2.1. Simple Ellagitannins
2.2. C-Glycosidic Ellagitannins
2.3. Complex Tannins
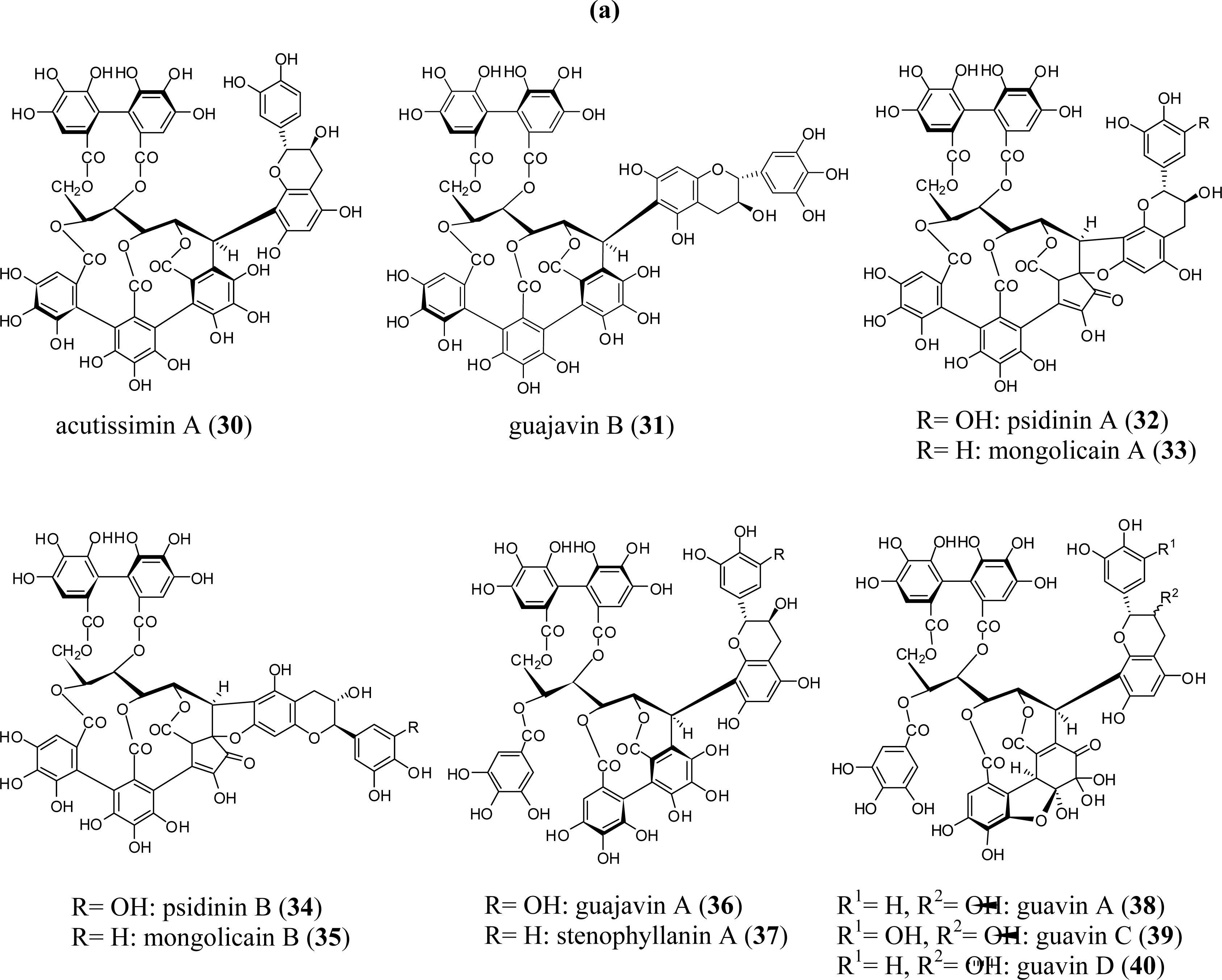
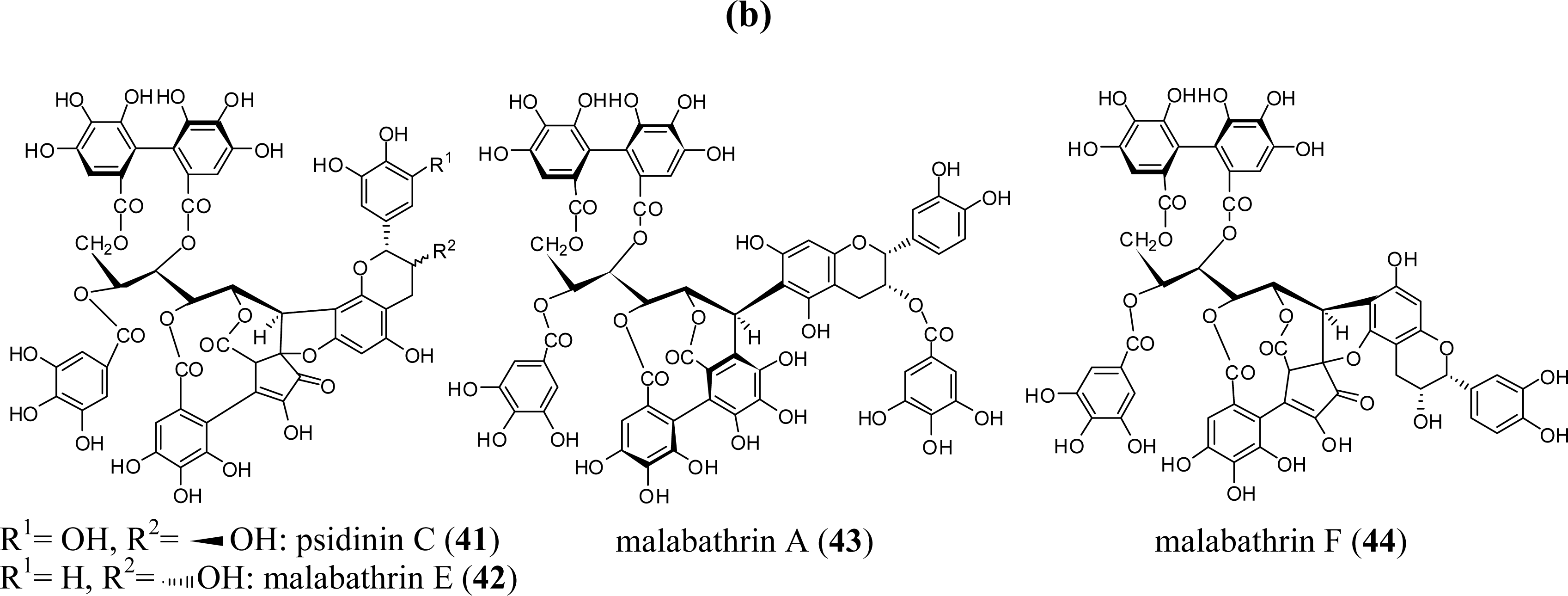
3. Oligomeric Ellagitannins
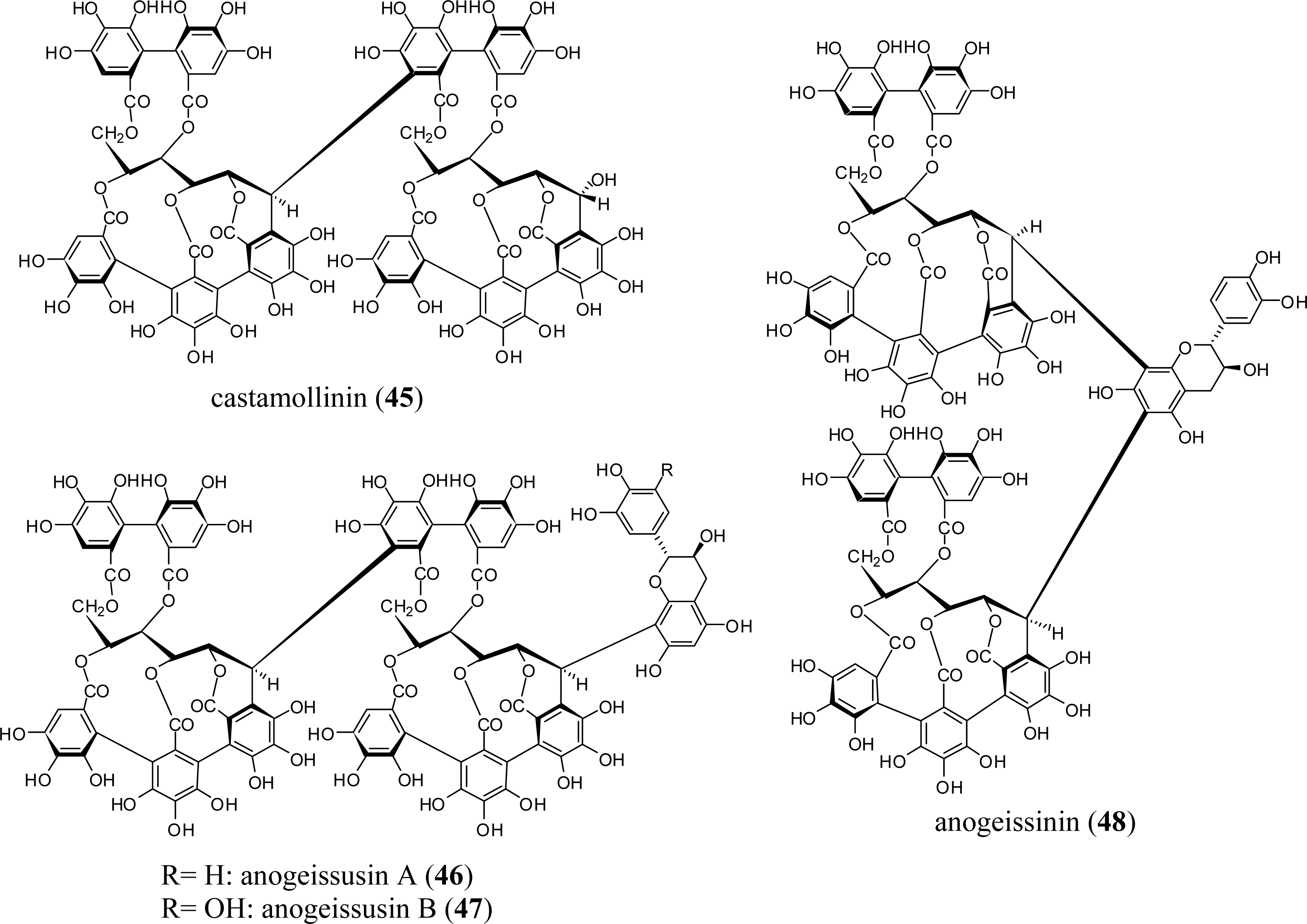
3.1. Oligomers from the Combretaceae
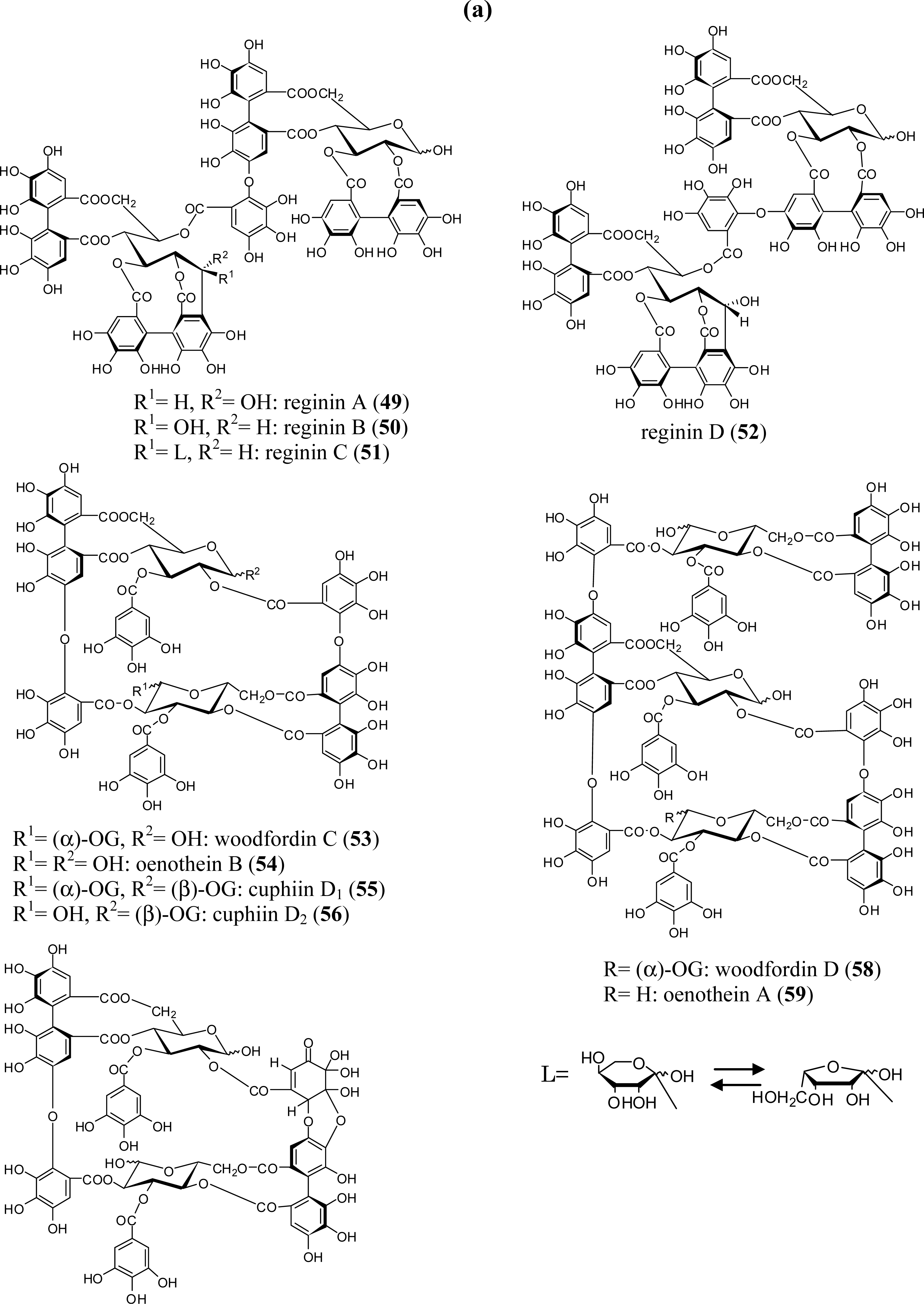
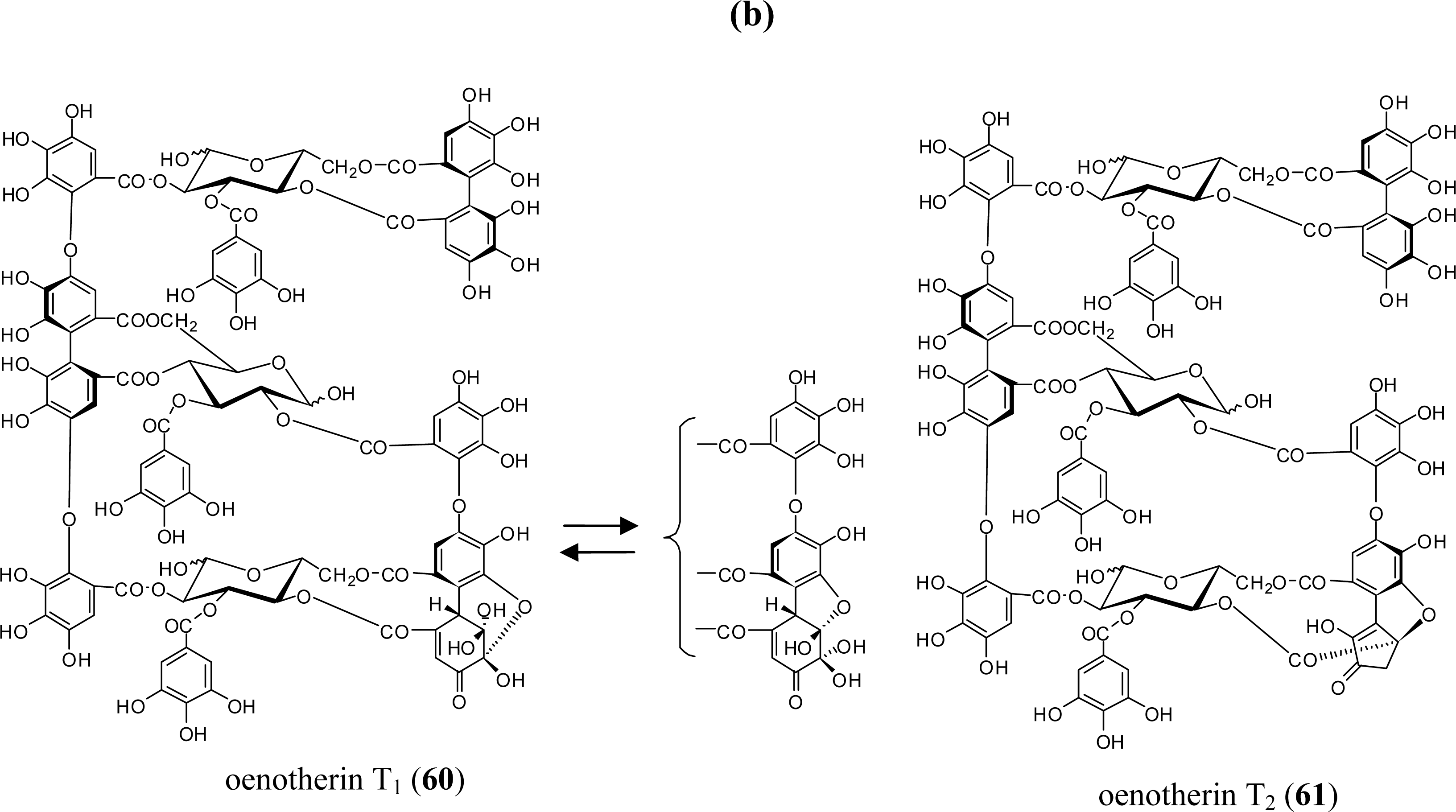
3.2. Oligomers from the Lythraceae and Onagraceae
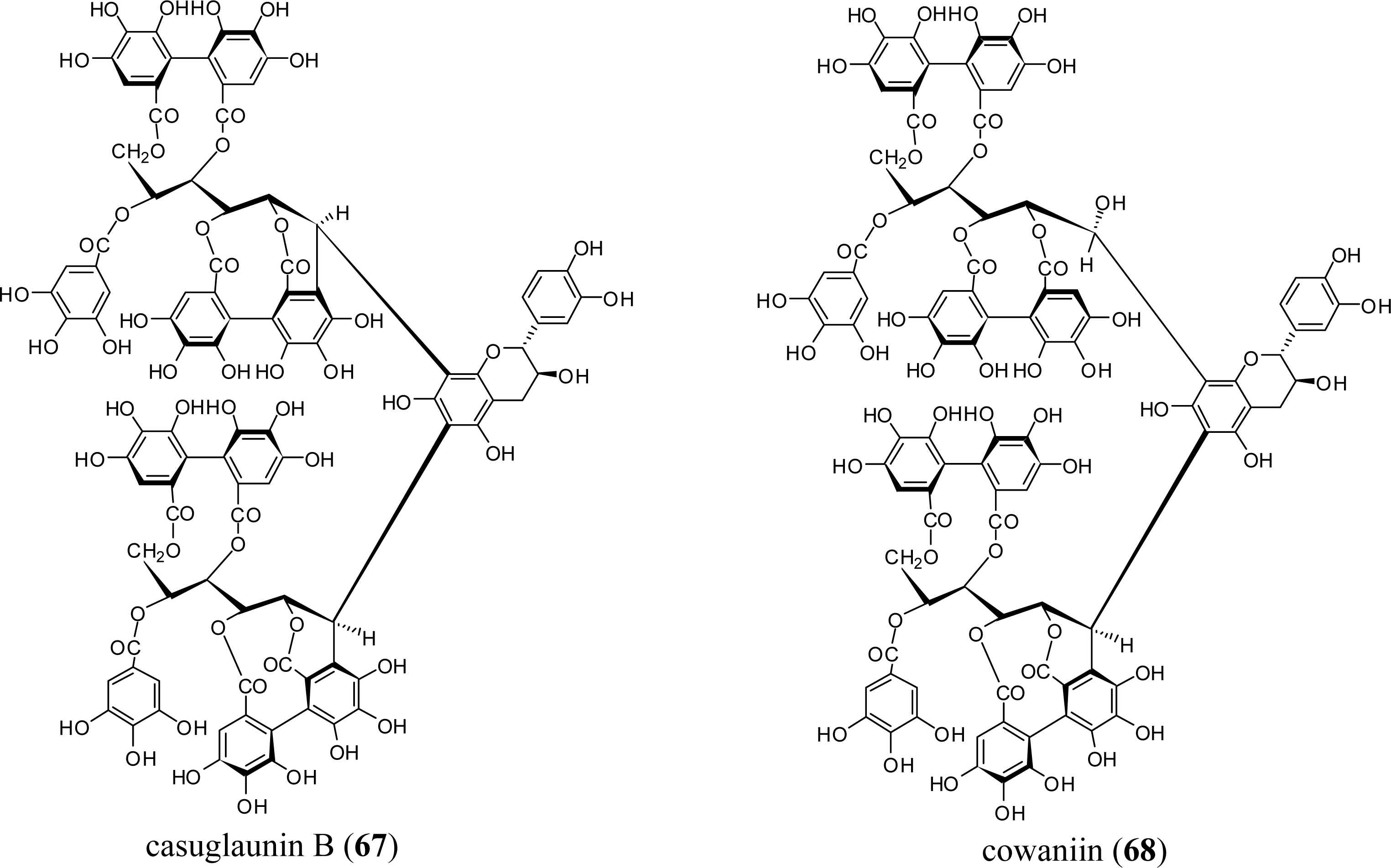
3.3. Oligomers from Myrtaceae
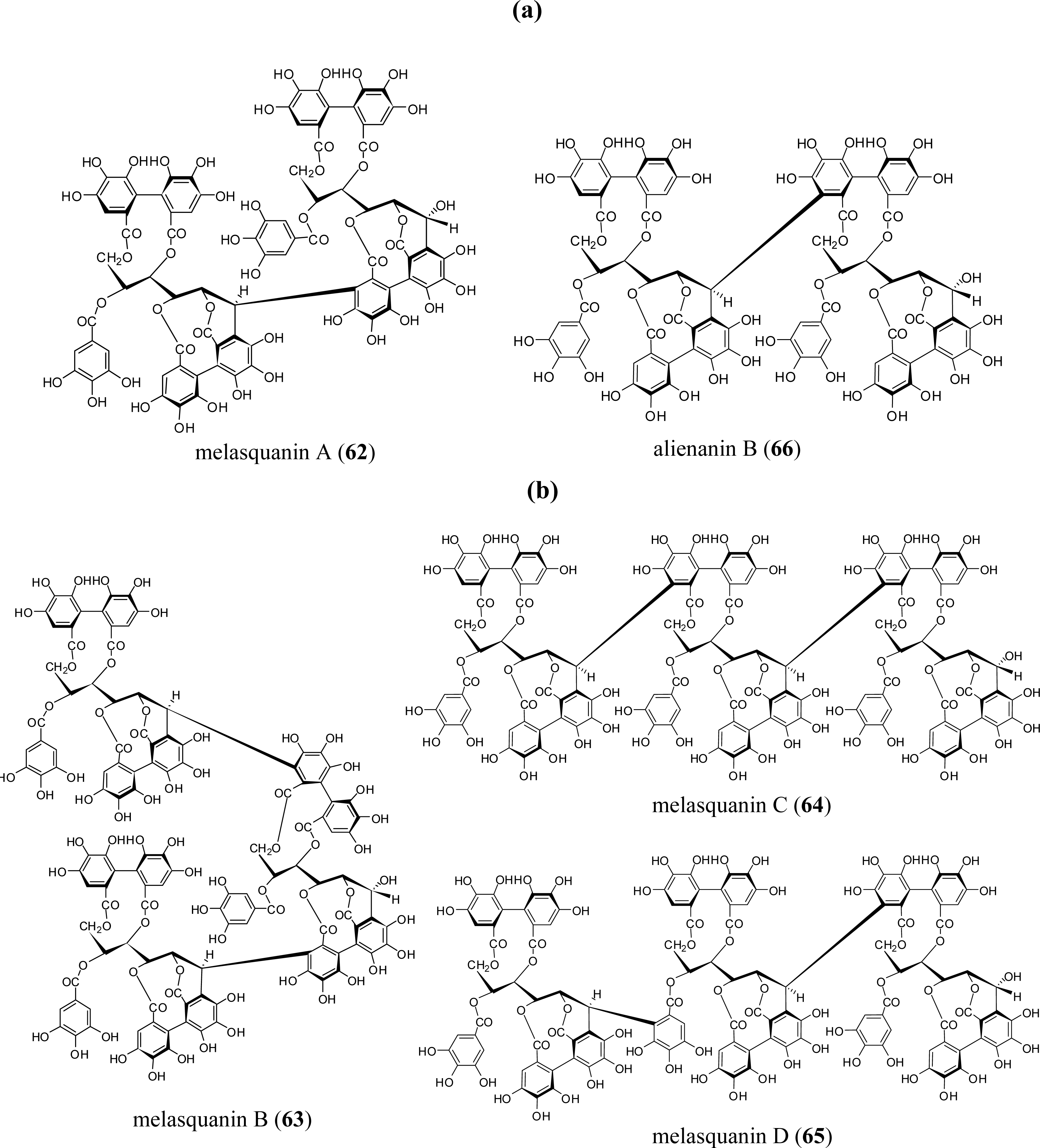
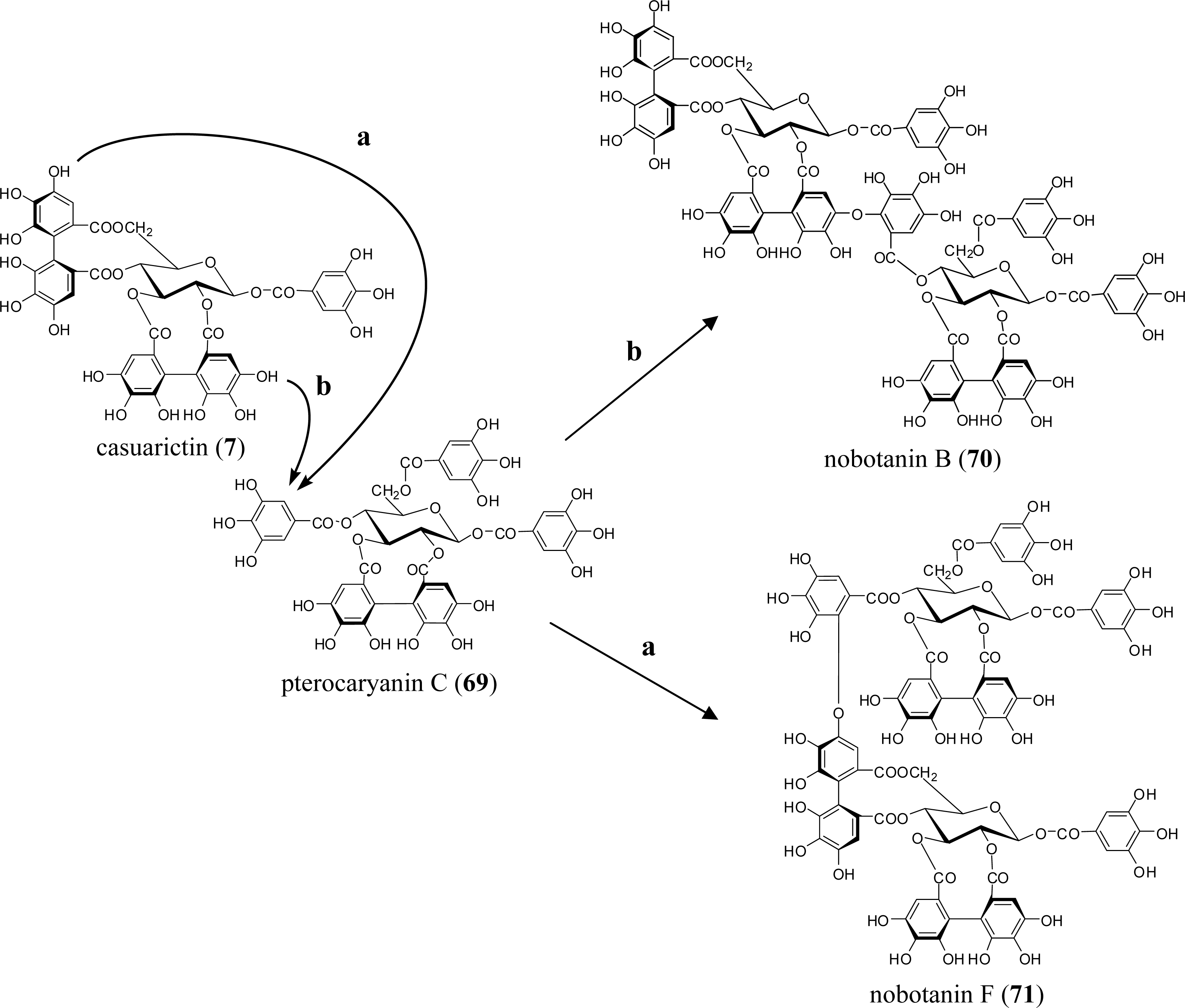
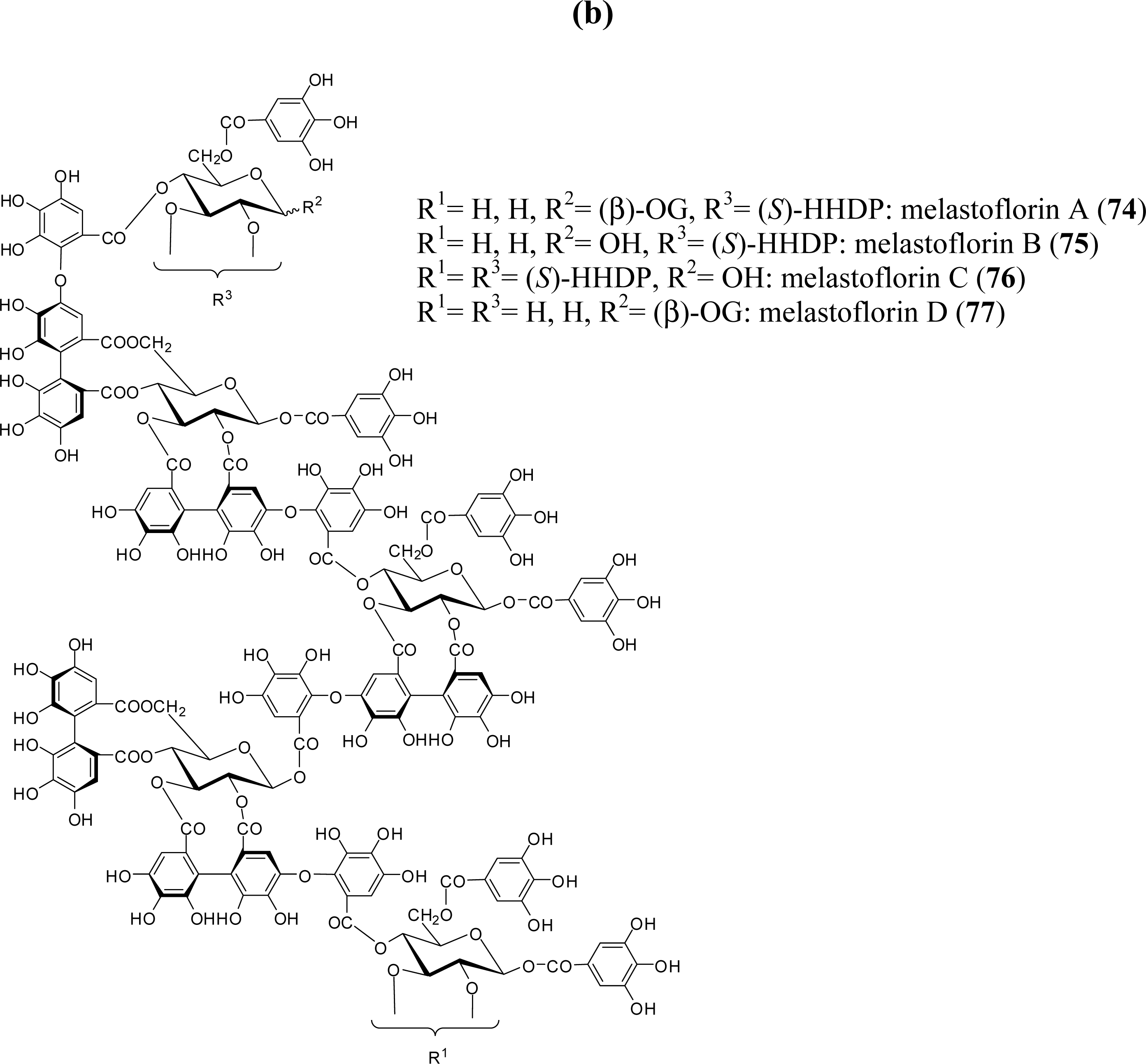
3.4. Oligomers from Melastomataceae
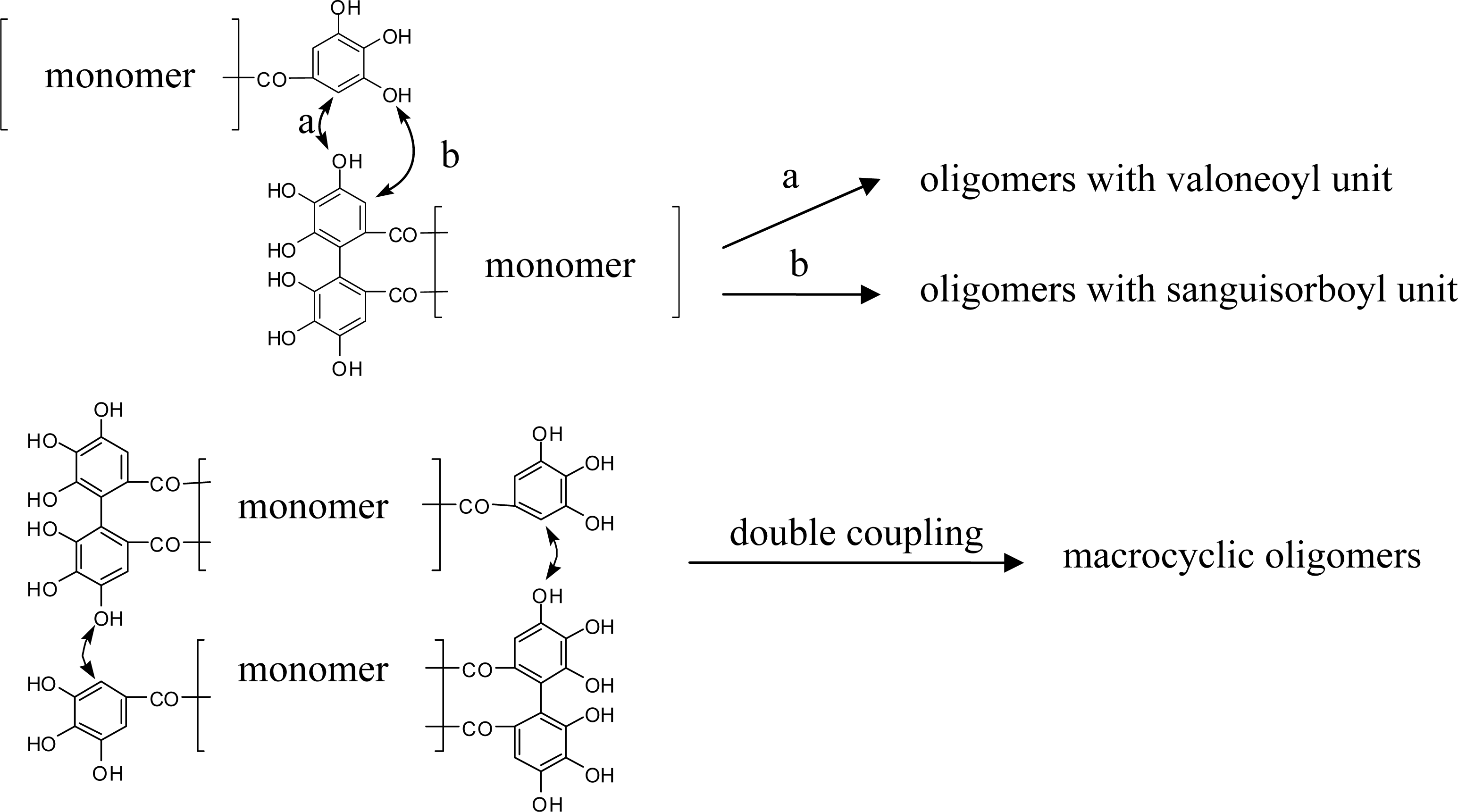
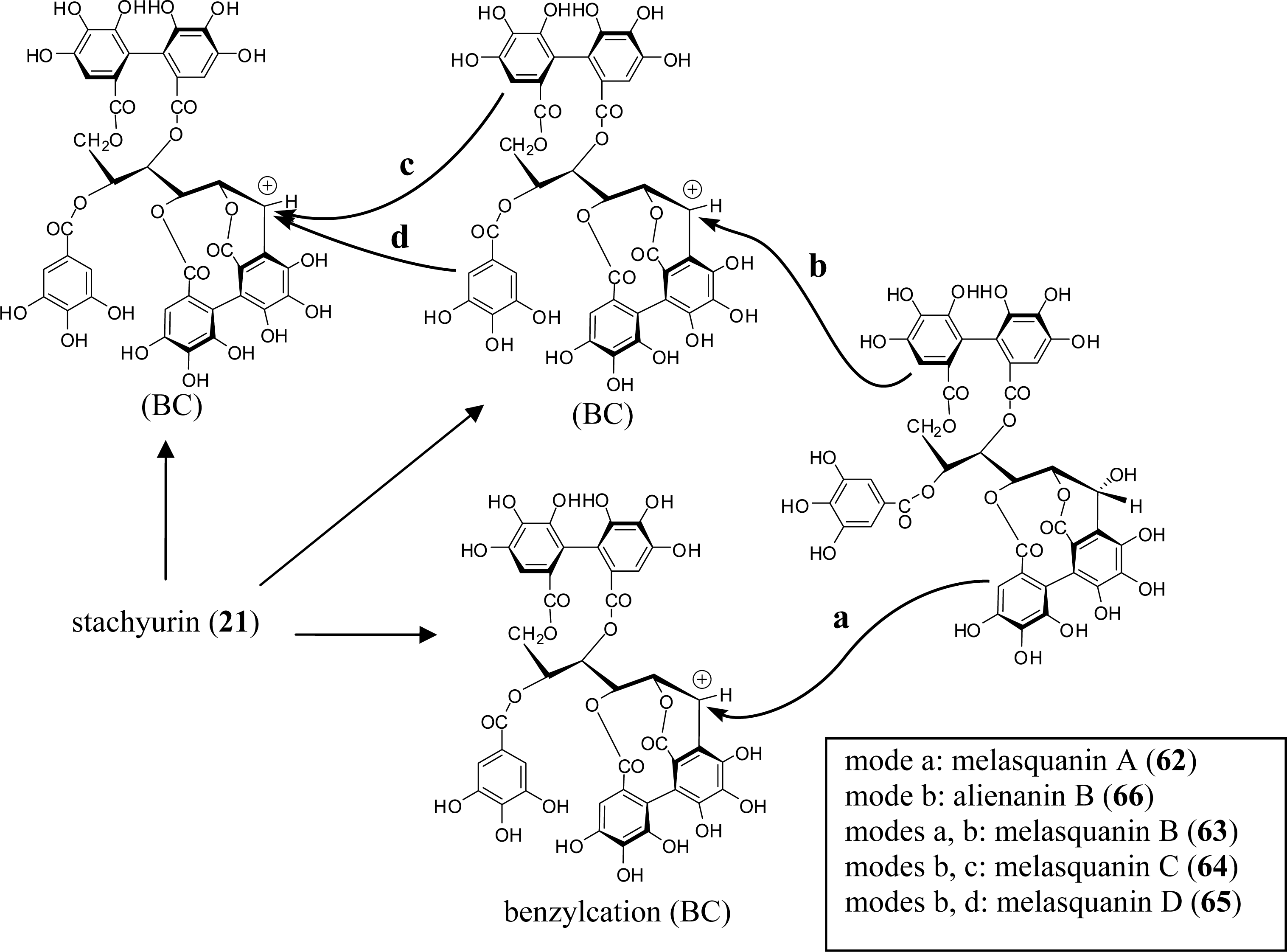
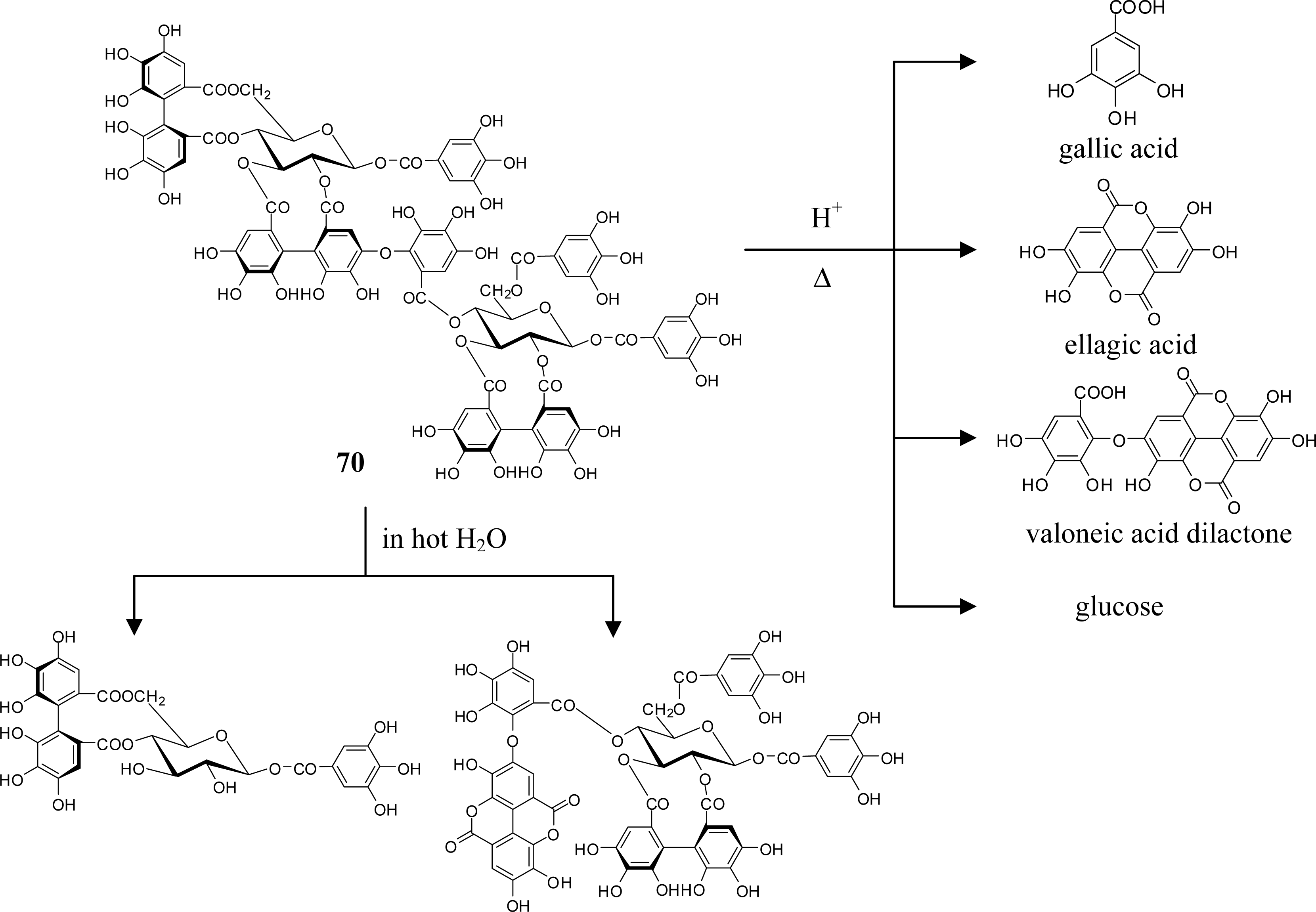
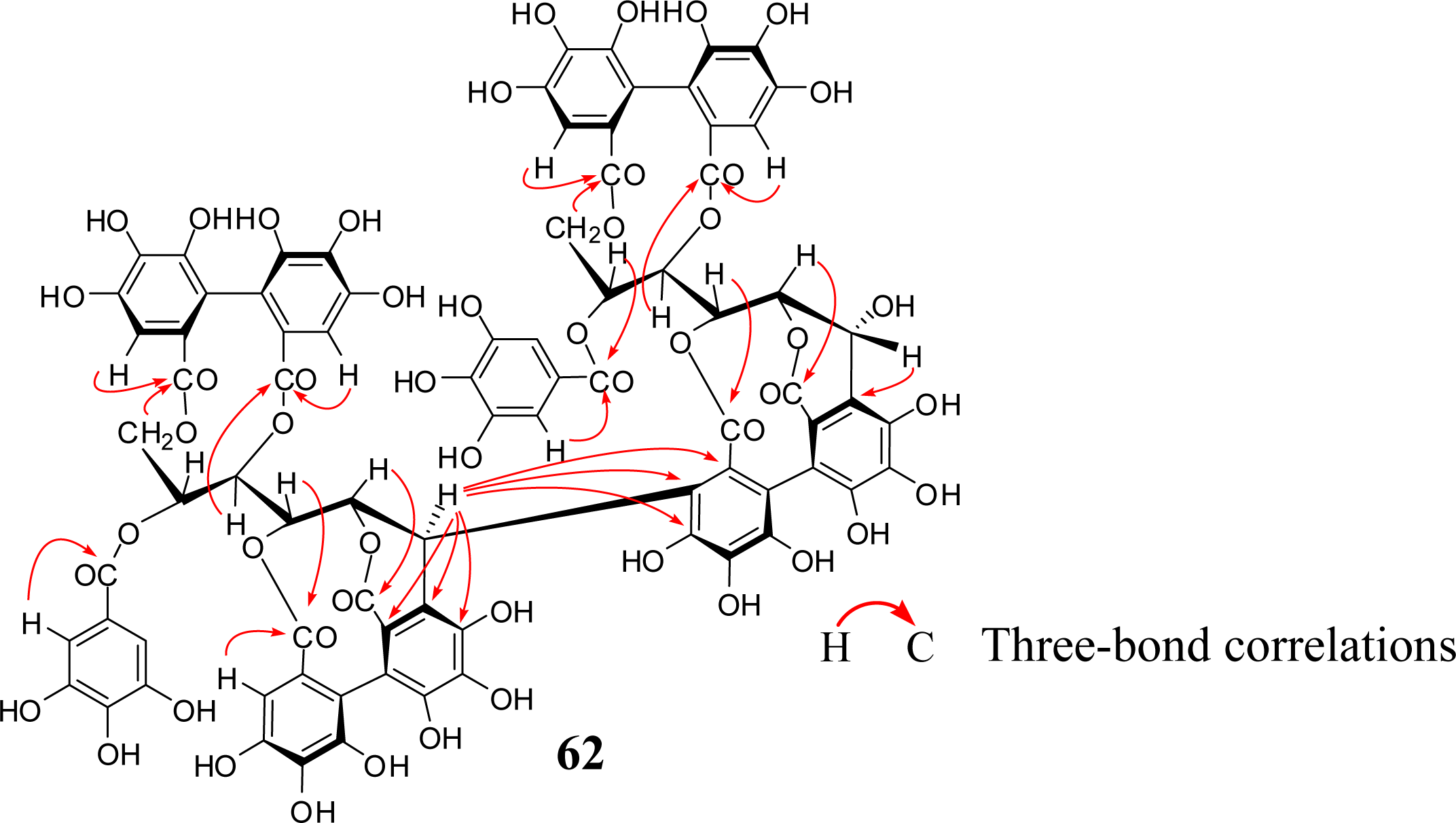
4. Structure Determination of the Oligomeric Ellagitannins
5. Biological Activities of Ellagitannins Found in the Myrtales
5.1. Casuarinin (20), Castalagin (21), and Related Tannins
5.2. Punicalagin (9) and Related Tannins
5.3. Lagerstroemin (29)
5.4. Oenothein B (54) and Related Macrocyclic Oligomers
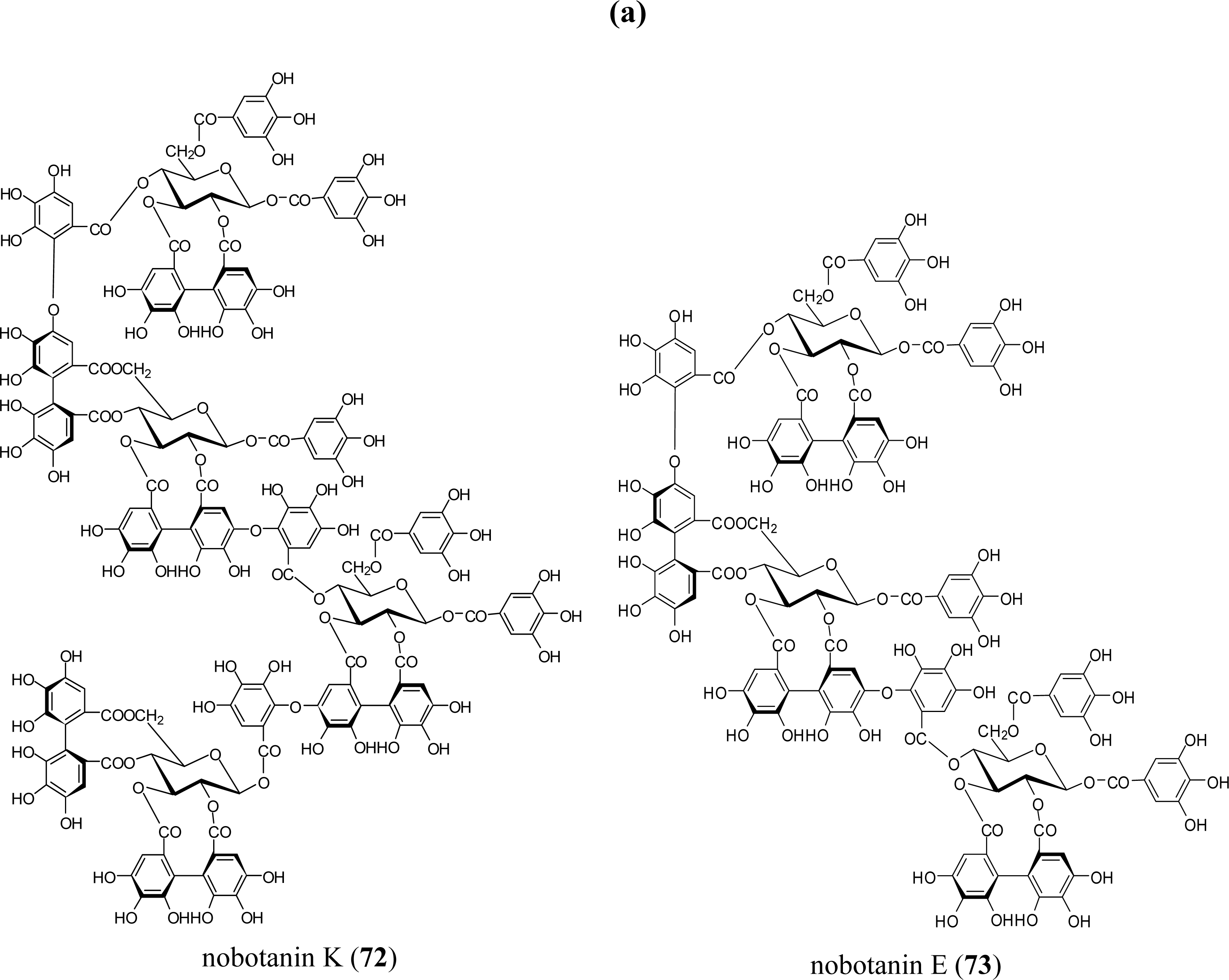
5.5. Nobotanins
6. Conclusions
References and Notes
- Okuda, T; Yoshida, T; Hatano, T. Progress in the Chemistry of Organic Natural Products; Springer: New York, NY, USA, 1995. [Google Scholar]
- Haslam, E. Plant Polyphenols; Cambridge University Press: Cambridge, UK, 1989. [Google Scholar]
- Okuda, T; Yoshida, T; Hatano, T. Classification of oligomeric hydrolysable tannins and specificity of their occurrence in plants. Phytochemistry 1993, 32, 507–521. [Google Scholar]
- Bremer, B; Bremer, K; Chase, MW; Reveal, JL; Soltis, DE; Soltis, PS; Stevens, PF; Anderberg, AA; Fay, MF; Goldblatt, P; Judd, WS; Källersjö, M; Kårehed, J; Kron, KA; Lundberg, J; Nickrent, DL; Olmstead, RG; Oxelman, B; Pires, JC; Rodman, JE; Rudall, PJ; Savolainen, V; Sytsma, KJ; Bank, MVD; Wurdack, K; Xiang, JQY; Zmarzty, S. An update of the angiosperm phylogeny group classification for the orders and families of flowering plants: APG II. Bot. J. Linn. Soc 2003, 141, 399–436. [Google Scholar]
- Niemetz, R; Gross, GG. Enzymology of gallotannin and ellagitannin biosynthesis. Phytochemistry 2005, 66, 2001–2011. [Google Scholar]
- Okuda, T; Yoshida, T; Ashida, M. Casuarictin and casuarinin, two new ellagitannins from Casuarina stricta. Heterocycles 1981, 16, 1681–1685. [Google Scholar]
- Yoshida, T; Okuda, T; Koga, T; Toh, N. Absolute configurations of chebulic, chebulinic and chebulagic acid. Chem. Pharm. Bull 1982, 30, 2655–2658. [Google Scholar]
- Okuda, T; Yoshida, T; Hatano, T; Koga, T; Toh, N; Kuriyama, K. Circular dichroism of hydrolyzable tannins. I. Ellagitannins and gallotannins. Tetrahedr. Lett 1982, 23, 3937–3940. [Google Scholar]
- Hatano, T; Okonogi, A; Yazaki, K; Okuda, T. Trapanins A and B: Oligomeric hydrolyzable tannins from Trapa japonica FLEROV. Chem. Pharm. Bull 1990, 38, 2707–2711. [Google Scholar]
- Yoshida, T; Arioka, H; Fujita, T; Chen, XM; Okuda, T. Monomeric and dimeric hydrolysable tannins from two melastomataceous species. Phytochemistry 1994, 37, 863–866. [Google Scholar]
- Yoshida, T; Haba, K; Nakata, F; Okano, Y; Shingu, T; Okuda, T. Tannins and related polyphenols of Melastomataceous plants. III. Nobotanins G, H and I, dimeric hydrolyzable tannins from Heterocentron roseum. Chem. Pharm. Bull 1992, 40, 66–71. [Google Scholar]
- Yoshida, T; Nakata, F; Hosotani, K; Nitta, A; Okuda, T. Dimeric hydrolysable tannins from Melastoma malabathricum. Phytochemistry 1992, 31, 2829–2833. [Google Scholar]
- Yoshida, T; Ohbayashi, H; Ishihara, K; Ohwashi, W; Haba, K; Okano, Y; Shingu, T; Okuda, T. Tannins and related polyphenols of Melastomataceous plants. I. Hydrolyzable tannins from Tibouchina semidecandra COGN. Chem. Pharm. Bull 1991, 39, 2233–2240. [Google Scholar]
- Mahmoud, II; Moharram, FA; Marzouk, MSA; Linscheid, MW; Saleh, MI. Polyphenolic constituents of Callistemon lanceolatus leaves. Pharmazie 2002, 57, 494–496. [Google Scholar]
- Yoshida, T; Maruyama, T; Nitta, A; Okuda, T. Eucalbanins A, B and C, monomeric and dimeric hydrolyzable tannins from Eucalyptus alba REINW. Chem. Pharm. Bull 1992, 40, 1750–1754. [Google Scholar]
- Santos, SC; Waterman, PG. Polyphenols from Eucalyptus consideniana and Eucalyptus viminalis. Fitoterapia 2001, 72, 95–97. [Google Scholar]
- Hou, AJ; Liu, YZ; Yang, H; Lin, ZW; Sun, HD. Hydrolyzable tannins and related polyphenols from Eucalyptus globulus. J. Asian Nat. Prod. Res 2000, 2, 205–212. [Google Scholar]
- Okamura, H; Mimura, A; Yakou, Y; Niwano, M; Takahara, Y. Antioxidant activity of tannins and flavonoids in Eucalyptus rostrata. Phytochemistry 1993, 33, 557–561. [Google Scholar]
- Yoshimura, M; Amakura, Y; Tokuhara, M; Yoshida, T. Polyphenolic compounds isolated from the leaves of Myrtus communis. J. Nat. Med 2008, 62, 366–368. [Google Scholar]
- Marzouk, MSA; Moharram, FA; Mohamed, MA; Gamal Eldeen, AM; Aboutabl, EA. Anticancer and antioxidant tannins from Pimenta dioica leaves. Z. Naturforsch., C, J. Biosci 2007, 62, 526–536. [Google Scholar]
- Nonaka, G; Aiko, Y; Aritake, K; Nishioka, I. Tannins and related compounds. CXIX: Samarangenins A and B, novel proanthocyanidins with doubly bonded structures, from Syzygium samarangens and S. aqueum. Chem. Pharm. Bull 1992, 40, 2671–2673. [Google Scholar]
- Tanaka, T; Orii, Y; Nonaka, G; Nishioka, I. Tannins and related compounds. CXXIII: Chromone, acetophenone and phenylpropanoid glycosides and their galloyl and/or hexahydroxydiphenoyl esters from the leaves of Syzygium aromaticum MERR.et PERRY. Chem. Pharm. Bull 1993, 41, 1232–1237. [Google Scholar]
- Liu, Y; Wang, C; Han, Q; Yu, B; Ding, G. Study on chemical constituents of Chamaenerion angustifolium II. Tannins and related polyphenolic compounds. Zhong Cao Yao 2003, 34, 967–969. [Google Scholar]
- Miyamoto, K; Kishi, N; Koshiura, R; Yoshida, T; Hatano, T; Okuda, T. Relationship between the structures and the antitumor activities of tannins. Chem. Pharm. Bull 1987, 35, 814–822. [Google Scholar]
- Yoshida, T; Chou, T; Shingu, T; Okuda, T. Oenotheins D, F and G, hydrolysable tannin dimers from Oenothera laciniata. Phytochemistry 1995, 40, 555–561. [Google Scholar]
- Taniguchi, S; Imayoshi, Y; Yoshida, T; Hatano, T. A new trimeric hydrolyzable tannin, oenotherin T2, isolated from aerial parts of Oenothera tetraptera Cav. Heterocycles 2009, 79, 617–626. [Google Scholar]
- Jossang, A; Pousset, JL; Bodo, B. Combreglutinin, a hydrolyzable tannin from Combretum glutinosum. J. Nat. Prod 1994, 57, 732–737. [Google Scholar]
- Asres, K; Bucar, F; Knauder, E; Yardley, V; Kendrick, H; Croft, SL. In vitro antiprotozoal activity of extract and compounds from the stem bark of Combretum molle. Phytother. Res 2001, 15, 613–617. [Google Scholar]
- Lin, TC; Ma, YT; Wu, J; Hsu, FL. Tannins and related compounds from Quisqualis indica. J. Chin. Chem. Soc. (Taipei) 1997, 44, 151–155. [Google Scholar]
- Lin, TC; Hsu, FL. Tannins and related compounds from Terminalia arborea. Chin. Pharm. J. (Taipei) 1996, 48, 167–175. [Google Scholar]
- Lin, TC; Ma, YT; Hsu, FL. Tannins from the bark of Terminalia arjuna. Chin. Pharm. J. (Taipei) 1996, 48, 25–35. [Google Scholar]
- Liu, M; Katerere, DR; Gray, AI; Seidel, V. Phytochemical and antifungal studies on Terminalia mollis and Terminalia brachystemma. Fitoterapia 2009, 80, 369–373. [Google Scholar]
- Tanaka, T; Morita, A; Nonaka, G; Lin, T; Nishioka, I; Ho, FC. Tannins and related compounds. CIII. Isolation and characterizatino of new monomeric, dimeric and trimeric ellagitannins, calamansanin and calamanins A, B and C, from Terminalia calamansanai (BLANCO) ROLFE. Chem. Pharm. Bull 1991, 39, 60–63. [Google Scholar]
- Tanaka, T; Nonaka, G; Nishioka, I. Tannins and related compounds. XLII: Isolation and characterization of four new hydrolyzable tannins, terflavins A and B, tergallagin and tercatain from the leaves of Terminalia catappa L. Chem. Pharm. Bull 1986, 34, 1039–1049. [Google Scholar]
- Lin, TC; Nonaka, G; Nishioka, I; Ho, FC. Tannins and related compounds. CII: Structures of terchebulin, an ellagitannin having a novel tetraphenylcarboxylic acid(terchebulic acid) moiety, and biogenetically related tannins from Terminalia chebula RETZ. Chem. Pharm. Bull 1990, 38, 3004–3008. [Google Scholar]
- Siriporn, B; Atchima, B. Antimicrobial activity of tannins from Terminalia citrina. Planta Med 1995, 61, 365–366. [Google Scholar]
- Conrad, J; Vogler, B; Reeb, S; Klaiber, I; Papajewski, S; Roos, G; Vasquez, E; Setzer, MC; Kraus, W. Isoterchebulin and 4,6-O-isoterchebuloyl-D-glucose, novel hydrolyzable tannins from Terminalia macroptera. J. Nat. Prod 2001, 64, 294–299. [Google Scholar]
- Marzouk, MSA; El-Toumy, SAA; Moharram, FA; Shalaby, NMM; Ahmed, AAE. Pharmacologically active ellagitannins from Terminalia myriocarpa. Planta Med 2002, 68, 523–527. [Google Scholar]
- Martino, V; Morales, J; Martinez Irujo, JJ; Font, MMA; Coussio, J. Two ellagitannins from the leaves of Terminalia triflora with inhibitory activity on HIV-1 reverse transcriptase. Phytother. Res 2004, 18, 667–669. [Google Scholar]
- Tanaka, T; Nonaka, G; Nishioka, I. Tannins and related compounds. XL: Revision of the structures of punicalin and punicalagin, and isolation and characterization of 2-O-galloylpunicalin from the bark of Punica granatum L. Chem. Pharm. Bull 1986, 34, 650–655. [Google Scholar]
- Okuda, T; Mori, K; Seno, K; Hatano, T. Constituents of Geranium thunbergii Sieb. et Zucc. VII. High-performance reversed-phase liquid chromatography of hydrolysable tannins and related polyphenols. J. Chromatogr 1979, 171, 313–320. [Google Scholar]
- Yoshida, T; Amakura, Y; Liu, YZ; Okuda, T. Tannins and related polyphenols of euphorbiaceous plants. XI. Three new hydrolyzable tannins and a polyphenol glucoside from Euphorbia humifusa. Chem. Pharm. Bull 1994, 42, 1803–1807. [Google Scholar]
- Lin, TC; Tanaka, T; Nonaka, G; Nishioka, I; Young, TJ. Tannins and related compounds. CVIII: Isolation and characterization of novel complex tannins (flavono-ellagitannins), anogeissinin and anogeissusins A and B, from Anogeissus acuminata (ROXB ex DC.) GUILL. et PERR. var. lanceolata WALL. ex CLARKE. Chem. Pharm. Bull 1991, 39, 1144–1147. [Google Scholar]
- Shuaibu, MN; Pandey, K; Wuyep, PA; Yanagi, T; Hirayama, K; Ichinose, A; Tanaka, T; Kouno, I. Castalagin from Anogeissus leiocarpus mediates the killing of Leishmania in vitro. Parasitol. Res 2008, 103, 1333–1338. [Google Scholar]
- Lin, TC; Hsu, FL; Cheng, JT. Antihypertensive activity of corilagin and chebulinic acid, tannins from Lumnitzera racemosa. J. Nat. Prod 1993, 56, 629–632. [Google Scholar]
- Itakura, Y; Habermehl, G; Mebs, D. Tannins occurring in the toxic Brazilian plant Thiloa glaucocarpa. Toxicon 1987, 25, 1291–1300. [Google Scholar]
- Xu, Y; Sakai, T; Tanaka, T; Nonaka, G; Nishioka, I. Tannins and related compounds. CVI. Preparation of aminoalditol derivatives of hydrolyzable tannins having α-and β-glucopyranose cores, and Its application to the structure elucidation of new tannins, reginins A and B and flosin A, isolated from Lagerstroemia flos-reginae RETZ. Chem. Pharm. Bull 1991, 39, 639–646. [Google Scholar]
- Tanaka, T; Tong, HH; Xu, Y; Ishimaru, K; Nonaka, G; Nishioka, I. Tannins and related compounds. CXVII. Isolation and characterization of three new ellagitannins, lagerstannins A, B and C, having a gluconic acid core, from Lagerstroemia speciosa (L.) PERS. Chem. Pharm. Bull 1992, 40, 2975–2980. [Google Scholar]
- Su, JD; Osawa, T; Kawakishi, S; Namiki, M. Tannin antioxidants from Osbeckia chinensis. Phytochemistry 1988, 27, 1315–1319. [Google Scholar]
- Nonaka, G; Ishimaru, K; Watanabe, M; Nishioka, I; Yamauchi, T; Wan, ASC. Tannins and related compounds. LI: Elucidation of the stereochemistry of the triphenoyl moiety in castalagin and vescalagin, and isolation of 1-O-galloyl Castalagin from Eugenia grandis. Chem. Pharm. Bull 1987, 35, 217–220. [Google Scholar]
- Kasajima, N; Ito, H; Hatano, T; Yoshida, T. Phloroglucinol diglycosides accompanying hydrolyzable tannins from Kunzea ambigua. Phytochemistry 2008, 69, 3080–3086. [Google Scholar]
- Yoshida, T; Ito, H; Yoshimura, M; Miyashita, K; Hatano, T. C-Glucosidic ellagitannin oligomers from Melaleuca squarrosa Donn ex Sm., Myrtaceae. Phytochemistry 2008, 69, 3070–3079. [Google Scholar]
- Gallo, MBC; Rocha, WC; da Cunha, US; Diogo, FA; da Silva, FC; Vieira, PC; Vendramim, JD; Fernandes, JB; da Silva, MFdGF; Batista Pereira, LG. Bioactivity of extracts and isolated compounds from Vitex polygama (Verbenaceae) and Siphoneugena densiflora (Myrtaceae) against Spodoptera frugiperda (Lepidoptera: Noctuidae). Pest Manag. Sci 2006, 62, 1072–1081. [Google Scholar]
- Tanaka, T; Ishida, N; Ishimatsu, M; Nonaka, G; Nishioka, I. Tannins and related compounds. CXVI. Six new complex tannins, guajavins, psidinins and psiguavin from the Bark of Psidium guajava L. Chem. Pharm. Bull 1992, 40, 2092–2098. [Google Scholar]
- Yoshida, T; Nakata, F; Hosotani, K; Nitta, A; Okuda, T. Tannins and related polyphenols of Melastomataceous plants. V. Three new complex tannins from Melastoma malabathricum L. Chem. Pharm. Bull 1992, 40, 1727–1732. [Google Scholar]
- Yoshida, T; Chou, T; Nitta, A; Miyamoto, K; Koshiura, R; Okuda, T. Woodfordin C, a macro-ring hydrolyzable tannin dimer with antitumor activity, and accompanying dimers from Woodfordia fruticosa flowers. Chem. Pharm. Bull 1990, 38, 1211–1217. [Google Scholar]
- Chen, LG; Yen, KY; Yang, LL; Hatano, T; Okuda, T; Yoshida, T. Macrocyclic ellagitannin dimers, cuphiins D1 and D2, and accompanying tannins from Cuphea hyssopifolia. Phytochemistry 1999, 50, 307–312. [Google Scholar]
- Yoshida, T; Chou, T; Matsuda, M; Yasuhara, T; Yazaki, K; Hatano, T; Nitta, A; Okuda, T. Woodfordin D and oenothein A, trimeric hydrolyzable tannins of macro-ring structure with anti-tumor activity. Chem. Pharm. Bull 1991, 39, 1157–1162. [Google Scholar]
- Ducrey, B; Marston, A; Gohring, S; Hartmann, RW; Hostettmann, K. Inhibition of 5 α-reductase and aromatase by the ellagitannins oenothein A and oenothein B from Epilobium species. Planta Med 1997, 63, 111–114. [Google Scholar]
- Taniguchi, S; Imayoshi, Y; Yabu-uchi, R; Ito, H; Hatano, T; Yoshida, T. A macrocyclic ellagitannin trimer, oenotherin T1, from Oenothera species. Phytochemistry 2002, 59, 191–195. [Google Scholar]
- Yoshida, T; Hatano, T; Ito, H. Chemistry and function of vegetable polyphenols with high molecular weights. BioFactors 2000, 13, 121–125. [Google Scholar]
- Lee, MH; Nishimoto, S; Yang, LL; Yen, KY; Hatano, T; Yoshida, T; Okuda, T. Two macrocyclic hydrolysable tannin dimers from Eugenia uniflora. Phytochemistry 1997, 44, 1343–1349. [Google Scholar]
- Yoshida, T; Maruyama, T; Nitta, A; Okuda, T. An hydrolysable tannin and accompanying polyphenols from Melaleuca leucadendron. Phytochemistry 1996, 42, 1171–1173. [Google Scholar]
- Ito, H; Miyake, M; Nishitani, E; Miyashita, K; Yoshimura, M; Yoshida, T; Takasaki, M; Konoshima, T; Kozuka, M; Hatano, T. Cowaniin, a C-glucosidic ellagitannin dimer linked through catechin from Cowania mexicana. Chem. Pharm. Bull 2007, 55, 492–494. [Google Scholar]
- Isaza, JH; Ito, H; Yoshida, T. Tetrameric and pentameric ellagitannins from Monochaetum multiflorum. Heterocycles 2000, 55, 29–32. [Google Scholar]
- Yoshida, T; Haba, K; Arata, R; Nakata, F; Shingu, T; Okuda, T. Tannins and related polyphenols of Melastomataceous plants. VII: Nobotanins J and K, trimeric and tetrameric hydrolyzable tanins from Heterocentron roseum. Chem. Pharm. Bull 1995, 43, 1101–1106. [Google Scholar]
- Cheng, HY; Lin, CC; Lin, TC. Antiherpes simplex virus type 2 activity of casuarinin from the bark of Terminalia arjuna Linn. Antiviral Res 2002, 55, 447–455. [Google Scholar]
- Po-Lin, K; Ya-Ling, H; Ta-Chen, L; Liang-Tzung, L; Jiunn-Kae, C; Chun-Ching, L. Casuarinin from the bark of Terminalia arjuna induces apoptosis and cell cycle arrest in human breast adenocarcinoma MCF-7 cells. Planta Medica 2005, 71, 237–243. [Google Scholar]
- Kolodziej, H; Kayser, O; Kiderlen, AF; Ito, H; Hatano, T; Yoshida, T; Foo, LY. Antileishmanial activity of hydrolyzable tannins and their modulatory effects on nitric oxide and tumor necrosis factor-α release in macrophages in vitro. Planta Med 2001, 67, 825–832. [Google Scholar]
- Toda, M; Kawabata, J; Kasai, T. α-Glucosidase inhibitors from clove (Syzgium aromaticum). Biosci. Biotechnol. Biochem 2000, 64, 294–298. [Google Scholar]
- Gao, H; Huang, YN; Gao, B; Kawabata, J. Chebulagic acid is a potent α-glucosidase inhibitor. Biosci. Biotechnol. Biochem 2008, 72, 601–603. [Google Scholar]
- Reddy, DB; Reddy, TCM; Jyotsna, G; Sharan, S; Priya, N; Lakshmipathi, V; Reddanna, P. Chebulagic acid, a COX-LOX dual inhibitor isolated from the fruits of Terminalia chebula Retz., induces apoptosis in COLO-205 cell line. J. Ethnopharmacol 2009, 124, 506–512. [Google Scholar]
- Reddy, DB; Reddanna, P. Chebulagic acid (CA) attenuates LPS-induced inflammation by suppressing NF-kappaB and MAPK activation in RAW 264.7 macrophages. Biochem. Biophys. Res. Commun 2009, 381, 112–117. [Google Scholar]
- Lin, CC; Hsu, YF; Lin, TC. Effects of punicalagin and punicalin on carrageenan-induced inflammation in rats. Am. J. Chin. Med 1999, 27, 371–376. [Google Scholar]
- Lin, CC; Hsu, YF; Lin, TC; Hsu, HY. Antioxidant and hepatoprotective effects of punicalagin and punicalin on acetaminophen-induced liver damage in rats. Phytother. Res 2001, 15, 206–212. [Google Scholar]
- Chen, PS; Li, JH; Liu, TY; Lin, TC. Folk medicine Terminalia catappa and its major tannin component, punicalagin, are effective against bleomycin-induced genotoxicity in Chinese hamster ovary cells. Cancer Lett 2000, 152, 115–122. [Google Scholar]
- Chen, PS; Li, JH. Chemopreventive effect of punicalagin, a novel tannin component isolated from Terminalia catappa, on H-ras-transformed NIH3T3 cells. Toxicol. Lett 2006, 163, 44–53. [Google Scholar]
- Hayashi, T; Maruyama, H; Kasai, R; Hattori, K; Takasuga, S; Hazeki, O; Yamasaki, K; Tanaka, T. Ellagitannins from Lagerstroemia speciosa as activators of glucose transport in fat cells. Planta Med 2002, 68, 173–175. [Google Scholar]
- Hattori, K; Sukenobu, N; Sasaki, T; Takasuga, S; Hayashi, T; Kasai, R; Yamasaki, K; Hazeki, O. Activation of insulin receptors by lagerstroemin. J. Pharmacol. Sci 2003, 93, 69–73. [Google Scholar]
- Bai, N; He, KR Marc; Zheng, B; Chen, X; Shao, Z; Peng, T; Zheng, Q. Active compounds from Lagerstroemia speciosa, insulin-like glucose uptake-stimulatory/inhibitory and adipocyte differentiation-inhibitory activities in 3T3-L1 cells. J. Agric. Food Chem 2008, 56, 11668–11674. [Google Scholar]
- Kadota, S; Takamori, Y; Khin, NN; Kikuchi, T; Tanaka, K; Ekimoto, H. Constituents of the Leaves of Woodfordia fruticosa KURZ. I: Isolation, structure, and proton and Carbon-13 nuclear magnetic resonance signal assignments of woodfruticosin (Woodfordin C), an inhibitor of deoxyribonucleic acid topoisomerase. Chem. Pharm. Bull 1990, 38, 2687–2697. [Google Scholar]
- Lee, MH; Chiou, JF; Yen, KY; Yang, LL. EBV DNA polymerase inhibition of tannins from Eugenia uniflora. Cancer Lett 2000, 154, 131–136. [Google Scholar]
- Kiss, A; Kowalski, J; Melzig, MF. induction of neutral endopeptidase activity in PC-3 cells but angiotensin converting enzyme is not influenced. Phytomedicine 2006, 13, 284–289. [Google Scholar]
- Wang, CC; Chen, LG; Yang, LL. In vitro immunomodulatory effects of cuphiin D1 on human mononuclear cells. Anticancer Res 2002, 22, 4233–4236. [Google Scholar]
- Wang, CC; Chen, LG; Yang, LL. Cuphiin D1, the macrocyclic hydrolyzable tannin induced apoptosis in HL-60 cell line. Cancer Lett 2000, 149, 77–83. [Google Scholar]
- Tsai, YJ; Aoki, T; Maruta, H; Abe, H; Sakagami, H; Hatano, T; Okuda, T; Tanuma, S. Mouse mammary tumor virus gene expression is suppressed by oligomeric ellagitannins, novel inhibitors of poly(ADP-ribose) glycohydrolase. J. Biol. Chem 1992, 267, 14436–14442. [Google Scholar]
- Miyamoto, K; Murayama, T; Nomura, M; Hatano, T; Yoshida, T; Furukawa, T; Koshiura, R; Okuda, T. Antitumor activity and interleukin-1 induction by tannins. Anticancer Res 1993, 13, 37–42. [Google Scholar]
- Hatano, T; Kusuda, M; Inada, K; Ogawa, T; Shiota, S; Tsuchiya, T; Yoshida, T. Effects of tannins and related polyphenols on methicillin-resistant Staphylococcus aureus. Phytochemistry 2005, 66, 2047–2055. [Google Scholar]
- Amakura, Y; Yoshimura, M; Sugimoto, N; Yamazaki, T; Yoshida, T. Marker constituents of the natural antioxidant Eucalyptus leaf extract for the evaluation of food additives. Biosci. Biotechnol. Biochem 2009, 73, 1060–1065. [Google Scholar]
- Doyle, B; Griffiths, LA. The metabolism of ellagic acid in the rat. Xenobiotica 1980, 10, 247–256. [Google Scholar]
- Ito, H; Iguchi, A; Hatano, T. Identification of urinary and intestinal bacterial metabolites of ellagitannin geraniin in rats. J. Agric. Food Chem 2008, 56, 393–400. [Google Scholar]
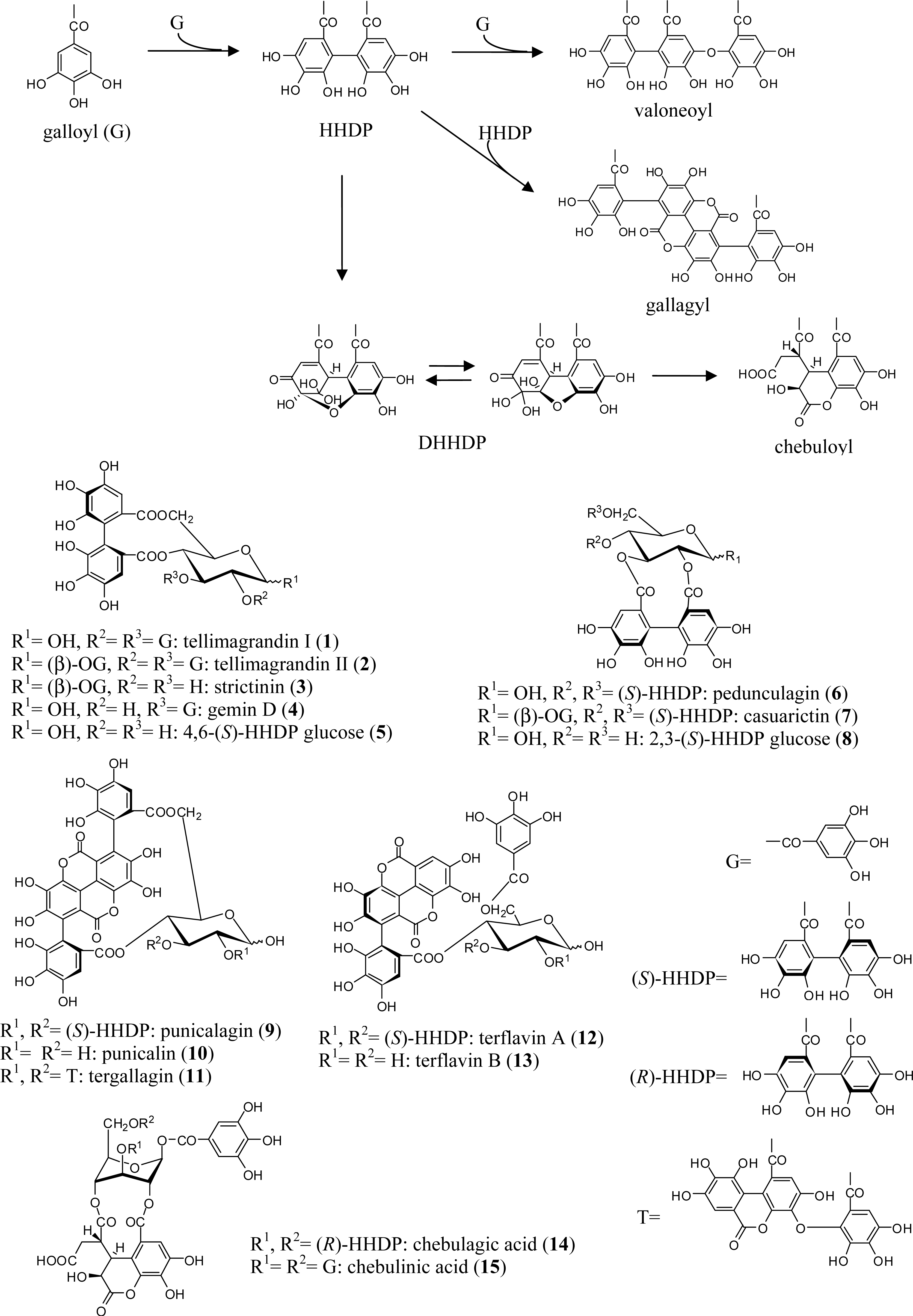
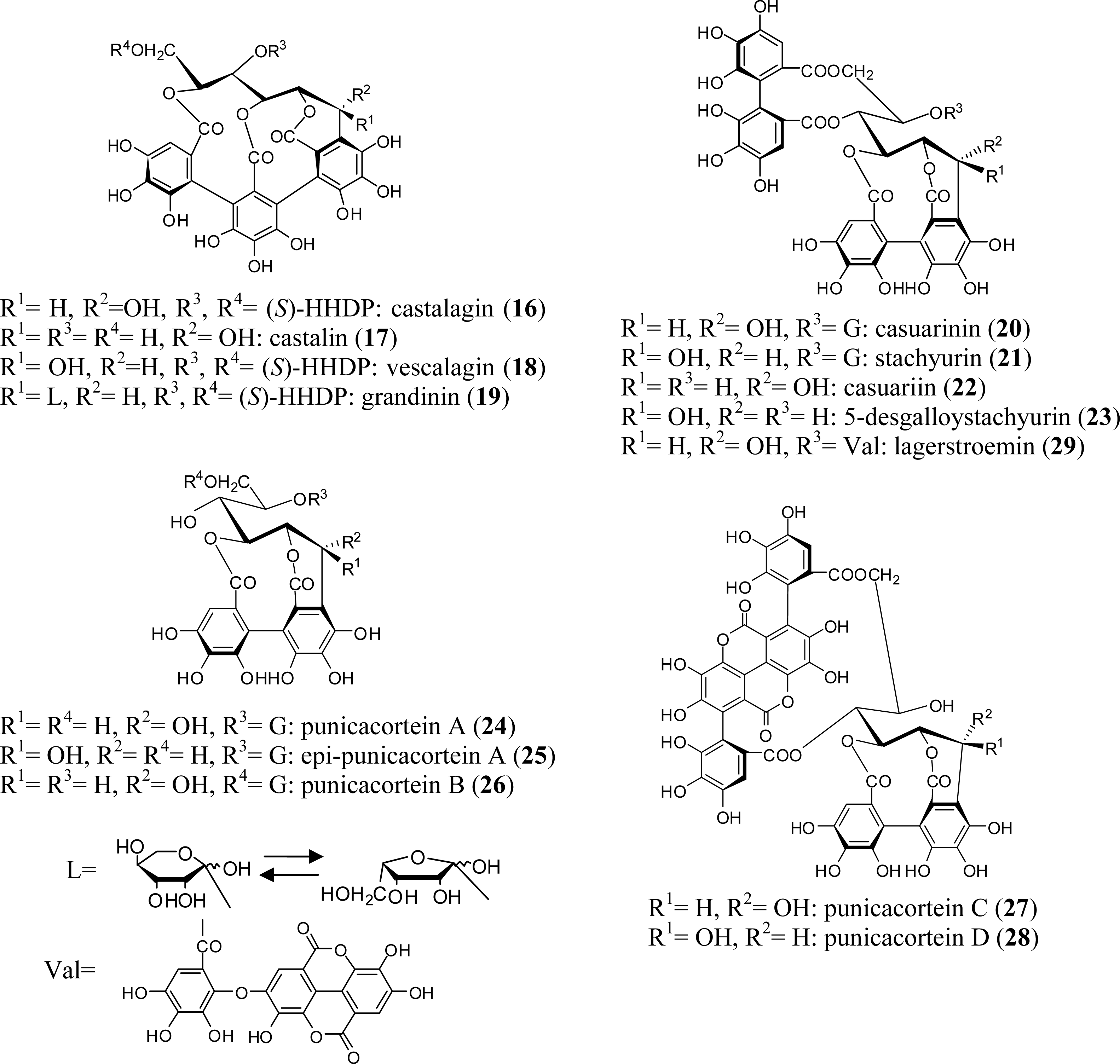
| Tannin | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Plant source [Ref] | |||||||||||||||
| Trapaceae | |||||||||||||||
| Trapa japonica [9] | + | + | + | + | |||||||||||
| Melastomataceae | |||||||||||||||
| Bredia tuberculata [10] | + | + | |||||||||||||
| Heterocentron roseum [11] | + | + | |||||||||||||
| Melastoma malabathricum [12] | + | + | + | ||||||||||||
| M. normale [10] | + | + | + | ||||||||||||
| Tibouchina semidecandra [13] | + | + | + | ||||||||||||
| Myrtaceae | |||||||||||||||
| Callistemon lanceolatus [14] | + | + | + | ||||||||||||
| Eucalyptus alba [15] | + | + | + | + | |||||||||||
| E. consideniana [16] | + | + | + | + | |||||||||||
| E. globulus [17] | + | ||||||||||||||
| E. rostrata [18] | + | ||||||||||||||
| E. viminalis [16] | + | + | + | + | |||||||||||
| Myrtus communis [19] | + | + | |||||||||||||
| Pimenta dioica [20] | + | + | + | + | |||||||||||
| Syzygium aqueum [21] | + | + | + | ||||||||||||
| S. aromaticum [22] | + | + | + | + | + | ||||||||||
| Onagraceae | |||||||||||||||
| Epilobium angustifolium [23] | + | + | + | + | |||||||||||
| Oenothera erythrosepala [24] | + | + | |||||||||||||
| O. laciniata [25] | + | ||||||||||||||
| O. tetraptera [26] | + | + | + | ||||||||||||
| Combretaceae | |||||||||||||||
| Combretum glutinosum [27] | + | + | + | ||||||||||||
| C. molle [28] | + | + | |||||||||||||
| Quisqualis indica [29] | + | + | + | + | + | + | |||||||||
| Terminalia arborea [30] | + | + | + | + | + | ||||||||||
| T. arjuna [31] | + | + | + | ||||||||||||
| T. brachystemma [32] | + | ||||||||||||||
| T. calamansanai [33] | + | + | + | + | + | ||||||||||
| T. catappa [34] | + | + | + | + | + | + | + | + | + | ||||||
| T. chebula [35] | + | + | + | + | + | + | |||||||||
| T. citrina [36] | + | + | |||||||||||||
| T. macroptera [37] | + | + | + | + | |||||||||||
| T. myriocarpa [38] | + | + | |||||||||||||
| T. triflora [39] | + | ||||||||||||||
| Punicaceae | |||||||||||||||
| Punica granatum [40] | + | + | + | + | + | + | |||||||||
| Family | Plant species | C-Glycosidic tannins | Ref. |
|---|---|---|---|
| Combretaceae | Anogeissus acuminata | 16, 17, 18, 19 | [43] |
| Anogeissus leiocarpus | 16 | [44] | |
| Lumnitzera racemosa | 16 | [45] | |
| Terminalia arjuna | 16, 20, 22 | [31] | |
| Terminalia macroptera | 27 | [37] | |
| Terminalia arborea | 28 | [30] | |
| Thiloa glaucocarpa | 16, 18, 20, 21 | [46] | |
| Lythraceae | Lagerstroemia flos-reginea | 16, 18, 20, 21, 22, 23, 24, 29 | [47] |
| Lagerstroemia speciosa | 16, 18, 19, 29 | [48] | |
| Melastomataceae | Osbeckia chinensis | 20, 22, 25 | [49] |
| Tibouchina semidecandra | 16, 18, 20 | [13] | |
| Myrtaceae | Callistemon lanceolatus | 20 | [14] |
| Eucalyptus alba | 21, 22 | [15] | |
| Eugenia grandis | 16, 18 | [50] | |
| Kunzea ambigua | 20 | [51] | |
| Melaleuca squarrosa | 20, 21 | [52] | |
| Pimenta dioica | 16, 18, 20, 22 | [20] | |
| Siphoneugena densiflora | 16, 20 | [53] | |
| Syzygium aqueum | 16, 18, 19 | [21] | |
| Syzygium aromaticum | 20, 22 | [22] | |
| Punicaceae | Punica granatum | 20, 22, 25, 26, 27, 28 | [40] |
| Trapaceae | Trapa japonica | 20 | [9] |
| Biological activity | Compound (source) | Ref. |
|---|---|---|
| Anti-Herpes simplex virus type 2 activity | casuarinin (20) (Terminalia arjuna) | [67] |
| Apoptosis in human breast adenocarcinoma MCF-7 cells | casuarinin (20) | [68] |
| Antileishmanial activity | casuarinin (20), castalagin (16) | [69] |
| castalagin (16) (Anogeissus leiocarpus) | [44] | |
| Antihypertensive activity (rats) | castalagin (16) (Lumnitzera racemosa) | [45] |
| corilagin, chebulinic acid (15) | ||
| α-Glucosidase inhibitor | casuarictin (7) (Syzygium aromaticum) | [70] |
| chebulagic acid (14) (Terminalia chebula) | [71] | |
| Dual inhibitor against COX and 5-LOX | chebulagic acid (14) (T. chebula) | [72] |
| Anti-inflammation in LPS-induced RAW 264.7 cells | chebulagic acid (14) (T. chebula) | [73] |
| Effect on carageenan-induced inflammation | punicalagin (9), punicalin (10) (T. catappa) | [74] |
| Antioxidant and hepatoprotective effects on acetaminophen-induced liver damage in rats | punicalagin (9), punicalin (10) (T. catappa) | [75] |
| Effect against bleomycin-induced genotoxicity in Chinese hamster ovary cells | punicalagin (9) (T. catappa) | [76] |
| Chemopreventive effect on H-ras-transformed NIH3T3 cells | punicalagin (9) (T. catappa) | [77] |
| Inhibitory effect on HIV-1 reverse transcriptase | punicalin (10), 2-O-galloylpunicalin (T. triflora) | [39] |
| Inhibitory effect on CCl4-induced hepatotoxicity | punicalagin (9) (T. myriocarpa) | [38] |
| Activators of glucose transport in fat cells | lagerstroemin (29), reginin A (49) (L. speciosa) | [78] |
| Activation of insulin receptors | lagerstroemin (29) | [79] |
| Insulin-like glucose uptake-stimulatory/inhibitory and adiposities differentiation inhibitory activity in 3T3-L1 cells | lagerstroemin (29) casuarinin (20), casuariin (22), stachyurin (21) | [80] |
| Host-mediated antitumor effect | oenothein B (54) (Oenothera erythrosepala) | [24] |
| Host-mediated antitumor | oenothein B (54) (Woodfordia fruticosa) | [56] |
| woodfordins A-C (53) | ||
| Inhibitor of deoxyribonucleic acid topoisomerase II | woodfruticosin [= woodfordin C (53)] | [81] |
| EBV DNA polymerase inhibitory effect | oenothein B (54) (Eugenia uniflora) | [82] |
| eugeniflorins D1, D2 (57) | ||
| 5α-reductase, aromatase inhibitory effect | oenotheins A (59), B (54) (Epilobium sp) | [59] |
| Induction of neutral endopeptidase activity in PC-3 cells | oenothein B (54) (Epilobium angustifolium) | [83] |
| In vitro immunomodulatory effect on human mononuclear cells | cuphiin D1 (55) (Cuphea sp) | [84] |
| Induce apoptosis in HL-60 cells | cuphiin D1 (55) | [85] |
| Poly (ADP-ribose) glycohydrolase inhibition | nobotanins B (70), K (72) (Tibouchina sp) | [86] |
© 2010 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Yoshida, T.; Amakura, Y.; Yoshimura, M. Structural Features and Biological Properties of Ellagitannins in Some Plant Families of the Order Myrtales. Int. J. Mol. Sci. 2010, 11, 79-106. https://doi.org/10.3390/ijms11010079
Yoshida T, Amakura Y, Yoshimura M. Structural Features and Biological Properties of Ellagitannins in Some Plant Families of the Order Myrtales. International Journal of Molecular Sciences. 2010; 11(1):79-106. https://doi.org/10.3390/ijms11010079
Chicago/Turabian StyleYoshida, Takashi, Yoshiaki Amakura, and Morio Yoshimura. 2010. "Structural Features and Biological Properties of Ellagitannins in Some Plant Families of the Order Myrtales" International Journal of Molecular Sciences 11, no. 1: 79-106. https://doi.org/10.3390/ijms11010079
APA StyleYoshida, T., Amakura, Y., & Yoshimura, M. (2010). Structural Features and Biological Properties of Ellagitannins in Some Plant Families of the Order Myrtales. International Journal of Molecular Sciences, 11(1), 79-106. https://doi.org/10.3390/ijms11010079




