Calcium Ions Regulate K+ Uptake into Brain Mitochondria: The Evidence for a Novel Potassium Channel
Abstract
:1. Introduction
2. Results
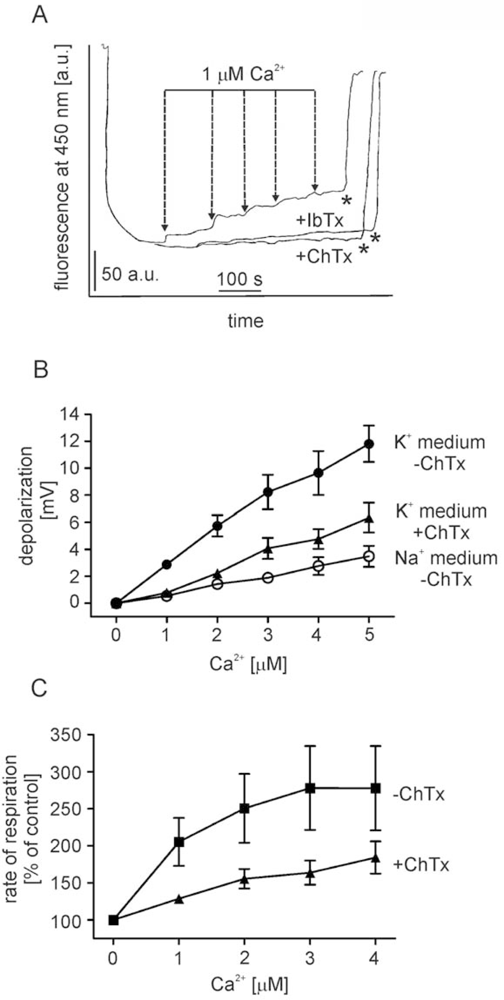
2.1. Ca2+ induces BK channel inhibitor-sensitive potassium transport in isolated brain mitochondria
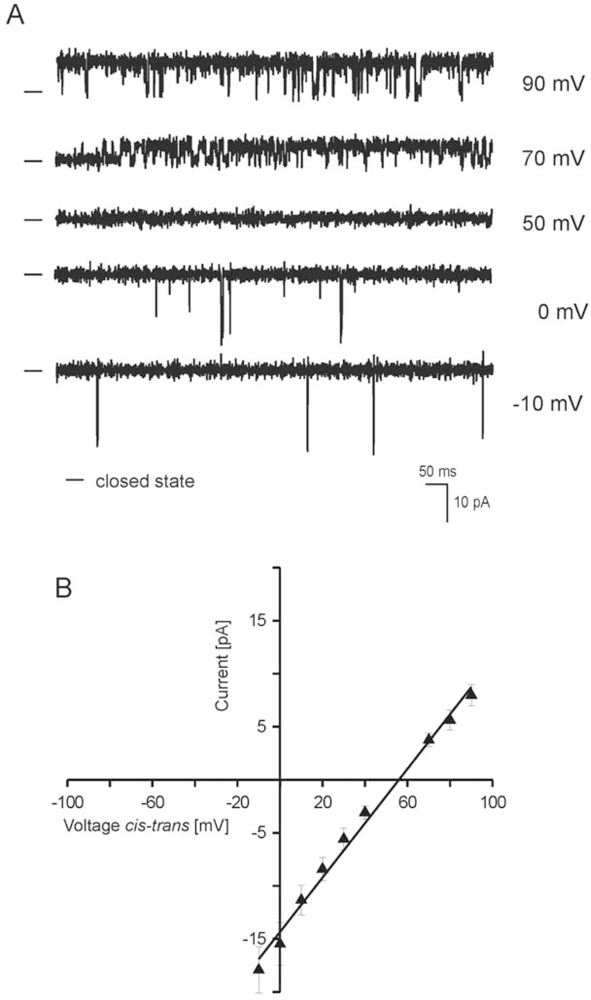
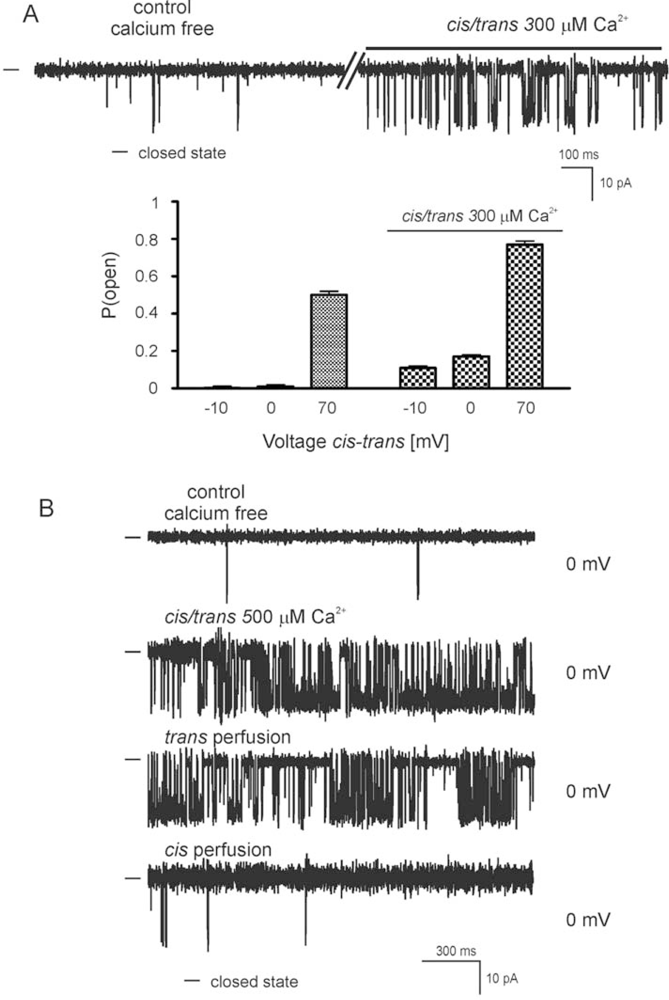
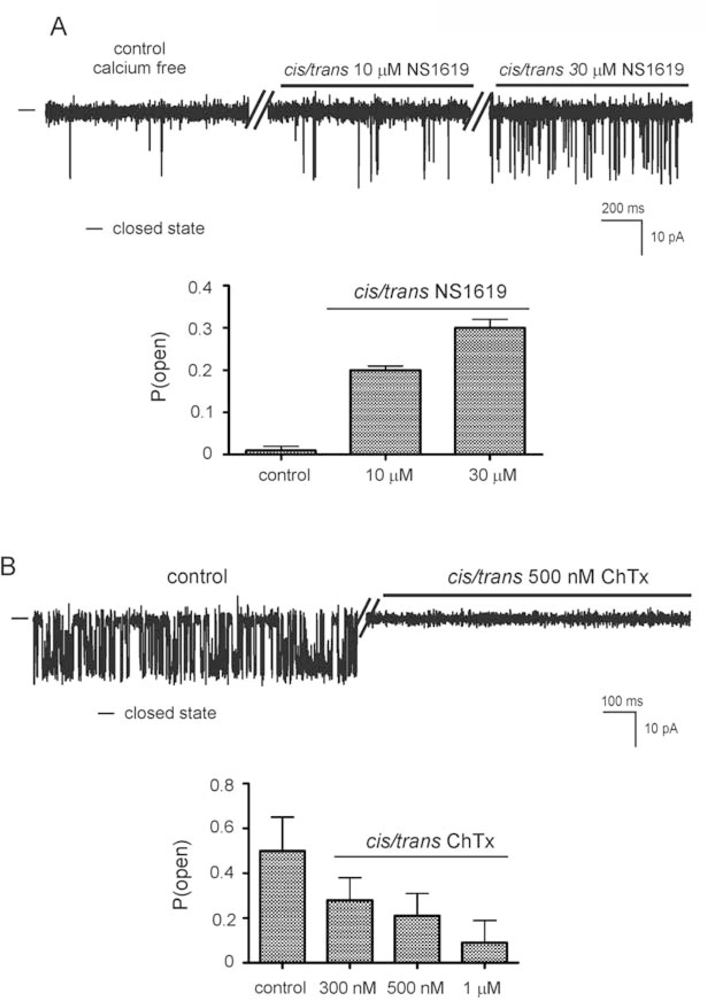
2.2. Reconstitution of inner mitochondrial membranes into planar lipid bilayers
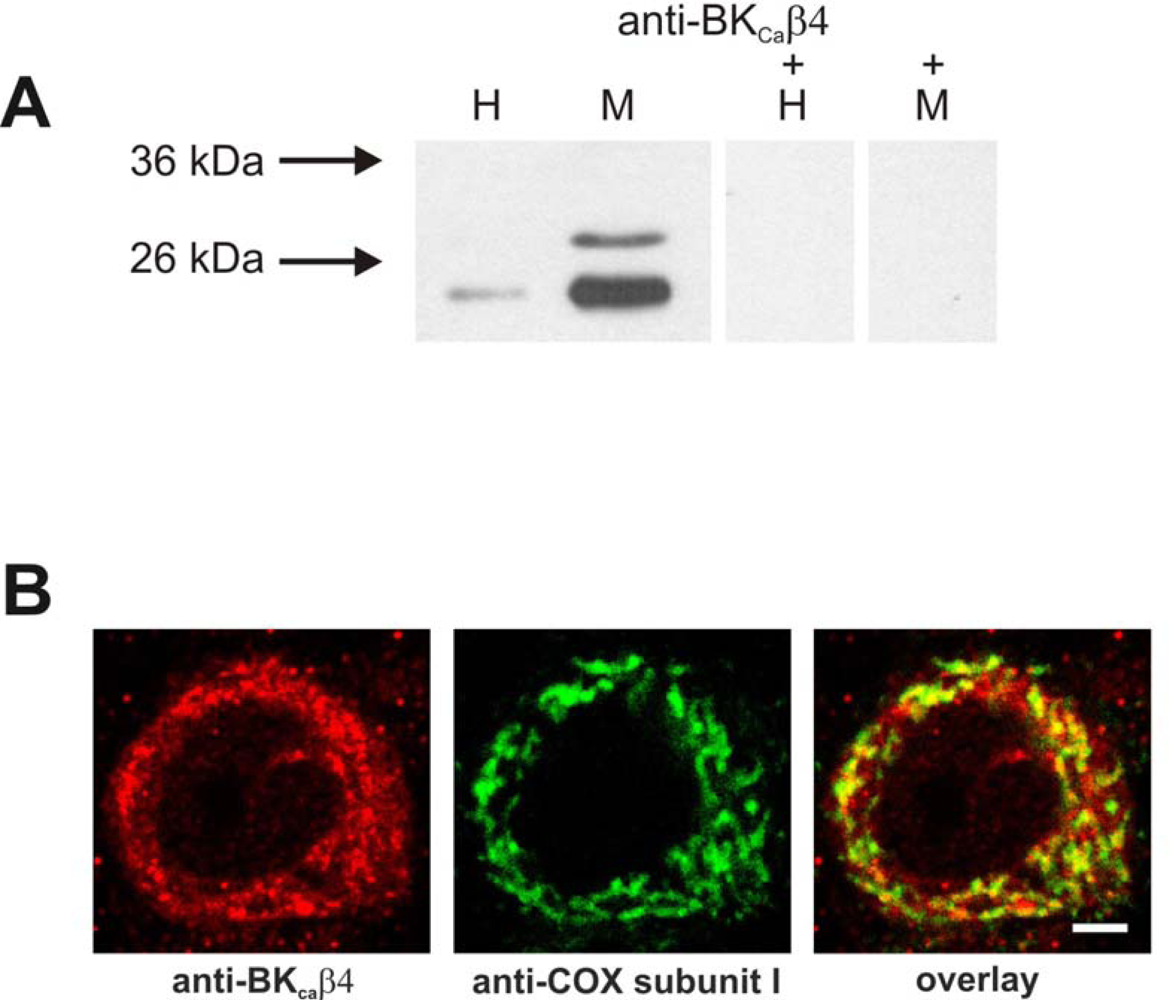
2.3. Identification of a BK channel subunit in brain mitochondria
3. Discussion
- - the channel (selective for potassium) is activated by calcium ions (probably from the matrix side) and is sensitive to ChTx and IbTx,
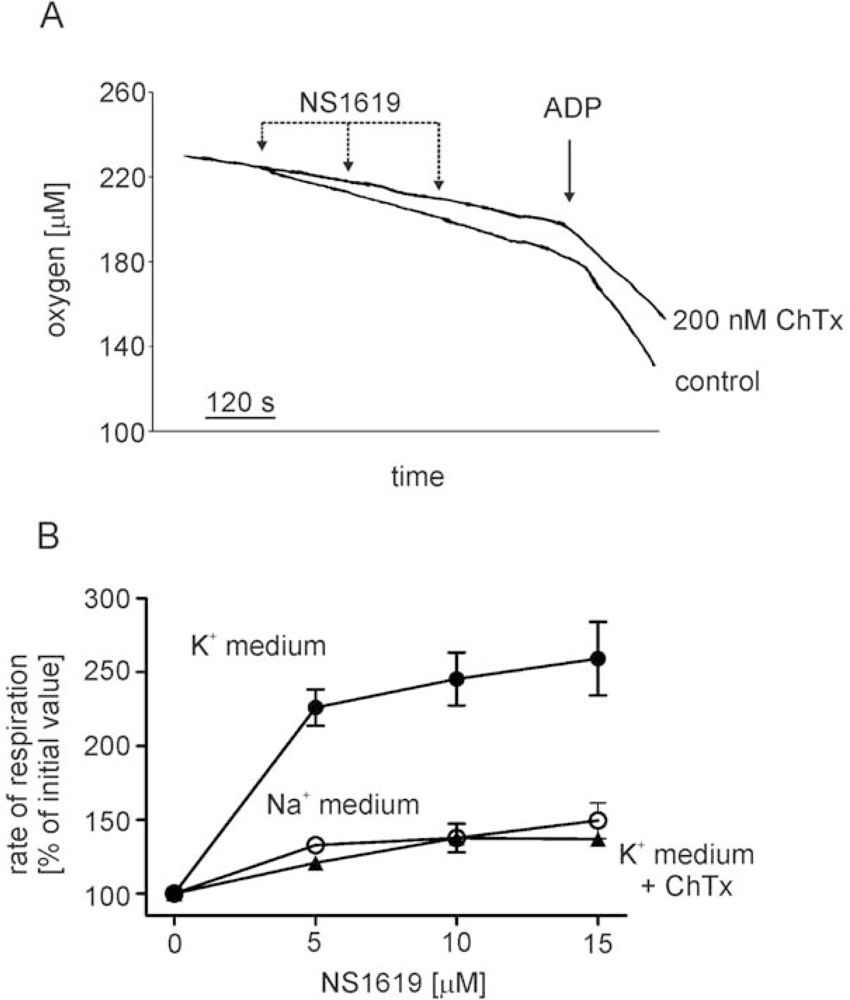
- - the channel is activated by the potassium channel opener NS1619,
- - the channel has a conductance of 265 pS and is voltage dependent.
4. Experimental Section
4.1. Materials
4.2. Isolation of rat brain mitochondria and submitochondrial particles
4.3. Mitochondrial membrane potential measurements
4.4. Mitochondrial respiration
4.5. Single channel measurements with black lipid membrane (BLM) technique
4.6. Data analysis
4.7. Western blot analysis
4.8. Immunochemistry
5. Conclusions
Acknowledgments
References
- Bajgar, R; Seetharaman, S; Kowaltowski, AJ; Garlid, KD; Paucek, P. Identification and properties of a novel intracellular (mitochondrial) ATP-sensitive potassium channel in brain. J. Biol Chem 2001, 276, 33369–33374. [Google Scholar]
- Bathori, G; Csordas, G; Garcia-Perez, C; Davies, E; Hajnoczky, G. Ca2+ dependent control of the permeability properties of mitochondrial outer membrane and voltage-dependent anion-selective channel (VDAC). J. Biol Chem 2006, 281, 17347–17358. [Google Scholar]
- Bednarczyk, P; Dołowy, K; Szewczyk, A. Matrix Mg2+ regulates mitochondrial ATP-dependent potassium channel from heart. FEBS Lett 2005, 579, 1625–1623. [Google Scholar]
- Bednarczyk, P; Kicinska, A; Kominkova, V; Ondrias, K; Dolowy, K; Szewczyk, A. Quinine inhibits mitochondrial ATP-regulated potassium channel from bovine heart. J. Membr. Biol 2004, 199, 63–72. [Google Scholar]
- Bernardi, P. Mitochondrial transport of cations: channels, exchangers, and permeability transition. Physiol. Rev 1999, 79, 1127–1155. [Google Scholar]
- Brookes, SP; Yoon, Y; Robotham, JL; Anders, MW; Sheu, SS. Calcium, ATP, and ROS: A mitochondrial love-hate triangle. Am J Cell Physiol 2004, 287, C817–C833. [Google Scholar]
- Budd, SL; Nicholls, DG. A reevaluation of the role of mitochondria in neuronal Ca2+ homeostasis. J. Neurochem 1996, 66, 403–411. [Google Scholar]
- Busija, DW; Lacza, Z; Rajapakse, N; Shimizu, K; Kis, B; Bari, F; Domoki, F; Horiguchi, T. Targeting mitochondrial ATP-sensitive potassium channels – a novel approach to neuroprotection. Brains Res. Brain Res. Rev 2004, 46, 282–294. [Google Scholar]
- Chan, PH. Mitochondria and neuronal death/survival signaling pathways in cerebral ischemia. Neurochem. Res 2004, 29, 1943–1949. [Google Scholar]
- Dahlem, YA; Horn, TFW; Buntinas, L; Gonoi, T; Wolf, G; Siemen, D. The human mitochondrialk KATP channel is modulated by calcium and nitric oxide: a patch-clamp approach. Biochim. Biophys. Acta 2004, 1656, 46–56. [Google Scholar]
- Debska, G; Kicinska, A; Dobrucki, J; Dworakowska, B; Nurowska, E; Skalska, J; Dolowy, K; Szewczyk, A. Large-conductance K+ channel openers NS1619 and NS004 as inhibitors of mitochondrial function in glioma cells. Biochem. Pharmacol 2003, 65, 1827–1834. [Google Scholar]
- Debska, G; Kicinska, A; Skalska, J; Szewczyk, A; May, R; Elger, CE; Kunz, WS. Opening of potassium channels modulates mitochondrial function in rat skeletal muscle. Biochim. Biophys. Acta 2002, 1556, 97–105. [Google Scholar]
- Debska, G; May, R; Kicinska, A; Szewczyk, A; Elger, CE; Kunz, WS. Potassium channel openers depolarize hippocampal mitochondria. Brain Res 2001, 892, 42–50. [Google Scholar]
- Douglas, RM; Lai, JC; Bian, S; Cummins, L; Moczydlowski, E; Haddad, GG. The calcium-sensitive large-conductance potassium channel (BK/MAXI K) is present in the inner mitochondrial membrane of rat brain. Neuroscience 2006, 13, 1249–1261. [Google Scholar]
- Inoue, I; Nagase, H; Kishi, K; Higuti, T. ATP-sensitive K+ channel in the mitochondrial inner membrane. Nature 1991, 352, 244–247. [Google Scholar]
- Jonas, E. Regulation of synaptic transmission by mitochondrial ion channels. J. Bioenerg. Biomembr 2004, 36, 357–361. [Google Scholar]
- Kicinska, A; Skalska, J; Szewczyk, A. Mitochondria and big-conductance potassium channel openers. Toxicol. Mech. Methods 2004, 14, 63–65. [Google Scholar]
- Kicinska, A; Szewczyk, A. Large-conductance potassium cation channel opener NS1619 inhibits cardiac mitochondria respiratory chain. Toxicol. Mech. Methods 2004, 14, 59–61. [Google Scholar]
- Kulawiak, B; Kudin, AP; Szewczyk, A; Kunz, WS. BK Channel openers inhibit ROS production of isolated rat brain mitochondria. Exp. Neurol 2008, 212, 543–547. [Google Scholar]
- Liu, D; Slevin, JR; Lu, C; Chan, SL; Hansson, M; Elmer, E; Mattson, MP. Involvement of mitochondrial K+ release and cellular efflux in ischemic and apoptotic neuronal death. J. Neurochem 2003, 86, 966–979. [Google Scholar]
- Nakagawa, I; Alessandri, B; Heimann, A; Kempski, O. MitoKATP-channel opener protects against neuronal death in rat venous ischemia. Neurosurgery 2005, 57, 334–340. [Google Scholar]
- Paucek, P; Mironova, G; Mahdi, F; Beavis, AD; Woldegiogis, G; Garlid, KD. Reconstitution and partial purification of the glibenclamide-sensitive, ATP-dependent K+ channel from rat liver and beef heart mitochondria. J. Biol Chem 1992, 267, 26062–26069. [Google Scholar]
- Piwonska, M; Wilczek, E; Szewczyk, A; Wilczyński, GM. Diferential distribution of Ca2+-activated channel β4 subunit in rat brain: immunolocalization in neuronal mitochondria. Neuroscience 2008, 153, 446–460. [Google Scholar]
- Sato, T; Saito, T; Saegusa, N; Nakaya, H. Mitochondrial Ca2+-activated K+ channels in cardiac myocytes: A mechanism of the cardioprotective effect and modulation by protein kinase A. Circulation 2005, 111, 198–203. [Google Scholar]
- Siemen, D; Loupatatzis, C; Borecky, J; Gulbins, E; Lang, F. Ca2+-activated K channel of the BK-type in the inner mitochondrial membrane of a human glioma cell line. Biochem. Biophys. Res. Commun 1999, 257, 549–554. [Google Scholar]
- Skalska, J; Piwonska, M; Wyroba, E; Surmacz, L; Wieczorek, R; Koszela-Piotrowska, I; Zielińska, J; Bednarczyk, P; Dołowy, K; Wilczynski, GM; Szewczyk, A; Kunz, WS. A novel potassium channel in skeletal muscle mitochondria. Biochim. Biophys. Acta 2008, 1777, 651–659. [Google Scholar]
- Smaili, SS; Hsu, YT; Youle, RJ; Russell, JT. Mitochondria in Ca2+ signaling and apoptosis. J. Bioenerg. Biomembr 2000, 32, 35–46. [Google Scholar]
- Stout, AK; Raphael, HM; Kanterewicz, BI; Klann, E; Reynolds, IJ. Glutamate-induced neuron death requires mitochondrial calcium uptake. Nat. Neurosci 1998, 1, 366–373. [Google Scholar]
- Szabo, I; Bock, J; Jekle, A; Soddemann, M; Adams, C; Lang, F; Zoratti, M; Gulbins, E. A novel potassium channel in lymphocyte mitochondria. J. Biol Chem 2005, 280, 12790–12798. [Google Scholar]
- Szewczyk, A. The intracellular potassium and chloride channels: Properties, pharmacology and function. Mol Membr. Biol 1998, 15, 49–58. [Google Scholar]
- Szewczyk, A; Marban, E. Mitochondria: A new target for K channel openers? Trends Pharmacol. Sci 1999, 20, 157–161. [Google Scholar]
- Szewczyk, A; Skalska, J; Glab, M; Kulawiak, B; Malinska, D; Koszela-Piotrowska, I; Kunz, WS. Mitochondrial potassium channels: from pharmacology to function. Biochim. Biophys. Acta 2006, 1757, 715–720. [Google Scholar]
- Szewczyk, A; Wojtczak, L. Mitochondria as pharmacological target. Pharmacol. Rev 2002, 54, 101–127. [Google Scholar]
- Xu, W; Liu, Y; Wang, S; McDonald, T; Van Eyk, JE; Sidor, A; O'Rourke, B. Cytoprotective role of Ca2+- activated K+ channels in the cardiac inner mitochondrial membrane. Science 2002, 298, 1029–1033. [Google Scholar]
- Zoratti, M; Szabo, I. The mitochondrial permeability transition. Biochim. Biophys. Acta 1995, 1241, 139–176. [Google Scholar]
Abbreviations:
| ANT | mitochondrial adenine nucleotide translocase; |
| BKCa | channel, large conductance Ca2+-activated potassium channel; |
| BLM | black lipid membrane; |
| ChTx | charybdotoxin; IbTx, iberiotoxin; |
| NS1619 | 1,3-dihydro-1-[2-hydroxy-5-(trifluoro-methyl)phenyl]-5-(trifluoromethyl)-2H-benzimidazole-2-one; |
| mitoBKCa | channel, mitochondrial large conductance Ca2+ - activated potassium channel; |
| mitoKATP | channel, mitochondrial ATP-regulated potassium channel; |
| ROS | reactive oxygen species; |
| SMP | submitochondrial particles; |
| EGTA | ethylene glycol–bis (β-amino–ethylether)–N,N,N’,N’, -tetraacetic acid; |
| TTFB | 4,5,6,7-tetrachloro-2-trifluoromethyl-benzimidazole. |
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/). This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Skalska, J.; Bednarczyk, P.; Piwońska, M.; Kulawiak, B.; Wilczynski, G.; Dołowy, K.; Kunz, W.S.; Kudin, A.P.; Szewczyk, A. Calcium Ions Regulate K+ Uptake into Brain Mitochondria: The Evidence for a Novel Potassium Channel. Int. J. Mol. Sci. 2009, 10, 1104-1120. https://doi.org/10.3390/ijms10031104
Skalska J, Bednarczyk P, Piwońska M, Kulawiak B, Wilczynski G, Dołowy K, Kunz WS, Kudin AP, Szewczyk A. Calcium Ions Regulate K+ Uptake into Brain Mitochondria: The Evidence for a Novel Potassium Channel. International Journal of Molecular Sciences. 2009; 10(3):1104-1120. https://doi.org/10.3390/ijms10031104
Chicago/Turabian StyleSkalska, Jolanta, Piotr Bednarczyk, Marta Piwońska, Bogusz Kulawiak, Grzegorz Wilczynski, Krzysztof Dołowy, Wolfram S. Kunz, Alexei P. Kudin, and Adam Szewczyk. 2009. "Calcium Ions Regulate K+ Uptake into Brain Mitochondria: The Evidence for a Novel Potassium Channel" International Journal of Molecular Sciences 10, no. 3: 1104-1120. https://doi.org/10.3390/ijms10031104




