Effect of Nanoparticles on Protein Folding and Fibrillogenesis
Abstract
:1. Introduction
2. Conformational changes occur when proteins adsorb onto nanoparticles
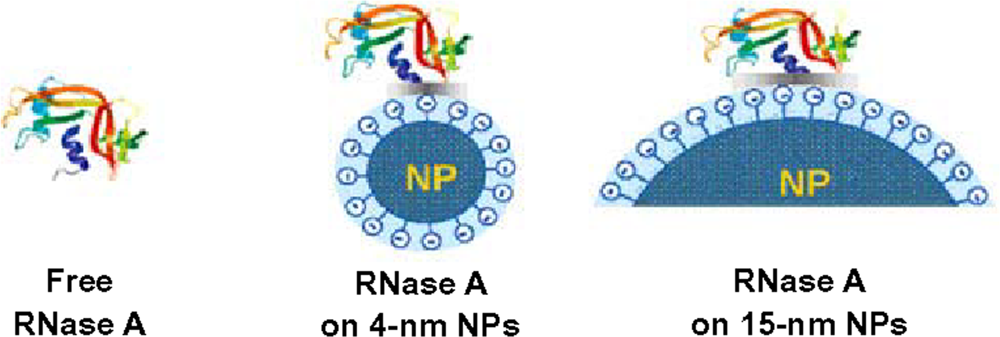
3. Size of nanoparticles influences both structure and function of adsorbed proteins
4. Surface concentration affects the extent of protein conformational changes
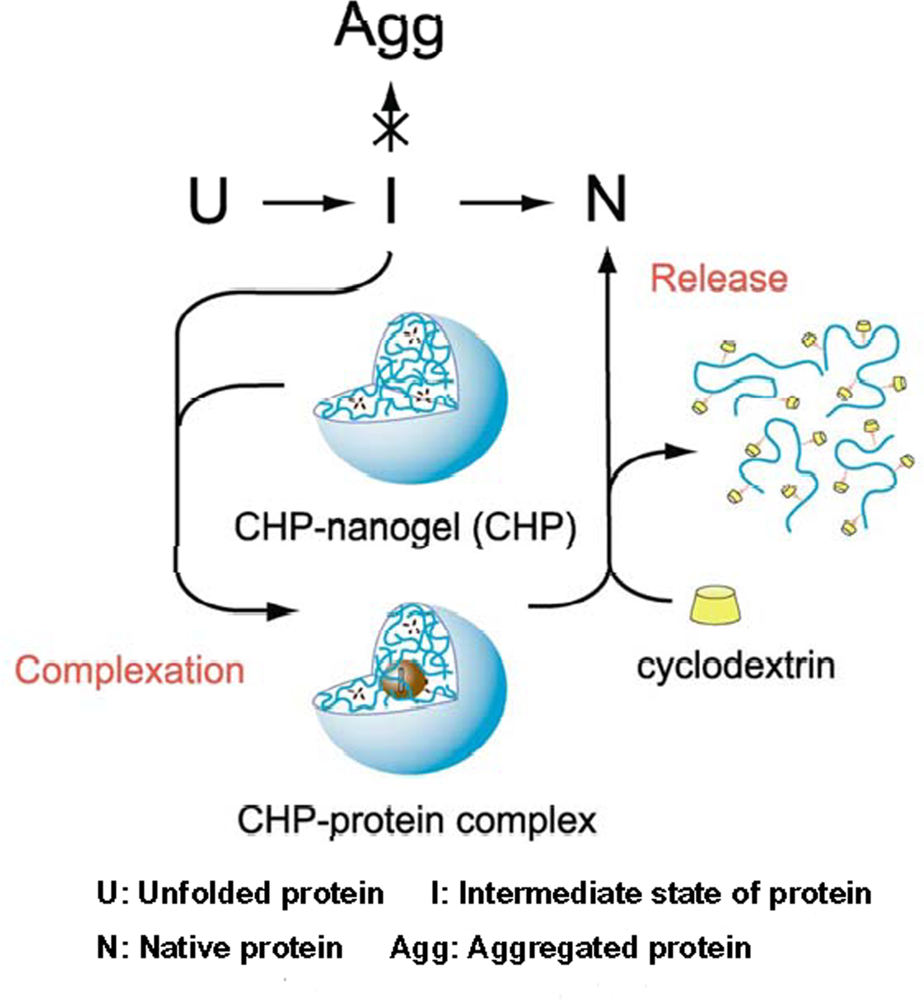
5. Chaperone activity of nanogel nanoparticles
6. Nanocrystals are used as a FRET probe to detect protein-folding intermediates
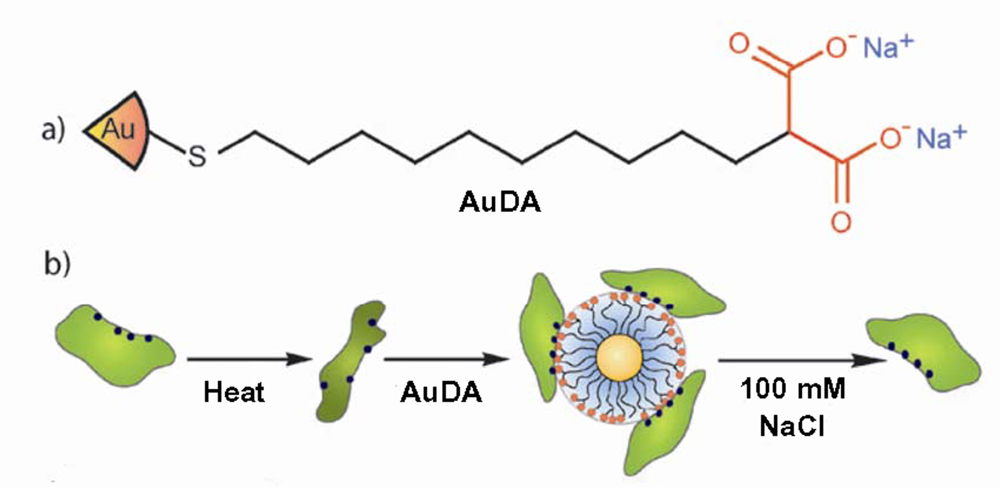
7. Effect of nanoparticles on fibrillogenesis
8. Conclusions
Acknowledgments
References
- Nel, A; Xia, T; Madler, L; Li, N. Toxic potential of materials at the nanolevel. Science 2006, 311, 622–627. [Google Scholar]
- Fischer, HC; Chan, WC. Nanotoxicity: The growing need for in vivo study. Curr. Opin. Biotechnol. 2007, 18, 565–571. [Google Scholar]
- Nie, S; Xing, Y; Kim, GJ; Simons, JW. Nanotechnology applications in cancer. Annu. Rev. Biomed. Eng. 2007, 9, 257–288. [Google Scholar]
- Marcato, PD; Duran, N. New aspects of nanopharmaceutical delivery systems. J. Nanosci. Nanotechnol. 2008, 8, 2216–2229. [Google Scholar]
- McBain, SC; Yiu, HH; Dobson, J. Magnetic nanoparticles for gene and drug delivery. Int J Nanomedicine 2008, 3, 169–180. [Google Scholar]
- De Jong, WH; Borm, PJ. Drug delivery and nanoparticles: applications and hazards. Int. J. Nanomedicine 2008, 3, 133–149. [Google Scholar]
- Heegaard, PM; Boas, U; Otzen, DE. Dendrimer effects on peptide and protein fibrillation. Macromol. Biosci. 2007, 7, 1047–1059. [Google Scholar]
- Crombez, L; Morris, MC; Deshayes, S; Heitz, F; Divita, G. Peptide-based nanoparticle for ex vivo and in vivo drug delivery. Curr. Pharm. Des. 2008, 14, 3656–3665. [Google Scholar]
- Aili, D; Enander, K; Rydberg, J; Nesterenko, I; Bjorefors, F; Baltzer, L; Liedberg, B. Folding induced assembly of polypeptide decorated gold nanoparticles. J. Am. Chem. Soc. 2008, 130, 5780–5788. [Google Scholar]
- Gao, L; Zhuang, J; Nie, L; Zhang, J; Zhang, Y; Gu, N; Wang, T; Feng, J; Yang, D; Perrett, S; Yan, X. Intrinsic peroxidase-like activity of ferromagnetic nanoparticles. Nat. Nanotechnol. 2007, 2, 577–583. [Google Scholar]
- Dobrovolskaia, MA; McNeil, SE. Immunological properties of engineered nanomaterials. Nat. Nanotechnol. 2007, 2, 469–478. [Google Scholar]
- Shang, L; Wang, Y; Jiang, J; Dong, S. pH-Dependent protein conformational changes in albumin: Gold nanoparticle bioconjugates: A spectroscopic study. Langmuir 2007, 23, 2714–2721. [Google Scholar]
- Wu, X; Narsimhan, G. Effect of surface concentration on secondary and tertiary conformational changes of lysozyme adsorbed on silica nanoparticles. Biochim. Biophys. Acta 2008, 1784, 1694–1701. [Google Scholar]
- Vertegel, AA; Siegel, RW; Dordick, JS. Silica nanoparticle size influences the structure and enzymatic activity of adsorbed lysozyme. Langmuir 2004, 20, 6800–6807. [Google Scholar]
- Shang, W; Nuffer, JH; Dordick, JS; Siegel, RW. Unfolding of ribonuclease A on silica nanoparticle surfaces. Nano Lett. 2007, 7, 1991–1995. [Google Scholar]
- Wu, X; Narsimhan, G. Characterization of secondary and tertiary conformational changes of beta-lactoglobulin adsorbed on silica nanoparticle surfaces. Langmuir 2008, 24, 4989–4998. [Google Scholar]
- Walter, S. Structure and function of the GroE chaperone. Cell Mol. Life Sci. 2002, 59, 1589–1597. [Google Scholar]
- Young, JC; Agashe, VR; Siegers, K; Hartl, FU. Pathways of chaperone-mediated protein folding in the cytosol. Nat. Rev. Mol. Cell Biol. 2004, 5, 781–791. [Google Scholar]
- Hoffmann, F; Rinas, U. Roles of heat-shock chaperones in the production of recombinant proteins in Escherichia coli. Adv. Biochem. Eng. Biotechnol. 2004, 89, 143–161. [Google Scholar]
- Akiyoshi, K; Sasaki, Y; Sunamoto, J. Molecular chaperone-like activity of hydrogel nanoparticles of hydrophobized pullulan: Thermal stabilization with refolding of carbonic anhydrase B. Bioconjug. Chem. 1999, 10, 321–324. [Google Scholar]
- Nomura, Y; Ikeda, M; Yamaguchi, N; Aoyama, Y; Akiyoshi, K. Protein refolding assisted by self-assembled nanogels as novel artificial molecular chaperone. FEBS Lett. 2003, 553, 271–276. [Google Scholar]
- Ikeda, K; Okada, T; Sawada, S; Akiyoshi, K; Matsuzaki, K. Inhibition of the formation of amyloid beta-protein fibrils using biocompatible nanogels as artificial chaperones. FEBS Lett. 2006, 580, 6587–6595. [Google Scholar]
- De, M; Rotello, VM. Synthetic “chaperones”: nanoparticle-mediated refolding of thermally denatured proteins. Chem. Commun. (Camb.) 2008, 3504–3506. [Google Scholar]
- Sarkar, R; Narayanan, SS; Palsson, LO; Dias, F; Monkman, A; Pal, SK. Direct conjugation of semiconductor nanocrystals to a globular protein to study protein-folding intermediates. J. Phys. Chem. B 2007, 111, 12294–12298. [Google Scholar]
- Bellotti, V; Nuvolone, M; Giorgetti, S; Obici, L; Palladini, G; Russo, P; Lavatelli, F; Perfetti, V; Merlini, G. The workings of the amyloid diseases. Ann. Med. 2007, 39, 200–207. [Google Scholar]
- Griffiths, HH; Morten, IJ; Hooper, NM. Emerging and potential therapies for Alzheimer’s disease. Expert Opin. Ther. Targets 2008, 12, 693–704. [Google Scholar]
- Chopra, D; Gulati, M; Saluja, V; Pathak, P; Bansal, P. Brain permeable nanoparticles. Recent Patents CNS Drug Discov 2008, 3, 216–225. [Google Scholar]
- Triulzi, RC; Dai, Q; Zou, J; Leblanc, RM; Gu, Q; Orbulescu, J; Huo, Q. Photothermal ablation of amyloid aggregates by gold nanoparticles. Colloids Surf. B Biointerfaces 2008, 63, 200–208. [Google Scholar]
- Pai, AS; Rubinstein, I; Onyuksel, H. PEGylated phospholipid nanomicelles interact with beta-amyloid((1–42)) and mitigate its beta-sheet formation, aggregation and neurotoxicity in vitro. Peptides 2006, 27, 2858–2866. [Google Scholar]
- Kim, JE; Lee, M. Fullerene inhibits beta-amyloid peptide aggregation. Biochem. Biophys. Res. Commun. 2003, 303, 576–579. [Google Scholar]
- Rocha, S; Thunemann, AF; Pereira Mdo, C; Coelho, M; Mohwald, H; Brezesinski, G. Influence of fluorinated and hydrogenated nanoparticles on the structure and fibrillogenesis of amyloid beta-peptide. Biophys. Chem. 2008, 137, 35–42. [Google Scholar]
- Wu, WH; Sun, X; Yu, YP; Hu, J; Zhao, L; Liu, Q; Zhao, YF; Li, YM. TiO2 nanoparticles promote beta-amyloid fibrillation in vitro. Biochem. Biophys. Res. Commun. 2008, 373, 315–318. [Google Scholar]
- Linse, S; Cabaleiro-Lago, C; Xue, WF; Lynch, I; Lindman, S; Thulin, E; Radford, SE; Dawson, KA. Nucleation of protein fibrillation by nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 8691–8696. [Google Scholar]
- Colvin, VL; Kulinowski, KM. Nanoparticles as catalysts for protein fibrillation. Proc. Natl. Acad. Sci. USA 2007, 104, 8679–8680. [Google Scholar]
- Cabaleiro-Lago, C; Quinlan-Pluck, F; Lynch, I; Lindman, S; Minogue, AM; Thulin, E; Walsh, DM; Dawson, KA; Linse, S. Inhibition of amyloid beta protein fibrillation by polymeric nanoparticles. J. Am. Chem. Soc. 2008, 130, 15437–15443. [Google Scholar]
- Cedervall, T; Lynch, I; Foy, M; Berggard, T; Donnelly, SC; Cagney, G; Linse, S; Dawson, KA. Detailed identification of plasma proteins adsorbed on copolymer nanoparticles. Angew. Chem. Int. Ed. Engl. 2007, 46, 5754–5756. [Google Scholar]
- Cedervall, T; Lynch, I; Lindman, S; Berggard, T; Thulin, E; Nilsson, H; Dawson, KA; Linse, S. Understanding the nanoparticle-protein corona using methods to quantify exchange rates and affinities of proteins for nanoparticles. Proc. Natl. Acad. Sci. USA 2007, 104, 2050–2055. [Google Scholar]
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Fei, L.; Perrett, S. Effect of Nanoparticles on Protein Folding and Fibrillogenesis. Int. J. Mol. Sci. 2009, 10, 646-655. https://doi.org/10.3390/ijms10020646
Fei L, Perrett S. Effect of Nanoparticles on Protein Folding and Fibrillogenesis. International Journal of Molecular Sciences. 2009; 10(2):646-655. https://doi.org/10.3390/ijms10020646
Chicago/Turabian StyleFei, Li, and Sarah Perrett. 2009. "Effect of Nanoparticles on Protein Folding and Fibrillogenesis" International Journal of Molecular Sciences 10, no. 2: 646-655. https://doi.org/10.3390/ijms10020646



