Biomass Production Potential of a Wastewater Alga Chlorella vulgaris ARC 1 under Elevated Levels of CO2 and Temperature
Abstract
:1. Introduction
2. Results
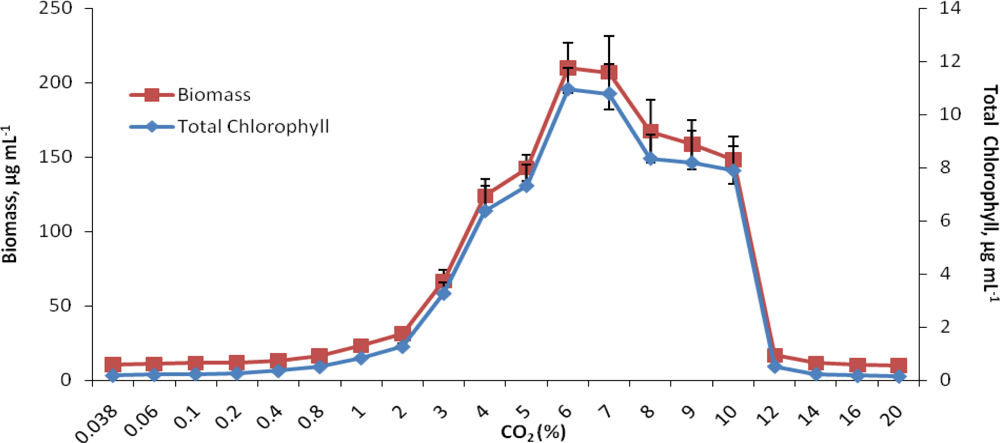
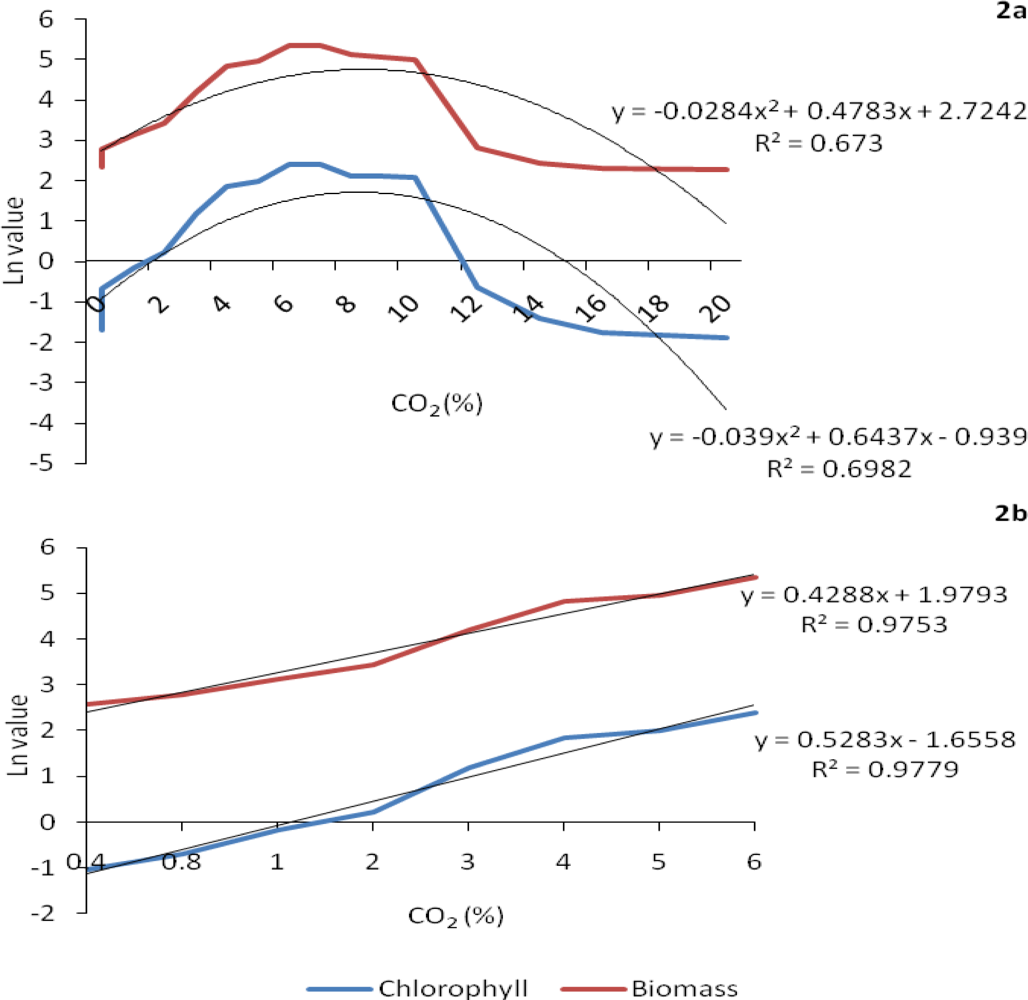
2.1. Growth response to increasing concentrations of CO2
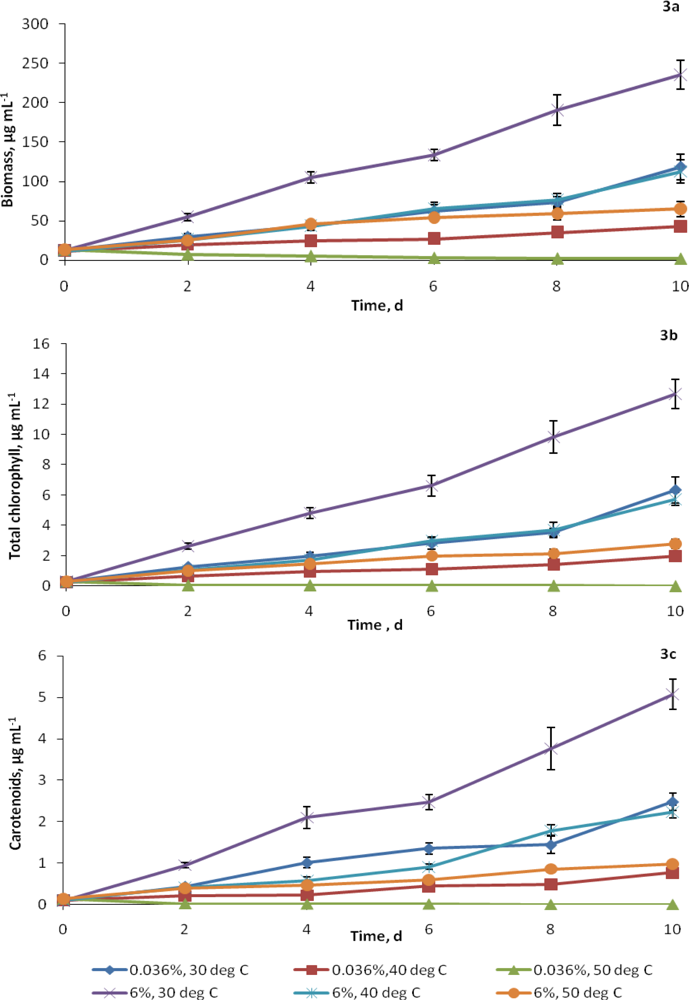
2.2. Growth at elevated CO2 and temperatures
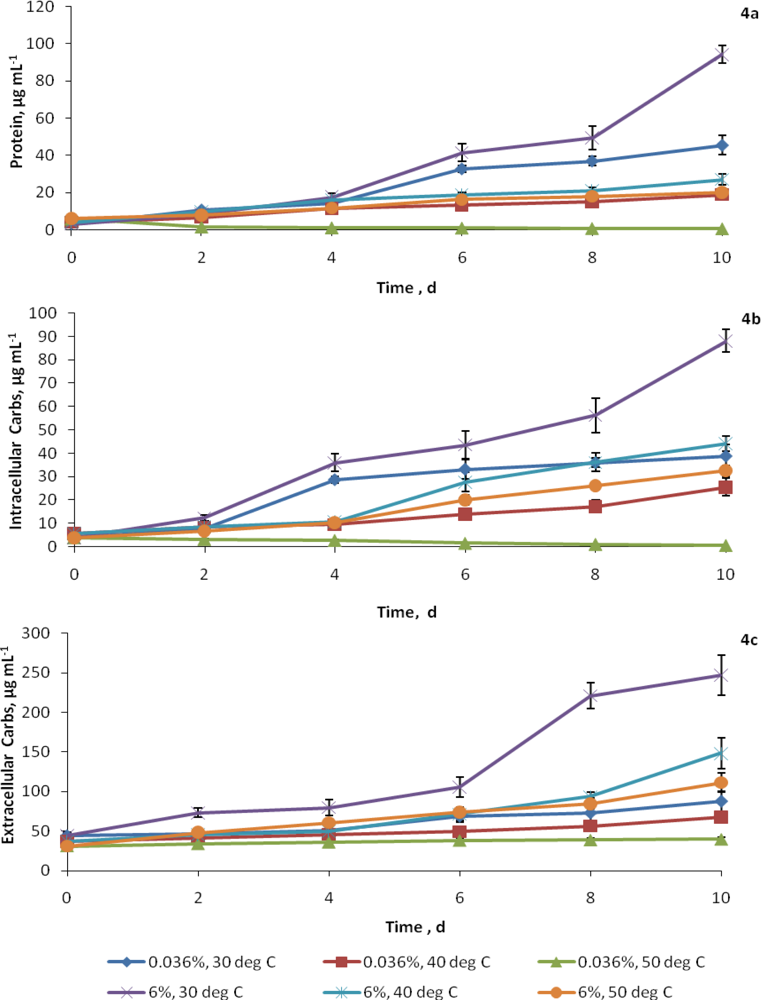
2.3. Synthesis of cellular constituents at elevated CO2 and temperatures
2.4. 14C uptake and carbonic anhydrase activity in response to elevated CO2 and temperatures
3. Discussion
4. Conclusions
5. Experimental Section
5.1. Experimental organism
5.2. Growth response to varied carbon dioxide concentrations
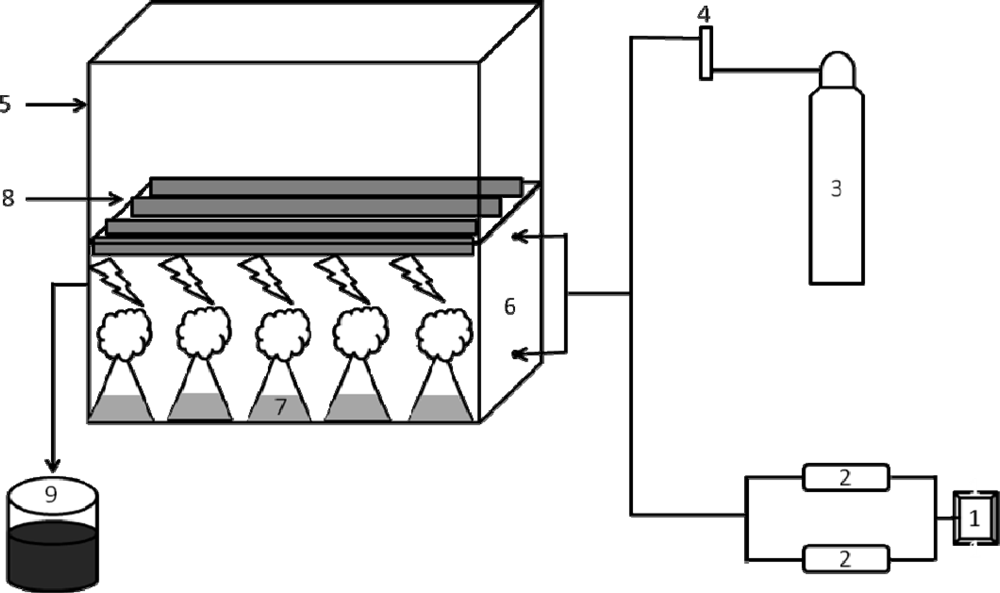
5.3. Experimental CO2 chamber set-up
4.4. Growth and physiological analyses
4.5. Statistical analysis
Acknowledgments
References and Notes
- Kondili, EM; Kaldellis, JK. Biofuel implementation in East Europe: Current status and future prospects. Renew. Sust. Energ. Rev 2007, 11, 2137–2151. [Google Scholar]
- Roman-Leshkov, Y; Barrett, CJ; Liu, ZY; Dumesic, JA. Production of dimethylfuran for liquid fuels from biomass-derived carbohydrates. Nature 2007, 447, 982–985. [Google Scholar]
- Gutierrez, R; Gutierrez-Sanchez, R; Nafidi, A. Trend analysis using nonhomogeneous stochastic diffusion processes. Emission of CO2; Kyoto protocol in Spain. Stoch. Environ. Res. Risk Assess 2008, 22, 57–66. [Google Scholar]
- Wang, B; Li, Y; Wu, N; Lan, CQ. CO2 bio-mitigation using microalgae. Appl. Microbiol. Biotechnol 2008, 79, 707–718. [Google Scholar]
- de Morais, MG; Costa, JAV. Biofixation of carbon dioxide by Spirulina sp. and Scenedesmus obliquus cultivated in a three-stage serial tubular photobioreactor. J. Biotechnol 2007, 129, 439–445. [Google Scholar]
- Skjanes, K; Lindblad, P; Muller, J. BioCO2- a multidisciplinary, biological approach using solar energy to capture CO2 while producing H2 and high value products. Biomol. Eng 2007, 24, 405–413. [Google Scholar]
- Li, Y; Horsman, M; Wu, N; Lan, CQ; Dubois-Calero, N. Biofuels from microalgae. Biotech. Prog 2008, 24, 815–820. [Google Scholar]
- Hsueh, HT; Chu, H; Yu, ST. A batch study on the bio-fixation of carbon dioxide in the absorbed solution from a chemical wet scrubber by hot spring and marine algae. Chemosphere 2007, 66, 878–886. [Google Scholar]
- Dismukes, GC; Carrieri, D; Bennette, N; Ananyev, GM; Posewitz, MC. Aquatic phototrophs: Efficient alternatives to land-based crops for biofuels. Curr. Opin. Biotechnol 2008, 19, 235–240. [Google Scholar]
- Kodama, M; Ikemoto, H; Miyachi, S. A new species of highly CO2-tolerant fast growing marine microalga suitable for high density culture. J. Mar. Biotechnol 1993, 1, 21–25. [Google Scholar]
- Miyachi, S; Iwasaki, I; Shiraiwa, Y. Historical perspective on microalgal and cyanobacterial acclimation to low- and extremely high-CO2 conditions. Photosynth. Res 2003, 77, 139–153. [Google Scholar]
- de Morais, MG; Costa, JAV. Isolation and selection of microalgae from coal fired thermoelectric power plant for biofixation of carbon dioxide. Energy Conv. Manag 2007, 41, 633–646. [Google Scholar]
- Papazi, A; Makridis, P; Divanach, P; Kotzabasis, K. Bioenergetic changes in the microalgal photosynthetic apparatus by extremely high CO2 concentrations induce an intense biomass production. Physiol. Plant 2008, 132, 338–349. [Google Scholar]
- Xia, JR; Gao, KS. Impacts of elevated CO2 concentration on biochemical composition, carbonic anhydrase and nitrate reductase activity of freshwater green algae. J. Integr. Plant Biol 2005, 47, 668–675. [Google Scholar]
- Chiu, SY; Kao, CY; Tsai, MT; Ong, SC; Chen, CH; Lin, CS. Lipid accumulation and CO2 utilization of Nannochloropsis oculata in response to CO2 aeration. Bioresour. Technol 2009, 100, 833–838. [Google Scholar]
- Sakai, N; Sakamoto, Y; Kishimoto, N; Chihara, M; Karube, I. Chlorella strains from hot springs tolerant to high temperature and high CO2. Energy Conv. Manag 1995, 36, 693–696. [Google Scholar]
- Mendes, RL; Nobre, BP; Cardoso, MT; Pereira, AP; Palavra, AF. Supercritical carbon dioxide extraction of compounds with pharmaceutical importance from microalgae. Inorg. Chim. Acta 2003, 356, 328–334. [Google Scholar]
- Moroney, JV; Husic, HD; Tolbert, NE. Effects of carbonic anhydrase inhibitors on inorganic carbon accumulation by Chlamydomonas reinhardtii. Plant J 1985, 9, 819–827. [Google Scholar]
- Moroney, JV; Somanchi, A. How do algae concentrate CO2 to increase the efficiency of photosynthetic carbon fixation? Plant Physiol 1999, 119, 9–16. [Google Scholar]
- Hanagata, N; Takeuchi, T; Fukuju, Y; Barnes, DJ; Karube, I. Tolerance of microalgae to high CO2 and high temperature. Phytochemistry 1992, 31, 3345–3348. [Google Scholar]
- DeLucia, EH; Sasek, TW; Strain, BR. Photosynthetic inhibition after long term exposure to elevated levels of atmospheric carbon dioxide. Photosynth. Res 1985, 7, 175–184. [Google Scholar]
- Badger, MR; Price, GD. The CO2 concentrating mechanism in cyanobacteria and green algae. Physiol. Plant 1994, 84, 606–615. [Google Scholar]
- FitzGerald, GP; Rohlich, GA. Biological removal of nutrients from treated sewage: Laboratory experiments. Verh Int Verein Theor Angew Limol 1962, XV, 597–608. [Google Scholar]
- Travieso, L; Pellón, A; Benítez, F; Sánchez, E; Borja, R; O’Farrill, N; Weiland, P. BIOALGA reactor: preliminary studies for heavy metals metal. Biochem. Eng. J 2002, 12, 87–91. [Google Scholar]
- Bhatnagar, A. Development of r- and K-selection model in the waste stabilisation pond system. J. Environ. Biol 1999, 20, 115–120. [Google Scholar]
- Stanier, RV; Kunisawa, R; Mandel, M; Cohen-Bazire, G. Purification and properties of unicellular blue-green algae (order: Chrococcales). Bacteriol. Rev 1971, 35, 171–205. [Google Scholar]
- Rogers, HH; Heck, WW; Heagle, AS. A field technique for the study of plant responses to elevated CO2 concentration. Air Pollut. Control Assoc. J 1983, 33, 42–44. [Google Scholar]
- MacKinney, G. Absorption of light by chlorophyll solutions. J. Biol. Chem 1941, 140, 315–322. [Google Scholar]
- Jensen, A. Chlorophylls and carotenoids. In Handbook of Physiological Methods; Hellebust, JA, Craige, JS, Eds.; Cambridge University Press: Cambridge, 1978; pp. 59–70. [Google Scholar]
- Lowry, OH; Rosebrough, NJ; Farr, AL; Randall, RJ. Protein measurement with Folin-Phenol reagent. J. Biol. Chem 1951, 193, 265–275. [Google Scholar]
- Dubois, M; Gilles, KA; Hamliton, JK; Rebers, PA; Smith, F. Colorimetric method for determination of sugars and related substances. Anal. Chem 1956, 28, 350–356. [Google Scholar]
- Kumar, A; Tabita, FR; Van Baalen, C. Isolation and characterization of heterocysts from Anabaena sp. Strain CA. Arch. Microbiol 1982, 133, 103–109. [Google Scholar]
- Dixon, GK; Patel, BN; Merrett, MJ. Role of intracellular carbonic anhydrase in inorganic carbon assimilation by Porphyridium purpureum. Planta 1987, 172, 508–513. [Google Scholar]
- Gomez, KA; Gomez, AA. Statistical procedures for agricultural research; John Wiley & Sons: Singapore, 1984. [Google Scholar]
| CO2 (%) | Temperature
(°C) | Biomass
(μbiomassd–1) | Chlorophyll
(μchl d–1) | Biomass
(% increase after 10 d) |
|---|---|---|---|---|
| 0.036
(ambient) | 30 | 0.128 | 0.148 | 879 |
| 40 | 0.082 | 0.121 | 361 | |
| 50 | NG | NG | NG | |
| 6
(elevated) | 30 | 0.222 | 0.262 | 1846 |
| 40 | 0.136 | 0.200 | 874 | |
| 50 | 0.065 | 0.106 | 379 | |
| CO2 (%) | Temperature
(°C) | 14CO2 uptake
(μmol NaH14CO3. mg chl–1 h–1) | Carbonic anhydrase activity
(Enzyme units. mg chl–1) | |
|---|---|---|---|---|
| 0.036
(ambient) | 30 | 7.833 | 14.811 | |
| 40 | 9.648 | 12.229 | ||
| 50 | n.d. | n.d. | ||
| 6
(elevated) | 30 | 6.033 | 7.861 | |
| 40 | 3.413 | 6.043 | ||
| 50 | 3.086 | 5.893 | ||
© 2009 by the authors; licensee Molecular Diversity Preservation International, Basel, Switzerland. This article is an open-access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/3.0/).
Share and Cite
Chinnasamy, S.; Ramakrishnan, B.; Bhatnagar, A.; Das, K.C. Biomass Production Potential of a Wastewater Alga Chlorella vulgaris ARC 1 under Elevated Levels of CO2 and Temperature. Int. J. Mol. Sci. 2009, 10, 518-532. https://doi.org/10.3390/ijms10020518
Chinnasamy S, Ramakrishnan B, Bhatnagar A, Das KC. Biomass Production Potential of a Wastewater Alga Chlorella vulgaris ARC 1 under Elevated Levels of CO2 and Temperature. International Journal of Molecular Sciences. 2009; 10(2):518-532. https://doi.org/10.3390/ijms10020518
Chicago/Turabian StyleChinnasamy, Senthil, Balasubramanian Ramakrishnan, Ashish Bhatnagar, and Keshav C. Das. 2009. "Biomass Production Potential of a Wastewater Alga Chlorella vulgaris ARC 1 under Elevated Levels of CO2 and Temperature" International Journal of Molecular Sciences 10, no. 2: 518-532. https://doi.org/10.3390/ijms10020518




