Assessment of Aqueous Extraction Methods on Extractable Organic Matter and Hydrophobic/Hydrophilic Fractions of Virgin Forest Soils
Abstract
:1. Introduction
2. Results
2.1. Physicochemical Analysis of Soils
2.2. Chemical Characterization of Different Aqueous Soil Extraction Methods
2.3. Hydrophobic and Hydrophilic Fraction
3. Discussion
3.1. Physicochemical Analysis of Soils
3.2. Extractable Organic Matter Using Autoclaved Methods
3.3. Hydrophobic and Hydrophilic Fractions of Soil Extracts
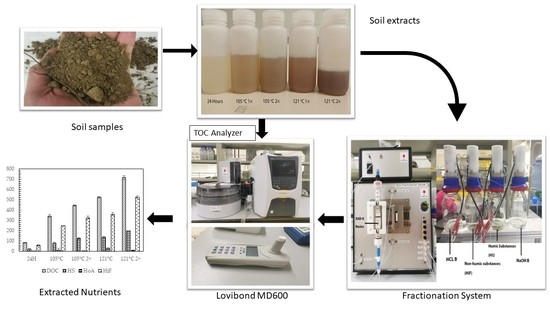
4. Materials and Methods
4.1. Soil Extraction
4.2. Macronutrient Analysis
4.3. Hydrophobic and Hydrophilic Fraction
- Vel = sample volume
- V0 = volume of XAD-8 resin (65% bulk column volume)
4.4. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- He, W.; Chen, M.; Schlautman, M.A.; Hur, J. Dynamic exchanges between DOM and POM pools in coastal and inland aquatic ecosystems: A review. Sci. Total Environ. 2016, 551–552, 415–428. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, M.M. Freshwater Culture media. In Algal Culture Techniques; Andersen, R.A., Ed.; Elsevier Academic Press: London, UK, 2005; p. 13. [Google Scholar]
- Marschner, B.; Kalbitz, K. Controls of bioavailability and biodegradability of dissolved organic matter in soils. Geoderma 2003, 113, 211–235. [Google Scholar] [CrossRef]
- Kalbitz, K.; Solinger, S.; Park, J.-H.; Michalzik, B.; Matzner, E. Controls on the dynamics of dissolved organic matter in soils: A review. Soil Sci. 2000, 165, 277–304. [Google Scholar] [CrossRef]
- Cotrufo, M.F.; Soong, J.L.; Horton, A.J.; Campbell, E.E.; Haddix, M.L.; Wall, D.H.; Parton, W.J. Formation of soil organic matter via biochemical and physical pathways of litter mass loss. Nat. Geosci. 2015, 8, 776–779. [Google Scholar] [CrossRef]
- Baldock, J.; Manning, N.; Vickerstaff, S. Social Policy, 3rd ed.; Oxford University Press: Oxford, UJ, USA, 2007; p. 722. [Google Scholar]
- Kleber, M.; Sollins, P.; Sutton, R. A conceptual model of organo-mineral interactions in soils: Self-assembly of organic molecular fragments into zonal structures on mineral surfaces. Biogeochemistry 2007, 85, 9–24. [Google Scholar] [CrossRef]
- Von Lützow, M.; Kögel-Knabner, I.; Ludwig, B.; Matzner, E.; Flessa, H.; Ekschmitt, K.; Guggenberger, G.; Marschner, B.; Kalbitz, K. Stabilization mechanisms of organic matter in four temperate soils: Development and application of a conceptual model. J. Plant Nutr. Soil Sci. 2008, 171, 111–124. [Google Scholar] [CrossRef]
- Piccolo, A. The supramolecular structure of humic substances: A novel understanding of humus chemistry and implications in soil science. Adv. Agron. 2002, 75, 57–134. [Google Scholar]
- McKee, G.A.; Soong, J.L.; Caldéron, F.; Borch, T.; Cotrufo, M.F. An integrated spectroscopic and wet chemical approach to investigate grass litter decomposition chemistry. Biogeochemistry 2016, 128, 107–123. [Google Scholar] [CrossRef] [Green Version]
- Huang, Y.; Eglinton, G.; Van Der Hage, E.R.E.; Boon, J.J.; Bol, R.; Ineson, P. Dissolved organic matter and its parent organic matter in grass upland soil horizons studied by analytical pyrolysis techniques. Eur. J. Soil Sci. 1998, 49, 1–15. [Google Scholar] [CrossRef]
- Hanke, D.; Dick, D.P. Organic Matter Stocks and the Interactions of Humic Substances with Metals in Araucaria Moist Forest Soil with Humic and Histic Horizons. Rev. Bras. Ciência Solo 2017, 41, 41. [Google Scholar] [CrossRef] [Green Version]
- Spaak, G. Investigating the Potential of Dissolved Organic Matter (DOM) Induced Denitrification in Dutch Groundwater. Ph.D. Thesis, Utrecht University, Utrecht, The Netherlands, April 2014. [Google Scholar]
- Thurman, E.M. Amount of Organic Carbon in Natural Waters BT-Organic Geochemistry of Natural Waters; Springer: Dordrecht, The Netherlands, 1985; pp. 7–65. [Google Scholar]
- McDonald, S.; Bishop, A.G.; Prenzler, P.D.; Robards, K. Analytical chemistry of freshwater humic substances. Anal. Chim. Acta 2004, 527, 105–124. [Google Scholar] [CrossRef]
- Orlov, D. Humic Substances of Soils and General Theory of Humification; Apple Academic Press: London, UK, 2020. [Google Scholar]
- Essington, M.E. Soil and Water Chemistry: An Integrative Approach; CRC Press: London, UK, 2015. [Google Scholar]
- Schwesig, D.; Göttlein, A.; Haumaier, L.; Blasek, R.; Ilgen, G. Soil Organic Matter Extraction Using Water at High Temperature and Elevated Pressure (ASE) as Compared to Conventional Methods. Int. J. Environ. Anal. Chem. 1999, 73, 253–268. [Google Scholar] [CrossRef]
- Masoom, H.; Courtier-Murias, D.; Farooq, H.; Soong, R.; Kelleher, B.P.; Zhang, C.; Maas, W.E.; Fey, M.; Kumar, R.; Monette, M.; et al. Soil Organic Matter in Its Native State: Unravelling the Most Complex Biomaterial on Earth. Environ. Sci. Technol. 2016, 50, 1670–1680. [Google Scholar] [CrossRef]
- Khairil, M.; Wan Juliana, W.A.; Nizam, M.S.; Razi Idris, W.M. Soil properties and variation between three forest types in tropical watershed forest of Chini Lake, Peninsular Malaysia. Sains Malays. 2014, 43, 1635–1643. [Google Scholar]
- Gasim, M.B.; Ismail, B.; Mir, S.-I.; Rahim, S.A.; Toriman, M.E. The Physico-chemical Properties of Four Soil Series in Tasik Chini, Pahang, Malaysia. Asian J. Earth Sci. 2011, 4, 75–84. [Google Scholar] [CrossRef] [Green Version]
- Rieuwerts, J.; Thornton, I.; Farago, M.; Ashmore, M. Factors influencing metal bioavailability in soils: Preliminary investigations for the development of a critical loads approach for metals. Chem. Speciat. Bioavailab. 1998, 10, 61–75. [Google Scholar] [CrossRef] [Green Version]
- Huang, B.; Li, Z.; Huang, J.; Guo, L.; Nie, X.; Wang, Y.; Zhang, Y.; Zeng, G. Adsorption characteristics of Cu and Zn onto various size fractions of aggregates from red paddy soil. J. Hazard. Mater. 2014, 264, 176–183. [Google Scholar] [CrossRef]
- Bech, J.; Abreu, M.M.; Chon, H.-T.; Roca, N. Remediation of Potentially Toxic Elements in Contaminated Soils. In PHEs, Environment and Human Health; Metzler, J.B., Ed.; Springer: Dordrecht, The Netherland, 2014; pp. 253–308. [Google Scholar]
- Kabata-Pendias, A. Trace Elements in Soils and Plants, Fourth ed.; Springer: Berlin/Heidelberg, Germany, 2010. [Google Scholar]
- Förstner, U. Land Contamination by Metals: Global Scope and Magnitude of Problem in Metal Speciation and Contamination of Soil; Lewis Publisher: Boca Raton, FL, USA, 1995. [Google Scholar]
- Cui, H.; Fan, Y.; Fang, G.; Zhang, H.; Su, B.; Zhou, J. Leachability, availability and bioaccessibility of Cu and Cd in a contaminated soil treated with apatite, lime and charcoal: A five-year field experiment. Ecotoxicol. Environ. Saf. 2016, 134, 148–155. [Google Scholar] [CrossRef]
- Asadu, C.L.A.; Chibuike, G.U. Contributions of organic matter, clay and silt to the effective CEC of soils of different land use history. Adv. Nat. Appl. Sci. 2015, 9, 110–115. [Google Scholar]
- Nciizah, A.D.; Wakindiki, I.I. Physical indicators of soil erosion, aggregate stability and erodibility. Arch. Agron. Soil Sci. 2014, 61, 827–842. [Google Scholar] [CrossRef]
- Moritsuka, N.; Matsuoka, K.; Katsura, K.; Sano, S.; Yanai, J. Soil color analysis for statistically estimating total carbon, total nitrogen and active iron contents in Japanese agricultural soils. Soil Sci. Plant Nutr. 2014, 60, 475–485. [Google Scholar] [CrossRef] [Green Version]
- Molino, A.; Mehariya, S.; Di Sanzo, G.; Larocca, V.; Martino, M.; Leone, G.P.; Marino, T.; Chianese, S.; Balducchi, R.; Musmarra, D. Recent developments in supercritical fluid extraction of bioactive compounds from microalgae: Role of key parameters, technological achievements and challenges. J. CO2 Util. 2020, 36, 196–209. [Google Scholar] [CrossRef]
- Da Silva, R.P.; Rocha-Santos, T.A.; Duarte, A.C. Supercritical fluid extraction of bioactive compounds. TrAC Trends Anal. Chem. 2016, 76, 40–51. [Google Scholar] [CrossRef] [Green Version]
- Berns, A.E.; Philipp, H.; Narres, H.-D.; Burauel, P.; Vereecken, H.; Tappe, W. Effect of gamma-sterilization and autoclaving on soil organic matter structure as studied by solid state NMR, UV and fluorescence spectroscopy. Eur. J. Soil Sci. 2008, 59, 540–550. [Google Scholar] [CrossRef]
- Teves-Costa, P.; Oliveira, C.S.; Senos, M.L. Effects of local site and building parameters on damage distribution in Angra do Heroísmo—Azores. Soil Dyn. Earthq. Eng. 2007, 27, 986–999. [Google Scholar] [CrossRef]
- Yaacob, N.; Ahmad, M.; Kawasaki, N.; Maniyam, M.; Abdullah, H.; Hashim, E.; Sjahrir, F.; Zamri, W.W.M.; Komatsu, K.; Kuwahara, V. Kinetics Growth and Recovery of Valuable Nutrients from SE-Langor Peat Swamp and Pristine Forest Soils Using Different Extraction Methods as Potential Microalgae Growth Enhancers. Molecules 2021, 26, 653. [Google Scholar] [CrossRef]
- De Oliveira, R.L.; da Silva, O.S.; Converti, A.; Porto, T.S. Thermodynamic and kinetic studies on pectinase extracted from Aspergillus aculeatus: Free and immobilized enzyme entrapped in alginate beads. Int. J. Biol. Macromol. 2018, 115, 1088–1093. [Google Scholar] [CrossRef]
- Chantigny, M.H.; Curtin, D.; Beare, M.H.; Greenfield, L.G. Influence of Temperature on Water-Extractable Organic Matter and Ammonium Production in Mineral Soils. Soil Sci. Soc. Am. J. 2010, 74, 517–524. [Google Scholar] [CrossRef]
- Jamien, F.M.; Embong, Z.; Tajudin, S.A.A.; Ahmad, S.; Lazim, A.M. The optimization of heating temperature for carbon extraction from peat soil. IOP Conf. Ser. Mater. Sci. Eng. 2018, 298, 12019. [Google Scholar] [CrossRef] [Green Version]
- Liegel, L.H. Effects of sterilization procedures on the biological, chemical, and physical properties of soils—A review. Turrialba 1986, 36, 11–19. [Google Scholar]
- McNamara, N.; Black, H.; Beresford, N.; Parekh, N. Effects of acute gamma irradiation on chemical, physical and biological properties of soils. Appl. Soil Ecol. 2003, 24, 117–132. [Google Scholar] [CrossRef]
- Truhlar, D.G. Interpretation of the activation energy. J. Chem. Educ. 1978, 55, 309. [Google Scholar] [CrossRef]
- Steinweg, J.M.; Jagadamma, S.; Frerichs, J.; Mayes, M.A. Activation Energy of Extracellular Enzymes in Soils from Different Biomes. PLoS ONE 2013, 8, e59943. [Google Scholar] [CrossRef] [PubMed]
- Inselsbacher, E. Recovery of individual soil nitrogen forms after sieving and extraction. Soil Biol. Biochem. 2014, 71, 76–86. [Google Scholar] [CrossRef]
- Martin, M.; Portetelle, D.; Michel, G.; Vandenbol, M. Microorganisms living on macroalgae: Diversity, interactions, and biotechnological applications. Appl. Microbiol. Biotechnol. 2014, 98, 2917–2935. [Google Scholar] [CrossRef] [Green Version]
- Razavi, D.S.; Lakzian, A. Evaluation of chemical and biological consequences of soil sterilization methods. Casp. J. Environ. Sci. 2007, 5, 87–91. [Google Scholar]
- Wolf, B.M.; Niedzwiedzki, D.M.; Magdaong, N.C.M.; Roth, R.; Goodenough, U.; Blankenship, R.E. Characterization of a newly isolated freshwater Eustigmatophyte alga capable of utilizing far-red light as its sole light source. Photosynth. Res. 2017, 135, 177–189. [Google Scholar] [CrossRef]
- Dietrich, P.; Cesarz, S.; Eisenhauer, N.; Roscher, C. Effects of steam sterilization on soil abiotic and biotic properties. Soil Org. 2000, 92, 99–108. [Google Scholar]
- Mulani, K.; Deshpande, H. Humic acid analysis for comparison of soil samples: A forensic perspective. Nucl. Magn. Reson. 2018, 11, 12. [Google Scholar]
- DouA, S. The thermodynamics stability of soil humic and fulvic acids. In Proceedings of the 19th World Congress of Soil Science: Soil Solutions for a Changing World, Brisbane, Australia, 1–6 August 2010; Symposium 2.2.1 Biogeochemical interfaces in soils. pp. 20–23. [Google Scholar]
- Kolokassidou, C.; Pashalidis, I.; Costa, C.; Efstathiou, A.; Buckau, G. Thermal stability of solid and aqueous solutions of humic acid. Thermochim. Acta 2007, 454, 78–83. [Google Scholar] [CrossRef]
- Fuentes, M.; González-Gaitano, G.; García-Mina, J.M. The usefulness of UV–visible and fluorescence spectroscopies to study the chemical nature of humic substances from soils and composts. Org. Geochem. 2006, 37, 1949–1959. [Google Scholar] [CrossRef]
- Francioso, O.; Montecchio, D.; Gioacchini, P.; Ciavatta, C. Thermal analysis (TG–DTA) and isotopic characterization (13C–15N) of humic acids from different origins. Appl. Geochem. 2005, 20, 537–544. [Google Scholar] [CrossRef]
- Katsumi, N.; Yonebayashi, K.; Okazaki, M. Effects of heating on composition, degree of darkness, and stacking nanostructure of soil humic acids. Sci. Total Environ. 2016, 541, 23–32. [Google Scholar] [CrossRef]
- González-Pérez, J.A.; González-Vila, F.J.; Almendros, G.; Knicker, H. The effect of fire on soil organic matter—A review. Environ. Int. 2004, 30, 855–870. [Google Scholar] [CrossRef]
- Martín, G.A.; Vila, F.J.G. Wildfires, soil carbon balance and resilient organic matter in Mediterranean ecosystems. A review. Span. J. Soil Sci. 2020, 2, 8–33. [Google Scholar] [CrossRef]
- Page-Dumroese, D.S.; Harvey, A.E.; Jurgensen, M.F. A Guide to Soil Sampling and Analysis on the National Forests of the Inland Northwest United States; Diane Publishing Company: Collingdale, PA, USA, 1995; pp. 1–11. [Google Scholar] [CrossRef]
- Provasoli, L.; McLaughlin, J.J.A.; Droop, M.R. The development of artificial media for marine algae. Arch. Microbiol. 1957, 25, 392–428. [Google Scholar] [CrossRef]
- Anderson, B.H.; Magdoff, F.R. Autoclaving Soil Samples Affects Algal-Available Phosphorus. J. Environ. Qual. 2005, 34, 1958–1963. [Google Scholar] [CrossRef] [Green Version]
- Komatsu, K.; Imai, A.; Kawasaki, N. Comparison between humic-like peaks in excitation-emission matrix spectra and resin-fractionated humic substances in aquatic environments. Limnology 2019, 20, 109–120. [Google Scholar] [CrossRef]
- Imai, A.; Fukushima, T.; Matsushige, K.; Kim, Y.-H.; Choi, K. Characterization of dissolved organic matter in effluents from wastewater treatment plants. Water Res. 2002, 36, 859–870. [Google Scholar] [CrossRef]
- Leenheer, J.A. Comprehensive approach to preparative isolation and fractionation of dissolved organic carbon from natural waters and wastewaters. Environ. Sci. Technol. 1981, 15, 578–587. [Google Scholar] [CrossRef]
- Thurman, E.M.; Malcolm, R.L. Preparative isolation of aquatic humic substances. Environ. Sci. Technol. 1981, 15, 463–466. [Google Scholar] [CrossRef] [PubMed]








| Soil Sample | Chini Lake | Langkawi Island | Kenyir Lake |
|---|---|---|---|
| pH | 3.97 ± 0.03 a | 4.23 ± 0.03 b | 4.83 ± 0.03 c |
| Sand (%) | 4.47 ± 0.06 a | 95 ± 0.00 b | 4.57 ± 0.00 a |
| Silt (%) | 94.89 ± 0.06 a | 0.00 ± 0.00 b | 94.91 ± 0.00 a |
| Clay (%) | 0.63 ± 0.01 a | 4.00 ± 0.00 b | 0.52 ± 0.00 c |
| Soil type | Silt loam | Sandy loam | Silt loam |
| Color | Dark brown | Light yellowish brown | Light yellowish brown |
| Soil Location | Chini Lake | Langkawi Island | Kenyir Lake |
|---|---|---|---|
| Total Organic Carbon (%) | 55.78 ± 1.29 a | 2.34 ± 0.02 b | 1.03 ± 0.02 b |
| Nitrogen (%) | 0.41 ± 0.05 a | 0.17 ± 0.00 b | 0.17 ± 0.00 b |
| Phosphorus (%) | 24.23 ± 0.84 a | 2.27 ± 0.01 b | 1.62 ± 0.01 b |
| Trace Metals (mg/kg) | |||
| Iron | 755.47 ± 16.15 a | 767.09 ± 1.57 a | 771.98 ± 2.21 a |
| Arsenic | ND < 1 | ND < 1 | ND < 1 |
| Cadmium | 0.20 ± 0.03 a | 0.25 ± 0.03 a | 0.27 ± 0.08 a |
| Chromium | 34.45 ± 3.92 | ND < 2 | ND < 2 |
| Chromium VII | 2.98 ± 0.18 | ND < 3 | ND < 3 |
| Lead | 3.06 ± 0.31 a | 3.52 ± 0.23 a | 33.83 ± 1.90 b |
| Copper | 5.26 ± 0.43 a | 1.23 ± 0.08 b | 1.65 ± 0.03 b |
| Nickel | 5.50 ± 0.33 a | 8.98 ± 0.54 b | 5.42a ± 0.29 a |
| Zinc | 5.67 ± 1.05 a | 8.56 ± 0.45 b | 27.09 ± 1.07 c |
| Mercury | ND < 1 | ND < 1 | ND < 1 |
| Silver | ND < 10 | ND < 10 | ND < 10 |
| Site | Treatments | Dissolved Organic Carbon, C (mg/L) | Total Dissolved Nitrogen, N (mg/L) | Total Dissolved Phosphorus, PO4 (mg/L) | Ammonia Concentrations, NH3 (mg/L) | Nitrate, NO3− (mg/L) |
|---|---|---|---|---|---|---|
| Chini Forest Reserve | 24 h | 74.15 ± 5.21 a | 6.63 ± 0.3 a | 0.20 ± 0.03 a | 3.50 ± 0.53 a | 0.67 ± 0.58 a |
| 105 °C 1× | 303.22 ± 17.19 b | 14.23 ± 1.65 b | 0.67 ± 0.3 b | 4.73 ± 0.12 b | 3.40 ± 0.61 b | |
| 105 °C 2× | 404.48 ± 24.11 c | 12.80 ± 1.9 b | 0.73 ± 0.2 b | 6.37 ± 0.21 c | 4.73 ± 0.40 c | |
| 121 °C 1× | 503.11 ± 39.99 d | 17.33 ± 2.0 b | 1.04 ± 0.06 c | 6.93 ± 1.01 d | 5.50 ± 0.78 d | |
| 121 °C 2× | 677.95 ± 69.10 e | 23.47 ± 4.0 c | 1.47 ± 0.17 d | 12.60 ± 0.40 e | 7.57 ± 0.32 e | |
| Langkawi Island | 24 h | 19.92 ± 1.26 a | 5.33 ± 1.89 a | 0.01 ± 0.02 a | 1.45 ± 0.07 a | NA |
| 105 °C 1× | 86.03 ± 4.62 b | 7.03 ± 0.15 a | 0.17 ± 0.01 b | 2.67 ± 0.23 b | 1.13 ± 0.15 a | |
| 105 °C 2× | 108.55 ± 5.61 c | 7.37 ± 0.23 a | 0.21 ± 0.05 b | 3.00 ± 0.20 b | 1.60 ± 0.10 b | |
| 121 °C 1× | 153.27 ± 6.44 d | 12.33 ± 1.89 b | 0.25 ± 0.03 c | 4.40 ± 0.53 c | 1.67 ± 0.31 c | |
| 121 °C 2× | 206.55 ± 3.23 e | 13.93 ± 2.16 b | 0.27± 0.03 c | 6.47 ± 0.12 d | 2.30 ± 0.44 d | |
| Sah Kecil Island (Kenyir) | 24 h | 22.80 ± 1.25 a | 4.13 ± 1.32 a | 0.11 ± 0.01 a | 2.23 ± 0.21 a | 1.43 ± 0.12 a |
| 105 °C 1× | 67.40 ± 8.97 b | 6.30 ± 0.82 b | 0.13 ± 0.03 b | 3.43 ± 0.35 b | 2.57 ± 0.15 b | |
| 105 °C 2× | 92.84 ± 27.23 b | 7.83 ± 1.12 b | 0.16 ± 0.03 b | 3.87 ± 0.21 b | 2.77 ± 0.15 b | |
| 121 °C 1× | 117.38 ± 3.25 c | 6.03 ± 1.08 b | 0.13 ± 0.01 c | 4.50 ± 0.30 c | 3.23 ± 0.31 c | |
| 121 °C 2× | 146.74 ± 4.00 d | 8.97 ± 0.55 b | 0.23 ± 0.03 d | 5.43 ± 0.47 d | 3.47 ± 0.15 c |
| Site | Treatments | Total DOC (mg C/L) | Hydrophobic Fractions (mg C/L) | Hydrophobic Neutrals (mg C/L) | Hydrophilic Fractions (mg C/L) |
|---|---|---|---|---|---|
| Chini | 24 h | 82.54 ± 11.16 a | 19.86 ± 7.40 a | 5.61 ± 8.09 a | 57.08 ± 15.07 a |
| 105 °C 1× | 343.69 ± 23.81 b | 80.38 ± 14.28 b | 14.34 ± 24.69 b | 248.95 ± 4.21 b | |
| 105 °C 2× | 444.95 ± 9.90 c | 125.19 ± 13.20 c | 0.86 ± 1.49 c | 325.54 ± 25.07 c | |
| 121 °C 1× | 525.83 ± 14.10 d | 136.84 ± 10.23 d | 29.33 ± 6.34 d | 359.66 ± 25.04 d | |
| 121 °C 2× | 715.32 ± 31.60 e | 200.25 ± 0.76 e | 8.48 ± 11.92 e | 506.69 ± 29.80 e | |
| Langkawi | 24 h | 19.53 ± 1.23 a | 13.90 ± 1.30 a | 1.06 ± 0.93 a | 5.36 ± 1.98 a |
| 105 °C 1× | 84.94 ± 6.66 b | 33.24 ± 7.93 b | 8.81 ± 10.93 a | 47.64 ± 20.68 b | |
| 105 °C 2× | 101.70 ± 14.1 0 b | 45.63 ± 9.13 c | 0.16 ± 0.27 a | 66.27 ± 7.19 b | |
| 121 °C 1× | 151.55 ± 18.51 c | 50.91 ± 4.55 d | 7.21 ± 12.49 a | 104.03 ± 34.93 c | |
| 121 °C 2× | 210.93 ± 11.18 d | 60.79 ± 2.48 d | 0.89 ± 0.99 a | 150.13 ± 13.28 d | |
| Kenyir | 24 h | 42.23 ± 0.90 a | 10.43 ± 2.25 a | 1.17 ± 1.16 a | 31.25 ± 4.40 a |
| 105 °C 1× | 59.81 ± 19.18 a | 19.18 ± 3.32 a | 8.33 ± 6.03 b | 32.30 ± 5.02 a | |
| 105 °C 2× | 93.60 ± 43.62 a | 26.44 ± 11.33 a | 0.00 ± 0.00 c | 73.70 ± 31.78 b | |
| 121 °C 1× | 105.21 ± 3.80 b | 29.07 ± 0.77 b | 0.00 ± 0.12 d | 79.49 ± 6.96 c | |
| 121 °C 2× | 135.18 ± 1.08 b | 33.57 ± 1.70 b | 0.00 ± 0.00 d | 109.20 ± 3.0 d |
| Parameters | Unit | Analysis Method |
|---|---|---|
| pH | - | APHA 4500-H B |
| Nitrogen, N | % | MS 417L PART 3: 1994 |
| Phosphorus, P | % | MS 678: PART 8 |
| Potassium, K | % | (MS 417: PART 3: 1994): MS 678: PART VI to IX: 1980 (APHA 3500 K)) |
| Total organic carbon | % | MS 678: PART 3A: 1980 |
| Arsenic, As | mg/kg | USEPA 1311 (APHA 3111-B) |
| Iron, Fe | mg/kg | USEPA 1311 (APHA 3111-B) |
| Cadmium, Cd | mg/kg | USEPA 1311 (APHA 3111-B) |
| Chromium, Cr | mg/kg | USEPA 1311 (APHA 3111-B) |
| Chromium Hexavalent | mg/kg | USEPA 1311 (APHA 3111-B) |
| Lead, Pb | mg/kg | USEPA 1311 (APHA 3111-B) |
| Copper, Cu | mg/kg | USEPA 1311 (APHA 3111-B) |
| Nickel, Ni | mg/kg | USEPA 1311 (APHA 3111-B) |
| Zinc, Zn | mg/kg | USEPA 1311 (APHA 3111-B) |
| Mercury, Hg | mg/kg | USEPA 1311 (APHA 3111-B) |
| Silver, Ag | mg/kg | USEPA 1311 (APHA 3111-B) |
| Particle Size Distribution (PSD) | ||
| Sand | % | USDA/NRCS Soil Taxonomy |
| Silt | % | USDA/NRCS Soil Taxonomy |
| Clay | % | USDA/NRCS Soil Taxonomy |
| Extraction Methods | Procedure |
|---|---|
| Natural Extraction 24 Hours (24 h) | Extracted at room temperature for 24 h incubation |
| Autoclave 105 °C (105 °C) | Extracted at 105 °C using autoclave for 1 h |
| Autoclave 121 °C (121 °C) | Extracted at 121 °C using autoclave for 1 h |
| Autoclave 105 °C (105 °C 2×) | Extracted at 105 °C using autoclave for 1 h, cooled (≈30 min) and autoclaved for another 1 h |
| Autoclave 121 °C (121 °C 2×) | Extracted at 121 °C using autoclave for 1 h, cooled (≈30 min) and autoclaved for another 1 h |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wan Mohd Zamri, W.M.I.; Sjahrir, F.; Yaacob, N.S.; Dzulkafli, N.F.; Ahmad, M.F.; Abdullah, H.; Maniyam, M.N.; Hashim, E.F.; Kawasaki, N.; Komatsu, K.; et al. Assessment of Aqueous Extraction Methods on Extractable Organic Matter and Hydrophobic/Hydrophilic Fractions of Virgin Forest Soils. Molecules 2021, 26, 2480. https://doi.org/10.3390/molecules26092480
Wan Mohd Zamri WMI, Sjahrir F, Yaacob NS, Dzulkafli NF, Ahmad MF, Abdullah H, Maniyam MN, Hashim EF, Kawasaki N, Komatsu K, et al. Assessment of Aqueous Extraction Methods on Extractable Organic Matter and Hydrophobic/Hydrophilic Fractions of Virgin Forest Soils. Molecules. 2021; 26(9):2480. https://doi.org/10.3390/molecules26092480
Chicago/Turabian StyleWan Mohd Zamri, Wan Muhammad Ikram, Fridelina Sjahrir, Nor Suhaila Yaacob, Noor Fazreen Dzulkafli, Mohd Fadzli Ahmad, Hasdianty Abdullah, Maegala Nallapan Maniyam, Emi Fazlina Hashim, Nobuyuki Kawasaki, Kazuhiro Komatsu, and et al. 2021. "Assessment of Aqueous Extraction Methods on Extractable Organic Matter and Hydrophobic/Hydrophilic Fractions of Virgin Forest Soils" Molecules 26, no. 9: 2480. https://doi.org/10.3390/molecules26092480