Synthesis, Enantiomeric Resolution and Biological Evaluation of HIV Capsid Inhibition Activity for Racemic, (S)- and (R)-PF74
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis and Characterisation
2.2. Biological Evaluation
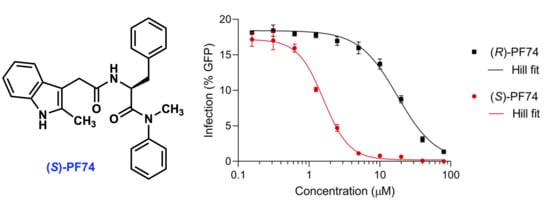
2.2.1. Antiviral Activity
2.2.2. Docking Studies
3. Materials and Methods
3.1. General Chemical Methods
3.2. Compound Synthesis and Characterization
3.2.1. General Procedure to Methyl Ester (3)
3.2.2. General Procedure to Acid (4)
3.2.3. Preparation of PF74 (5)
3.3. Biological Assays—Materials and Methods
3.3.1. Cells and Viruses
3.3.2. Virus Titrations
3.3.3. Compound Fixed Dose (Virus Titration) Assay
3.3.4. Compound Dose Response (Fixed Virus Dose) Assay
3.4. Molecular Docking Methods
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Sample Availability
References
- Bean, P. New drug targets for HIV. Clin. Infect. Dis. 2005, 41 (Suppl. S1), S96–S100. [Google Scholar] [CrossRef] [Green Version]
- UNAIDS. Global HIV & AIDS Statistics–2020 Fact Sheet. Available online: https://www.unaids.org/en/resources/fact-sheet (accessed on 16 May 2021).
- Zhan, P.; Pannecouque, C.; De Clercq, E.; Liu, X. Anti-HIV Drug Discovery and Development: Current Innovations and Future Trends. J. Med. Chem. 2016, 59, 2849–2878. [Google Scholar] [CrossRef] [PubMed]
- Le Sage, V.; Mouland, A.J.; Valiente-Echeverría, F. Roles of HIV-1 capsid in viral replication and immune evasion. Virus Res. 2014, 193, 116–129. [Google Scholar] [CrossRef] [PubMed]
- Blair, W.S.; Pickford, C.; Irving, S.L.; Brown, D.G.; Anderson, M.; Bazin, R.; Cao, J.; Ciaramella, G.; Isaacson, J.; Jackson, L.; et al. HIV Capsid is a Tractable Target for Small Molecule Therapeutic Intervention. PLoS Pathog. 2010, 6, e1001220. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bhattacharya, A.; Alam, S.L.; Fricke, T.; Zadrozny, K.; Sedzicki, J.; Taylor, A.B.; Demeler, B.; Pornillos, O.; Ganser-Pornillos, B.K.; Diaz-Griffero, F.; et al. Structural basis of HIV-1 capsid recognition by PF74 and CPSF6. Proc. Natl. Acad. Sci. USA 2014, 111, 18625–18630. [Google Scholar] [CrossRef] [Green Version]
- Chen, B. HIV Capsid Assembly, Mechanism, and Structure. Biochemistry 2016, 55, 2539–2552. [Google Scholar] [CrossRef] [PubMed]
- Shi, J.; Zhou, J.; Shah, V.B.; Aiken, C.; Whitby, K. Small-molecule inhibition of human immunodeficiency virus type 1 infection by virus capsid destabilization. J. Virol. 2011, 85, 542–549. [Google Scholar] [CrossRef] [Green Version]
- Gres, A.T.; Kirby, K.A.; KewalRamani, V.N.; Tanner, J.J.; Pornillos, O.; Sarafianos, S.G. X-ray crystal structures of native HIV-1 capsid protein reveal conformational variability. Science 2015, 349, 99–103. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rankovic, S.; Ramalho, R.; Aiken, C.; Rousso, I. PF74 Reinforces the HIV-1 Capsid To Impair Reverse Transcription-Induced Uncoating. J. Virol. 2018, 92, e00845-18. [Google Scholar] [CrossRef] [Green Version]
- Sun, L.; Dick, A.; Meuser, M.E.; Huang, T.; Zalloum, W.A.; Chen, C.-H.; Cherukupalli, S.; Xu, S.; Ding, X.; Gao, P.; et al. Design, Synthesis, and Mechanism Study of Benzenesulfonamide-Containing Phenylalanine Derivatives as Novel HIV-1 Capsid Inhibitors with Improved Antiviral Activities. J. Med. Chem. 2020, 63, 4790–4810. [Google Scholar] [CrossRef] [PubMed]
- Price, A.J.; Jacques, D.A.; McEwan, W.A.; Fletcher, A.J.; Essig, S.; Chin, J.W.; Halambage, U.D.; Aiken, C.; James, L.C. Host cofactors and pharmacologic ligands share an essential interface in HIV-1 capsid that is lost upon disassembly. PLoS Pathog. 2014, 10, e1004459. [Google Scholar] [CrossRef]
- Halambage, U.D.; Wong, J.P.; Melancon, B.J.; Lindsley, C.W.; Aiken, C. Microplate-based assay for identifying small molecules that bind a specific intersubunit interface within the assembled HIV-1 capsid. Antimicrob. Agents Chemother. 2015, 59, 5190–5195. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Meuser, M.E.; Reddy, P.A.N.; Dick, A.; Maurancy, J.M.; Salvino, J.M.; Cocklin, S. Rapid Optimization of the Metabolic Stability of a Human Immunodeficiency Virus Type-1 Capsid Inhibitor Using a Multistep Computational Workflow. J. Med. Chem. 2021, 64, 3747–3766. [Google Scholar] [CrossRef] [PubMed]
- El-Faham, A.; Albericio, F. Peptide Coupling Reagents, More than a Letter Soup. Chem. Rev. 2011, 111, 6557–6602. [Google Scholar] [CrossRef]
- Nguyen, L.A.; He, H.; Pham-Huy, C. Chiral drugs: An overview. Int. J. Biomed. Sci. 2006, 2, 85–100. [Google Scholar]
- Rosales-Hurtado, M.; Lebeau, A.; Bourouh, C.; Cebrian-Torrejon, G.; Albalat, M.; Jean, M.; Naubron, J.V.; Annicotte, J.S.; Benfodda, Z.; Meffre, P. Improved synthesis, resolution, absolute configuration determination and biological evaluation of HLM006474 enantiomers. Bioorg. Med. Chem. Lett. 2019, 29, 380–382. [Google Scholar] [CrossRef] [PubMed]
- Bechthold, E.; Schreiber, J.A.; Lehmkuhl, K.; Frehland, B.; Schepmann, D.; Bernal, F.A.; Daniliuc, C.; Álvarez, I.; Garcia, C.V.; Schmidt, T.J.; et al. Ifenprodil Stereoisomers: Synthesis, Absolute Configuration, and Correlation with Biological Activity. J. Med. Chem. 2021, 64, 1170–1179. [Google Scholar] [CrossRef] [PubMed]
- Beutner, G.L.; Young, I.S.; Davies, M.L.; Hickey, M.R.; Park, H.; Stevens, J.M.; Ye, Q. TCFH–NMI: Direct Access to N-Acyl Imidazoliums for Challenging Amide Bond Formations. Org. Lett. 2018, 20, 4218–4222. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kemp, D.S.; Rebek, J. Peptide racemization mechanism. Kinetic isotope effect as a means of distinguishing enolization from oxazolone formation. J. Am. Chem. Soc. 1970, 92, 5792–5793. [Google Scholar] [CrossRef]
- Fray, M.J. Investigation of Epimer Formation in Amide-Coupling Reactions: An Experiment for Advanced Undergraduate Students. J. Chem. Educ. 2014, 91, 136–140. [Google Scholar] [CrossRef]
- Demaison, C.; Parsley, K.; Brouns, G.; Scherr, M.; Battmer, K.; Kinnon, C.; Grez, M.; Thrasher, A.J. High-level transduction and gene expression in hematopoietic repopulating cells using a human immunodeficiency virus type 1-based lentiviral vector containing an internal spleen focus forming virus promoter. Hum. Gene Ther. 2002, 13, 803–813. [Google Scholar] [CrossRef] [PubMed]
- Rihn, S.J.; Aziz, M.A.; Stewart, D.G.; Hughes, J.; Turnbull, M.L.; Varela, M.; Sugrue, E.; Herd, C.S.; Stanifer, M.; Sinkins, S.P.; et al. TRIM69 Inhibits Vesicular Stomatitis Indiana Virus. J. Virol. 2019, 93, e00951-19. [Google Scholar] [CrossRef] [PubMed] [Green Version]







Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ruddell, S.; Sugrue, E.; Memarzadeh, S.; Hellam, L.M.; Wilson, S.J.; France, D.J. Synthesis, Enantiomeric Resolution and Biological Evaluation of HIV Capsid Inhibition Activity for Racemic, (S)- and (R)-PF74. Molecules 2021, 26, 3919. https://doi.org/10.3390/molecules26133919
Ruddell S, Sugrue E, Memarzadeh S, Hellam LM, Wilson SJ, France DJ. Synthesis, Enantiomeric Resolution and Biological Evaluation of HIV Capsid Inhibition Activity for Racemic, (S)- and (R)-PF74. Molecules. 2021; 26(13):3919. https://doi.org/10.3390/molecules26133919
Chicago/Turabian StyleRuddell, Stuart, Elena Sugrue, Sarah Memarzadeh, Lorna Mae Hellam, Sam J. Wilson, and David J. France. 2021. "Synthesis, Enantiomeric Resolution and Biological Evaluation of HIV Capsid Inhibition Activity for Racemic, (S)- and (R)-PF74" Molecules 26, no. 13: 3919. https://doi.org/10.3390/molecules26133919