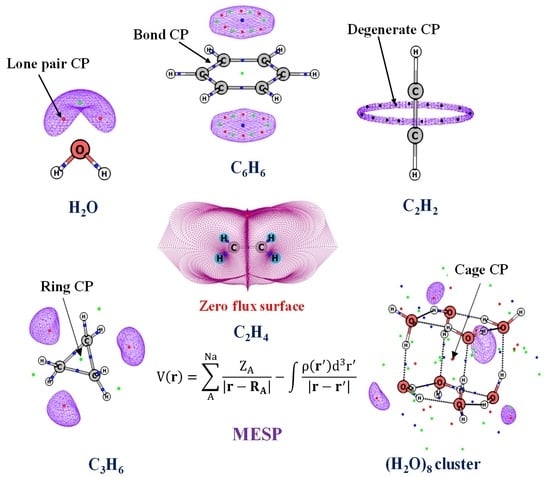
Electrostatic Potential Topology for Probing Molecular Structure, Bonding and Reactivity
Abstract
:1. Introduction
2. MESP Topology: Early Basic Studies and Algorithm Development
3. Recent Studies on MESP Topology and Algorithm Development
4. Applications of MESP Topology to Face Selectivity, Cation Binding and Reaction Mechanisms
5. Electrostatic Potential for Intermolecular Complexation (EPIC) Model and Its Applications
6. MESP Topography, π Systems and Substituent Effects
7. MESP as a Tool for the Quantification and Characterization of Noncovalent Complexes
8. Concluding Remarks and Future Outlook
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Bader, R.F.W. Atoms in Molecules. A Quantum Theory; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Gadre, S.R. Computational Chemistry: Reviews of Current Trends; Leszczynski, J., Ed.; World Scientific: Singapore, 2000; Volume 4. [Google Scholar]
- Gadre, S.R.; Shirsat, R.N. Electrostatics of Atoms and Molecules; Universities Press: Hyderabad, India, 2000. [Google Scholar]
- Hohenberg, P.; Kohn, W. Inhomogeneous electron gas. Phys. Rev. B 1964, 136, B864–B871. [Google Scholar] [CrossRef] [Green Version]
- Bader, R.F.W. A Quantum Theory of Molecular Structure and Its Applications. Chem. Rev. 1991, 91, 893–928. [Google Scholar] [CrossRef]
- Bader, R.F.W. Definition of molecular structure: By choice or by appeal to observation? J. Phys. Chem. A 2010, 114, 7431–7444. [Google Scholar] [CrossRef] [PubMed]
- Bader, R.F.W.; Laidig, K.E. The definition of a chemical bond, of molecular structure and of its change as exemplified by the structure diagram for C4H7+. J. Mol. Struct. Theochem. 1992, 261, 1–20. [Google Scholar] [CrossRef]
- Bader, R.F.W.; MacDougall, P.J. Toward a Theory of Chemical Reactivity Based on the Charge Density. J. Am. Chem. Soc. 1985, 107, 6788–6795. [Google Scholar] [CrossRef]
- Bader, R.F.W.; Nguyen-Dang, T.T.; Tal, Y. A topological theory of molecular structure. Rep. Prog. Phys. 1981, 44, 893–948. [Google Scholar] [CrossRef]
- Bader, R.F.W.; Nguyen-Dang, T.T. Quantum Theory of Atoms in Molecules–Dalton Revisited. In Advances in Quantum Chemistry; Löwdin, P.-O., Ed.; Academic Press: New York, NY, USA, 1981; Volume 14, pp. 63–124. [Google Scholar]
- Cremer, D.; Kraka, E. A Description of the Chemical Bond in Terms of Local Properties of Electron Density and Energy. Croat. Chem. Acta 1984, 57, 1259–1281. [Google Scholar]
- Kraka, E.; Cremer, D. Description of chemical reactions in terms of the properties of the electron density. J. Mol. Struct. Theochem. 1992, 255, 189–206. [Google Scholar] [CrossRef]
- Edgecombe, K.E.; Smith, V.H. Nonnuclear Maxima in the Charge Density. Z. Nat. Sect. A J. Phys. Sci. 1993, 48, 127–133. [Google Scholar] [CrossRef]
- Cao, W.L.; Gatti, C.; MacDougall, P.J.; Bader, R.F.W. On the presence of non-nuclear attractors in the charge distributions of Li and Na clusters. Chem. Phys. Lett. 1987, 141, 380–385. [Google Scholar] [CrossRef]
- Gatti, C.; Fantucci, P.; Pacchioni, G. Charge density topological study of bonding in lithium clusters—Part I: Planar Lin clusters (n = 4, 5, 6). Theor. Chim. Acta 1987, 72, 433–458. [Google Scholar] [CrossRef]
- Bertini, L.; Cargnoni, F.; Gatti, C. Chemical insight into electron density and wave functions: Software developments and applications to crystals, molecular complexes and materials science. Theor. Chem. Acc. 2007, 117, 847–884. [Google Scholar] [CrossRef]
- Cioslowski, J.; Mixon, S.T. Universality among topological properties of electron density associated with the hydrogen–hydrogen nonbonding interactions. Can. J. Chem. 1992, 70, 443–449. [Google Scholar] [CrossRef]
- Haaland, A.; Shorokhov, D.J.; Tverdova, N.V. Topological analysis of electron densities: Is the presence of an atomic interaction line in an equilibrium geometry a sufficient condition for the existence of a chemical bond? Chem. Eur. J. 2004, 10, 4416–4421. [Google Scholar] [CrossRef]
- Krapp, A.; Frenking, G. Is this a chemical bond? A theoretical study of Ng2@C60 (Ng = He, Ne, Ar, Kr, Xe). Chem. Eur. J. 2007, 13, 8256–8270. [Google Scholar] [CrossRef]
- Poater, J.; Visser, R.; Solà, M.; Bickelhaupt, F.M. Polycyclic benzenoids: Why kinked is more stable than straight. J. Org. Chem. 2007, 72, 1134–1142. [Google Scholar] [CrossRef]
- Shahbazian, S. Revisiting the foundations of the quantum theory of atoms in molecules: Some open problems. Int. J. Quantum Chem. 2018, 118, 1–10. [Google Scholar] [CrossRef]
- Bader, R.F.W. Bond paths are not chemical bonds. J. Phys. Chem. A 2009, 113, 10391–10396. [Google Scholar] [CrossRef] [Green Version]
- Politzer, P.; Truhlar, D.G. (Eds.) Chemical Applications of Atomic and Molecular Electrostatic Potentials; Plenum: New York, NY, USA, 1981. [Google Scholar]
- Murray, J.S.; Sen, K. (Eds.) Molecular Electrostatic Potentials: Concepts and Applications; Elsevier: Amsterdam, The Netherlands, 1996. [Google Scholar]
- Politzer, P.; Abrahmsen, L.; Sjoberg, P. Effects of amino and nitro substituents upon the electrostatic potential of an aromatic ring. J. Am. Chem. Soc. 1984, 106, 855–860. [Google Scholar] [CrossRef]
- Politzer, P.; Laurence, P.R. Relationships between the electrostatic potential, epoxide hydrase inhibition and carcinogenicity for some hydrocarbon and halogenated hydrocarbon epoxides. Carcinogenesis 1984, 5, 845–848. [Google Scholar] [CrossRef]
- Pathak, R.K.; Gadre, S.R. Maximal and minimal characteristics of molecular electrostatic potentials. J. Chem. Phys. 1990, 93, 1770–1773. [Google Scholar] [CrossRef]
- Popelier, P.L.A.; Brémond, É.A.G. Geometrically faithful homeomorphisms between the electron density and the bare nuclear potential. Int. J. Quantum Chem. 2009, 109, 2542–2553. [Google Scholar] [CrossRef]
- Bonaccorsi, R.; Pullman, A.; Scrocco, E.; Tomasi, J. The molecular electrostatic potentials for the nucleic acid bases: Adenine, thymine, and cytosine. Theor. Chim. Acta 1972, 24, 51–60. [Google Scholar] [CrossRef]
- Bonaccorsi, R.; Scrocco, E.; Tomasi, J. Molecular SCF calculations for the ground state of some three-membered ring molecules: (CH2)3, (CH2)2NH, (CH2,)2NH2, (CH2)2O, (CH2)2S, (CH)2CH2, and N2CH2. J. Chem. Phys. 1970, 52, 5270. [Google Scholar] [CrossRef]
- Scrocco, E.; Tomasi, J. The electrostatic molecular potential as a tool for the interpretation of molecular properties. Topcurrchem 1973, 42, 95–170. [Google Scholar]
- Scrocco, E.; Tomasi, J. Electronic Molecular Structure, Reactivity and Intermolecular Forces: An Euristic Interpretation by Means of Electrostatic Molecular Potentials. In Advances in Quantum Chemistry; Löwdin, P.-O., Ed.; Academic Press: New York, NY, USA, 1978; Volume 11, pp. 115–193. [Google Scholar]
- Politzer, P.; Daiker, K.C.; Donnelly, R.A. Molecular electrostatic potentials: A new approach to the study of the metabolic and carcinogenic activities of hydrocarbons. Cancer Lett. 1976, 2, 17–23. [Google Scholar] [CrossRef]
- Politzer, P.; Donnelly, R.A.; Daiker, K.C. Molecular electrostatic potentials. Mechanistic aspects of electrophilic attack on furan. J. Chem. Soc. Chem. Commun. 1973, 617–618. [Google Scholar] [CrossRef]
- Politzer, P.; Sukumar, N.; Jayasuriya, K.; Ranganathan, S. Computational evaluation and comparison of some nitramine properties. J. Am. Chem. Soc. 1988, 110, 3425–3430. [Google Scholar] [CrossRef]
- Brinck, T.; Murray, J.S.; Politzer, P. Quantitative determination of the total local polarity (charge separation) in molecules. Mol. Phys. 1992, 76, 609–617. [Google Scholar] [CrossRef]
- Haeberlein, M.; Murray, J.S.; Brinck, T.; Politzer, P. Calculated electrostatic potentials and local surface ionization energies of para-substituted anilines as measures of substituent effects. Can. J. Chem. 1992, 70, 2209–2214. [Google Scholar] [CrossRef]
- Murray, J.S.; Paulsen, K.; Politzer, P. Molecular surface electrostatic potentials in the analysis of non-hydrogen-bonding noncovalent interactions. Proc. Indian Acad. Sci. Chem. Sci. 1994, 106, 267–275. [Google Scholar]
- Bonnaccorsi, R.; Scrocco, E.; Tomasi, J.; Pullman, A. Ab initio molecular electrostatic potentials—Guanine compared to adenine. Theor. Chim. Acta 1975, 36, 339–344. [Google Scholar] [CrossRef]
- Bulat, F.A.; Toro-Labbé, A.; Brinck, T.; Murray, J.S.; Politzer, P. Quantitative analysis of molecular surfaces: Areas, volumes, electrostatic potentials and average local ionization energies. J. Mol. Model. 2010, 16, 1679–1691. [Google Scholar] [CrossRef]
- Nagy, P.; Náray-Szabó, G. Electrostatic lock-and-key model for the analysis of inhibitor recognition by dihydrofolate reductase. Can. J. Chem. 1985, 63, 1694–1698. [Google Scholar] [CrossRef]
- Náray-Szabó, G. Analysis of molecular recognition: Steric electrostatic and hydrophobic complementarity. J. Mol. Recognit. 1993, 6, 205–210. [Google Scholar] [CrossRef]
- Gadre, S.R.; Bendale, R.D. On the similarity between molecular electron densities, electrostatic potentials and bare nuclear potentials. Chem. Phys. Lett. 1986, 130, 515–521. [Google Scholar] [CrossRef]
- Malcolm, N.O.J.; Popelier, P.L.A. The full topology of the Laplacian of the electron density: Scrutinising a physical basis for the VSEPR model. Faraday Discuss. 2003, 124, 353–363. [Google Scholar] [CrossRef]
- Popelier, P.L.A. On the full topology of the laplacian of the electron density. Coord. Chem. Rev. 2000, 197, 169–189. [Google Scholar] [CrossRef]
- Popelier, P.L.A. The Nature of the Chemical Bond. Revisited; Frenking, G., Shaik, S., Eds.; Wiley-VCH: Weinheim, Germany, 2014; pp. 271–308. [Google Scholar]
- Silvi, B.; Savin, A. Classification of chemical bonds based on topological analysis of electron localization functions. Nature 1994, 371, 683–686. [Google Scholar] [CrossRef]
- Polo, V.; Andres, J.; Berski, S.; Domingo, L.R.; Silvi, B. Understanding reaction mechanisms in organic chemistry from catastrophe theory applied to the electron localization function topology. J. Phys. Chem. A 2008, 112, 7128–7136. [Google Scholar] [CrossRef]
- Keith, T.A.; Bader, R.F.W. Topological analysis of magnetically induced molecular current distributions. J. Chem. Phys. 1993, 99, 3669–3682. [Google Scholar] [CrossRef]
- Pendas, A.M.; Hernandez-Trujillo, J. The Ehrenfest force field: Topology and consequences for the definition of an atom in a molecule. J. Chem. Phys. 2012, 137, 134101. [Google Scholar] [CrossRef]
- Pendas, A.M.; Francisco, E.; Blanco, M.A.; Gatti, C. Bond paths as privileged exchange channels. Chem. A Eur. J. 2007, 13, 9362–9371. [Google Scholar] [CrossRef]
- Weinstein, H.; Politzer, P.; Srebrenik, S. A misconception concerning the electronic density distribution of an atom. Theor. Chim. Acta 1975, 38, 159–163. [Google Scholar] [CrossRef]
- Sen, K.D.; Politzer, P. Characteristic features of the electrostatic potentials of singly negative monoatomic ions. J. Chem. Phys. 1989, 90, 4370. [Google Scholar] [CrossRef]
- Sen, K.D.; Politzer, P. Approximate radii for singly negative ions of 3d, 4d, and 5d metal atoms. J. Chem. Phys. 1989, 91, 5123. [Google Scholar] [CrossRef]
- Gadre, S.R.; Pathak, R.K. Nonexistence of local maxima in molecular electrostatic potential maps. Proc. Indian Acad. Sci. Chem Sci. 1990, 102, 189–192. [Google Scholar]
- Gadre, S.R.; Kulkarni, S.A.; Pathak, R.K. Reply to the comment on: Maximal and minimal characteristics of molecular electrostatic potentials: Some further extensions. J. Chem. Phys. 1991, 94, 8639. [Google Scholar] [CrossRef] [Green Version]
- Silberbach, H. Comment on: Maximal and minimal characteristics of molecular electrostatic potentials. J. Chem. Phys. 1991, 94, 8638. [Google Scholar] [CrossRef] [Green Version]
- Shirsat, R.N.; Bapat, S.V.; Gadre, S.R. Molecular electrostatics. A comprehensive topographical approach. Chem. Phys. Lett. 1992, 200, 373–378. [Google Scholar] [CrossRef]
- Gadre, S.R.; Shrivastava, I.H. Shapes and sizes of molecular anions via topographical analysis of electrostatic potential. J. Chem. Phys. 1991, 94, 4384–4390. [Google Scholar] [CrossRef]
- Gadre, S.R.; Koelmel, C.; Shrivastava, I.H. Deriving chemical parameters from electrostatic potential maps of molecular anions. Inorg. Chem. 1992, 31, 2279–2281. [Google Scholar] [CrossRef]
- Gadre, S.R.; Kulkarni, S.A.; Shrivastava, I.H. Molecular electrostatic potentials: A topographical study. J. Chem. Phys. 1992, 96, 5253–5260. [Google Scholar] [CrossRef]
- Popelier, P.L.A. Atoms in Molecules. An. Introduction; Pearson Education: Harlow, UK, 2000. [Google Scholar]
- Malcolm, N.O.J.; Popelier, P.L.A. On the Full Topology of the Laplacian of the Electron Density II: Umbrella Inversion of the Ammonia Molecule. J. Phys. Chem. A 2001, 105, 7638–7645. [Google Scholar] [CrossRef]
- Luque, F.J.; Illas, F.; Orozco, M. Comparative study of the molecular electrostatic potential obtained from different wavefunctions. Reliability of the semiempirical MNDO wavefunction. J. Comput. Chem. 1990, 11, 416–430. [Google Scholar] [CrossRef]
- Luque, F.J.; Orozco, M.; Illas, F.; Rubio, J. Effect of electron correlation on the electrostatic potential distribution of molecules. J. Am. Chem. Soc. 1991, 113, 5203–5211. [Google Scholar] [CrossRef]
- Orozco, M.; Luque, F.J. On the use of mixed basis sets to compute accurate molecular electrostatic potentials. Chem. Phys. Lett. 1989, 160, 305–310. [Google Scholar] [CrossRef]
- Gadre, S.R.; Kulkarni, S.A.; Suresh, C.H.; Shrivastava, I.H. Basis set dependence of the molecular electrostatic potential topography. A case study of substituted benzenes. Chem. Phys. Lett. 1995, 239, 273–281. [Google Scholar] [CrossRef]
- Kulkarni, S.A. Electron correlation effects on the topography of molecular electrostatic potentials. Chem. Phys. Lett. 1996, 254, 268–273. [Google Scholar] [CrossRef]
- Gadre, S.R.; Shrivastava, I.H.; Kulkarni, S.A. Application of rigorous bounds for efficient evaluation of molecular electrostatic potentials. Chem. Phys. Lett. 1990, 170, 271–276. [Google Scholar] [CrossRef]
- Gadre, S.R.; Bapat, S.; Shrivastava, I. Computation of molecular electrostatic potential: An efficient algorithm and parallelization. J. Comput. Chem. 1991, 15, 203–206. [Google Scholar] [CrossRef]
- Gadre, S.R.; Bapat, S.V.; Sundararajan, K.; Shrivastava, I.H. A general parallel algorithm for the generation of moleular electrostatic potential maps. Chem. Phys. Lett. 1990, 175, 307–312. [Google Scholar] [CrossRef]
- Malcolm, N.O.J.; Popelier, P.L.A. An improved algorithm to locate critical points in a 3D scalar field as implemented in the program MORPHY. J. Comput. Chem. 2003, 24, 437–442. [Google Scholar] [CrossRef]
- Leboeuf, M.; Köster, A.M.; Jug, K.; Salahub, D.R. Topological analysis of the molecular electrostatic potential. J. Chem. Phys. 1999, 111, 4893–4905. [Google Scholar] [CrossRef]
- Hopf, H. Vektorfelder in n-dimensionalen Mannigfaltigkeiten. Math. Ann. 1926, 96, 225–250. [Google Scholar] [CrossRef]
- Balanarayan, P.; Gadre, S.R. Topography of molecular scalar fields. I. Algorithm and Poincaré-Hopf relation. J. Chem. Phys. 2003, 119, 5037–5043. [Google Scholar] [CrossRef]
- Roy, D.; Balanarayan, P.; Gadre, S.R. An appraisal of Poincaŕ-Hopf relation and application to topography of molecular electrostatic potentials. J. Chem. Phys. 2008, 129, 174103. [Google Scholar] [CrossRef]
- Gadre, S.R.; Shirsat, R.N.; Limaye, A.C. Molecular Tailoring Approach for Simulation of Electrostatic Properties. J. Phys. Chem. 1994, 98, 9165–9169. [Google Scholar] [CrossRef]
- Babu, K.; Gadre, S.R. Ab initio quality one-electron properties of large molecules: Development and testing of molecular tailoring approach. J. Comput. Chem. 2003, 24, 484–495. [Google Scholar] [CrossRef]
- Babu, K.; Ganesh, V.; Gadre, S.R.; Ghermani, N.-E. Tailoring approach for exploring electron densities and electrostatic potentials of molecular crystals. Theor. Chem. Acc. 2004, 111, 255–263. [Google Scholar] [CrossRef]
- Yeole, S.D.; Gadre, S.R. On the applicability of fragmentation methods to conjugated π systems within density functional framework. J. Chem. Phys. 2010, 132, 094102. [Google Scholar] [CrossRef]
- Sahu, N.; Yeole, S.D.; Gadre, S.R. Appraisal of molecular tailoring approach for large clusters. J. Chem. Phys. 2013, 138, 104101. [Google Scholar] [CrossRef]
- Sahu, N.; Gadre, S.R. Molecular tailoring approach: A route for ab initio treatment of large clusters. Acc. Chem. Res. 2014, 47, 2739–2747. [Google Scholar] [CrossRef]
- Yeole, S.D.; Gadre, S.R. Topography of scalar fields: Molecular clusters and π-conjugated systems. J. Phys. Chem. A 2011, 115, 12769–12779. [Google Scholar] [CrossRef]
- Yeole, S.D.; López, R.; Gadre, S.R. Rapid topography mapping of scalar fields: Large molecular clusters. J. Chem. Phys. 2012, 137, 74116–74122. [Google Scholar] [CrossRef] [Green Version]
- Fernández Rico, J.; López, R.; Ema, I.; Ramírez, G. Deformed atoms in molecules: Analytical representation of atomic densities for Gaussian type orbitals. J. Mol. Struct. Theochem. 2005, 727, 115–121. [Google Scholar] [CrossRef]
- Ganesh, V.; Kavathekar, R.; Rahalkar, A.; Gadre, S.R. WebProp: Web interface for ab initio calculation of molecular one-electron properties. J. Comput. Chem. 2008, 29, 488–495. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Yeole, S.D.; Gadre, S.R.; López, R.; Rico, J.F.; Ramírez, G.; Ema, I.; Zorrilla, D. DAMQT 2.1. 0: A new version of the DAMQT package enabled with the topographical analysis of electron density and electrostatic potential in molecules. J. Comput. Chem. 2015, 36, 2350–2359. [Google Scholar] [CrossRef] [PubMed]
- Limaye, A.C.; Gadre, S.R. UNIVIS-2000: An indigenously developed comprehensive visualization package. Curr. Sci. 2001, 80, 1296–1301. [Google Scholar]
- Ganesh, V. MeTA studio: A cross platform, programmable IDE for computational chemist. J. Comput. Chem. 2009, 30, 661–672. [Google Scholar] [CrossRef]
- Kumar, A.; Gadre, S.R.; Mohan, N.; Suresh, C.H. Lone pairs: An electrostatic viewpoint. J. Phys. Chem. A 2014, 118, 526–532. [Google Scholar] [CrossRef]
- Bijina, P.V.; Suresh, C.H.; Gadre, S.R. Electrostatics for probing lone pairs and their interactions. J. Comput. Chem. 2018, 39, 488–499. [Google Scholar] [CrossRef]
- Kumar, A.; Gadre, S.R. On the electrostatic nature of electrides. Phys. Chem. Chem. Phys. 2015, 17, 15030–15035. [Google Scholar] [CrossRef]
- Kumar, A.; Gadre, S.R. Exploring the Gradient Paths and Zero Flux Surfaces of Molecular Electrostatic Potential. J. Chem. Theory Comput. 2016, 12, 1705–1713. [Google Scholar] [CrossRef]
- Popelier, P.L.A. A fast algorithm to compute atomic charges based on the topology of the electron density. Theoret. Chem. Acc. 2001, 105, 393–399. [Google Scholar] [CrossRef]
- Mata, I.; Molins, E.; Espinosa, E. Zero-Flux Surfaces of the Electrostatic Potential: The Border of Influence Zones of Nucleophilic and Electrophilic Sites in Crystalline Environment. J. Phys. Chem. A 2007, 111, 9859–9870. [Google Scholar] [CrossRef]
- Kumar, A.; Gadre, S.R. Molecular Electrostatic Potential-Based Atoms in Molecules: Shielding Effects and Reactivity Patterns. Aust. J. Chem. 2016, 69, 975–982. [Google Scholar] [CrossRef]
- Mehta, G.; Khan, F.A.; Gadre, S.R.; Shirsat, R.N.; Ganguly, B.; Chandrasekhar, J. Electrostatic vs. orbital control of facial selectivities in π systems: Experimental and theoretical study of electrophilic additions to 7-isopropylidenenorbornanes. Angew. Chem. Int. Ed. Engl. 1994, 33, 1390–1392. [Google Scholar] [CrossRef]
- Mehta, G.; Gunasekaran, G.; Gadre, S.R.; Shirsat, R.N.; Ganguly, B.; Chandrasekhar, J. Electrophilic additions to 7-methylenenorbornenes and 7-isopropylidenenorbornenes: Can remote substituents swamp electrostatic control of π-face selectivity? J. Org. Chem. 1994, 59, 1953–1955. [Google Scholar] [CrossRef]
- Mehta, G.; Singh, S.R.; Balanarayan, P.; Gadre, S.R. Electrophilic additions to a 2-methylenebicyclo [2.1.1] hexane system: Probing π-face selectivity for electrostatic and orbital effects. Org. Lett. 2002, 4, 2297–2300. [Google Scholar] [CrossRef]
- Mehta, G.; Ravikrishna, C.; Gadre, S.R.; Suresh, C.H.; Kalyanaraman, P.; Chandrasekhar, J. Face selectivity in electrophilic additions to methylenenorsnoutanes: Relative importance of through-space, through-bond and electrostatic interactions. Chem. Commun. 1998, 975–976. [Google Scholar] [CrossRef]
- Suresh, C.H.; Koga, N.; Gadre, S.R. Molecular Electrostatic Potential and Electron Density Topography: Structure and Reactivity of (substituted arene)Cr(CO)3 Complexes. Organometallics 2000, 19, 3008–3015. [Google Scholar] [CrossRef]
- Suresh, C.H.; Koga, N.; Gadre, S.R. Revisiting Markovnikov addition to alkenes via molecular electrostatic potential. J. Org. Chem. 2001, 66, 6883–6890. [Google Scholar] [CrossRef] [PubMed]
- Balanarayan, P.; Gadre, S.R. Why are carborane acids so acidic? An electrostatic interpretation of Brønsted acid strengths. Inorg. Chem. 2005, 44, 9613–9615. [Google Scholar] [CrossRef]
- Balanarayan, P.; Kavathekar, R.; Gadre, S.R. Electrostatic potential topography for exploring electronic reorganizations in 1,3 dipolar cycloadditions. J. Phys. Chem. A 2007, 111, 2733–2738. [Google Scholar] [CrossRef]
- Phukan, A.K.; Kalagi, R.P.; Gadre, S.R.; Jemmis, E.D. Structure, reactivity and aromaticity of acenes and their BN analogues: A density functional and electrostatic investigation. Inorg. Chem. 2004, 43, 5824–5832. [Google Scholar] [CrossRef]
- Sandhya, K.S.; Remya, G.S.; Suresh, C.H. Pincer ligand modifications to tune the activation barrier for H2 elimination in water splitting Milstein catalyst. Inorg. Chem. 2015, 54, 11150–11156. [Google Scholar] [CrossRef]
- Mathew, J.; Suresh, C.H. Assessment of stereoelectronic effects in Grubbs first-generation olefin metathesis catalysis using molecular electrostatic potential. Organometallics 2011, 30, 1438–1444. [Google Scholar] [CrossRef]
- Sandhya, K.S.; Suresh, C.H. Designing metal hydride complexes for water splitting reactions: A molecular electrostatic potential approach. Dalton Trans. 2014, 43, 12279–12287. [Google Scholar] [CrossRef]
- Anjali, B.A.; Suresh, C.H. Interpreting oxidative addition of Ph–X (X = CH3, F, Cl, and Br) to monoligated Pd(0) catalysts using molecular electrostatic potential. ACS Omega 2017, 2, 4196–4206. [Google Scholar] [CrossRef] [Green Version]
- Gejji, S.P.; Suresh, C.H.; Bartolotti, L.J.; Gadre, S.R. Electrostatic potential as a harbinger of cation coordination: CF3SO3- ion as a model example. J. Phys. Chem. A 1997, 101, 5678–5686. [Google Scholar] [CrossRef]
- Gejji, S.P.; Suresh, C.H.; Babu, K.; Gadre, S.R. Ab initio structure and vibrational frequencies of (CF3SO2)2N-Li+ ion pairs. J. Phys. Chem. A 1999, 103, 7474–7480. [Google Scholar] [CrossRef]
- Gejji, S.P.; Gadre, S.R.; Barge, V.J. Theoretical investigations on structure, electrostatic potentials and vibrational frequencies of diglyme and Li+–(diglyme) conformers. Chem. Phys. Lett. 2001, 344, 527–535. [Google Scholar] [CrossRef]
- Suresh, C.H.; Gadre, S.R.; Gejji, S.P. Theoretical studies on the structure of M+BF−4 ion pairs M = Li+, NH+4: The role of electrostatics and electron correlation. Theor. Chem. Acc. 1998, 99, 151–157. [Google Scholar] [CrossRef]
- Gadre, S.R.; Pundlik, S.S. Complementary electrostatics for the study of DNA base-pair interactions. J. Phys. Chem. B 1997, 101, 3298–3303. [Google Scholar] [CrossRef]
- Pundlik, S.S.; Gadre, S.R. Structure and stability of DNA base trimers: An electrostatic approach. J. Phys. Chem. B 1997, 101, 9657–9662. [Google Scholar] [CrossRef]
- Pingale, S.S.; Gadre, S.R.; Bartolotti, L.J. Electrostatic insights into molecular hydration process: A case study of crown ethers. J. Phys. Chem. A 1998, 102, 9987–9992. [Google Scholar] [CrossRef]
- Gadre, S.R.; Kulkarni, A.D. Molecular electrostatics for exploring hydration patterns of molecules: 2—Formamide. Ind. J. Chem. A 2000, 39, 50–59. [Google Scholar]
- Gadre, S.R.; Babu, K.; Rendell, A.P. Electrostatics for exploring hydration patterns of molecules. 3. Uracil. J. Phys. Chem. A 2000, 104, 8976–8982. [Google Scholar] [CrossRef]
- Jovan Jose, K.V.; Gadre, S.R. Ab initio study on (CO2)n clusters via electrostatics- and molecular tailoring-based algorithm. Int. J. Quantum Chem. 2009, 109, 2238–2247. [Google Scholar] [CrossRef]
- Jovan Jose, K.V.; Gadre, S.R. An ab initio investigation on (CO2)n and CO2(Ar)m clusters: Geometries and IR spectra. J. Chem. Phys. 2008, 128, 124310. [Google Scholar] [CrossRef] [PubMed]
- Yeole, S.D.; Gadre, S.R. Molecular cluster building algorithm: Electrostatic guidelines and molecular tailoring approach. J. Chem. Phys. 2011, 134, 084111. [Google Scholar] [CrossRef] [PubMed]
- Hammett, L.P. Some relations between reaction rates and equilibrium constants. Chem. Rev. 1935, 17, 125–136. [Google Scholar] [CrossRef]
- Taft, R.W. The General Nature of the Proportionality of Polar Effects of Substituent Groups in Organic Chemistry. J. Am. Chem. Soc. 1953, 75, 4231–4238. [Google Scholar] [CrossRef]
- Murray, J.S.; Politzer, P. Electrostatic potentials of amine nitrogens as a measure of the total electron-attracting tendencies of substituents. Chem. Phys. Lett. 1988, 152, 364–370. [Google Scholar] [CrossRef]
- Gadre, S.R.; Suresh, C.H. Electronic Perturbations of the Aromatic Nucleus: Hammett Constants and Electrostatic Potential Topography. J. Org. Chem. 1997, 62, 2625–2627. [Google Scholar] [CrossRef]
- Suresh, C.H.; Gadre, S.R. A novel electrostatic approach to substituent constants: Doubly substituted benzenes. J. Am. Chem. Soc. 1998, 120, 7049–7055. [Google Scholar] [CrossRef]
- Suresh, C.H.; Gadre, S.R. Electrostatic potential minimum of the aromatic ring as a measure of substituent constant. J. Phys. Chem. A 2007, 111, 710–714. [Google Scholar] [CrossRef]
- Galabov, B.; Ilieva, S.; Schaefer, H.F. An efficient computational approach for the evaluation of substituent constants. J. Org. Chem. 2006, 71, 6382–6387. [Google Scholar] [CrossRef]
- Suresh, C.H.; Alexander, P.; Vijayalakshmi, K.P.; Sajith, P.K.; Gadre, S.R. Use of molecular electrostatic potential for quantitative assessment of inductive effect. Phys. Chem. Chem. Phys. 2008, 10, 6492–6499. [Google Scholar] [CrossRef]
- Sayyed, F.B.; Suresh, C.H.; Gadre, S.R. Appraisal of through-bond and through-space substituent effects via molecular electrostatic potential topography. J. Phys. Chem. A 2010, 114, 12330–12333. [Google Scholar] [CrossRef]
- Sayyed, F.B.; Suresh, C.H. Quantification of substituent effects using molecular electrostatic potentials: Additive nature and proximity effects. New J. Chem. 2009, 33, 2465–2471. [Google Scholar] [CrossRef]
- Sayyed, F.B.; Suresh, C.H. Quantitative Assessment of Substituent Effects on Cation−π Interactions Using Molecular Electrostatic Potential Topography. J. Phys. Chem. A 2011, 115, 9300–9307. [Google Scholar] [CrossRef]
- Sayyed, F.B.; Suresh, C.H. Accurate Prediction of Cation−π Interaction Energy Using Substituent Effects. J. Phys. Chem. A 2012, 116, 5723–5732. [Google Scholar] [CrossRef]
- Remya, G.S.; Suresh, C.H. Quantification and classification of substituent effects in organic chemistry: A theoretical molecular electrostatic potential study. Phys. Chem. Chem. Phys. 2016, 18, 20615–20626. [Google Scholar] [CrossRef]
- Clar, E. The Aromatic Sextet; Wiley: London, UK, 1972. [Google Scholar]
- Suresh, C.H.; Gadre, S.R. Clar’s aromatic sextet theory revisited via molecular electrostatic potential topography. J. Org. Chem. 1999, 64, 2505–2512. [Google Scholar] [CrossRef]
- Jiang, Y.; Zhang, H. Stability and reactivities based on moment analysis. Theor. Chim. Acta 1989, 75, 279–297. [Google Scholar] [CrossRef]
- Zhou, Z.; Parr, R.G. New measures of aromaticity: Absolute hardness and relative hardness. J. Am. Chem. Soc. 1989, 111, 7371–7379. [Google Scholar] [CrossRef]
- Vijayalakshmi, K.P.; Suresh, C.H. Pictorial representation and validation of Clar’s aromatic sextet theory using molecular electrostatic potentials. New J. Chem. 2010, 34, 2132–2138. [Google Scholar] [CrossRef]
- Anjalikrishna, P.K.; Suresh, C.H.; Gadre, S.R. Electrostatic Topographical Viewpoint of π-Conjugation and Aromaticity of Hydrocarbons. J. Phys. Chem. A 2019, 123, 10139–10151. [Google Scholar] [CrossRef] [PubMed]
- Kollman, P.; McKelvey, J.; Johansson, A.; Rothenberg, S. Theoretical studies of hydrogen-bonded dimers. Complexes involving HF, H2O, NH3, CH1, H2S, PH3, HCN, HNC, HCP, CH2NH, H2CS, H2CO, CH4, CF3, H, C2H2, C2H4, C6H6, F- and H3O+. J. Am. Chem. Soc. 1975, 97, 955–965. [Google Scholar] [CrossRef]
- Espinosa, E.; Lecomte, C.; Ghermani, N.E.; Deve’my, J.; Rohmer, M.M.; Be’nard, M.; Molins, E. Hydrogen Bonds: First Quantitative Agreement between Electrostatic Potential Calculations from Experimental X-(X + N) and Theoretical ab Initio SCF Models. J. Am. Chem. Soc. 1996, 118, 2501–2502. [Google Scholar] [CrossRef]
- Mata, I.; Molins, E.; Alkorta, I.; Espinosa, E. Topological Properties of the Electrostatic Potential in Weak and Moderate N…H Hydrogen Bonds. J. Phys. Chem. A 2007, 111, 6425–6433. [Google Scholar] [CrossRef] [PubMed]
- Bobadova-Parvanova, P.; Galabov, B. Ab Initio Molecular-Orbital Study of Hydrogen-Bonded Complexes of Carbonyl Aliphatic Compounds and Hydrogen Fluoride. J. Phys. Chem. A 1998, 102, 1815–1819. [Google Scholar] [CrossRef]
- Dimitrova, V.; Ilieva, S.; Galabov, B. Electrostatic Potential at Atomic Sites as a Reactivity Descriptor for Hydrogen Bonding. Complexes of Monosubstituted Acetylenes and Ammonia. J. Phys. Chem. A 2002, 106, 11801–11805. [Google Scholar] [CrossRef]
- Galabov, B.; Bobadova-Parvanova, P. Molecular Electrostatic Potential as Reactivity Index in Hydrogen Bonding: Ab Initio Molecular Orbital Study of Complexes of Nitrile and Carbonyl Compounds with Hydrogen Fluoride. J. Phys. Chem. A 1999, 103, 6793–6799. [Google Scholar] [CrossRef]
- Gadre, S.R.; Bhadane, P.K. Patterns in hydrogen bonding via electrostatic potential topography. J. Chem. Phys. 1997, 107, 22–24. [Google Scholar] [CrossRef]
- Gadre, S.R. Molecular electrostatics for exploring complexes of carbonyl compounds and hydrogen fluoride. J. Phys. Chem. A 1999, 103, 3512–3517. [Google Scholar] [CrossRef]
- Brinck, T.; Murray, J.S.; Politzer, P. Surface electrostatic potentials of halogenated methanes as indicators of directional intermolecular interactions. Int. J. Quantum Chem. 1992, 44, 57–64. [Google Scholar] [CrossRef]
- Clark, T.; Hennemann, M.; Murray, J.S.; Politzer, P. Halogen bonding: The σ-hole. J. Mol. Model. 2007, 13, 291–296. [Google Scholar] [CrossRef]
- Politzer, P.; Murray, J.S.; Peralta-Inga, Z. Molecular surface electrostatic potentials in relation to noncovalent interactions in biological systems. Int. J. Quantum Chem. 2001, 85, 676–684. [Google Scholar]
- Murray, J.S.; Politzer, P. Molecular electrostatic potentials and noncovalent interactions. WIREs Comput. Mol. Sci. 2017, 7, e1326. [Google Scholar] [CrossRef]
- Roy, D.K.; Balanarayan, P.; Gadre, S.R. Signatures of molecular recognition from the topography of electrostatic potential. J. Chem. Sci. 2009, 121, 815–821. [Google Scholar] [CrossRef]
- Mohan, N.; Suresh, C.H. A molecular electrostatic potential analysis of hydrogen, halogen, and dihydrogen bonds. J. Phys. Chem. A 2014, 118, 1697–1705. [Google Scholar] [CrossRef]
- Mohan, N.; Suresh, C.H.; Kumar, A.; Gadre, S.R. Molecular electrostatics for probing lone pair–π interactions. Phys. Chem. Chem. Phys. 2013, 15, 18401–18409. [Google Scholar] [CrossRef]
- Mohan, N.; Suresh, C.H. Anion Receptors Based on Highly Fluorinated Aromatic Scaffolds. J. Phys. Chem. A 2014, 118, 4315–4324. [Google Scholar] [CrossRef]
- Luque, F.J.; Orozco, M.; Bhadane, P.K.; Gadre, S.R. Effect of solvation on the shapes, sizes, and anisotropies of polyatomic anions via molecular electrostatic potential topography: An ab initio self-consistent reaction field approach. J. Chem. Phys. 1994, 100, 6718. [Google Scholar]
- Kermack, W.O.; Robinson, R.J. An explanation of the property of induced polarity of atoms and an interpretation of the theory of partial valencies on an electronic basis. Chem. Soc. Trans. 1922, 121, 427. [Google Scholar] [CrossRef] [Green Version]
- Bartolotti, L.J.; S.R., G.; Parr, R.G. Electronegativities of the elements from simple Xα theory. J. Am. Chm. Soc. 1980, 102, 2945–2948. [Google Scholar] [CrossRef]
- Chattaraj, P.K.; Maiti, B.; Sarkar, U. Philicity: A Unified Treatment of Chemical Reactivity and Selectivity. J. Phys. Chem. A 2003, 25, 4973–4975. [Google Scholar] [CrossRef]
- De Proft, F.; Geerlings, P. Conceptual and computational DFT in the study of aromaticity. Chem. Rev. 2001, 101, 1451–1464. [Google Scholar] [CrossRef]
- Geerlings, P.; De Proft, F. Conceptual DFT: The chemical relevance of higher response functions. Phys. Chem. Chem. Phys. 2008, 10, 3028–3042. [Google Scholar] [CrossRef]
- Matito, E.; Putz, M.V. New link between conceptual density functional theory and electron delocalization. J. Phys. Chem. A 2011, 115, 12459–12462. [Google Scholar] [CrossRef]
- Mortier, W.J.; Genechten, K.V.; Gasteiger, J. Electronegativity equalization: Application and parametrization. J. Am. Chem. Soc. 1985, 107, 829–835. [Google Scholar] [CrossRef]
- Parr, R.G.; Pearson, R.G. Absolute hardness: Companion parameter to absolute electronegativity. J. Am. Chem. Soc. 1983, 105, 7512–7516. [Google Scholar] [CrossRef]
- Putz, M.V.; Chattaraj, P.K. Electrophilicity kernel and its hierarchy through softness in conceptual density functional theory. Int. J. Quantum Chem. 2013, 113, 2163–2171. [Google Scholar] [CrossRef]











Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gadre, S.R.; Suresh, C.H.; Mohan, N. Electrostatic Potential Topology for Probing Molecular Structure, Bonding and Reactivity. Molecules 2021, 26, 3289. https://doi.org/10.3390/molecules26113289
Gadre SR, Suresh CH, Mohan N. Electrostatic Potential Topology for Probing Molecular Structure, Bonding and Reactivity. Molecules. 2021; 26(11):3289. https://doi.org/10.3390/molecules26113289
Chicago/Turabian StyleGadre, Shridhar R., Cherumuttathu H. Suresh, and Neetha Mohan. 2021. "Electrostatic Potential Topology for Probing Molecular Structure, Bonding and Reactivity" Molecules 26, no. 11: 3289. https://doi.org/10.3390/molecules26113289