Influence of Different Frying Processes on the Flavor Characteristics and Sensory Profile of Garlic Oil
Abstract
:1. Introduction
2. Results
2.1. Effects of Different Initial and Terminational Temperatures on Volatile Flavor Compounds
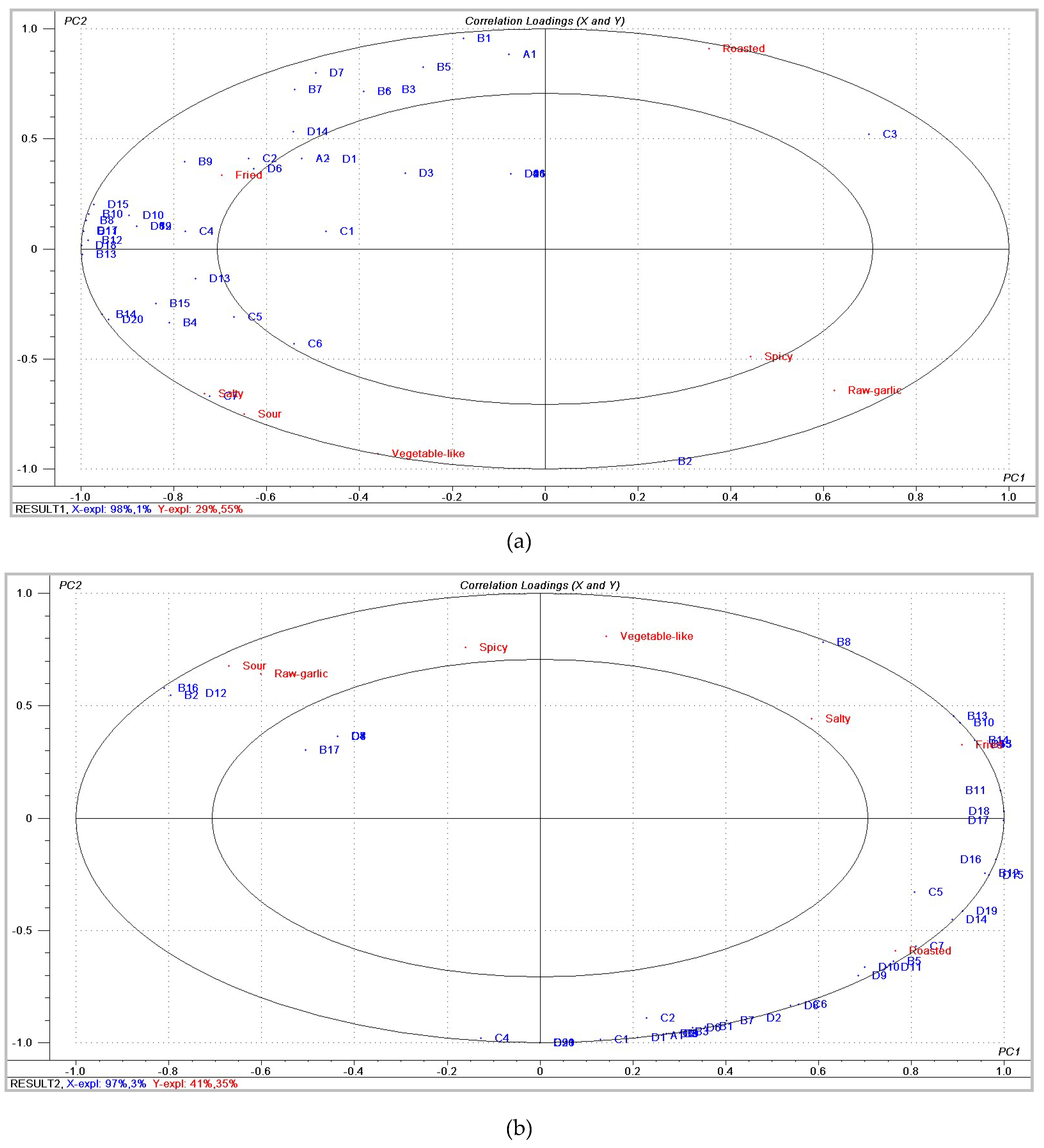
2.2. Relationship between Sensory Descriptors and Flavor Compounds
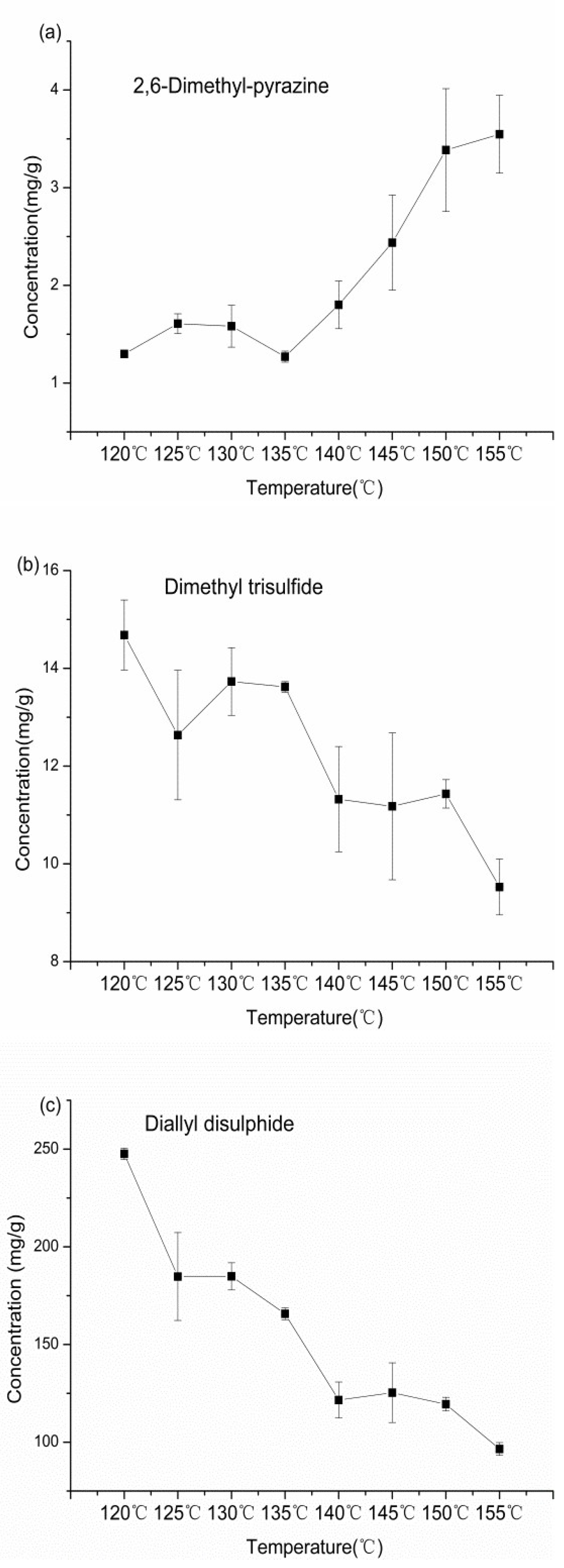
2.3. Generating the Patterns of 2,6-dimethylpyrazine, Dimethyl Trisulfide, and Diallyl Disulfide in Garlic Oil
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Sample Preparation
4.3. Solvent-Assisted Flavor Evaporation (SAFE)
4.4. Gas Chromatography–Mass Spectrometry (GC–MS)
4.5. Quantitative Analysis
4.6. Sensory Evaluation
4.7. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Lanzotti, V. The analysis of onion and garlic. J. Chromatogr. A 2006, 1112, 3–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Miron, T.; Mironchik, M.; Mirelman, D.; Wilchek, M.; Rabinkov, A. Inhibition of tumor growth by a novel approach: In situ allicin generation using targeted alliinase delivery. Mol. Cancer Ther. 2003, 2, 1295–1301. [Google Scholar] [PubMed]
- Tsai, J.C.; Chen, Y.A.; Wu, J.T.; Cheng, K.C.; Lai, P.S.; Liu, K.F.; Lin, Y.K.; Huang, Y.T.; Hsieh, C.W. Extracts from fermented black garlic exhibit a hepatoprotective effect on acute hepatic injury. Molecules 2019, 24, 6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petropoulos, S.; Fernandes, Â.; Barros, L.; Ćirić, A.; Sokovic, M.; Ferreira, I.C.F.R. Antimicrobial and antioxidant properties of various greek garlic genotypes. Food Chem. 2018, 245, 7–12. [Google Scholar] [CrossRef] [Green Version]
- Contardo, I.; Parada, J.; Leiva, A.; Bouchon, P. The effect of vacuum frying on starch gelatinization and its in vitro digestibility in starch–gluten matrices. Food Chem. 2016, 197, 353–358. [Google Scholar] [CrossRef]
- Parkash Kochhar, S.; Gertz, C. New theoretical and practical aspects of the frying process. Eur. J. Lipid. Sci. Tech. 2004, 106, 722–727. [Google Scholar] [CrossRef]
- Song, J.; Kim, M.J.; Kim, Y.J.; Lee, J. Monitoring changes in acid value, total polar material, and antioxidant capacity of oils used for frying chicken. Food Chem. 2017, 220, 306–312. [Google Scholar] [CrossRef]
- Khor, Y.P.; Sim, B.I.; Abas, F.; Lai, O.M.; Wang, Y.; Nehdi, I.A.; Sbihi, H.M.; Gewik, M.M.; Tan, C.P. Influence of carbohydrate- and protein-based foods on the formation of polar lipid fraction during deep-frying. Food Con. 2020, 107, 1–7. [Google Scholar] [CrossRef]
- Yu, T.H.; Wu, C.M.; Rosen, R.T.; Hartman, T.G.; Ho, C.T. Volatile compounds generated from thermal degradation of alliin and deoxyalliin in an aqueous solution. J. Agric. Food Chem. 1993, 42, 146–153. [Google Scholar] [CrossRef]
- Condurso, C.; Cincotta, F.; Tripodi, G.; Merlino, M.; Verzera, A. Influence of drying technologies on the aroma of Sicilian red garlic. LWT–Food Sci. Technol. 2019, 104, 180–185. [Google Scholar] [CrossRef]
- Kim, S.M.; Wu, C.M.; Kobayashi, A.; Kubota, K.; Okumura, J. Volatile compounds in stir-fried garlic. J. Agric. Food Chem. 1995, 43, 2951–2955. [Google Scholar] [CrossRef]
- Yu, T.H.; Lin, L.Y.; Ho, C.T. Volatile compounds of blanched, fried blanched, and baked blanched garlic slices. J. Agric. Food Chem. 1994, 42, 1342–1347. [Google Scholar] [CrossRef]
- Chen, H.T.; Li, M.; Sun, J.; Zhang, N.; Sun, B.G.; Tian, H.Y. Analysis of volatile flavor constituents of fresh garlic and fried garlic oil. Fine. Chem. 2018, 8, 1355–1362. [Google Scholar]
- Kim, N.Y.; Park, M.H.; Jang, E.Y.; Lee, J.H. Volatile distribution in garlic (allium sativum l.) by solid phase microextraction (SPME) with different processing conditions. Food Sci. Biotechnol. 2011, 20, 775–782. [Google Scholar] [CrossRef]
- Xiao, Z.B.; Yu, D.; Niu, Y.W.; Chen, F.; Song, S.Q.; Zhu, J.C.; Zhu, G.Y. Characterization of aroma compounds of Chinese famous liquors by gas chromatography–mass spectrometry and flash GC electronic-nose. J. Chromatogr. B. 2014, 945, 92–100. [Google Scholar] [CrossRef]
- Block, E.; Dane, A.J.; Thomas, S.; Cody, R.B. Applications of direct analysis in real time mass spectrometry (DART-MS) in allium chemistry. 2-propenesulfenic and 2-propenesulfinic acids, diallyl trisulfane s-oxide, and other reactive sulfur compounds from crushed garlic and other alliums. J. Agric. Food Chem. 2010, 5, 4617–4625. [Google Scholar] [CrossRef]
- Bae, S.E.; Cho, S.Y.; Won, Y.D.; Lee, S.H.; Park, H.J. Changes in s-allyl cysteine contents and physicochemical properties of black garlic during heat treatment. LWT–Food Sci. Technol. 2014, 55, 397–402. [Google Scholar] [CrossRef]
- Lee, Y.R.; Yun, K.L.; Hwang, I.G.; Lee, Y.J.; Woo, K.S.; Han, C.S.; Kim, E.S.; Jeong, H.S. Optimization of the processing conditions for heated garlic juice by response surface methodology. Food Sci. Nutr. 2008, 13, 334–339. [Google Scholar] [CrossRef] [Green Version]
- Wang, J.; Cao, Y.; Sun, B.; Wang, C.; Mo, Y. Effect of ultrasound on the activity of alliinase from fresh garlic. Ultrason. Sonochem. 2011, 18, 534–540. [Google Scholar] [CrossRef]
- Calvo-Gómez, O.; Morales-López, J.; López, M.G. Solid-phase microextraction-gaschromatographic-mass spectrometric analysis of garlic oil obtained by hydrodistillation. J. Chromatogr. A. 2004, 1036, 91–93. [Google Scholar] [CrossRef]
- Kimbaris, A.C.; Siatis, N.G.; Daferera, D.J.; Tarantilis, P.A.; Polissiou, M.G. Comparison of distillation and ultrasound-assisted extraction methods for the isolation of sensitive aroma compounds from garlic (allium sativum). Ultrason. Sonochem. 2006, 13, 54–60. [Google Scholar] [CrossRef] [PubMed]
- Block, E.; Cody, R.B.; Dane, A.J.; Sheridan, R.; Vattekkatte, A.; Wang, K. Allium chemistry: Use of new instrumental techniques to “see” reactive organosulfur species formed upon crushing garlic and onion. Pure Appl. Chem. 2010, 82, 535–539. [Google Scholar] [CrossRef]
- Ilić, D.; Nikolić, V.; Stanković, M.; Nikolić, L.; Stanojević, L.; Mladenović-Ranisavljević, I.; Smelcerović, A. Transformation of synthetic allicin: The influence of ultrasound, microwaves, different solvents and temperatures, and the products isolation. Sci. World J. 2012, 2012, 1–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, J.W.; Choi, J.H.; Youn-Soon, K.; Kyung, K.H. Antiyeast potency of heated garlic in relation to the content of allyl alcohol thermally generated from alliin. J. Food Sci. 2006, 71, 1–5. [Google Scholar] [CrossRef]
- Rizzi, G.P. Formation of sulfur-containing flavor compounds from allylic alcohol precursors. J. Food Sci. 1995, 37, 289–302. [Google Scholar]
- Molina-Calle, M.; Priego-Capote, F.; Luque de Castro, M.D. Headspace-GC–MS volatile profile of black garlic, vs, fresh garlic: Evolution along fermentation and behavior under heating. LWT–Food Sci. Technol. 2017, 80, 98–105. [Google Scholar] [CrossRef]
- Tocmo, R.; Wu, Y.; Liang, D.; Fogliano, V.; Huang, D. Boiling enriches the linear polysulfdes and the hydrogen sulfde-releasing activity of garlic. Food Chem. 2017, 221, 1867–1873. [Google Scholar] [CrossRef]
- Zhang, N.; Sun, B.G.; Mao, X.Y.; Chen, H.T.; Zhang, Y.Y. Flavor formation in frying process of green onion (Allium fistulosum L.) deep-fried oil. Food Res. Int. 2019, 121, 296–306. [Google Scholar] [CrossRef]
- Cadwallader, K.; Potts, D.; Brisske-BeVier, L.; Mirarefi, S. Contribution of volatile sulfur compounds to the characteristic aroma of roasted garlic. Acs Symposium. 2011, 1068, 137–151. [Google Scholar]
Sample Availability: Samples of the compounds are available from the authors. |

| Number | Compounds a | Concentration (mg/g) b | RI | Identification Method c | |||
|---|---|---|---|---|---|---|---|
| 110 °C | 115 °C | 120 °C | 125 °C | ||||
| Alcohols | |||||||
| A1 | 2-Propen-1-ol | 0.049 ± 0.008 a | 0.038 ± 0.021 a | 0.075 ± 0.05 b | — | 1123 | MS/RI |
| A2 | (E)-3-Penten-2-ol | — | 0.015 ± 0.011 a | 0.031 ± 0.019 b | — | 1179 | MS/RI |
| Thioethers | |||||||
| B1 | Dimethyl disulfide | 0.021 ± 0.001 a | 0.019 ± 0.011 a | 0.026 ± 0.02 a | — | 1075 | MS/RI |
| B2 | 1-Allyloxy-2,3-epoxypropane | — | — | — | 0.026 ± 0.018 | 1123 | MS |
| B3 | Diallyl sulfide | 0.059 ± 0.006 a | 0.073 ± 0.052a | 0.115 ± 0.078 b | 0.024 ± 0.016 c | 1147 | MS/RI |
| B4 | (E)-Allyl(prop-1-en-1-yl)sulfane | — | 0.023 ± 0.009 a | — | 0.01 ± 0.002 b | 1188 | MS |
| B5 | (E)-1-Methyl-2-(prop-1-en-1-yl)disulfane | 0.101 ± 0.016 a | 0.107 ± 0.067 a | 0.157 ± 0.098 a,b | 0.038 ± 0.018 a,c | 1206 | MS |
| B6 | Methyl allyl disulfide | 0.246 ± 0.006 a | 0.315 ± 0.189 a | 0.445 ± 0.27 a,b | 0.133 ± 0.06 a,c | 1278 | MS/RI |
| B7 | Dimethyl trisulfide | 0.178 ± 0.008 a | 0.25 ± 0.134 b | 0.293 ± 0.163 b | 0.098 ± 0.048 c | 1369 | MS/RI |
| B8 | 1-Allyl-2-isopropyldisulfane | 0.015 ± 0.003 a | 0.042 ± 0.012 b,c | 0.03 ± 0.011 b | 0.02 ± 0.001 a,d | 1421 | MS |
| B9 | Diallyl disulfide | 3.149 ± 0.055 a | 9.334 ± 2.569 b | 6.475 ± 1.543 c,d | 4.538 ± 0.416 a,d | 1468 | MS/RI |
| B10 | (E)-1-Allyl-2-(prop-1-en-1-yl)disulfane | 1.059 ± 0.117 a | 3.219 ± 0.809 b,c | 2.305 ± 0.588 b | 1.415 ± 0.056 a,d | 1457 | MS |
| B11 | Allyl n-propyl sulfide | — | 0.021 ± 0.014 a | 0.024 ± 0.018 a | — | 1450 | MS |
| B12 | Methyl allyl trisulfide | 1.727 ± 0.096 a | 4.59 ± 0.419 b | 3.577 ± 0.554 c | 2.586 ± 0.13 d | 1574 | MS/RI |
| B13 | 1,2-Di-((E)-prop-1-en-1-yl)disulfane | 0.027 ± 0.001 a | 0.098 ± 0.006 b | 0.058 ± 0.011 c | 0.047 ± 0.001 c | 1741 | MS |
| B14 | Diallyl trisulfide | 1.502 ± 0.188 a | 6.926 ± 0.353 b | 4.252 ± 1.646 c | 4.409 ± 0.635 c | 1771 | MS/RI |
| B15 | (Z)-1-Allyl-3-(prop-1-en-1-yl)trisulfane | — | 0.023 ± 0.005 a | — | 0.008 ± 0.001 b | 1776 | MS |
| Aldehydes | |||||||
| C1 | Hexanal | — | 0.022 ± 0.018 a | 0.048 ± 0.037 b | 0.016 ± 0.016 a | 1085 | MS/RI |
| C2 | 4-Heptenal | — | 0.026 ± 0.014 a | 0.041 ± 0.028 b | — | 1169 | MS/RI |
| C3 | (E)-2-Heptenal | 0.029 ± 0.009 | — | — | — | 1321 | MS/RI |
| C4 | (Z)-2-Heptenal | — | 0.04 ± 0.019 a | 0.049 ± 0.027 a | 0.018 ± 0.007 b | 1322 | MS/RI |
| C5 | (E)-2-Octenal | — | 0.011 ± 0.004 a | 0.014 ± 0.002 a | 0.01 ± 0.002 b | 1425 | MS/RI |
| C6 | (E,E)-2,4-Heptadienal | 0.069 ± 0.004a | 0.134 ± 0.022 b | 0.163 ± 0.012 b | 0.146 ± 0.019 b | 1465 | MS/RI |
| C7 | (E,E)-2,4-Decadienal | — | 0.162 ± 0.019 a | 0.104 ± 0.04 b | 0.159 ± 0.001 a | 1759 | MS/RI |
| Heterocyclic compounds | |||||||
| D1 | 2-Pentylfuran | — | 0.01 ± 0.005 a | 0.024 ± 0.002 b | — | 1233 | MS/RI |
| D2 | Methylpyrazine | — | — | 0.046 ± 0.03 | — | 1266 | MS/RI |
| D3 | 4-Methylpyrimidine | 0.025 ± 0.003 a | 0.032 ± 0.012 a | — | 0.006 ± 0.002 b | 1267 | MS |
| D4 | 2-Methyl-2-thiazolidine | — | — | 0.014 ± 0.001 | — | 1295 | MS/RI |
| D5 | 2-Ethenylthiophene | — | 0.019 ± 0.011 | — | — | 1296 | MS/RI |
| D6 | 2,6-Dimethylpyrazine | 0.041 ± 0.001 a | 0.082 ± 0.03 b | 0.108 ± 0.028 b | 0.045 ± 0.009 a | 1319 | MS/RI |
| D7 | 2,5-Dimethylpyrazine | 0.044 ± 0.008 a | 0.061 ± 0.033 a,b | 0.071 ± 0.033 b | 0.014 ± 0.002 c | 1356 | MS/RI |
| D8 | 4-Ethylpyridine | — | 0.006 ± 0.001 | — | — | 1356 | MS/RI |
| D9 | 3-Ethylpyridine | — | — | 0.014 ± 0.006 | — | 1374 | MS/RI |
| D10 | 2-Ethyl-6-methylpyrazine | 0.072 ± 0.003 a | 0.187 ± 0.043 b | 0.178 ± 0.017 b | 0.104 ± 0.013 c | 1381 | MS/RI |
| D11 | 3-Methyl-2-ethylpyrazine | — | — | 0.045 ± 0.019 | — | 1397 | MS/RI |
| D12 | 3,5-Dimethyl-2-ethylpyrazine | — | 0.006 ± 0.001 | — | — | 1428 | MS/RI |
| D13 | 2,5-Dimethyl-3-ethylpyrazine | 0.044 ± 0.001 a | 0.126 ± 0.009 b | 0.143 ± 0.017 b | 0.1 ± 0.008 c | 1440 | MS/RI |
| D14 | 3H-1,2-Dithiole | 0.047 ± 0.008 a | 0.073 ± 0.043 a | 0.097 ± 0.031 a,b | 0.038 ± 0.018 a,c | 1510 | MS/RI |
| D15 | 3-Methyl-3H-1,2-dithiole | 0.485 ± 0.025 a | 1.29 ± 0.264 b | 0.791 ± 0.178 a | 0.523 ± 0.079 a | 1570 | MS |
| D16 | 2-Furanmethanol | — | — | 0.051 ± 0.012 | — | 1670 | MS/RI |
| D17 | 3-Vinyl-4H-1,2-dithiin | 2.868 ± 0.031 a | 8.074 ± 0.731 b | 5.476 ± 0.751 c | 3.958 ± 0.131 d | 1711 | MS/RI |
| D18 | 2-Vinyl-4H-1,2-dithiin | 5.646 ± 0.956 a | 16.572 ± 1.891 b | 10.591 ± 2.486 c,d | 8.398 ± 2.045 a,d | 1821 | MS/RI |
| D19 | Furaneol | — | 0.095 ± 0.011 | — | — | 2045 | MS/RI |
| D20 | 2,3-Dihydro-3,5-dihydroxy-6-methyl-4(H)-pyran-4-one | 0.109 ± 0.005 a | 0.664 ± 0.082 b | 0.429 ± 0.165 c,d | 0.432 ± 0.142 b,d | 2282 | MS/RI |
| Number | Compounds a | Concentration (mg/g) b | RI | Identification Method c | |||
|---|---|---|---|---|---|---|---|
| 145 °C | 150 °C | 155 °C | 160 °C | ||||
| Alcohols | |||||||
| A1 | 2-Propen-1-ol | — | — | 0.038 ± 0.021 a | 0.151 ± 0.033 b | 1123 | MS/RI |
| Thioethers | |||||||
| B1 | Dimethyl disulfide | — | 0.005 ± 0.004 a | 0.019 ± 0.011 b | 0.049 ± 0.003 c | 1075 | MS/RI |
| B2 | 1-Allyloxy-2,3-epoxypropane | 0.036 ± 0.007 a | 0.024 ± 0.012 a | — | — | 1122 | MS |
| B3 | Diallyl sulfide | 0.031 ± 0.001 a | 0.039 ± 0.005 a | 0.073 ± 0.052 b | 0.16 ± 0.001 c | 1147 | MS/RI |
| B4 | (E)-Allyl(prop-1-en-1-yl)sulfane | — | — | 0.023 ± 0.009 | — | 1188 | MS |
| B5 | (E)-1-Methyl-2-(prop-1-en-1-yl)disulfane | 0.032 ± 0.004 a | 0.045 ± 0.014 a | 0.107 ± 0.067 b | 0.116 ± 0.003 b | 1284 | MS |
| B6 | Methyl allyl disulfide | 0.179 ± 0.015 a | 0.206 ± 0.033 a | 0.315 ± 0.189 a | 0.642 ± 0.023 b | 1277 | MS/RI |
| B7 | Dimethyl trisulfide | 0.12 ± 0.015 a | 0.164 ± 0.048 a | 0.25 ± 0.134 a | 0.411 ± 0.001 b | 1368 | MS/RI |
| B8 | 1-Allyl-2-isopropyldisulfane | 0.022 ± 0.003 a | 0.028 ± 0.009 a | 0.042 ± 0.012 b | 0.01 ± 0.001 c | 1420 | MS |
| B9 | Allyl n-propyl sulfide | — | — | 0.021 ± 0.014 a | 0.074 ± 0.003 b | 1450 | MS |
| B10 | (E)-1-Allyl-2-(prop-1-en-1-yl)disulfane | 1.06 ± 0.201 a | 1.24 ± 0.357 a | 3.219 ± 0.809 b | 0.918 ± 0.065 a | 1456 | MS |
| B11 | Diallyl disulfide | 4.94 ± 0.649 a | 5.378 ± 1.634 a | 9.334 ± 2.569 b | 6.065 ± 0.082 a | 1473 | MS/RI |
| B12 | Methyl allyl trisulfide | 1.905 ± 0.504 a | 2.427 ± 0.853 a | 4.59 ± 0.419 b | 3.571 ± 0.179 b | 1574 | MS/RI |
| B13 | 1,2-Di-((E)-prop-1-en-1-yl)disulfane | 0.038 ± 0.007 a | 0.043 ± 0.012 a | 0.098 ± 0.006 b | 0.032 ± 0.005 a | 1741 | MS |
| B14 | Diallyl trisulfide | 2.763 ± 0.929 a | 3.186 ± 0.966 a | 6.926 ± 0.353 b | 2.907 ± 0.117 a | 1770 | MS/RI |
| B15 | (Z)-1-Allyl-3-(prop-1-en-1-yl)trisulfane | — | — | 0.023 ± 0.005 | — | 2282 | MS/RI |
| B16 | (Z)-Allyl(prop-1-en-1-yl)sulfane | 0.009 ± 0.002 a | 0.01 ± 0.006 a | — | — | 1187 | MS |
| B17 | 1-Allyl-3-propyltrisulfane | 0.003 ± 0.001 | — | — | — | 1707 | MS/RI |
| Aldehydes | |||||||
| C1 | Hexanal | — | 0.012 ± 0.004 a | 0.022 ± 0.018 a | 0.128 ± 0.007 b | 1085 | MS/RI |
| C2 | 4-Heptenal | — | 0.026 ± 0.002 a | 0.026 ± 0.014 a | 0.069 ± 0.004 b | 1169 | MS/RI |
| C3 | (Z)-2-Heptenal | — | — | 0.04 ± 0.019 a | 0.143 ± 0.008 b | 1322 | MS/RI |
| C4 | Nonanal | 0.014 ± 0.007 a | — | — | 0.059 ± 0.01 b | 1391 | MS/RI |
| C5 | (E)-2-Octenal | — | 0.006 ± 0.001 a | 0.011 ± 0.004 b | 0.009 ± 0.013 b | 1425 | MS/RI |
| C6 | (E,E)-2,4-Heptadienal | — | — | 0.134 ± 0.022 a | 0.232 ± 0.006 b | 1465 | MS/RI |
| C7 | (E,E)-2,4-Decadienal | — | 0.031 ± 0.003 a | 0.162 ± 0.019 b | 0.163 ± 0.009 b | 1798 | MS/RI |
| C8 | (E)-2-Heptenal | — | 0.012 ± 0.005 | — | — | 1321 | MS/RI |
| C9 | Heptanal | — | — | — | 0.005 ± 0.001 | 1187 | MS/RI |
| Heterocyclic compounds | |||||||
| D1 | 2-Pentylfuran | — | — | 0.01 ± 0.005 a | 0.047 ± 0.013 b | 1233 | MS/RI |
| D2 | 4-Methylpyrimidine | — | — | 0.032 ± 0.012 a | 0.069 ± 0.003 b | 1267 | MS |
| D3 | 3-Methylpyridine | — | — | — | 0.01 ± 0.001 | 1217 | MS |
| D4 | 2-Methyl-2-thiazolidine | — | 0.005 ± 0.001 | — | — | 1294 | MS/RI |
| D5 | 2-Ethenylthiophene | — | — | 0.019 ± 0.011 | — | 1296 | MS/RI |
| D6 | 2,5-Dimethylpyrazine | 0.018 ± 0.002 a | 0.041 ± 0.009 a | 0.082 ± 0.03 b | 0.19 ± 0.007 c | 1319 | MS/RI |
| D7 | 3-Methyl-2-ethylpyrazine | — | 0.012 ± 0.013 | — | — | 1402 | MS |
| D8 | 4-Ethypyridine | — | — | 0.006 ± 0.001 | — | 1356 | MS/RI |
| D9 | 6-Methyl-2-ethyl-pyrazine | 0.039 ± 0.001 a | 0.081 ± 0.012 b | 0.187 ± 0.043 c | 0.227 ± 0.027ac | 1380 | MS/RI |
| D10 | 2,5-Dimethyl-3-ethylpyrazine | 0.053 ± 0.004 a | 0.08 ± 0.018 a | 0.126 ± 0.009 b | 0.141 ± 0.011 b | 1440 | MS/RI |
| D11 | 3,5-Dimethyl-2-ethylpyrazine | — | — | 0.006 ± 0.001 a | 0.007 ± 0.003 a | 1428 | MS/RI |
| D12 | 3-Ethylpyridine | 0.01 ± 0.001 a | 0.017 ± 0.008 a | — | — | 1374 | MS/RI |
| D13 | 2,6-Dimethylpyrazine | 0.01 ± 0.006 a | 0.02 ± 0.007 a | 0.061 ± 0.033 b | 0.098 ± 0.015 c | 1326 | MS/RI |
| D14 | 3H-1,2-Dithiole | 0.021 ± 0.003 a | 0.027 ± 0.019 a | 0.073 ± 0.043 b | 0.064 ± 0.011 b | 1510 | MS/RI |
| D15 | 3-Methyl-3H-1,2-dithiole | 0.491 ± 0.105 a | 0.557 ± 0.199 a | 1.29 ± 0.264 b | 0.969 ± 0.03 b | 1570 | MS |
| D16 | 3-Vinyl-4H-1,2-dithiin | 2.674 ± 0.564 a | 3.019 ± 1.116 a | 8.074 ± 0.731 b | 5.515 ± 0.918 c | 1711 | MS/RI |
| D17 | 2-Vinyl-4H-1,2-dithiin | 6.626 ± 1.858 a | 6.733 ± 2.29 a | 16.572 ± 1.891 b | 10.043 ± 0.525 c | 1822 | MS/RI |
| D18 | 2,3-Dihydro-3,5-dihydroxy-6-methyl-4H-pyran-4-one | 0.115 ± 0.046 a | 0.146 ± 0.095 a | 0.664 ± 0.082 b | 0.293 ± 0.001 c | 2281 | MS/RI |
| D19 | Furaneol | — | — | 0.095 ± 0.011 a | 0.072 ± 0.014 b | 2045 | MS/RI |
| D20 | 3,4-Dihydro-2H-pyran | — | — | — | 0.015 ± 0.001 | 1133 | MS |
| D21 | 2-Ethyltetrahydrothiophene | — | — | — | 0.059 ± 0.004 | 1494 | MS |
| Sample | Salty | Fried | Roasted | Vegetable-Like | Spicy | Sour | Raw Garlic |
|---|---|---|---|---|---|---|---|
| 110Sam | 5.00 a | 6.00 ab | 6.14 a | 3.86 a | 6.14 | 1.86 | 5.14 |
| 115Sam | 7.00 bc | 7.57 a | 4.14 bc | 5.43 ab | 5.64 | 2.29 | 4.00 |
| 120Sam | 6.29 c | 5.29 b | 4.57 b | 4.71 a | 4.86 | 2.00 | 3.57 |
| 125Sam | 6.93 c | 5.14 b | 2.71 c | 6.43 b | 6.29 | 2.29 | 5.86 |
| p | 0.025 | 0.025 | 0.010 | 0.032 | 0.388 | 0.892 | 0.066 |
| Sample | Salty | Fried | Roasted | Vegetable-Like | Spicy | Sour | Raw Garlic |
|---|---|---|---|---|---|---|---|
| 145Sam | 4.67 a | 5.83 | 4.00 a | 5.00 | 5.33 | 3.00 | 5.67 a |
| 150Sam | 6.67 bc | 6.17 | 4.83 a | 6.00 | 6.00 | 3.33 | 4.50 b |
| 155Sam | 7.00 b | 6.83 | 6.50 b | 5.67 | 5.50 | 2.50 | 3.83 bc |
| 160Sam | 5.17 ac | 6.00 | 6.67 b | 4.50 | 5.00 | 2.33 | 3.33 c |
| p | 0.013 | 0.623 | 0.01 | 0.21 | 0.778 | 0.656 | 0 |
| Compounds | Calibration Equations (Y*10^6) | R2 | Concentration (mg/g) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| 120 °C | 125 °C | 130 °C | 135 °C | 140 °C | 145 °C | 150 °C | 155 °C | |||
| Dimethyl trisulfide | y = 2.625 x − 3.113 | 0.999 | 14.68 ± 0.72 | 12.63 ± 1.32 | 13.73 ± 0.69 | 13.62 ± 0.11 | 11.32 ± 1.08 | 11.18 ± 1.5 | 11.43 ± 0.29 | 9.53 ± 0.57 |
| Diallyl disulfide | y = 1.893 x + 42.220 | 0.997 | 247.6 ± 2.88 | 184.8 ± 22.47 | 184.9 ± 6.95 | 165.75 ± 3.03 | 121.6 ± 9.17 | 125.35 ± 15.3 | 119.46 ± 3.52 | 96.54 ± 3.31 |
| 2,6-Dimethyl-pyrazine | y = 1.945 x + 36.005 | 0.999 | 1.3 ± 0.01 | 1.61 ± 0.10 | 1.4 ± 0.26 | 1.27 ± 0.06 | 1.8 ± 0.24 | 2.44 ± 0.49 | 3.39 ± 0.63 | 3.55 ± 0.40 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sun, J.; Sun, B.; Ren, F.; Chen, H.; Zhang, N.; Zhang, Y. Influence of Different Frying Processes on the Flavor Characteristics and Sensory Profile of Garlic Oil. Molecules 2019, 24, 4456. https://doi.org/10.3390/molecules24244456
Sun J, Sun B, Ren F, Chen H, Zhang N, Zhang Y. Influence of Different Frying Processes on the Flavor Characteristics and Sensory Profile of Garlic Oil. Molecules. 2019; 24(24):4456. https://doi.org/10.3390/molecules24244456
Chicago/Turabian StyleSun, Jie, Baoguo Sun, Fazheng Ren, Haitao Chen, Ning Zhang, and Yuyu Zhang. 2019. "Influence of Different Frying Processes on the Flavor Characteristics and Sensory Profile of Garlic Oil" Molecules 24, no. 24: 4456. https://doi.org/10.3390/molecules24244456




