Formation and Stabilization of Gold Nanoparticles in Bovine Serum Albumin Solution
Abstract
1. Introduction
2. Results and Discussion
2.1. UV-Vis Measurements
2.2. TEM and DLS Measurements
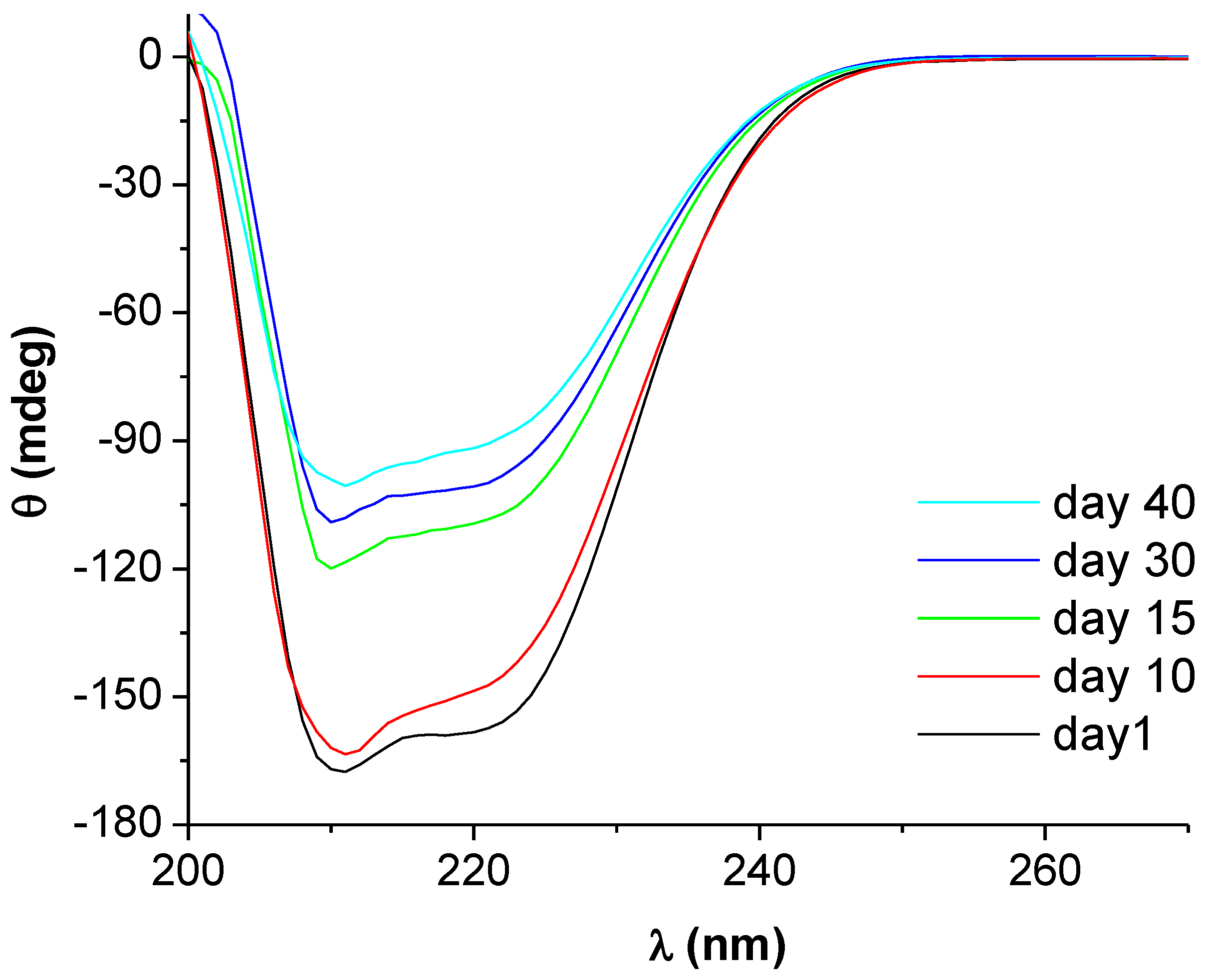
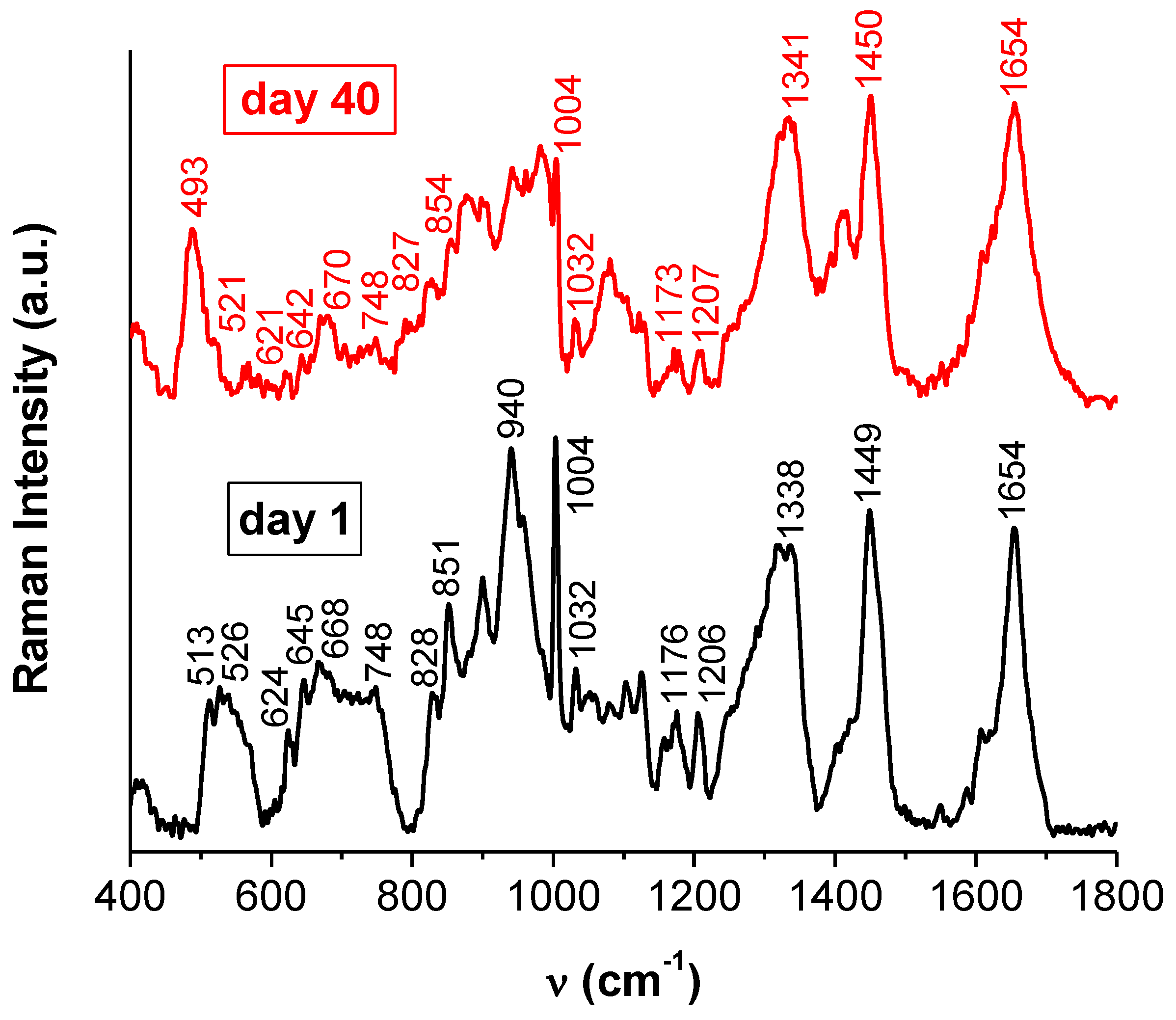
2.3. Circular Dichroism and Raman Spectroscopy
2.3.1. Modes of Adsorption of BSA onto Gold Nanoparticles
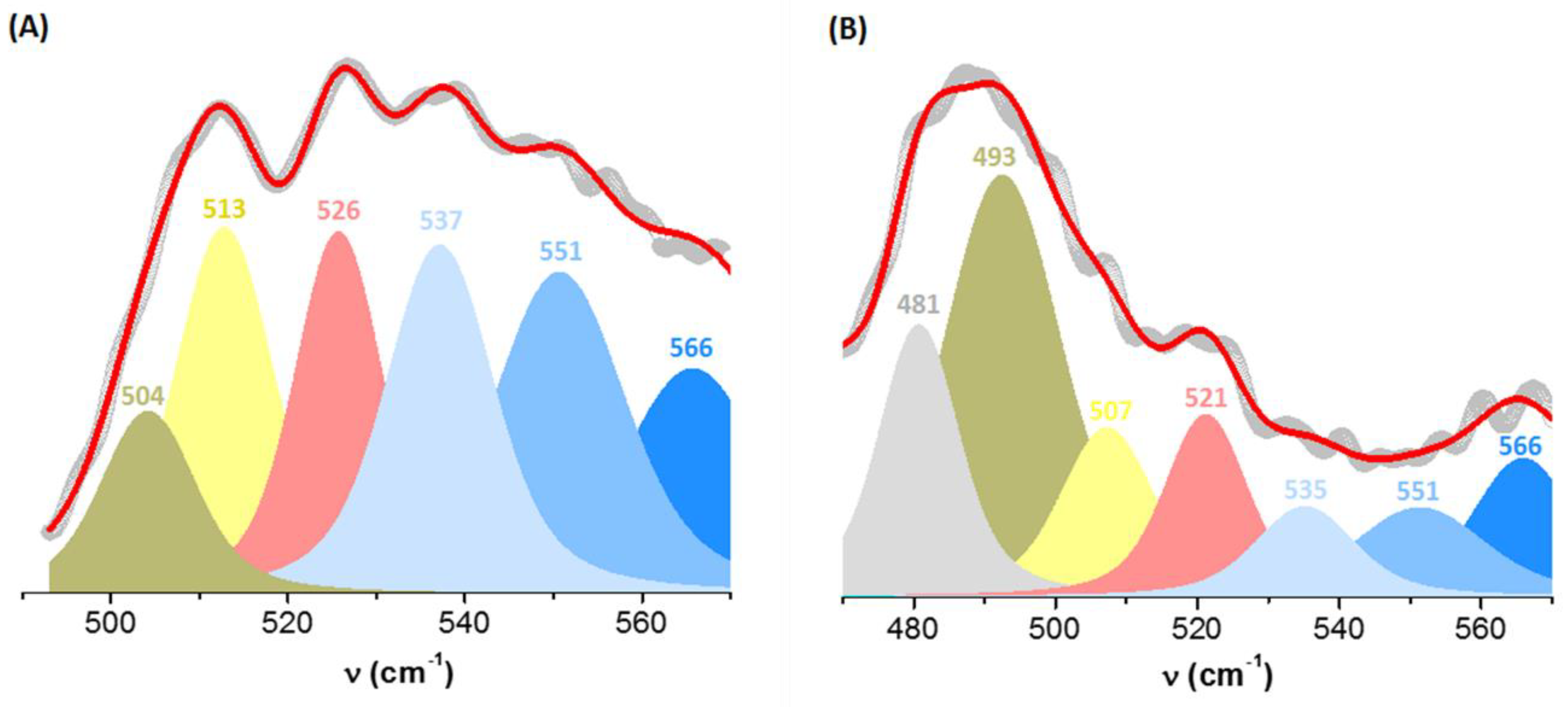
2.3.2. The Configuration of Disulfide Bridges
2.3.3. Estimating the Change in BSA Secondary Structure by Raman Spectroscopy
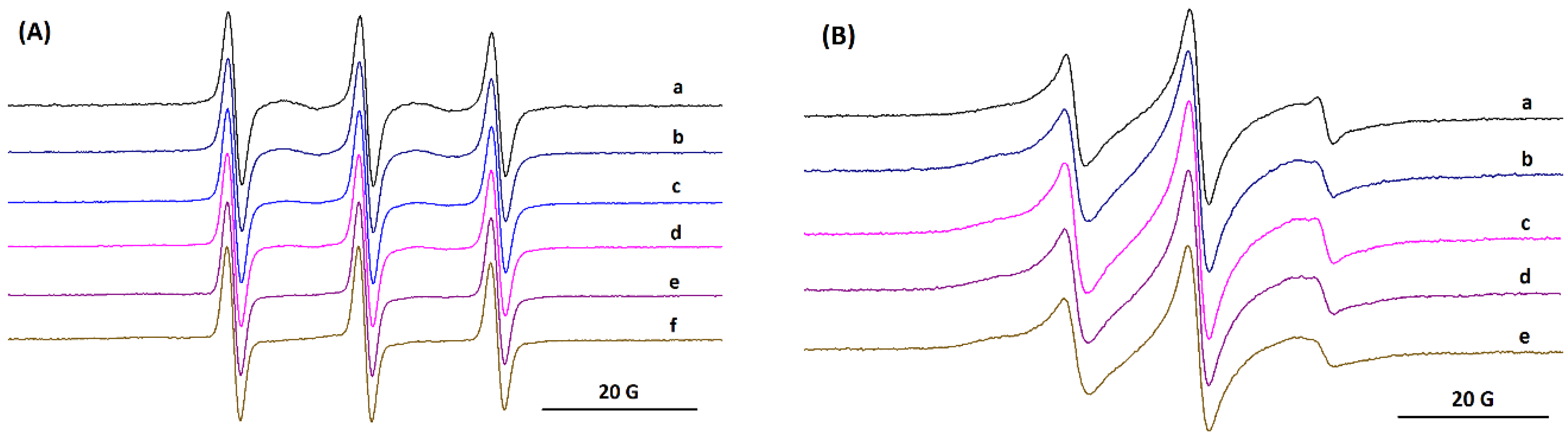
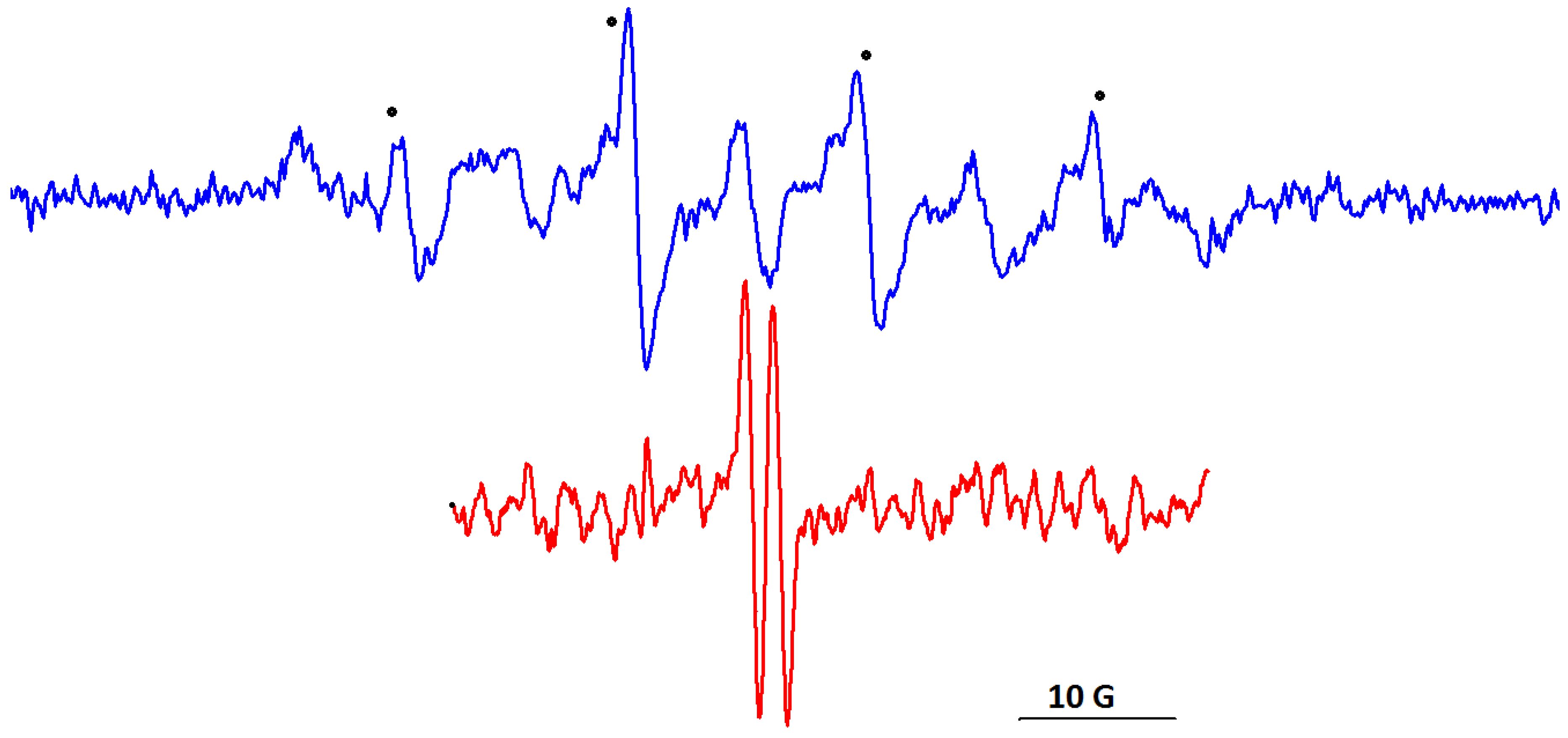
2.4. Electron Paramagnetic Resonance Measurements
3. Materials and Methods
3.1. Sample Preparation
3.2. Instrumentation
3.2.1. UV-Vis Absorption
3.2.2. DLS Measurements
3.2.3. Circular Dichroism Measurements
3.2.4. Raman Spectroscopy
3.2.5. Transmission Electron Microscopy
3.2.6. EPR Spectroscopy
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Sotnikov, D.V.; Berlina, A.N.; Ivanov, V.S.; Zherdev, A.V.; Dzantiev, B.B. Adsorption of proteins on gold nanoparticles: One or more layers? Coll. Surf. B 2019, 173, 557–563. [Google Scholar] [CrossRef] [PubMed]
- Dominguez-Medina, S.; McDonough, S.; Swanglap, P.; Landes, C.F.; Link, S. In situ measurement of bovine serum albumin interaction with gold nanospheres. Langmuir 2012, 28, 9131–9139. [Google Scholar] [CrossRef] [PubMed]
- Casals, E.; Pfaller, T.; Duschl, A.; Oostingh, G.J.; Puntes, V. Time evolution of the nanoparticle protein corona. ACS Nano 2010, 4, 3623–3632. [Google Scholar] [CrossRef] [PubMed]
- Murawala, P.; Tirmale, A.; Shiras, A.; Prasad, B.L.V. In situ synthesized BSA capped gold nanoparticles: Effective carrier of anticancer drug Methotrexate to MCF-7 breast cancer cells. Mater. Sci. Eng. C 2014, 34, 158–167. [Google Scholar] [CrossRef] [PubMed]
- Sardar, R.; Funston, A.M.; Mulvaney, P.; Murray, R.W. Gold nanoparticles: Past, present and future. Langmuir 2009, 25, 13840–13851. [Google Scholar] [CrossRef] [PubMed]
- Turkevich, J.; Stevenson, P.C.; Hillier, J.A. Study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc. 1951, 11, 55–59. [Google Scholar] [CrossRef]
- Brewer, S.H.; Glomm, W.R.; Johnson, M.C.; Knag, M.K.; Franzen, S. Probing BSA binding to citrate-coated gold nanoparticles and surfaces. Langmuir 2005, 21, 9303–9307. [Google Scholar] [CrossRef]
- Nakata, S.; Kido, N.; Hayashi, M.; Hara, M.; Sasabe, H.; Sugawara, T.; Matsuda, T. Chemisorption of proteins and their thiol derivatives onto gold surfaces: Characterization based on electrochemical nonlinearity. Biophys. Chem. 1996, 62, 63–72. [Google Scholar] [CrossRef]
- Silin, V.; Weetall, H.; Vanderah, D.J. SPR studies of the nonspecific adsorption kinetics of human IgG and BSA on gold surfaces modified by self-assembled monolayers (SAMs). J. Colloid Interface Sci. 1997, 185, 94–103. [Google Scholar] [CrossRef]
- De Paoli Lacerda, A.H.; Park, J.J.; Curt Meuse, C.; Pristinski, D.; Becker, M.L.; Karim, A.; Douglas, J.F. Interaction of gold nanoparticles with common human blood proteins. ACS Nano 2010, 4, 365–379. [Google Scholar] [CrossRef]
- Khandelia, R.; Jaiswal, A.; Ghosh, S.S.; Chattopadhyay, A. Gold nanoparticle–protein agglomerates as versatile nanocarriers for drug delivery. Small 2013, 9, 3494–3505. [Google Scholar] [CrossRef] [PubMed]
- Khullar, P.; Singh, V.; Mahal, A.; Dave, P.N.; Thakur, S.; Kaur, G.; Singh, J.; Kamboj, S.S.; Bakshi, M.S. Bovine serum albumin bioconjugated gold nanoparticles: Synthesis, Hemolysis, and cytotoxicity toward cancer cell lines. J. Phys. Chem. C 2012, 116, 8834–8843. [Google Scholar] [CrossRef]
- Xie, J.; Lee, J.Y.; Wang, D.I.C. Synthesis of single-crystalline gold nanoplates in aqueous solutions through biomineralization by serum albumin protein. J. Phys. Chem. C 2007, 111, 10226–10232. [Google Scholar] [CrossRef]
- Basu, N.; Bhattacharya, R.; Mukherjee, P. Protein-mediated autoreduction of gold salts to gold nanoparticles. Biomed. Mater. 2008, 3, 034105. [Google Scholar] [CrossRef] [PubMed]
- Rogozea, A.; Matei, I.; Turcu, I.M.; Ionita, G.; Sahini, V.E.; Salifoglou, A. EPR and circular dichroism solution studies on the interactions of bovine serum albumin with ionic surfactants and β-cyclodextrin. J. Phys. Chem. B 2012, 116, 14245–14253. [Google Scholar] [CrossRef] [PubMed]
- Neacsu, M.V.; Matei, I.; Micutz, M.; Staicu, T.; Precupas, A.; Popa, V.T.; Salifoglou, A.; Ionita, G. Interaction between albumin and Pluronic F127 block copolymer revealed by global and local physicochemical profiling. J. Phys. Chem. B 2016, 120, 4258–4267. [Google Scholar] [CrossRef] [PubMed]
- Matei, I.; Ariciu, A.M.; Neacsu, M.V.; Collauto, A.; Salifoglou, A.; Ionita, G. Cationic spin probe reporting on thermal denaturation and complexation−decomplexation of BSA with SDS. Potential applications in protein purification processes. J. Phys. Chem. B 2014, 118, 11238–11252. [Google Scholar] [CrossRef]
- Ionita, P.; Caragheorgheopol, A.; Gilbert, B.C.; Chechik, V. EPR study of a place-exchange reaction on Au nanoparticles: Two branches of a disulfide molecule do not adsorb adjacent to each other. J. Am. Chem. Soc. 2002, 124, 9048–9049. [Google Scholar] [CrossRef]
- Chechik, V.; Wellsted, H.J.; Korte, A.; Gilbert, B.C.; Horia Caldararu, H.; Ionita, P.; Caragheorgheopol, A. Spin-labelled Au nanoparticles. Faraday Discuss. 2004, 125, 279–291. [Google Scholar] [CrossRef]
- Leslie Au, L.; Lim, B.; Colletti, P.; Jun, Y.-S.; Xia, Y. Synthesis of gold microplates using bovine serum albumin as a reductant and a stabilizer. Chem. Asian J. 2010, 5, 123–129. [Google Scholar]
- Hirayama, K.; Akashi, S.; Furuya, M.; Fukuhara, K.I. Rapid confirmation and revision of the primary structure of bovine serum albumin by ESIMS and Frit-FAB LC/MS. Biochem. Biophys. Res. Commun. 1990, 173, 639–646. [Google Scholar] [CrossRef]
- Haiss, W.; Thanh, N.T.K.; Aveyard, J.; Fernig, D.G. Determination of size and concentration of gold nanoparticles from UV-Vis spectra. Anal. Chem. 2007, 79, 4215–4221. [Google Scholar] [CrossRef] [PubMed]
- Das, T.; Ghosh, P.; Shanavas, M.S.; Maity, A.; Mondal, S.; Purkayastha, P. Protein-templated gold nanoclusters: Size dependent inversion of fluorescence emission in the presence of molecular oxygen. Nanoscale 2012, 4, 6018–6024. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, N.J. Applications of circular dichroism in protein and peptide analysis. Trends Anal. Chem. 1999, 18, 236–244. [Google Scholar] [CrossRef]
- Peters, T. All about Albumin. Biochemistry, Genetics and Medical Application; Academic Press: San Diego, CA, USA, 1996. [Google Scholar]
- Paris, G.; Kraszewski, S.; Ramseyer, C.; Enescu, M. About the structural role of disulfide bridges in serum albumins: Evidence from protein simulated unfolding. Biopolymers 2012, 97, 889–898. [Google Scholar] [CrossRef] [PubMed]
- Tu, A.T. Advances in Spectroscopy, Spectroscopy of Biological Systems; Clark, R.J.H., Hester, R.E., Eds.; Wiley: Chichester, UK, 1986; Volume 13, pp. 47–112. [Google Scholar]
- Hernandez, B.; Pfluger, F.; Lopez-Tobar, E.; Kruglik, S.G.; Garcia-Ramos, J.V.; Sanchez-Cortes, S.; Ghomi, M. Disulfide linkage Raman markers: A reconsideration attempt. J. Raman Spectrosc. 2014, 45, 657–664. [Google Scholar] [CrossRef]
- Fleury, F.; Ianoul, A.; Berjot, M.; Feofanov, A.; Alix, A.J.P.; Nabiev, I. Camptothecin-binding site in human serum albumin and protein transformations induced by drug binding. FEBS Lett. 1997, 411, 215–220. [Google Scholar] [CrossRef]
- Nakamura, K.; Era, S.; Ozaki, Y.; Sogami, M.; Hayashi, T.; Murakami, M. Conformational changes in seventeen cystine disulfide bridges of bovine serum albumin proved by Raman spectroscopy. FEBS Lett. 1997, 417, 375–378. [Google Scholar] [CrossRef]
- Jurasekova, Z.; Marconi, G.; Sanchez-Cortes, S.; Torreggiani, A. Spectroscopic and molecular modeling studies on the binding of the flavonoid luteolin and human serum albumin. Biopolymers 2009, 91, 917–927. [Google Scholar] [CrossRef]
- Navarra, G.; Tinti, A.; Di Foggia, M.; Leone, M.; Militello, V.; Torreggiani, A. Metal ions modulate thermal aggregation of beta-lactoglobulin: A joint chemical and physical characterization. J. Inorg. Biochem. 2014, 137, 64–73. [Google Scholar] [CrossRef]
- Van Wart, H.E.; Scheraga, H.A. Raman spectra of strained disulfides. Effect of rotation about sulfur-sulfur bonds on sulfur-sulfur stretching frequencies. J. Phys. Chem. 1976, 80, 1823–1832. [Google Scholar] [CrossRef]
- Liu, R.; Zi, X.; Kang, Y.; Si, M.; Wu, Y. Surface-enhanced Raman scattering study of human serum on PVA–Ag nanofilm prepared by using electrostatic self-assembly. J. Raman Spectrosc. 2011, 42, 137–144. [Google Scholar] [CrossRef]
- Jurasekova, Z.; Tinti, A.; Torreggiani, A. Use of Raman spectroscopy for the identification of radical-mediated damages in human serum albumin. Anal. Bioanal. Chem. 2011, 400, 2921–2931. [Google Scholar] [CrossRef] [PubMed]
- Podstawka, E.; Ozaki, Y.; Proniewicz, L.M. Adsorption of S–S containing proteins on a colloidal silver surface studied by surface-enhanced Raman spectroscopy. Appl. Spectrosc. 2004, 58, 1147–1156. [Google Scholar] [CrossRef] [PubMed]
- David, C.; Foley, S.; Mavon, C.; Enescu, E. Reductive unfolding of serum albumins uncovered by Raman spectroscopy. Biopolymers 2008, 89, 623–634. [Google Scholar] [CrossRef] [PubMed]
- Pelton, J.T.; McLean, L.R. Spectroscopic methods for analysis of protein secondary structure. Anal. Biochem. 2000, 277, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Ma, Y.; Chechik, V. Aging of Gold Nanoparticles: Ligand Exchange with Disulfides. Langmuir 2011, 27, 14432–14437. [Google Scholar] [CrossRef] [PubMed]
- Brust, M.; Walker, M.; Bethell, D.; Schiffrin, D.J.; Whyman, R. Synthesis of thiol-derivatised gold nanoparticles in a two-phase liquid–liquid system. Chem. Commun. 1994. [Google Scholar] [CrossRef]
- Junk, M.J.N.; Spiess, H.W.; Hinderberger, D. The distribution of fatty acids reveals the functional structure of human serum albumin. Angew. Chem. Int. Ed. 2010, 49, 8755–8759. [Google Scholar] [CrossRef]
- Muravsky, V.; Gurachevskaya, T.; Berezenko, S.; Schnurr, K.; Gurachevsky, A. Fatty acid binding sites of human and bovine albumins: Differences observed by spin probe ESR. Spectrochim. Acta A 2009, 74, 42–47. [Google Scholar] [CrossRef]
- Ionita, P.; Conte, M.; Gilbert, B.C.; Chechik, V. Gold nanoparticle-initiated free radical oxidations and halogen abstractions. Org. Biomol. Chem. 2007, 5, 3504–3509. [Google Scholar] [CrossRef] [PubMed]
- Conte, M.; Miyamura, H.; Kobayashi, S.; Chechik, V. Spin trapping of Au-H intermediate in the alcohol oxidation by supported and unsupported gold catalysts. J. Am. Chem. Soc. 2009, 131, 7189–7196. [Google Scholar] [CrossRef] [PubMed]
- Conte, M.; Liu, X.; Murphy, D.M.; Whiston, K.; Hutchings, G.J. Cyclohexane oxidation using Au/MgO: An investigation of the reaction mechanism. Phys. Chem. Chem. Phys. 2012, 14, 16279–16285. [Google Scholar] [CrossRef] [PubMed]
- Spasojević, I. Free radicals and antioxidants at a glance using EPR spectroscopy. Crit. Rev. Clin. Lab. Sci. 2011, 48, 114–142. [Google Scholar] [CrossRef] [PubMed]
- Buettner, G.R.; Jurkiewicz, B.A. Catalytic metals, ascorbate and free radicals: Combinations to avoid. Radiation Res. 1996, 145, 532–541. [Google Scholar] [CrossRef] [PubMed]
- Buettner, G.R.; Jurkiewicz, B.A. Ascorbate free radical as a marker of oxidative stress: An EPR study. Free Radic. Biol. Med. 1993, 14, 49–55. [Google Scholar] [CrossRef]
- Dichroweb. On-line Analysis for Protein Circular Dichroism Spectra. Available online: http://dichroweb.cryst.bbk.ac.uk (accessed on 2 October 2018).
- Whitmore, L.; Wallace, B.A. DICHROWEB: An online server for protein secondary structure analyses from circular dichroism spectroscopic data. Nucleic Acids Res. 2004, 32, W668–W673. [Google Scholar] [CrossRef] [PubMed]
- Andrade, M.A.; Chacon, P.; Merelo, J.J.; Moran, F. Evaluation of secondary structure of proteins from UV circular dichroism using an unsupervised learning neural network. Prot. Eng. 1993, 6, 383–390. [Google Scholar] [CrossRef] [PubMed]
- Mao, D.; Wachter, E.; Wallace, B.A. Folding of the mitochondrial proton adenosine triphosphatase proteolipid channel in phospholipid vesicles. Biochemistry 1982, 21, 4960–4968. [Google Scholar] [CrossRef]
- Whitmore, L.; Wallace, B.A. Methods of Analysis for Circular Dichroism Spectroscopy of Proteins and the DichroWeb Server. In Advances in Biomedical Spectroscopy, vol. 1, Modern Techniques for Circular Dichroism and Synchrotron Radiation Circular Dichroism Spectroscopy; Wallace, B.A., Janes, R.W., Eds.; IOS Press: Amsterdam, The Netherlands, 2009; pp. 165–182. [Google Scholar]
- Budil, D.E.; Lee, S.; Saxena, S.; Freed, J.H. Nonlinear-least squares analysis of slow-motion EPR spectra in one and two dimensions using a modified Levenberg−Marquardt algorithm. J. Magn. Reson. Ser. A 1996, 120, 155–189. [Google Scholar] [CrossRef]
Sample Availability: Samples of AuNPs/BSA are available from the authors. |




| Sample | Day | Diameter (nm) (Intensity Distribution) | Diameter (nm) (Volume Distribution) |
|---|---|---|---|
| BSA | Initial | 20.4 ± 11.9 | 9.6 ± 4.5 |
| BSA/Au3+ | Initial | 31.1 ± 20.4 | 12.3 ± 6.3 |
| BSA/Au0 | 21 | 38.6 ± 19.4 | 21.5 ± 8.9 |
| BSA/Au0 | 30 | 73.3 ± 30.1 | 46.5 ± 17.7 |
| Day | α-Helix | β-Sheet | Random Coil |
|---|---|---|---|
| 1 | 0.60 | 0.07 | 0.33 |
| 10 | 0.59 | 0.08 | 0.33 |
| 15 | 0.40 | 0.21 | 0.39 |
| 30 | 0.36 | 0.18 | 0.46 |
| 40 | 0.30 | 0.18 | 0.52 |
| Day 1 | Day 40 | ||
|---|---|---|---|
| ν (cm−1) | Area (%) | ν (cm−1) | Area (%) |
| – | – | 481 | 16 |
| 504 | 9 | 493 | 36 |
| 513 | 18 | 507 | 11 |
| 526 | 16 | 521 | 11 |
| 537 | 19 | 535 | 7 |
| 551 | 22 | 551 | 9 |
| 566 | 16 | 566 | 10 |
| Day 1 | Day 40 | Assignment | ||
|---|---|---|---|---|
| ν (cm−1) | Area (%) | ν (cm−1) | Area (%) | |
| 1617 | 11 | 1623 | 13 | β-sheet |
| 1639 | 9 | 1637 | 13 | random coil |
| 1655 | 62 | 1654 | 31 | α-helix |
| 1670 | 8 | 1671 | 19 | β-turn |
| 1688 | 10 | 1694 | 24 | β-turn |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Matei, I.; Buta, C.M.; Turcu, I.M.; Culita, D.; Munteanu, C.; Ionita, G. Formation and Stabilization of Gold Nanoparticles in Bovine Serum Albumin Solution. Molecules 2019, 24, 3395. https://doi.org/10.3390/molecules24183395
Matei I, Buta CM, Turcu IM, Culita D, Munteanu C, Ionita G. Formation and Stabilization of Gold Nanoparticles in Bovine Serum Albumin Solution. Molecules. 2019; 24(18):3395. https://doi.org/10.3390/molecules24183395
Chicago/Turabian StyleMatei, Iulia, Cristina Maria Buta, Ioana Maria Turcu, Daniela Culita, Cornel Munteanu, and Gabriela Ionita. 2019. "Formation and Stabilization of Gold Nanoparticles in Bovine Serum Albumin Solution" Molecules 24, no. 18: 3395. https://doi.org/10.3390/molecules24183395
APA StyleMatei, I., Buta, C. M., Turcu, I. M., Culita, D., Munteanu, C., & Ionita, G. (2019). Formation and Stabilization of Gold Nanoparticles in Bovine Serum Albumin Solution. Molecules, 24(18), 3395. https://doi.org/10.3390/molecules24183395








