Exploring Nanosilver-Coated Hollow Fiber Microfiltration to Mitigate Biofouling for High Loading Membrane Bioreactor
Abstract
:1. Introduction
2. Results and Discussion
2.1. Characterization of the Modified AgNPs-MF Membrane
2.2. Performance of Membrane Bioreactor
2.3. Membrane Fouling Analysis
2.3.1. Permeate Flux Variation
2.3.2. Transmembrane Pressure Variation Test
2.3.3. Resistance Model Analysis
2.3.4. Biofouling Analysis by SEM-EDS
2.3.5. Stability of AgNPs Coated Membrane and Silver Leaching Analysis
3. Material and Methods
3.1. Nanosilver Coated Membrane Fabrication
3.2. MF-MBR Experimental Setup
3.3. Membrane Fouling Analysis
3.4. Analysis of Chemical and Physical Parameters
4. Conclusions
- (1)
- AgNPs coating membrane was successfully fabricated by a simple novel technique. TEM, SEM, EDS and ICP analysis showed that the membrane was coated by high density AgNPs, and there was no significant leaching observed from the AgNPs-MF membrane during the experimental period. Thus, the stability of the AgNPs on the membrane suggested that the antibacterial effects of AgNPs could exist for a longer time in an aerobic bioreactor under high MLSS condition.
- (2)
- There was no adverse effect of the AgNPs to the MF-MBR treatment efficiency on the bioreactor performance. The effluent water quality from the AgNPs-MF and the unmodified MF were similar after 60 days of operation.
- (3)
- The AgNPs-MF membrane module was effective and had direct impacts on reducing the biofouling of the membrane. The AgNPs-MF membrane was not only having antimicrobial effects, but also prevented the bacteria attachment to the membrane surface, thus, reducing biofilm formation. The current study also showed that the membrane in operation mode caused the membrane clogging and biofouling faster than the membrane just submerged in the bioreactor.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Radjenović, J.; Matošić, M.; Mijatović, I.; Petrović, M.; Barceló, D. Membrane Bioreactor (MBR) as an Advanced Wastewater Treatment Technology. In Emerging Contaminants from Industrial and Municipal Waste: Removal Technologies; Barceló, D., Petrovic, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2008; pp. 37–101. [Google Scholar]
- Judd, S. The status of membrane bioreactor technology. Trends Biotechnol. 2008, 26, 109–116. [Google Scholar] [CrossRef]
- Bagheri, M.; Mirbagheri, S.A.; Kamarkhani, A.M.; Bagheri, Z. Modeling of effluent quality parameters in a submerged membrane bioreactor with simultaneous upward and downward aeration treating municipal wastewater using hybrid models. Desalin. Water Treat. 2016, 57, 8068–8089. [Google Scholar] [CrossRef]
- Bagheri, M.; Mirbagheri, S.A. Critical review of fouling mitigation strategies in membrane bioreactors treating water and wastewater. Bioresour. Technol. 2018, 258, 318–334. [Google Scholar] [CrossRef]
- Meng, F.; Shi, B.; Yang, F.; Zhang, H. Effect of hydraulic retention time on membrane fouling and biomass characteristics in submerged membrane bioreactors. Bioprocess Biosyst. Eng. 2007, 30, 359–367. [Google Scholar] [CrossRef]
- Trussell, R.S.; Merlo, R.P.; Hermanowicz, S.W.; Jenkins, D. The effect of organic loading on process performance and membrane fouling in a submerged membrane bioreactor treating municipal wastewater. Water Res. 2006, 40, 2675–2683. [Google Scholar] [CrossRef]
- Menniti, A.; Morgenroth, E. The influence of aeration intensity on predation and EPS production in membrane bioreactors. Water Res. 2010, 44, 2541–2553. [Google Scholar] [CrossRef]
- Bolto, B.A.; Xie, Z. Recent Developments in Fouling Minimization of Membranes Modifed with Silver Nanoparticles. J. Membr. Sci. Res. 2018, 4, 111–120. [Google Scholar]
- Yang, H.-L.; Lin, J.C.-T.; Huang, C. Application of nanosilver surface modification to RO membrane and spacer for mitigating biofouling in seawater desalination. Water Res. 2009, 43, 3777–3786. [Google Scholar] [CrossRef]
- Zodrow, K.; Brunet, L.; Mahendra, S.; Li, D.; Zhang, A.; Li, Q.; Alvarez, P.J.J. Polysulfone ultrafiltration membranes impregnated with silver nanoparticles show improved biofouling resistance and virus removal. Water Res. 2009, 43, 715–723. [Google Scholar] [CrossRef] [Green Version]
- Zhang, M.; Zhang, K.; de Gusseme, B.; Verstraete, W. Biogenic silver nanoparticles (bio-Ag0) decrease biofouling of bio-Ag0/PES nanocomposite membranes. Water Res. 2012, 46, 2077–2087. [Google Scholar] [CrossRef]
- Alpatova, A.; Kim, E.-S.; Sun, X.; Hwang, G.; Liu, Y.; Gamal El-Din, M. Fabrication of porous polymeric nanocomposite membranes with enhanced anti-fouling properties: Effect of casting composition. J. Membr. Sci. 2013, 444, 449–460. [Google Scholar] [CrossRef]
- Liu, X.; Qi, S.; Li, Y.; Yang, L.; Cao, B.; Tang, C.Y. Synthesis and characterization of novel antibacterial silver nanocomposite nanofiltration and forward osmosis membranes based on layer-by-layer assembly. Water Res. 2013, 47, 3081–3092. [Google Scholar] [CrossRef]
- Zhang, C.; Liang, Z.; Hu, Z. Bacterial response to a continuous long-term exposure of silver nanoparticles at sub-ppm silver concentrations in a membrane bioreactor activated sludge system. Water Res. 2014, 50, 350–358. [Google Scholar] [CrossRef]
- Yin, T.; Walker, H.W.; Chen, D.; Yang, Q. Influence of pH and ionic strength on the deposition of silver nanoparticles on microfiltration membranes. J. Membr. Sci. 2014, 449, 9–14. [Google Scholar] [CrossRef]
- Mecha, C.A.; Pillay, V.L. Development and evaluation of woven fabric microfiltration membranes impregnated with silver nanoparticles for potable water treatment. J. Membr. Sci. 2014, 458, 149–156. [Google Scholar] [CrossRef]
- Sun, X.-F.; Qin, J.; Xia, P.-F.; Guo, B.-B.; Yang, C.-M.; Song, C.; Wang, S.-G. Graphene oxide–silver nanoparticle membrane for biofouling control and water purification. Chem. Eng. J. 2015, 281, 53–59. [Google Scholar] [CrossRef]
- Dolina, J.; Dlask, O.; Lederer, T.; Dvořák, L. Mitigation of membrane biofouling through surface modification with different forms of nanosilver. Chem. Eng. J. 2015, 275, 125–133. [Google Scholar] [CrossRef]
- Ronen, A.; Lerman, S.; Ramon, G.Z.; Dosoretz, C.G. Experimental characterization and numerical simulation of the anti-biofuling activity of nanosilver-modified feed spacers in membrane filtration. J. Membr. Sci. 2015, 475, 320–329. [Google Scholar] [CrossRef]
- Li, J.; Liu, X.; Lu, J.; Wang, Y.; Li, G.; Zhao, F. Anti-bacterial properties of ultrafiltration membrane modified by graphene oxide with nano-silver particles. J. Colloid Interface Sci. 2016, 484, 107–115. [Google Scholar] [CrossRef]
- Liu, L.-F.; Wu, H.; Li, R.-H.; Yu, C.-y.; Zhao, X.-T.; Gao, C.-J. Modification of poly (amide-urethane-imide)(PAUI) thin film composite reverse osmosis membrane with nano-silver particles. Rsc Adv. 2018, 8, 37817–37827. [Google Scholar] [CrossRef]
- Ben Hamouda, S.; Nguyen, Q.T.; Langevin, D.; Chappey, C.; Roudesli, S. Polyamide 12-polytetramethyleneoxide block copolymer membranes with silver nanoparticles – Synthesis and water permeation properties. React. Funct. Polym. 2007, 67, 893–904. [Google Scholar] [CrossRef]
- Mohd Dahan, R.; Arshad, A.N.; Mohamed, N.A.; Zawawi, E.; Zaharah, E.; Kamarun, D.; Wahid, M.H.; Sarip, M.N.; Rusop Mahmood, M. ATR-FTIR Analysis on Polymorphism of PVDF/MgO Nanocomposite Thin Films. Adv. Mater. Res. Trans. Tech. Pub. 2016, 1134, 39–43. [Google Scholar] [CrossRef]
- Stuart, B. Infrared spectroscopy: Fundamentals and applications; John Wiley & Sons: The Atrium, Southern Gate, Chichester, West Sussex, England, 2004; ISBN 9780470011140. [Google Scholar]
- Prince, J.A.; Bhuvana, S.; Boodhoo, K.V.K.; Anbharasi, V.; Singh, G. Synthesis and characterization of PEG-Ag immobilized PES hollow fiber ultrafiltration membranes with long lasting antifouling properties. J. Membr. Sci. 2014, 454, 538–548. [Google Scholar] [CrossRef] [Green Version]
- Amouamouha, M.; Badalians Gholikandi, G. Characterization and Antibiofouling Performance Investigation of Hydrophobic Silver Nanocomposite Membranes: A Comparative Study. Membranes 2017, 7, 64. [Google Scholar] [CrossRef] [PubMed]
- Zularisam, A.W.; Ismail, A.F.; Salim, R. Behaviours of natural organic matter in membrane filtration for surface water treatment —a review. Desalination 2006, 194, 211–231. [Google Scholar] [CrossRef]
- Crittenden, J.C.; Trussell, R.R.; Hand, D.W.; Howe, K.J.; Tchobanoglous, G. MWH’s Water Treatment: Principles and Design; John Wiley & Sons: Hoboken, NJ, USA, 2012. [Google Scholar]
- Rice, E.W.; Bridgewater, L.; American Public Health Association; American Water Works Association; Water Environment Federation. Standard Methods for the Examination of Water and Wastewate; American Public Health Association: Washington, DC, USA, 2012. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |
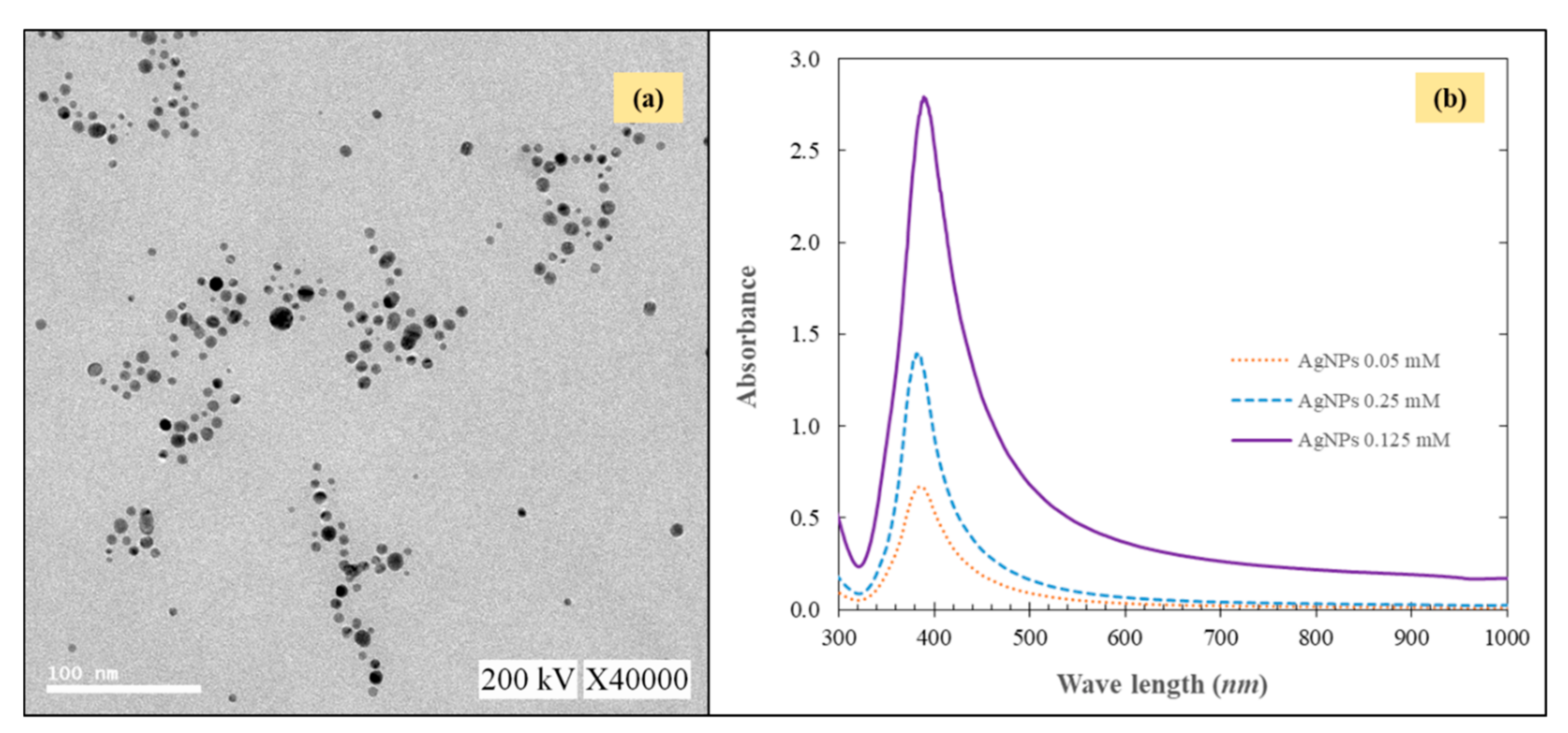
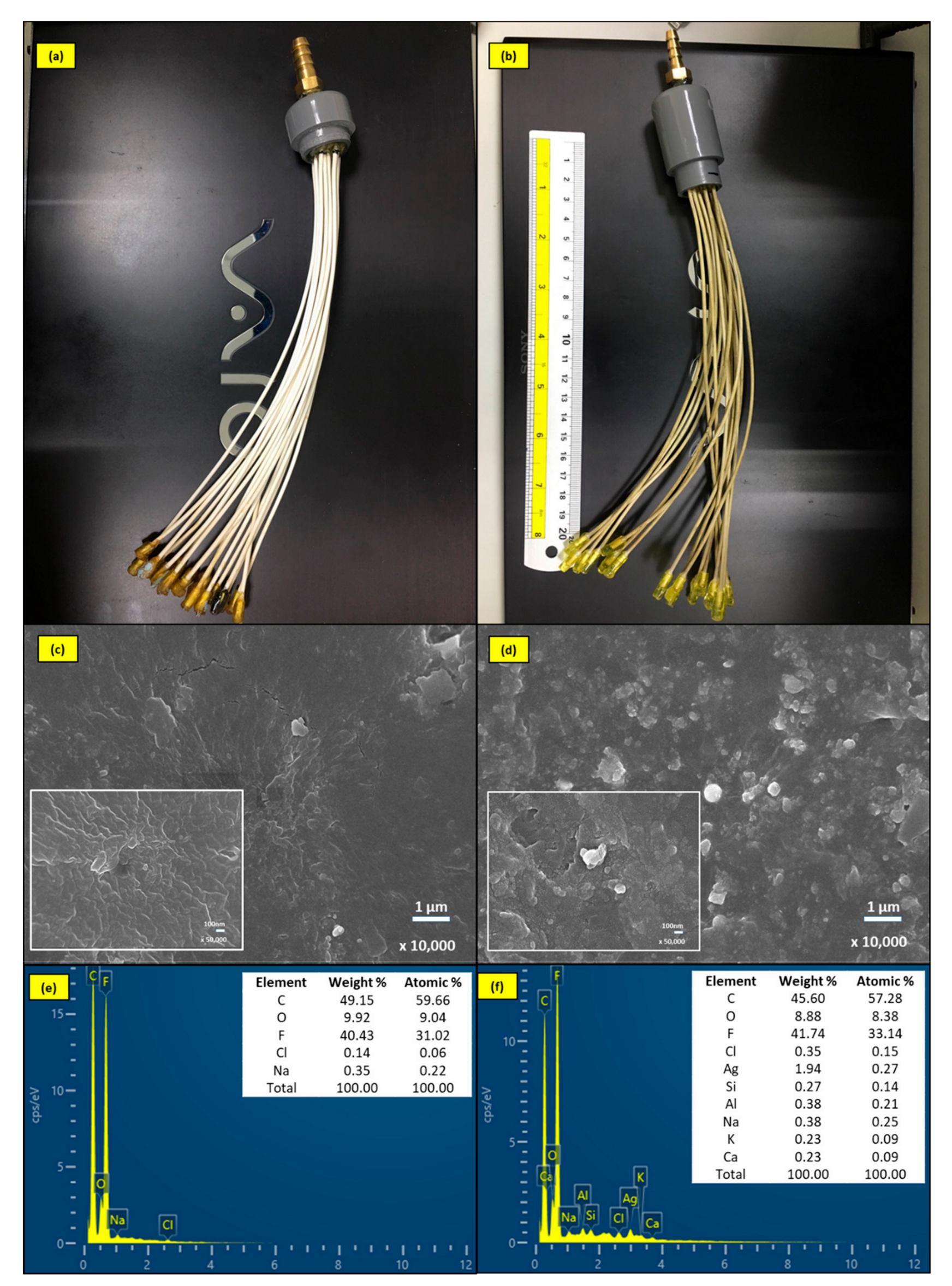



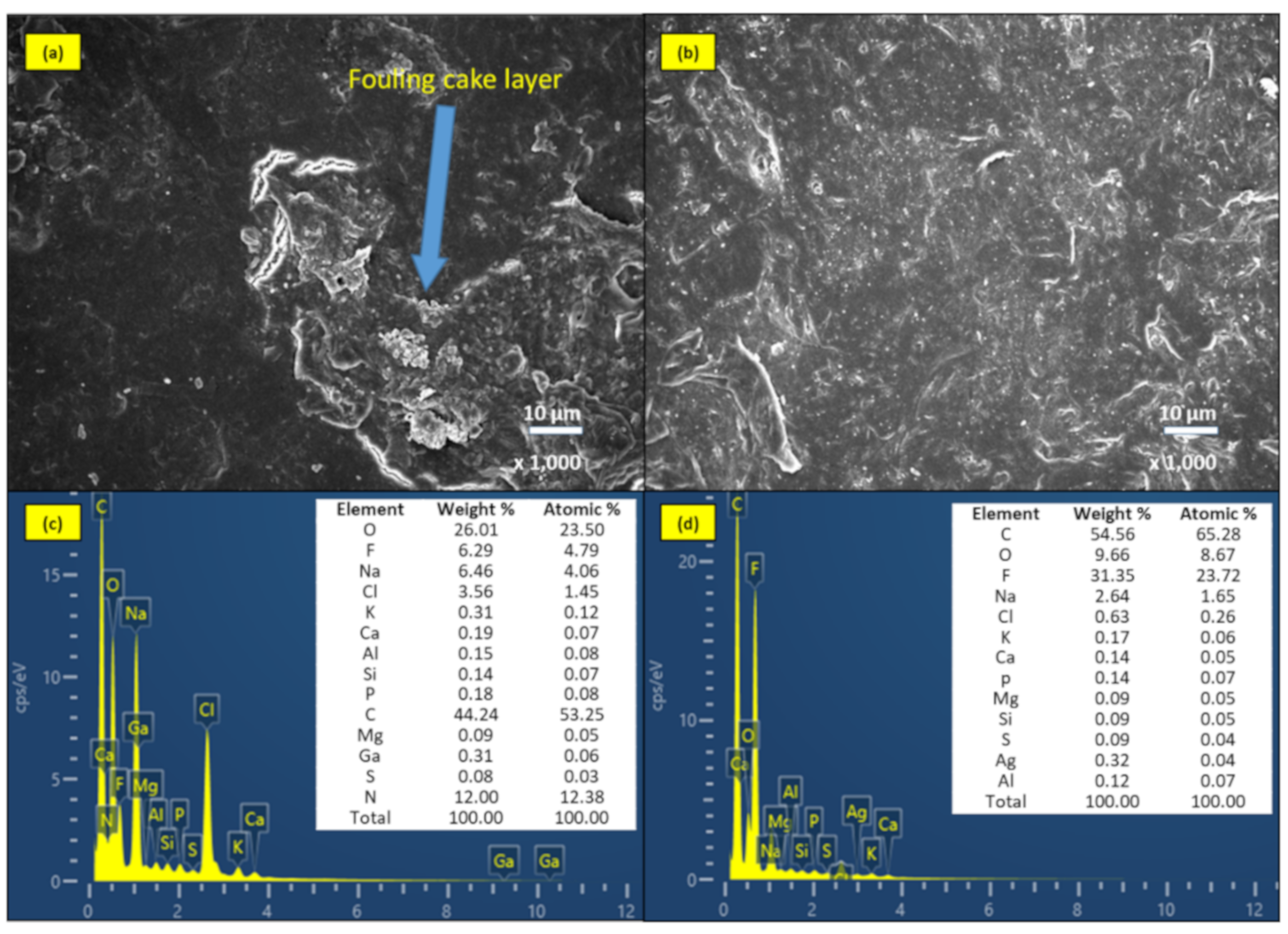



| Type of Membrane | Application | Scale | Membrane Fabrication/Modification Techniques | Quantity of AgNPs on Membrane | Duration | Mitigating Membrane Biofouling | References |
|---|---|---|---|---|---|---|---|
| RO | Seawater desalination | Flat-sheet membrane cell in lab scale | Membrane modification with chemical reduction method (silver nitrate and formaldehyde solutions). | - | 20 days | Yes | [9] |
| UF | Antibacterial and anti-biofouling using bacterial strain | Flat-sheet vacuum filtration cell in lab scale | Membrane fabrication with the wet phase-inversion process (polysulfone with commercial AgNPs) | 0.9% (by weight) | 24—72 h | Yes | [10] |
| UF | Antibacterial and anti-biofouling using bacterial strain | disc diffusion method | Membrane modification with biogenic AgNPs | 0.1—1.0% (by weight) | 12 h | Yes | [11] |
| NF | Antibacterial and anti-biofouling using bacterial strain | Flat-sheet membrane cell in lab scale | Membrane fabrication (polymeric composite + commercial AgNPs) | 0.05—10% (by weight) | 24 h | Yes | [12] |
| NF and FO | Antibacterial and anti-biofouling using bacterial strain | crossflow filtration model in lab scale | Membrane fabrication (fabricated by layer-by-layer (LbL) assembly + commercial AgNPs) | 0.22–1.19 (by weight) | 48 h | Yes | [13] |
| UF | Wastewater treatment | Aerobic MBR with submerged hollow fiber UF membrane module in lab scale | No (feeding solution: Synthesis wastewater + 0.10 mg/L AgNPs) | 0 | 65 days | No | [14] |
| MF | pH and ionic strength study | Dead-end microfiltration system in lab scale | Membrane modification (deposition of AgNPs synthesis by chemical reduction silver nitrate with ammonia solution) | - | - | Didn’t study | [15] |
| MF | Water disinfection potable water | Dead-end filtration cell in lab scale with raw river water and synthetic feed containing E. coli | Membrane modification (AgNPs synthesis by chemical reduction (Silver nitrate, sodium borohydride and ethanol)) | 0.0139 mg cm−2 | 5 days | Didn’t study | [16] |
| UF | Antibacterial and anti-biofouling using bacterial strain | Flat sheet bench-scale module | Graphene oxide -nanosilver UF membrane fabrication (Hummers’ method + Turkevich method) | - | 24 h | Yes | [17] |
| UF | Antibacterial and anti-biofouling using wastewater | Flat-sheet vacuum filtration cell in lab scale | Membrane modification with (i) diffusion of silver ions with subsequent reduction, (ii) addition of polyethyleneimine-capped AgNPs, and (iii) thermal-pressure fixation of silver-modified nanofibres | < 0.07–0.92% (by weight) | 0.5–8.0 h | Yes | [18] |
| RO | Water filtration | Flat-sheet membrane cell in lab scale | modification of the spacer y sonochemical deposition (deposition of AgNPs synthesis by chemical reduction silver nitrate with ammonia solution) | 9.0% (by weight) on spacer | 10 days | Yes | [19] |
| UF | Antibacterial and anti-biofouling using bacterial strain | Dead-end filtration cell in lab scale | Graphene oxide-nanosilver/PVDF UF membrane fabrication (Hummers’ method + AgNPs synthesis by chemical reduction silver nitrate with ammonia solution) | - | 24 h | Yes | [20] |
| RO | Lake water purification | Flat-sheet membrane cell in lab scale | Membrane fabrication (polyamide-urethane-imide + commercial AgNPs) | - | 7 days | Yes | [21] |
| Time (Day) | AgNPs-MF Membrane Module km = 8.46 × 1012 (m−1) | Unmodified MF Membrane Module km = 7.47 × 1012 (m−1) | ||||
|---|---|---|---|---|---|---|
| kh (m−1) | kc (m−1) | Total Resistance (m−1) | kh (m−1) | kc (m−1) | Total Resistance (m−1) | |
| 0 | 0 | 0 | 8.46 × 1012 | 0 | 0 | 7.47 × 1012 |
| 12 | 2.15 × 1013 | 6.35 × 1011 | 3.06 × 1013 | 1.08 × 1013 | 1.81 × 1012 | 2.01 × 1013 |
| 22 | 2.19 × 1013 | 3.76 × 1012 | 3.41 × 1013 | 1.52 × 1013 | 3.87 × 1012 | 2.66 × 1013 |
| 36 | 2.22 × 1013 | 2.73 × 1012 | 3.34 × 1013 | 2.09 × 1013 | 5.72 × 1012 | 3.41 × 1013 |
| 49 | 2.57 × 1013 | 7.30 × 1012 | 4.15 × 1013 | 5.13 × 1013 | 2.32 × 1013 | 8.20 × 1013 |
| 60 | 1.80 × 1013 | 1.24 × 1013 | 3.89 × 1013 | 4.43 × 1013 | 3.34 × 1013 | 8.51 × 1013 |
| Membrane Module | Material/AgNPs Coating | Operation Mode | Shape/Pore Size/Surface Area |
|---|---|---|---|
| 1 | Polyethylene/unmodified membrane | In operation mode | Hollow fiber/0.1 μm/19.1 cm2 |
| 2 | Polyethylene/AgNPs coated membrane | In operation mode | Hollow fiber/0.1 μm/19.1 cm2 |
| 3 | Polyethylene/unmodified membrane | In submerged mode—dipping in bioreactor | Hollow fiber/0.1 μm/19.1 cm2 |
| 4 | Polyethylene/AgNPs coated membrane | in submerged mode—dipping in bioreactor | Hollow fiber/0.1 μm/19.1 cm2 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Le, H.Q.; Sowe, A.; Chen, S.-S.; Duong, C.C.; Ray, S.S.; Cao, T.N.-D.; Nguyen, N.C. Exploring Nanosilver-Coated Hollow Fiber Microfiltration to Mitigate Biofouling for High Loading Membrane Bioreactor. Molecules 2019, 24, 2345. https://doi.org/10.3390/molecules24122345
Le HQ, Sowe A, Chen S-S, Duong CC, Ray SS, Cao TN-D, Nguyen NC. Exploring Nanosilver-Coated Hollow Fiber Microfiltration to Mitigate Biofouling for High Loading Membrane Bioreactor. Molecules. 2019; 24(12):2345. https://doi.org/10.3390/molecules24122345
Chicago/Turabian StyleLe, Huy Quang, Alieu Sowe, Shiao-Shing Chen, Chinh Cong Duong, Saikat Sinha Ray, Thanh Ngoc-Dan Cao, and Nguyen Cong Nguyen. 2019. "Exploring Nanosilver-Coated Hollow Fiber Microfiltration to Mitigate Biofouling for High Loading Membrane Bioreactor" Molecules 24, no. 12: 2345. https://doi.org/10.3390/molecules24122345






