Beneficial Effects of Tomato Juice Fermented by Lactobacillus Plantarum and Lactobacillus Casei: Antioxidation, Antimicrobial Effect, and Volatile Profiles
Abstract
:1. Introduction
2. Results and Discussion
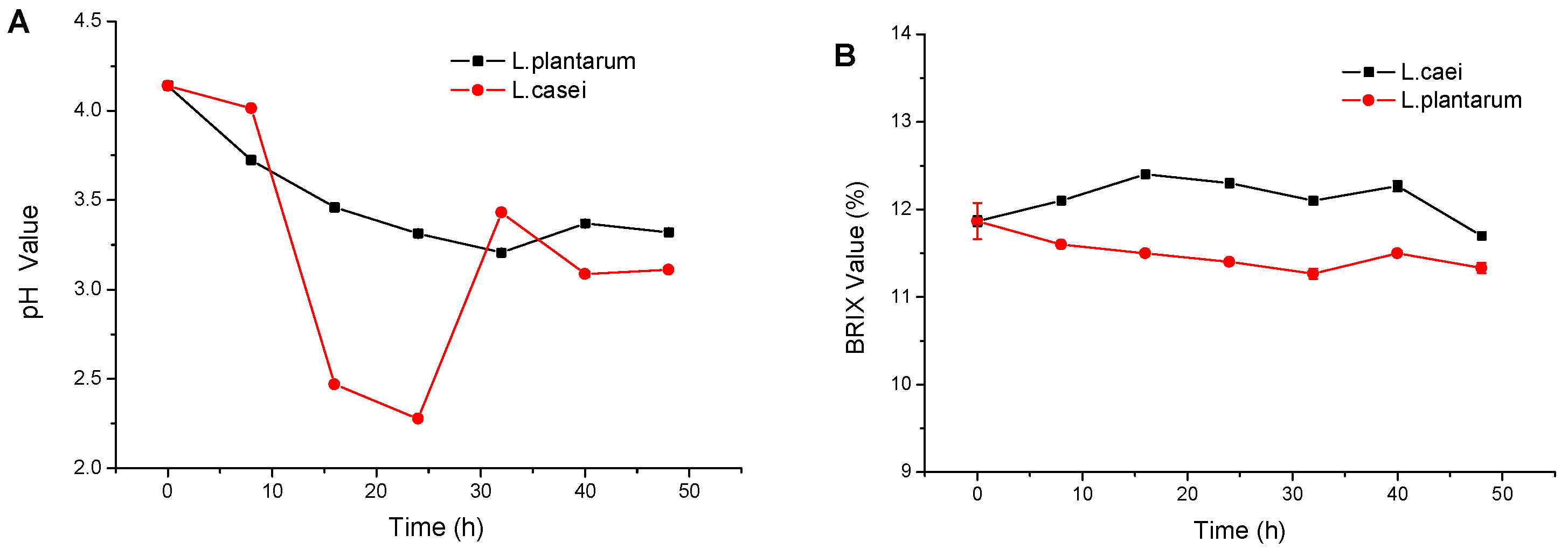
2.1. Juice Yield, Solid Content and pH Value
2.2. Chemical Characteristics of the Tomato Juice
2.3. Copper-Induced Human LDL-Cholesterol Oxidation
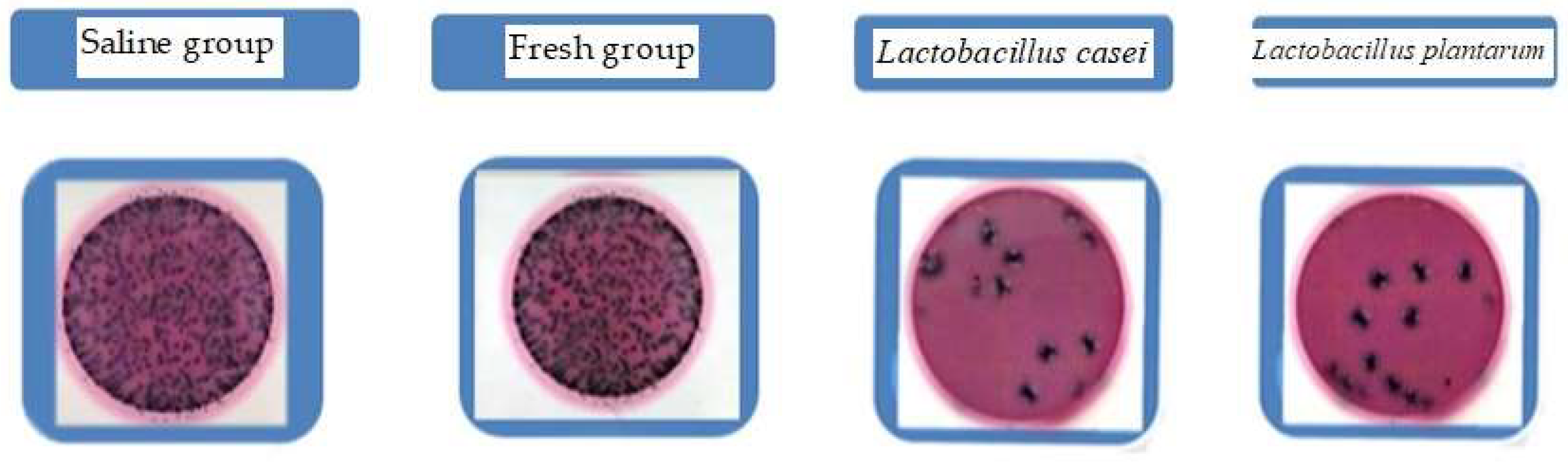
2.4. Gut Bacteria Inhibition
2.5. Changes of Total Phenolics during Fermentation
2.6. Changes in Lycopene, Total Carotenoids, and Ascorbic Acid
2.7. Volatile Compositions of Tomato Juice before and after Fermentation
3. Materials and Methods
3.1. Materials
3.2. Juice Preparation
3.3. Microbial Cultivation
3.3.1. Open Ampoule
3.3.2. Strain Recovery Training
3.3.3. Strain-Activated Cultivation
3.4. Pretreatment Method
| Use a pipette to suck a 5 mL sample into the centrifugal tube |
| ↓ |
| 20 mL 80% ethyl alcohol |
| ↓ |
| Ultrasound bath at 40 °C for 30 min |
| ↓ |
| Pour 25 mL of supernatant liquor into the volumetric flask until constant volume |
| ↓ |
| Seal the tube to avoid light preservation (4 °C) |
3.5. Chemical Characteristics of the Fruit Juice Blends
3.5.1. Determination of pH and Total Soluble Solids (°Brix)
3.5.2. Determination of DPPH Radical Scavenging Activity
3.5.3. Determination of ABTS· Radical Scavenging Activity
3.5.4. FRAP Assay
3.5.5. Determination of Total Phenolic Compounds (TPC)
3.5.6. Determination of Lycopene and Total Carotenoids
3.5.7. Ascorbic Acid Content (AA)
3.5.8. Copper-Induced Human LDL-Cholesterol Oxidation
3.5.9. Effects of Fermented Fruit and Vegetable on the Intestinal Flora
3.5.10. Determination of Volatile Compounds
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nazir, N.; Adrian, M.R. The improvement lycopene availability and antioxidant activities of tomato (Lycopersicum Esculentum, Mill) Jelly Drink. Agric. Agric. Sci. Proc. 2016, 9, 328–334. [Google Scholar] [CrossRef]
- Durante, M.; Montefusco, A.; Marrese, P.P.; Soccio, M.; Pastore, D.; Piro, G.; Lenucci, M.S. Seeds of pomegranate, tomato and grapes: An underestimated source of natural bioactive molecules and antioxidants from agri-food by-products. J. Food Compos. Anal. 2017, 63, 65–72. [Google Scholar] [CrossRef]
- Bergougnoux, V. The history of tomato: From domestication to biopharming. Biotechnol. Adv. 2014, 32, 170–189. [Google Scholar] [CrossRef] [PubMed]
- Kips, L.; De Paepe, D.; Van Meulebroek, L.; Van Poucke, C.; Larbat, R.; Bernaert, N.; Van Pamel, E.; De Loose, M.; Raes, K.; Van Droogenbroeck, B. A novel spiral-filter press for tomato processing: Process impact on phenolic compounds, carotenoids and ascorbic acid content. J. Food Eng. 2017, 213, 27–37. [Google Scholar] [CrossRef]
- Montesano, D.; Fallarino, F.; Cossignani, L.; Bosi, A.; Simonetti, M.S.; Puccetti, P.; Damiani, P. Innovative extraction procedure for obtaining high pure lycopene from tomato. Eur. Food Res. Technol. 2008, 226, 327–335. [Google Scholar] [CrossRef]
- Fattore, M.; Montesano, D.; Pagano, E.; Teta, R.; Borrelli, F.; Mangoni, A.; Albrizio, S. Carotenoid and flavonoid profile and antioxidant activity in “Pomodorino Vesuviano” tomatoes. J. Food Compos. Anal. 2016, 53, 61–68. [Google Scholar] [CrossRef]
- Montesano, D.; Gennari, O.; Seccia, S.; Albrizio, S. A Simple and Selective Analytical Procedure for the Extraction and Quantification of Lutein from Tomato By-Products by HPLC-DAD. Food Anal. Method. 2012, 5, 710–715. [Google Scholar] [CrossRef]
- Chua, K.J.; Kwok, W.C.; Aggarwal, N.; Sun, T.; Chang, M.W. Designer probiotics for the prevention and treatment of human diseases. Curr. Opin. Chem. Biol. 2017, 40, 8–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Luo, M.; Wang, X.; Xiaohui, H.U.; Zhao, J.; Cui, L.; Amp, N.A.; University, F. Optimization of SPME-GC/MS to Determine Tomato Flavoring Components. North. Hortic. Chn. 2017, 13, 7–13. [Google Scholar] [CrossRef]
- Li, S.; Huang, R.; Shah, N.P.; Tao, X.; Xiong, Y.; Wei, H. Antioxidant and antibacterial activities of exopolysaccharides from Bifidobacterium bifidum WBIN03 and Lactobacillus plantarum R315. J. Dairy Sci. 2014, 97, 7334–7343. [Google Scholar] [CrossRef] [PubMed]
- Nishino, N.; Hattori, H.; Wada, H.; Touno, E. Biogenic amine production in grass, maize and total mixed ration silages inoculated with Lactobacillus casei or Lactobacillus buchneri. J. Appl. Microbio. 2007, 103, 325–332. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kleinveld, H.A.; Hak-Lemmers, H.L.; Stalenhoef, A.F.; Demacker, P.N. Improved measurement of low-density-lipoprotein susceptibility to copper-induced oxidation: application of a short procedure for isolating low-density lipoprotein. Clin. Chem. 1992, 38, 2066–2072. [Google Scholar] [PubMed]
- de Camargo, A.C.; Regitanod’Arce, M.A.; Biasoto, A.C.; Shahidi, F. Low molecular weight phenolics of grape juice and winemaking byproducts: antioxidant activities and inhibition of oxidation of human low-density lipoprotein cholesterol and DNA strand breakage. J. Agr. Food Chem. 2014, 62, 12159–12171. [Google Scholar] [CrossRef] [PubMed]
- Khan, N.; Monagas, M.; Andres-Lacueva, C.; Casas, R.; Urpí-Sardà, M.; Lamuela-Raventós, R.M.; Estruch, R. Regular consumption of cocoa powder with milk increases HDL cholesterol and reduces oxidized LDL levels in subjects at high-risk of cardiovascular disease. Nutr. Metab. Cardiov. Dis. 2012, 22, 1046–1053. [Google Scholar] [CrossRef] [PubMed]
- Terahara, M.; Kurama, S.; Takemoto, N. Prevention by lactic acid bacteria of the oxidation of human LDL. Biosci. Biotech. Bioch. 2001, 65, 1864–1868. [Google Scholar] [CrossRef]
- Lee, D.K.; Jang, S.; Baek, E.H.; Mi, J.K.; Lee, K.S.; Shin, H.S.; Chung, M.J.; Jin, E.K.; Kang, O.L.; Ha, N.J. Lactic acid bacteria affect serum cholesterol levels, harmful fecal enzyme activity, and fecal water content. Lipids Health Dis. 2009, 8, 21. [Google Scholar] [CrossRef] [PubMed]
- Astuti, A.; Umniyati, S.; Rakhmawati, A.; Yulianti, E. Utilization probiotic lactic acid bacteria fish from waste faeces on LDL level blood broiler chickens strain lohmann. Jurnal Sains Dasar 2016, 5, 48–51. [Google Scholar] [CrossRef]
- Silaste, M.L.; Alfthan, G.; Aro, A.; Kesäniemi, Y.A.; Hörkkö, S. Tomato juice decreases LDL cholesterol levels and increases LDL resistance to oxidation. Brit. J. Nutr. 2007, 98, 1251–1258. [Google Scholar] [CrossRef] [PubMed]
- Ullah, N.; Wang, X.; Wu, J.; Guo, Y.; Ge, H.; Li, T.; Khan, S.; Li, Z.; Feng, X. Purification and primary characterization of a novel bacteriocin, LiN333, from Lactobacillus casei, an isolate from a Chinese fermented food. Food Sci. Technol. 2017, 84, 867–875. [Google Scholar] [CrossRef]
- Puertollano, E.; Puertollano, M.A.; Cruz-Chamorro, L.; De Cienfuegos, G.Á.; Ruiz-Bravo, A.; De Pablo, M.A. Effects of concentrated supernatants recovered from Lactobacillus plantarum on Escherichia coli growth and on the viability of a human promyelocytic cell line. J. Appl. Microbiol. 2010, 106, 1194–1203. [Google Scholar] [CrossRef] [PubMed]
- Chingwaru, W.; Vidmar, J. Potential of Zimbabwean commercial probiotic products and strains of Lactobacillus plantarum as prophylaxis and therapy against diarrhoea caused by Escherichia coli in children. Asian Pac. J. trop. Med. 2017, 10, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Mangell, P.; Nejdfors, P.; Wang, M.; Ahrné, S.; Weström, B.; Thorlacius, H.; Jeppsson, B. Lactobacillus plantarum 299v inhibits escherichia coli-induced intestinal permeability. Digest. Dis. Sci. 2002, 47, 511–516. [Google Scholar] [CrossRef] [PubMed]
- Bamidele, O.P.; Fasogbon, M.B. Chemical and antioxidant properties of snake tomato (Trichosanthes cucumerina) juice and Pineapple (Ananas comosus) juice blends and their changes during storage. Food Chem. 2017, 220, 184–189. [Google Scholar] [CrossRef] [PubMed]
- Gao, D.; Gao, Z.; Zhu, G. Antioxidant effects of lactobacillus plantarum via activation of transcription factor nrf2. Food Funct. 2013, 4, 982–989. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Li, W.; Rui, X.; Chen, X.; Jiang, M.; Dong, M. Structural characterization and bioactivity of released exopolysaccharides from Lactobacillus plantarum 70810. Int. J. Biol. Macromol. 2014, 67, 71–78. [Google Scholar] [CrossRef] [PubMed]
- Sumby, K.M.; Grbin, P.R.; Jiranek, V. Microbial modulation of aromatic esters in wine: Current knowledge and future prospects. Food Chem. 2010, 121, 1–16. [Google Scholar] [CrossRef]
- Cagno, R.D.; Surico, R.F.; Paradiso, A.; Angelis, M.D.; Salmon, J.C.; Buchin, S.; Gara, L.D.; Gobbetti, M. Effect of autochthonous lactic acid bacteria starters on health-promoting and sensory properties of tomato juices. Int. J. Food Microbiol. 2009, 128, 473–483. [Google Scholar] [CrossRef] [PubMed]
- Brandwilliams, W.; Cuvelier, M.E.; Berset, C. Use of a free radical method to evaluate antioxidant activity. LWT - Food Sci. Technol. 1995, 28, 25–30. [Google Scholar] [CrossRef]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Riceevans, C. Antioxidant activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Benzie, I.F.; Strain, J.J. The ferric reducing ability of plasma (FRAP) as a measure of "antioxidant power": the frap assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Wang, S.Y. Antioxidant activity and phenolic compounds in selected herbs. J. Agr. Food Chem. 2001, 49, 5165–5170. [Google Scholar] [CrossRef]
- AOAC (2005): Official Methods of Analysis. 18th ed. Association of Official Analytical Chemists, Gaithersburg, USA.
- Abuamsha, R.; Croft, K.D.; Puddey, I.B.; Proudfoot, J.M.; Beilin, L.J. Phenolic content of various beverages determines the extent of inhibition of human serum and low-density lipoprotein oxidation in vitro: Identification and mechanism of action of some cinnamic acid derivatives from red wine. Clin. Sci. 1996, 91, 449–458. [Google Scholar] [CrossRef]
- Manohar, G.K.; Khan, S.; Jashbhai, P.B. Comparison of 3MTM PetrifilmTM E. coli/Coliform Count (EC) Plates vs. Is methods for enumeration of Coliforms (IS-5401 Part-1) and E. coli (IS 5887: Part-1) to evaluate quality of Indian milk and milk products. Indian J. Dairy Sci. 2017, 70, 193–199. [Google Scholar]
Sample Availability: Samples of the compounds are not available from the authors. |


| Hours of Fermentation | TPC 2,c | Lycopene c | β-Carotinoid c | Ascorbic Acid c | TPC 2,d | Lycopened | Total Carotenoids d | Ascorbic Acid d |
|---|---|---|---|---|---|---|---|---|
| (ug GAE/mL) | (ug/mL) | (ug/mL) | (ug/mL) | (ug GAE/mL) | (ug/mL) | (ug/mL) | (ug/mL) | |
| 0 | 166.05 ± 2.12 d | 191.51 ± 2.14 c | 153.55 ± 0.81 d | 9.77 ± 2.97 a,b | 166.05 ± 9.25 a | 191.51 ± 8.75 a | 153.55 ± 2.62 a | 11.65 ± 1.82 a |
| 8 | 204.94 ± 4.75 a | 210.34 ± 3.96 a | 161.32 ± 1.42 a | 7.94 ± 0.22 b | 167.42 ± 10.97 a,b | 182.59 ± 1.91 a | 149.67 ± 0.71 a,b | 12.94 ± 4.98 a |
| 16 | 184.16 ± 6.47 b,c | 193.69 ± 3.96 c | 154.47 ± 1.59 c,d | 7.75 ± 0.77 b | 165.41 ± 5.81 a,b | 179.23 ± 3.57 a,b | 148.24 ± 1.23 a,b | 11.38 ± 1.42 a |
| 24 | 153.65 ± 8.19 d | 200.63 ± 2.48 b | 157.03 ± 0.99 b,c | 8.56 ± 0.93 b | 161.69 ± 4.61 b | 188.14 ± 4.49 a,b | 152.12 ± 2.18 a | 12.06 ± 1.23 a |
| 32 | 168.07 ± 7.27 c,d | 202.21 ± 5.45 b | 158.46 ± 2.73 a,b | 7.86 ± 0.12 b | 152.34 ± 8.37 b | 192.7 ± 8.92 a,b | 154.17 ± 2.73 a | 14.09 ± 1.38 a |
| 40 | 182.82 ± 10.07 b,c | 197.46 ± 2.06 b,c | 156.41 ± 0.99b c,d | 7.79 ± 0.12 b | 169.03 ± 2.09 a,b | 179.62 ± 4.22 a,b | 148.64 ± 1.85 a,b | 13.08 ± 1.66 a |
| 48 | 195.55 ± 1.59 a,b | 202.41 ± 3.96 b | 158.56 ± 1.87 a,b | 11.11 ± 1.02 a | 170.04 ± 6.06 a,b | 161.39 ± 5.95 b | 142.27 ± 2.77 b | 12.13 ± 0.82 a |
| Category | Volatile Compounds | Tomato Juice | Fermented Tomato Juice (L. casei) | Fermented Tomato Juice (L. Plantarum) | |||
|---|---|---|---|---|---|---|---|
| Peak Area | RPA (%) | Peak Area | RPA (%) | Peak Area | RPA (%) | ||
| Alcohols | Ethanol | 45.04 ± 1 | 3.83 | 36.8 ± 0.16 | 4.33 | 16.95 ± 0.42 | 1.61 |
| 1-Butanol | 2.07 ± 0.03 | 0.18 | ND | 0 | ND | 0 | |
| 1-Butanol, 3-methyl- | 58.93 ± 0.35 | 5 | 43.8 ± 0.08 | 5.15 | 116.16 ± 0.73 | 11.04 | |
| 1-Pentanol | 68.14 ± 1.35 | 5.79 | 54.16 ± 0.9 | 6.37 | 27.58 ± 0.6 | 2.62 | |
| 2-Tridecen-1-ol, (E)- | 16.65 ± 0.75 | 1.41 | ND | 0 | ND* | 0 | |
| 1-Penten-3-ol | 15.95 ± 0.77 | 1.35 | 4.69 ± 0.21 | 0.55 | ND | 0 | |
| (S)-(+)-3-Methyl-1-pentanol | 4.79 ± 0.1 | 0.41 | ND | 0 | ND | 0 | |
| 1-Hexanol | 200.44 ± 0.4 | 17.02 | 157.42 ± 6.08 | 18.51 | 102.69 ± 0.83 | 9.76 | |
| 3-Hexen-1-ol, (Z)- | 43.93 ± 0.97 | 3.73 | 29.85 ± 0.28 | 3.51 | 16.14 ± 0.08 | 1.53 | |
| 2-Hepten-1-ol, (E)- | 7.05 ± 0.1 | 0.6 | 3.43 ± 0 | 0.4 | ND | 0 | |
| 1-Hexanol, 4-methyl-, (S)- | 1.1 ± 0.04 | 0.09 | 0.32 ± 0.01 | 0.04 | 2.17 ± 0 | 0.21 | |
| 1-Octen-3-ol | 9.39 ± 0.13 | 0.8 | 4.19 ± 0.16 | 0.49 | ND | 0 | |
| 1-Heptanol | 4.6 ± 0.06 | 0.39 | 4.44 ± 0.15 | 0.52 | 3.01 ± 0.07 | 0.29 | |
| 1-Pentanol, 2-methyl- | 0.44 ± 0.01 | 0.04 | ND | 0 | ND | 0 | |
| 6-Hepten-1-ol, 2-methyl- | 2.17 ± 0.1 | 0.18 | 3.26 ± 0.14 | 0.38 | ND | 0 | |
| 2-Propyl-1-pentanol | 1.67 ± 0.04 | 0.14 | ND | 0 | ND | 0 | |
| 1-Octanol | 6.82 ± 0.09 | 0.58 | 9.93 ± 0.22 | 1.17 | 5.4 ± 0.07 | 0.51 | |
| Bicyclo[2.2.1]heptan-2-ol, 1,3,3-trimethyl- | 2.35 ± 0.08 | 0.2 | ND | 0 | ND | 0 | |
| 5-Isopropenyl-2-methyl-7-oxabicyclo[4.1.0]heptan-2-ol | 0.48 ± 0.02 | 0.04 | ND | 0 | ND | 0 | |
| 1-Methylcycloheptanol | 0.65 ± 0 | 0.05 | 0.72 ± 0.04 | 0.08 | ND | 0 | |
| 2-Octen-1-ol, (E)- | 5.47 ± 0.11 | 0.46 | ND | 0 | ND | 0 | |
| Phenylethyl Alcohol | 0.94 ± 0.05 | 0.08 | 1.29 ± 0.02 | 0.15 | ND | 0 | |
| 2-Hexen-1-ol, (Z)- | 81.11 ± 2.17 | 6.89 | 56.7 ± 0.27 | 6.67 | 36.09 ± 1.12 | 3.43 | |
| .alpha.-Terpineol | ND | 0 | 6.16 ± 0.02 | 0.72 | ND | 0 | |
| 3-Nonen-1-ol, (Z)- | ND | 0 | 0.59 ± 0.01 | 0.07 | ND | 0 | |
| trans-2-Undecen-1-ol | ND | 0 | 6.92 ± 0.16 | 0.81 | ND | 0 | |
| 2-Propyl-1-pentanol | ND | 0 | 6.48 ± 0.28 | 0.76 | ND | 0 | |
| 1-Pentanol, 3-methyl- | ND | 0 | 10.19 ± 0.12 | 1.2 | 46.72 ± 1.3 | 4.44 | |
| 2-Penten-1-ol, (Z)- | ND | 0 | 3.27 ± 0.08 | 0.38 | 0.93 ± 0.05 | 0.09 | |
| 3-Hexen-1-ol, acetate, (Z)- | ND | 0 | 6.07 ± 0.13 | 0.71 | ND | 0 | |
| 2-Penten-1-ol, (Z)- | ND | 0 | 13.74 ± 0.16 | 1.62 | ND | 0 | |
| 3-Buten-1-ol, 3-methyl- | ND | 0 | 7.24 ± 0.31 | 0.85 | 7.49 ± 0.36 | 0.71 | |
| 4-Hexen-1-ol | ND | 0 | 7.09 ± 0.01 | 0.83 | ND | 0 | |
| 2-Nonanol | ND | 0 | 4.12 ± 0 | 0.49 | ND | 0 | |
| trans-(2-Ethylcyclopentyl)methanol | ND | 0 | 3.21 ± 0.06 | 0.38 | ND | 0 | |
| cis-9-Tetradecen-1-ol | ND | 0 | 0.59 ± 0.02 | 0.07 | ND | 0 | |
| 2-Furanmethanol | ND | 0 | 1 ± 0.05 | 0.12 | ND | 0 | |
| 2-Nonen-1-ol, (E)- | ND | 0 | 0.22 ± 0.01 | 0.03 | ND | 0 | |
| Geraniol | ND | 0 | 1.77 ± 0.06 | 0.21 | ND | 0 | |
| 5,9-Undecadien-2-ol, 6,10-dimethyl- | ND | 0 | 1.07 ± 0.03 | 0.13 | ND | 0 | |
| Bicyclo[3.1.1]hept-2-ene-2-methanol, 6,6-dimethyl- | ND | 0 | 0.42 ± 0.02 | 0.05 | ND | 0 | |
| 1,6-Octadien-3-ol, 3,7-dimethyl- | ND | 0 | 18.45 ± 0.25 | 2.17 | 14.23 ± 0.27 | 1.35 | |
| Acetoin | ND | 0 | ND | 0 | 24.71 ± 0.64 | 2.35 | |
| 3-Hexen-1-ol, (E)- | ND | 0 | ND | 0 | 4.15 ± 0.12 | 0.39 | |
| 1-Hexanol, 2-ethyl- | ND | 0 | ND | 0 | 41.16 ± 1.18 | 3.91 | |
| 3-Furanmethanol | ND | 0 | ND | 0 | 1.12 ± 0.05 | 0.11 | |
| Phenylethyl Alcohol | ND | 0 | ND | 0 | 4.51 ± 0.17 | 0.43 | |
| 2-Buten-1-ol, 3-methyl- | ND | 0 | ND | 0 | 12.62 ± 0.29 | 1.2 | |
| 1-Pentanol, 4-methyl- | ND | 0 | ND | 0 | 9.46 ± 0.37 | 0.9 | |
| 3-Hexen-1-ol, acetate, (E)- | ND | 0 | ND | 0 | 2.02 ± 0.05 | 0.19 | |
| 6-Hepten-1-ol, 2-methyl- | ND | 0 | ND | 0 | 16.29 ± 0.01 | 1.55 | |
| 1-Methylcycloheptanol | ND | 0 | ND | 0 | 1.09 ± 0.04 | 0.1 | |
| 2-Tridecen-1-ol, (E)- | ND | 0 | ND | 0 | 3.9 ± 0.04 | 0.37 | |
| Citronellol | ND | 0 | ND | 0 | 0.49 ± 0 | 0.05 | |
| 3-Nonen-1-ol, (Z)- | ND | 0 | ND | 0 | 0.64 ± 0.02 | 0.06 | |
| Geraniol | ND | 0 | ND | 0 | 2.56 ± 0.02 | 0.24 | |
| 5,9-Undecadien-2-ol, 6,10-dimethyl- | ND | 0 | ND | 0 | 2.74 ± 0.11 | 0.26 | |
| subtotal | 580.18 | 49.26 | 509.64 | 59.92 | 523.04 | 49.7 | |
| Hydrocarbons | Ethylbenzene | 2.2 ± 0.07 | 0.19 | ND | 0 | ND | 0 |
| Benzene, 1,3-dimethyl- | 0.53 ± 0 | 0.04 | 0.51 ± 0.01 | 0.06 | ND | 0 | |
| p-Xylene | 2.14 ± 0.05 | 0.18 | ND | 0 | ND | 0 | |
| 3-Carene | 7.02 ± 0.17 | 0.6 | ND | 0 | 5.27 ± 0.04 | 0.5 | |
| o-Xylene | 0.87 ± 0.02 | 0.07 | 3.4 ± 0.02 | 0.4 | ND | 0 | |
| Styrene | 3.88 ± 0.1 | 0.33 | 1.15 ± 0.01 | 0.13 | ND | 0 | |
| Cyclohexane, ethylidene- | 2.29 ± 0.02 | 0.19 | 1.32 ± 0.01 | 0.16 | ND | 0 | |
| Propylene Carbonate | 20.37 ± 0.03 | 1.73 | ND | 0 | ND | 0 | |
| Toluene | 2.43 ± 0.01 | 0.21 | ND | 0 | ND | 0 | |
| 2-Octene, 2-methyl-6-methylene- | 2.75 ± 0.02 | 0.23 | ND | 0 | ND | 0 | |
| 3-Hexen-1-ol, acetate, (Z)- | 1.5 ± 0.01 | 0.13 | ND | 0 | ND | 0 | |
| Pentane, 1-nitro- | 24.29 ± 0.9 | 2.06 | ND | 0 | ND | 0 | |
| Pentane, 2-nitro- | ND | 0 | 14.43 ± 0.21 | 1.7 | ND | 0 | |
| 5-Hepten-2-one, 6-methyl- | 90.79 ± 3.29 | 7.71 | 0 ± 0 | 0 | ND | 0 | |
| Undecyne | 0.35 ± 0.01 | 0.03 | ND | 0 | ND | 0 | |
| Bicyclo[4.1.0]heptane, 7-(1-methylethylidene)- | 1.21 ± 0.04 | 0.1 | ND | 0 | ND | 0 | |
| Bicyclo[2.2.1]hept-2-ene, 1,7,7-trimethyl- | ND | 0 | 0.84 ± 0.02 | 0.1 | ND | 0 | |
| 1,6-Octadien-3-ol, 3,7-dimethyl- | 5.17 ± 0.22 | 0.44 | ND | 0 | ND | 0 | |
| 2-Furanmethanol | 1.67 ± 0.08 | 0.14 | ND | 0 | ND | 0 | |
| alpha.-Terpineol | 6.34 ± 0.24 | 0.54 | ND | 0 | ND | 0 | |
| Hexadecane, 2,6,10,14-tetramethyl- | 2.39 ± 0.03 | 0.2 | 4.7 ± 0.23 | 0.55 | 7.08 ± 0.3 | 0.67 | |
| 1-Tetradecyne | ND | 0 | 4.95 ± 0.09 | 0.58 | ND | 0 | |
| 1-Hexene, 3,3,5-trimethyl- | ND | 0 | 2.52 ± 0.01 | 0.3 | ND | 0 | |
| Heneicosane | ND | 0 | 1.37 ± 0.07 | 0.16 | ND | 0 | |
| 4-Tridecene, (Z)- | ND | 0 | 0.84 ± 0.03 | 0.1 | ND | 0 | |
| d-Limonene | ND | 0 | ND | 0 | 1.87 ± 0.05 | 0.18 | |
| Butylated Hydroxytoluene | ND | 0 | ND | 0 | 1.85 ± 0.04 | 0.18 | |
| Benzene, 1-methoxy-4-methyl- | ND | 0 | ND | 0 | 0.26 ± 0.01 | 0.02 | |
| 1-Hexene, 3,3-dimethyl- | ND | 0 | ND | 0 | 1.99 ± 0.02 | 0.19 | |
| Heneicosane | ND | 0 | ND | 0 | 0.82 ± 0.01 | 0.08 | |
| 3-Nonen-5-yne, 4-ethyl- | ND | 0 | ND | 0 | 4.09 ± 0.06 | 0.39 | |
| 3-Buten-2-one, 4-(2,6,6-trimethyl-1-cyclohexen-1-yl)- | ND | 0 | ND | 0 | 2.4 ± 0.03 | 0.23 | |
| subtotal | 178.2 | 15.12 | 36.03 | 4.24 | 25.61 | 2.26 | |
| Aldehydes | Hexanal | 175 ± 0.4 | 14.86 | ND | 0 | 7.7 ± 0.03 | 0.73 |
| 2-Hexenal, (E)- | 93.72 ± 3.31 | 7.96 | ND | 0 | 0.85 ± 0.02 | 0.08 | |
| Octanal | 1.4 ± 0.07 | 0.12 | ND | 0 | ND | 0 | |
| Nonanal | 8.5 ± 0.34 | 0.72 | ND | 0 | 3.55 ± 0.18 | 0.34 | |
| Butanal, 3-methyl- | 8.41 ± 0.12 | 0.71 | ND | 0 | ND | 0 | |
| 2,4-Hexadienal, (E,E)- | 3.66 ± 0.06 | 0.31 | ND | 0 | ND | 0 | |
| Decanal | 0.87 ± 0.01 | 0.07 | ND | 0 | 0.28 ± 0.01 | 0.03 | |
| 2-Nonenal, (E)- | 1.7 ± 0.08 | 0.14 | 0.8 ± 0.02 | 0.09 | 0.17 ± 0 | 0.02 | |
| Benzene acetaldehyde | 1.05 ± 0.02 | 0.09 | ND | 0 | ND | 0 | |
| 2,6-Octadienal, 3,7-dimethyl-, (Z)- | 0.84 ± 0.01 | 0.07 | ND | 0 | ND | 0 | |
| 1-Cyclohexene-1-carboxaldehyde, 2,6,6-trimethyl- | 2.45 ± 0.1 | 0.21 | ND | 0 | ND | 0 | |
| 2,6-Octadienal, 3,7-dimethyl-, (E)- | 2.78 ± 0.09 | 0.24 | ND | 0 | 4.85 ± 0.09 | 0.46 | |
| 1H-Pyrrole-2-carboxaldehyde | 0.57 ± 0.02 | 0.05 | 0.45 ± 0.02 | 0.05 | 0.78 ± 0.03 | 0.07 | |
| 2-Undecenal, E- | ND | 0 | 0.58 ± 0.02 | 0.07 | ND | 0 | |
| 2-Octenal, (E)- | ND | 0 | ND | 0 | 1.08 ± 0.05 | 0.1 | |
| 2,6-Octadienal, 3,7-dimethyl-, (Z)- | ND | 0 | ND | 0 | 0.65 ± 0.01 | 0.06 | |
| subtotal· | 300.97 | 25.55 | 1.84 | 0.21 | 19.91 | 1.89 | |
| Ketones | Cyclohexanone, 2,2,6-trimethyl- | 0.56 ± 0.02 | 0.05 | ND | 0 | ND | 0 |
| Acetone | 3.67 ± 0.07 | 0.31 | 1.79 ± 0.05 | 0.21 | ND | 0 | |
| 4’-(Trifluoromethyl)acetophenone | 0.51 ± 0.02 | 0.04 | 3.67 ± 0.02 | 0.43 | ND | 0 | |
| trans-beta.-Ionone | 1 ± 0.02 | 0.08 | ND | 0 | ND | 0 | |
| 3-Buten-2-one, 4-(2,2,6-trimethyl-7-oxabicyclo[4.1.0]hept-1-yl)- | 0.38 ± 0.01 | 0.03 | 1.46 ± 0.03 | 0.17 | 0.47 ± 0.02 | 0.04 | |
| 4-Hydroxy-2-methylacetophenone | ND | 0 | 0.25 ± 0.01 | 0.03 | 0.61 ± 0.03 | 0.06 | |
| 2-Undecanone | ND | 0 | 0.25 ± 0 | 0.03 | ND | 0 | |
| 5-Hepten-2-one, 6-methyl- | ND | 0 | 85.9 ± 3.47 | 10.1 | 91.68 ± 3.49 | 8.71 | |
| 2-Nonanone | ND | 0 | 8.29 ± 0.4 | 0.98 | 1.47 ± 0.06 | 0.14 | |
| 2-Pentanone | ND | 0 | 23.03 ± 0.25 | 2.71 | ND | 0 | |
| 2-Propanone, 1-hydroxy- | ND | 0 | 0.47 ± 0.02 | 0.06 | 1.65 ± 0.08 | 0.16 | |
| Acetoin | ND | 0 | 0.99 ± 0.01 | 0.12 | ND | 0 | |
| 2-Cyclopenten-1-one, 3,4,4-trimethyl- | ND | 0 | ND | 0 | 3.05 ± 0.06 | 0.29 | |
| 2-Heptanone | ND | 0 | ND | 0 | 7.75 ± 0.2 | 0.91 | |
| 2,3-Butanedione | ND | 0 | ND | 0 | 14.73 ± 0.24 | 1.4 | |
| 2-Heptanone | ND | 0 | ND | 0 | 2.62 ± 0.04 | 0.25 | |
| 2H-Pyran-2-one, tetrahydro- | ND | 0 | ND | 0 | 0.51 ± 0 | 0.05 | |
| 3,5,9-Undecatrien-2-one, 6,10-dimethyl- | ND | 0 | ND | 0 | 0.2 ± 0 | 0.02 | |
| 2(4H)-Benzofuranone, 5,6,7,7a-tetrahydro-4,4,7a-trimethyl-, (R)- | ND | 0 | ND | 0 | 0.37 ± 0 | 0.04 | |
| subtotal | 6.12 | 0.51 | 126.1 | 14.84 | 125.09 | 12.07 | |
| Acids | Acetic acid | 32.74 ± 1.15 | 2.78 | ND | 0 | 281.29 ± 3.92 | 26.72 |
| Butanoic acid, 3-methyl- | 7.41 ± 0.31 | 0.63 | ND | 0 | 22.28 ± 0.06 | 2.12 | |
| Butanoic acid, anhydride | ND | 0 | 0.3 ± 0 | 0.04 | ND | 0 | |
| Hexanoic acid | 3.84 ± 0.17 | 0.33 | 9.17 ± 0.44 | 1.08 | 12.85 ± 0.63 | 1.22 | |
| Octanoic acid | ND | 0 | 10.6 ± 0.46 | 1.25 | 0.36 ± 0 | 0.03 | |
| Cyclohexylmethyl formate | ND | 0 | 0.36 ± 0 | 0.04 | ND | 0 | |
| Ammonium acetate | ND | 0 | 87.11 ± 0.25 | 10.25 | ND | 0 | |
| subtotal· | 43.99 | 3.74 | 107.54 | 12.66 | 316.78 | 30.09 | |
| Esters | Ethyl Acetate | 22 ± 0.36 | 1.87 | ND | 0 | ND | 0 |
| 3-Nonen-5-yne, 4-ethyl- | 4.86 ± 0.23 | 0.41 | 5.33 ± 0.2 | 0.63 | ND | 0 | |
| Methyl undecyl ether | ND | 0 | 2.86 ± 0.04 | 0.34 | ND | 0 | |
| Ethyl tridecanoate | ND | 0 | 3.93 ± 0.01 | 0.46 | ND | 0 | |
| Acetic acid, butyl ester | ND | 0 | 4.76 ± 0.13 | 0.56 | 3.05 ± 0.15 | 0.29 | |
| Dodecanoic acid, methyl ester | ND | 0 | ND | 0 | 0.49 ± 0 | 0.05 | |
| Tetradecanoic acid, ethyl ester | ND | 0 | ND | 0 | 0.32 ± 0.01 | 0.03 | |
| subtotal | 26.85 | 2.28 | 16.88 | 1.99 | 3.87 | 0.37 | |
| Others | Pyridine | 1.01 ± 0.05 | 0.09 | ND | 0 | ND | 0 |
| 2-Isobutylthiazole | 7.72 ± 0.35 | 0.66 | 6.95 ± 0.13 | 0.82 | ND | 0 | |
| Oxime-, methoxy-phenyl-_ | 24.13 ± 1.04 | 2.05 | 11.44 ± 0.3 | 1.35 | ND | 0 | |
| 1H-Isoindole, 3-methoxy-4,7-dimethyl- | 0.63 ± 0.01 | 0.05 | ND | 0 | ND | 0 | |
| Benzofuran, 2,3-dihydro- | 1.11 ± 0.04 | 0.09 | 1.13 ± 0.01 | 0.13 | 17.49 ± 0.8 | 1.66 | |
| Phenol, 2,4-bis(1,1-dimethylethyl)- | 2.23 ± 0.11 | 0.19 | 1.75 ± 0.06 | 0.21 | 2.44 ± 0.07 | 0.23 | |
| D-Limonene | ND | 0 | 20.34 ± 0.69 | 2.39 | ND | 0 | |
| Butanenitrile, 3-methyl- | ND | 0 | 1.95 ± 0.01 | 0.23 | ND | 0 | |
| Oxepine, 2,7-dimethyl- | ND | 0 | 0.87 ± 0.04 | 0.1 | ND | 0 | |
| Furfural | ND | 0 | ND | 0 | 6.92 ± 0.13 | 0.66 | |
| Ethyl tridecanoate | ND | 0 | ND | 0 | 8.17 ± 0.02 | 0.78 | |
| Phenol | ND | 0 | ND | 0 | 0.49 ± 0.02 | 0.05 | |
| Methyl tetradecanoate | ND | 0 | ND | 0 | 0.54 ± 0.02 | 0.05 | |
| Bicyclo[3.1.1]hept-2-ene-2-methanol, 6,6-dimethyl- | ND | 0 | ND | 0 | 1.26 ± 0.05 | 0.12 | |
| 2-Ethylhexyl salicylate | ND | 0 | ND | 0 | 1.31 ± 0.03 | 0.12 | |
| subtotal | 36.820377 | 3.13 | 44.435835 | 5.23 | 38.626639 | 3.67 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Y.; Chen, H.; Chen, W.; Zhong, Q.; Zhang, G.; Chen, W. Beneficial Effects of Tomato Juice Fermented by Lactobacillus Plantarum and Lactobacillus Casei: Antioxidation, Antimicrobial Effect, and Volatile Profiles. Molecules 2018, 23, 2366. https://doi.org/10.3390/molecules23092366
Liu Y, Chen H, Chen W, Zhong Q, Zhang G, Chen W. Beneficial Effects of Tomato Juice Fermented by Lactobacillus Plantarum and Lactobacillus Casei: Antioxidation, Antimicrobial Effect, and Volatile Profiles. Molecules. 2018; 23(9):2366. https://doi.org/10.3390/molecules23092366
Chicago/Turabian StyleLiu, Yiyun, Haiming Chen, Wenxue Chen, Quipping Zhong, Guanfei Zhang, and Weijun Chen. 2018. "Beneficial Effects of Tomato Juice Fermented by Lactobacillus Plantarum and Lactobacillus Casei: Antioxidation, Antimicrobial Effect, and Volatile Profiles" Molecules 23, no. 9: 2366. https://doi.org/10.3390/molecules23092366





