“On-Water” Synthesis of Quinazolinones and Dihydroquinazolinones Starting from o-Bromobenzonitrile
Abstract
:1. Introduction
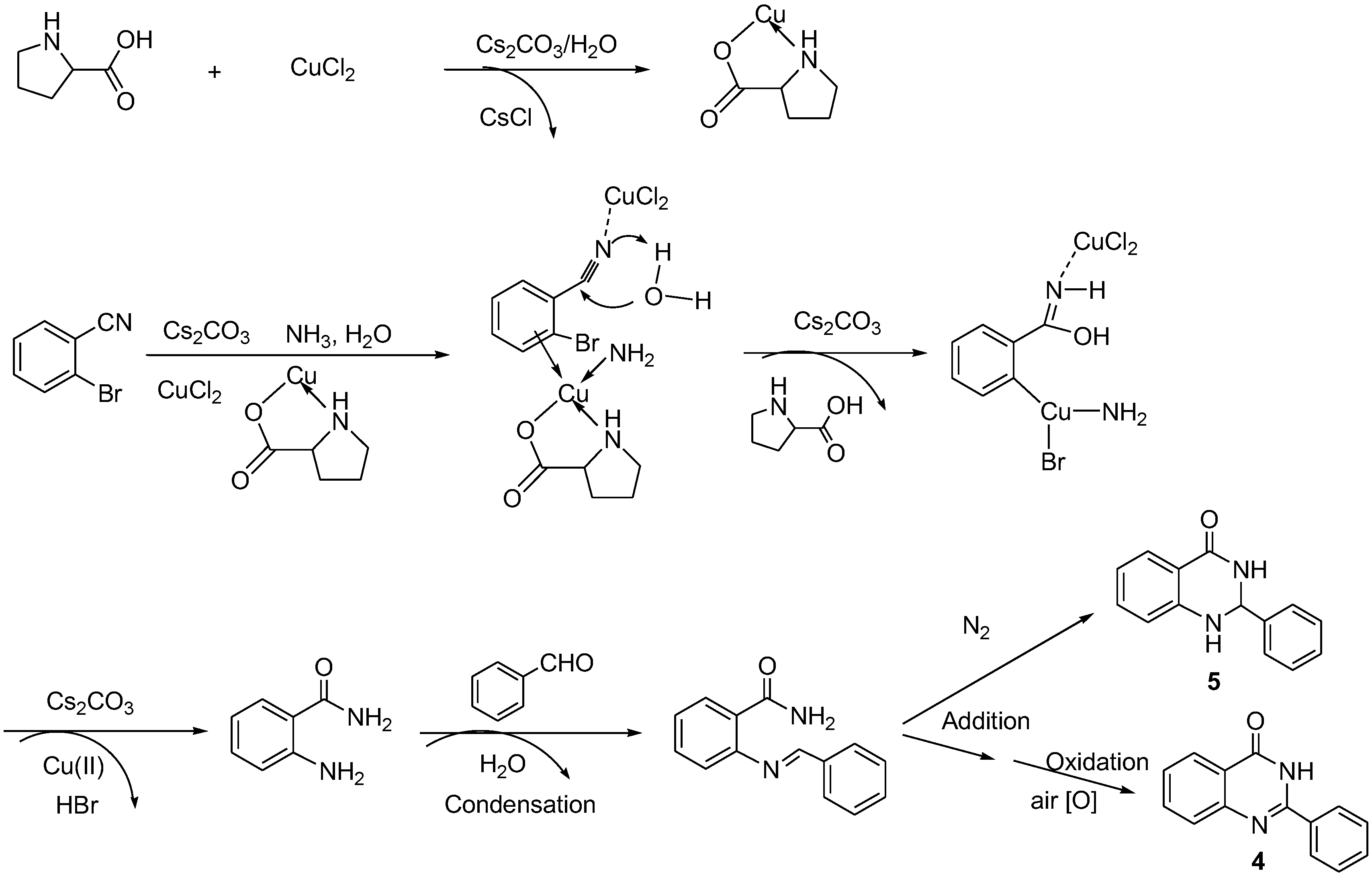
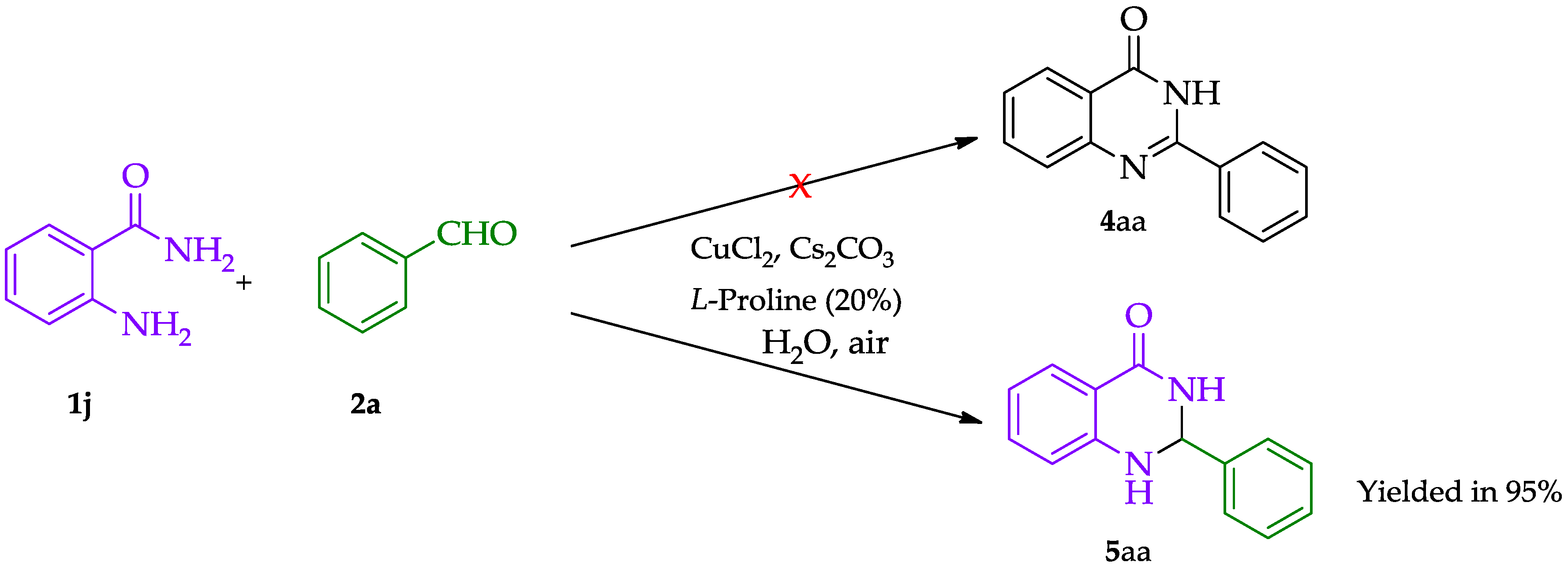
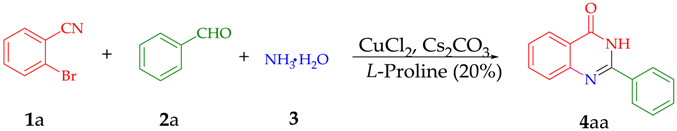
2. Discussion and Results
3. Conclusions
4. Materials and Methods
4.1. General
4.2. General Procedure for the Synthesis of 2-Phenylquinazolin-4(3H)-one (4aa)
4.3. General Procedure for the Synthesis of 2-Phenyl-2,3-dihydroquinazolin-4(1H)-one (5aa)
4.4. General Procedure for the Synthesis of 2-Phenylquinazolin-4(3H)-one (4aa) with Scheme 3
4.5. General Procedure for the Synthesis of 2-Phenyl-2,3-dihydroquinazolin-4(1H)-one (5aa) with Scheme 3
4.6. General Procedure for the Synthesis of 2-Phenyl-2,3-dihydroquinazolin-4(1H)-one (5aa) with Scheme 4
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Safavi, M.; Ashtari, A.; Khalili, F.; Mirfazli, S.S.; Saeedi, M.; Ardestani, S.K.; Rashidi, R.P.; Barazandeh, T.M.; Larijani, B.; Mahdavi, M. Novel quinazolin-4(3H)-one linked to 1,2,3-triazoles: Synthesis and anticancer activity. Chem. Biol. Drug Des. 2018, 92, 1373–1381. [Google Scholar] [CrossRef] [PubMed]
- Logé, C.; Testard, A.; Thiéry, V.; Lozach, O.; Blairvacq, M.; Robert, J.-M.; Meijer, L.; Besson, T. Novel 9-oxo-thiazolo [5, 4-f] quinazoline-2-carbonitrile derivatives as dual cyclin-dependent kinase 1 (CDK1)/glycogen synthase kinase-3 (GSK-3) inhibitors: Synthesis, biological evaluation and molecular modeling studies. Eur. J. Med. Chem. 2008, 43, 1469–1477. [Google Scholar] [CrossRef] [PubMed]
- Moussa, G.; Alaaeddine, R.; Alaeddine, L.M.; Nassra, R.; Belal, A.S.F.; Ismail, A.; El-Yazbi, A.F.; Abdel-Ghany, Y.S.; Hazzaa, A. Novel click modifiable thioquinazolinones as anti-inflammatory agents: Design, synthesis, biological evaluation and docking study. Eur. J. Med. Chem. 2018, 144, 635–650. [Google Scholar] [CrossRef] [PubMed]
- Zhan, X.; Xu, Y.; Qi, Q.; Wang, Y.; Shi, H.; Mao, Z. Synthesis, cytotoxic, and antibacterial evaluation of quinazolinone derivatives with substituted amino moiety. Chem. Biodivers. 2018, 15, e1700513. [Google Scholar] [CrossRef] [PubMed]
- Liao, B.-L.; Pan, L.W.; Pan, Y.-J.; Zhang, W. Four natural compounds were separated from folium isatidis: Crystal structures and antibacterial activity. Chem. Biodivers. 2018, e1800152. [Google Scholar] [CrossRef] [PubMed]
- Wolfe, J.F.; Rathman, T.L.; Sleevi, M.C.; Campbell, J.A.; Greenwood, T.D. Synthesis and anticonvulsant activity of some new 2-substituted 3-aryl-4(3H)-quinazolinones. J. Med. Chem. 1990, 33, 161–166. [Google Scholar] [CrossRef] [PubMed]
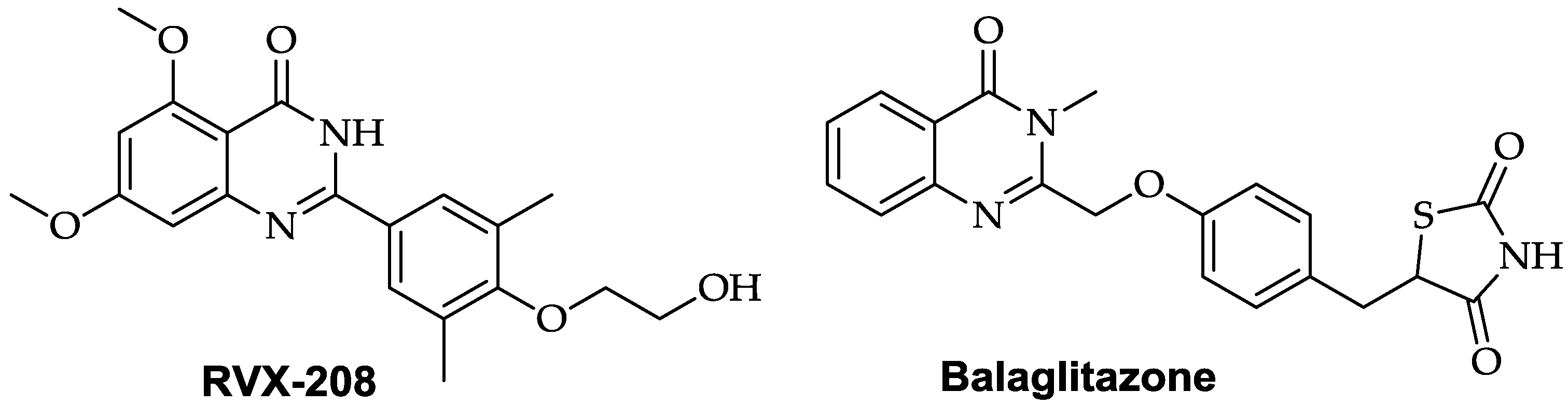
- Ghosh, G.C.; Bhadra, R.; Ghosh, R.K.; Banerjee, K.; Gupta, A. RVX 208: A novel BET protein inhibitor, role as an inducer of apo A.-I./HDL and beyond. Cardiovasc. Ther. 2017, 35, e12265. [Google Scholar] [CrossRef] [PubMed]
- Larsen, P.J.; Lykkegaard, K.; Larsen, L.K.; Fleckner, J.; Sauerberg, P.; Wassermann, K.; Wulff, E.M. Dissociation of antihyperglycaemic and adverse effects of partial perioxisome proliferator-activated receptor (PPAR-γ) agonist balaglitazone. Eur. J. Pharmacol. 2008, 596, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-X.; Lin, J.; Liang, T.-Z.; Duan, H.; Tan, X.-H.; Xi, B.-M.; Li, L.; Liu, S.-W. The BET bromodomain inhibitor apabetalone induces apoptosis of latent HIV-1 reservoir cells following viral reactivation. Acta Pharmacol. Sin. 2018. [Google Scholar] [CrossRef] [PubMed]
- Connolly, D.J.; Cusack, D.; O’Sullivan, T.P.; Guiry, P.J. Synthesis of quinazolinones and quinazolines. Tetrahedron 2005, 61, 10153–10202. [Google Scholar] [CrossRef]
- He, L.; Li, H.; Chen, J.; Wu, X.-F. Recent advances in 4(3H)-quinazolinone syntheses. RSC Adv. 2014, 4, 12065–12077. [Google Scholar] [CrossRef]
- Upadhyaya, K.; Thakur, R.K.; Shukla, S.K.; Tripathi, R.P. One-pot copper(i)-catalyzed ligand/base-free tandem cyclooxidative synthesis of quinazolinones. J. Org. Chem. 2016, 81, 5046–5055. [Google Scholar] [CrossRef] [PubMed]
- Laclef, S.; Harari, M.; Godeau, J.; Schmitz-Afonso, I.; Bischoff, L.; Hoarau, C.; Levacher, V.; Fruit, C.; Besson, T. Ligand-free Pd-catalyzed and copper-assisted C–H arylation of quinazolin-4-ones with aryl iodides under microwave heating. Org. Lett. 2015, 17, 1700–1703. [Google Scholar] [CrossRef] [PubMed]
- Cheng, R.; Guo, T.; Zhang-Negrerie, D.; Du, Y.; Zhao, K. One-pot synthesis of quinazolinones from anthranilamides and aldehydes via p-toluenesulfonic acid catalyzed cyclocondensation and phenyliodine diacetate mediated oxidative dehydrogenation. Synthesis 2013, 45, 2998–3006. [Google Scholar] [CrossRef]
- Ge, W.; Zhu, X.; Wei, Y. Iodine-catalyzed oxidative system for cyclization of primary alcohols with o-aminobenzamides to quinazolinones using DMSO as the oxidant in dimethyl carbonate. RSC Adv. 2013, 3, 10817–10822. [Google Scholar] [CrossRef]
- Jiang, X.; Tang, T.; Wang, J.-M.; Chen, Z.; Zhu, Y.-M.; Ji, S.-J. Palladium-Catalyzed One-Pot Synthesis of Quinazolinones via tert-Butyl Isocyanide Insertion. J. Org. Chem. 2014, 79, 5082–5087. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.Y.; Cheon, C.-H. Synthesis of quinazolinones from anthranilamides and aldehydes via metal-free aerobic oxidation in DMSO. Tetrahedron Lett. 2014, 55, 2340–2344. [Google Scholar] [CrossRef]
- Sharif, M.; Opalach, J.; Langer, P.; Beller, M.; Wu, X.-F. Oxidative synthesis of quinazolinones and benzothiadiazine 1,1-dioxides from 2-aminobenzamide and 2-aminobenzenesulfonamide with benzyl alcohols and aldehydes. RSC Adv. 2014, 4, 8–17. [Google Scholar] [CrossRef]
- Wu, X.-F.; He, L.; Neumann, H.; Beller, M. Palladium-catalyzed carbonylative synthesis of quinazolinones from 2-aminobenzamide and aryl bromides. Chem.-Eur. J. 2013, 19, 12635–12638. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Zhu, X.-R.; Qian, P.-C.; Zhang, X.-G.; Deng, C.-L. Copper-catalyzed tandem reaction of 2-aminobenzamides with tertiary amines for the synthesis of quinazolinone derivatives. Synlett 2016, 27, 2851–2857. [Google Scholar] [CrossRef]
- Zhan, D.; Li, T.; Wei, H.; Weng, W.; Ghandi, K.; Zeng, Q. A recyclable CuO-catalyzed synthesis of 4(3H)-quinazolinones. RSC Adv. 2013, 3, 9325–9329. [Google Scholar] [CrossRef]
- Li, F.; Lu, L.; Liu, P. Acceptorless dehydrogenative coupling of o-aminobenzamides with the activation of methanol as a C1 source for the construction of quinazolinones. Org. Lett. 2016, 18, 2580–2583. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.-Q.; Wang, L.-X.; Xiang, J.-F.; Yang, L.; Tang, Y.-L. Cu(II)-catalyzed domino reaction of 2-halobenzamide and arylmethanamine to construct 2-aryl quinazolinone. Chin. Chem. Lett. 2015, 26, 369–372. [Google Scholar] [CrossRef]
- Huang, C.; Fu, Y.; Fu, H.; Jiang, Y.; Zhao, Y. Highly efficient copper-catalyzed cascade synthesis of quinazoline and quinazolinone derivatives. Chem. Commun. 2008, 6333–6335. [Google Scholar] [CrossRef] [PubMed]
- Kotipalli, T.; Kavala, V.; Janreddy, D.; Bandi, V.; Kuo, C.-W.; Yao, C.-F. Synthesis of 2,3-disubstituted quinazolinone derivatives through copper catalyzed C–H amidation reactions. Eur. J. Org. Chem. 2016, 2016, 1182–1193. [Google Scholar] [CrossRef]
- Larksarp, C.; Alper, H. Palladium-catalyzed cyclocarbonylation of o-iodoanilines with heterocumulenes: Regioselective preparation of 4(3H)-quinazolinone derivatives. J. Org. Chem. 2000, 65, 2773–2777. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Jiang, Y.; Ma, D. Synthesis of 3-substituted and 2,3-disubstituted quinazolinones via cu-catalyzed aryl amidation. Org. Lett. 2012, 14, 1150–1153. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Fu, H. Amino acids as the nitrogen-containing motifs in copper-catalyzed domino synthesis of n-heterocycles. J. Org. Chem. 2011, 76, 3846–3852. [Google Scholar] [CrossRef] [PubMed]
- Xu, W.; Jin, Y.; Liu, H.; Jiang, Y.; Fu, H. Copper-catalyzed domino synthesis of quinazolinones via ullmann-type coupling and aerobic oxidative C.−H. amidation. Org. Lett. 2011, 13, 1274–1277. [Google Scholar] [CrossRef] [PubMed]
- Yu, L.; Wang, M.; Li, P.; Wang, L. Fe3O4 nanoparticle-supported copper(I): Magnetically recoverable and reusable catalyst for the synthesis of quinazolinones and bicyclic pyrimidinones. Appl. Organomet. Chem. 2012, 26, 576–582. [Google Scholar] [CrossRef]
- Guo, S.; Li, Y.; Tao, L.; Zhang, W.; Fan, X. Rapid assembly of quinazolinone scaffold via copper-catalyzed tandem reaction of 2-bromobenzamides with aldehydes and aqueous ammonia: Application to the synthesis of the alkaloid tryptanthrin. RSC Adv. 2014, 4, 59289–59296. [Google Scholar] [CrossRef]
- Liu, X.; Fu, H.; Jiang, Y.; Zhao, Y. A simple and efficient approach to quinazolinones under mild copper-catalyzed conditions. Angew. Chem. Int. Ed. 2009, 48, 348–351. [Google Scholar] [CrossRef] [PubMed]
- He, L.; Li, H.; Neumann, H.; Beller, M.; Wu, X.-F. Highly efficient four-component synthesis of 4(3 H)-quinazolinones: Palladium-catalyzed carbonylative coupling reactions. Angew. Chem. Int. Ed. 2014, 53, 1420–1424. [Google Scholar] [CrossRef] [PubMed]
- Zeng, L.-Y.; Cai, C. Iodine: Selectively promote the synthesis of mono substituted quinazolin-4(3H)-ones and 2,3-dihydroquinazolin-4(1H)-ones in one-pot. J. Heterocycl. Chem. 2010, 47, 1035–1039. [Google Scholar] [CrossRef]
- Li, H.; He, L.; Neumann, H.; Beller, M.; Wu, X.-F. Cascade synthesis of quinazolinones from 2-aminobenzonitriles and aryl bromides via palladium-catalyzed carbonylation reaction. Green Chem. 2014, 16, 1336–1343. [Google Scholar] [CrossRef]
- Wu, X.-F.; Oschatz, S.; Block, A.; Spannenberg, A.; Langer, P. Base mediated synthesis of 2-aryl-2,3-dihydroquinazolin-4(1H)-ones from 2-aminobenzonitriles and aromatic aldehydes in water. Org. Biomol. Chem. 2014, 12, 1865–1870. [Google Scholar] [CrossRef] [PubMed]
- Wu, X.-F.; Oschatz, S.; Sharif, M.; Beller, M.; Langer, P. Palladium-catalyzed carbonylative synthesis of N-(2-cyanoaryl)benzamides and sequential synthesis of quinazolinones. Tetrahedron 2014, 70, 23–29. [Google Scholar] [CrossRef]
- Schmid, T.E.; Gómez-Herrera, A.; Songis, O.; Sneddon, D.; Révolte, A.; Nahra, F.; Cazin, C.S. Selective NaOH-catalysed hydration of aromatic nitriles to amides. Catal. Sci. Technol. 2015, 5, 2865–2868. [Google Scholar] [CrossRef] [Green Version]
- Sahnoun, S.; Messaoudi, S.; Peyrat, J.-F.; Brion, J.-D.; Alami, M. Cs2CO3 in pyrrolidinone promoted hydration of functionalized (hetero)aryl nitriles under metal-free conditions. Tetrahedron Lett. 2012, 53, 2860–2863. [Google Scholar] [CrossRef]
- Zeng, L.-Y.; Liu, T.; Yang, J.; Yang, Y.; Cai, C.; Liu, S. “On-Water” facile synthesis of novel pyrazolo[3,4-b]pyridinones possessing anti-influenza virus activity. ACS Comb. Sci. 2017, 19, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Yadong, F.; Yudong, L.; Guolin, C.; Lianhui, W.; Xiuling, C. Copper-catalyzed synthesis of 2-arylquinazolinones from 2-arylindoles with amines or ammoniums. J. Org. Chem. 2015, 80, 7099–7107. [Google Scholar] [CrossRef]
- Mitsuaki, Y.; Yukari, N.; Seiya, H.; Akira, I. One-pot synthesis of polyhydropyrido [1, 2-a] indoles and tetracyclic quinazolinones from 2-arylindoles using copper-mediated oxidative tandem reactions. Tetrahedron 2016, 72, 4123–4131. [Google Scholar] [CrossRef]
- Khan, I.; Zaib, S.; Batool, S.; Abbas, N.; Ashraf, Z.; Iqbal, J.; Saeed, A. Quinazolines and quinazolinones as ubiquitous structural fragments in medicinal chemistry: An update on the development of synthetic methods and pharmacological diversification. Bioorgan. Med. Chem. 2016, 24, 2361–2381. [Google Scholar] [CrossRef] [PubMed]
- Butler, R.N.; Coyne, A.G. Water: Nature’s reaction enforcer—Comparative effects for organic synthesis “in-water” and “on-water”. Chem. Rev. 2010, 110, 6302–6337. [Google Scholar] [CrossRef] [PubMed]
- Dam, B.; Nandi, S.; Pal, A.K. An efficient ‘on-water’ synthesis of 1,4-dihydropyridines using Fe3O4@SiO2 nanoparticles as a reusable catalyst. Tetrahedron Lett. 2014, 55, 5236–5240. [Google Scholar] [CrossRef]
- Saikia, B.; Boruah, P.R.; Ali, A.A.; Sarma, D. ‘On-water’ organic synthesis: A. green, highly efficient, low cost and reusable catalyst system for biaryl synthesis under aerobic conditions at room temperature. RSC Adv. 2015, 5, 50655–50659. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are not available from the authors. |

| Entry | Catal. (10 mol %) | Base (2 eqv.) | Solvent | Temp (°C) | Yield (%) b |
|---|---|---|---|---|---|
| 1 | CuBr | Cs2CO3 | DMSO | 100 | 54 |
| 2 | CuBr | Cs2CO3 | DMSO | 80 | 49 |
| 3 | CuBr | Cs2CO3 | DMSO | 120 | 42 |
| 4 | CuBr | - | DMSO | 100 | 38 |
| 5 | CuBr | K2CO3 | DMSO | 100 | 47 |
| 6 | CuBr | K3PO4 | DMSO | 100 | 51 |
| 7 | CuBr | NaOH | DMSO | 100 | 37 |
| 8 | CuBr | Et3N | DMSO | 100 | 21 |
| 9 | CuCl | Cs2CO3 | DMSO | 100 | 54 |
| 10 | - | Cs2CO3 | DMSO | 100 | 16 |
| 11 | CuI | Cs2CO3 | DMSO | 100 | 33 |
| 12 | CuCl2 | Cs2CO3 | DMSO | 100 | 62 |
| 13 | FeCl3 | Cs2CO3 | DMSO | 100 | 28 |
| 14 | CuO | Cs2CO3 | DMSO | 100 | 63 |
| 15 | ZnCl2 | Cs2CO3 | DMSO | 100 | 45 |
| 16 | CuCl2 | Cs2CO3 | DMF | 100 | 24 |
| 17 | CuCl2 | Cs2CO3 | DMA | 100 | 41 |
| 18 | CuCl2 | Cs2CO3 | H2O | 100 | 75 |
| 19 | CuCl2 | Cs2CO3 | H2O/DMSO (4:1) | 100 | 47 |
| 20 | CuCl2 | Cs2CO3 | H2O/PEG400 (4:1) | 100 | 55 |
| 21 | CuCl2 | Cs2CO3 | DMSO | 100 | 67 |
| 22 | CuCl2 | Cs2CO3 | H2O | 100 | Trace c |
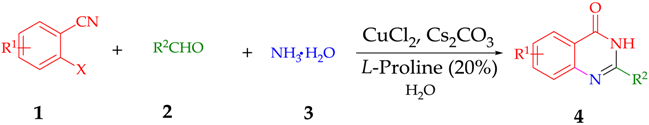
| Entry | X | R1 | R2 | Product | Yield b |
|---|---|---|---|---|---|
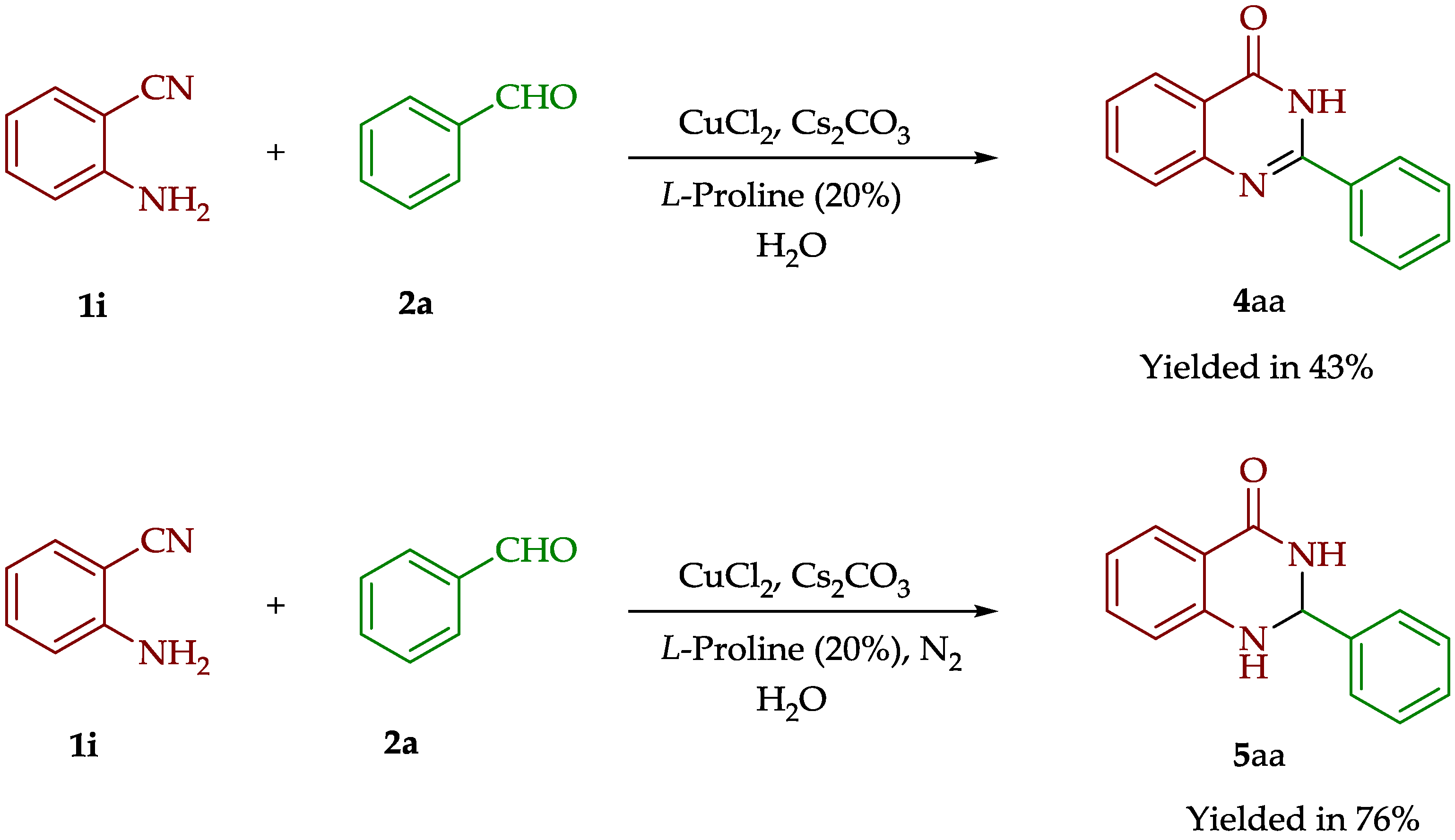
| 1 | Br | - | C6H5 | 4aa | 75% |
| 2 | F | - | C6H5 | 4aa | 0 |
| 3 | Cl | - | C6H5 | 4aa | 21% |
| 4 | I | - | C6H5 | 4aa | 17% |
| 5 | Br | - | 4-ClC6H5 | 4ab | 44% |
| 6 | Br | - | 2-ClC6H5 | 4ac | 41% |
| 7 | Br | - | 4-CH3C6H5 | 4ad | 71% |
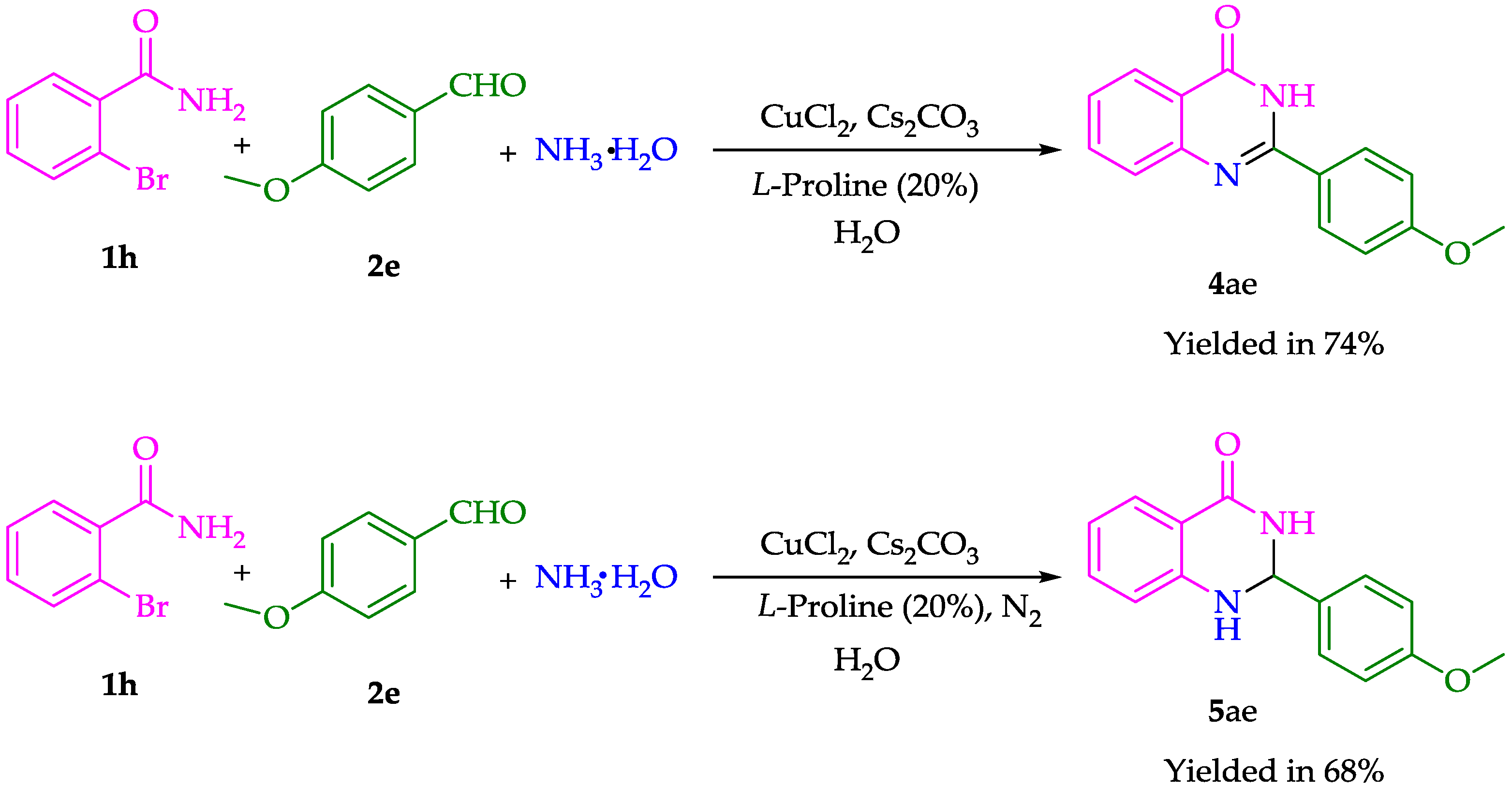
| 8 | Br | - | 4-MeOC6H5 | 4ae | 73% |
| 9 | Br | - | 2-MeOC6H5 | 4af | 83% |
| 10 | Br | - | 4-HOC6H5 | 4ag | 42% |
| 11 | Br | - | 2-HOC6H5 | 4ah | 0 |
| 12 | Br | - | 4-CF3C6H5 | 4ai | 27% |
| 13 | Br | - | 4-N(CH3)2C6H5 | 4aj | 30% |
| 14 | Br | - | naphthalene | 4ak | 66% |
| 15 | Br | - | 4-Pyridine | 4al | 48% |
| 16 | Br | - | 2-Pyridine | 4am | 0 |
| 17 | Br | - | 2-furan | 4an | 0 |
| 18 | Br | - | 2-thiophene | 4ao | 0 |
| 19 | Br | - | 2-pyrrole | 4ap | 27% |
| 20 | Br | 5-F | C6H5 | 4ea | 60% |
| 21 | Br | 5-F | 4-MeOC6H5 | 4ee | 73% |
| 22 | Br | 5-F | 2-MeOC6H5 | 4ef | 92% |
| 23 | Br | 5-F | 3-MeO-4-HOC6H5 | 4eq | 60% |
| 24 | Br | 5-CH3 | C6H5 | 4fa | 63% |
| 25 | Br | 5-CH3 | 4-ClC6H5 | 4fb | 47% |
| 26 | Br | 5-CH3 | 4-MeOC6H5 | 4fe | 51% |
| 27 | Br | 5-CH3 | 2-MeOC6H5 | 4ff | 62% |
| 28 | Br | 5-MeO | 4-MeOC6H5 | 4ge | 67% |
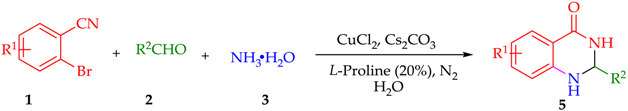
| Entry | R1 | R2 | Product | Yield b |
|---|---|---|---|---|
| 1 | - | C6H5 | 5aa | 74% |
| 2 | - | 4-ClC6H5 | 5ab | 96% |
| 3 | - | 4-MeOC6H5 | 5ae | 71% |
| 4 | - | 2-MeOC6H5 | 5af | 54% |
| 5 | 5-F | C6H5 | 5ea | 39% |
| 6 | 5-F | 4-ClC6H5 | 5eb | 27% |
| 7 | 5-F | 4-MeOC6H5 | 5ee | 53% |
| 8 | 5-F | 2-MeOC6H5 | 5eq | 53% |
| 9 | 5-CH3 | C6H5 | 5fa | 77% |
| 10 | 5-CH3 | 4-ClC6H5 | 5fb | 0 |
| 11 | 5-CH3 | 4-MeOC6H5 | 5fe | 50% |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Liu, Z.; Zeng, L.-Y.; Li, C.; Yang, F.; Qiu, F.; Liu, S.; Xi, B. “On-Water” Synthesis of Quinazolinones and Dihydroquinazolinones Starting from o-Bromobenzonitrile. Molecules 2018, 23, 2325. https://doi.org/10.3390/molecules23092325
Liu Z, Zeng L-Y, Li C, Yang F, Qiu F, Liu S, Xi B. “On-Water” Synthesis of Quinazolinones and Dihydroquinazolinones Starting from o-Bromobenzonitrile. Molecules. 2018; 23(9):2325. https://doi.org/10.3390/molecules23092325
Chicago/Turabian StyleLiu, Zibin, Li-Yan Zeng, Chao Li, Fubiao Yang, Fensheng Qiu, Shuwen Liu, and Baomin Xi. 2018. "“On-Water” Synthesis of Quinazolinones and Dihydroquinazolinones Starting from o-Bromobenzonitrile" Molecules 23, no. 9: 2325. https://doi.org/10.3390/molecules23092325







