Structure–Activity Relationship of the Tyrosinase Inhibitors Kuwanon G, Mulberrofuran G, and Albanol B from Morus Species: A Kinetics and Molecular Docking Study
Abstract
:1. Introduction
2. Results
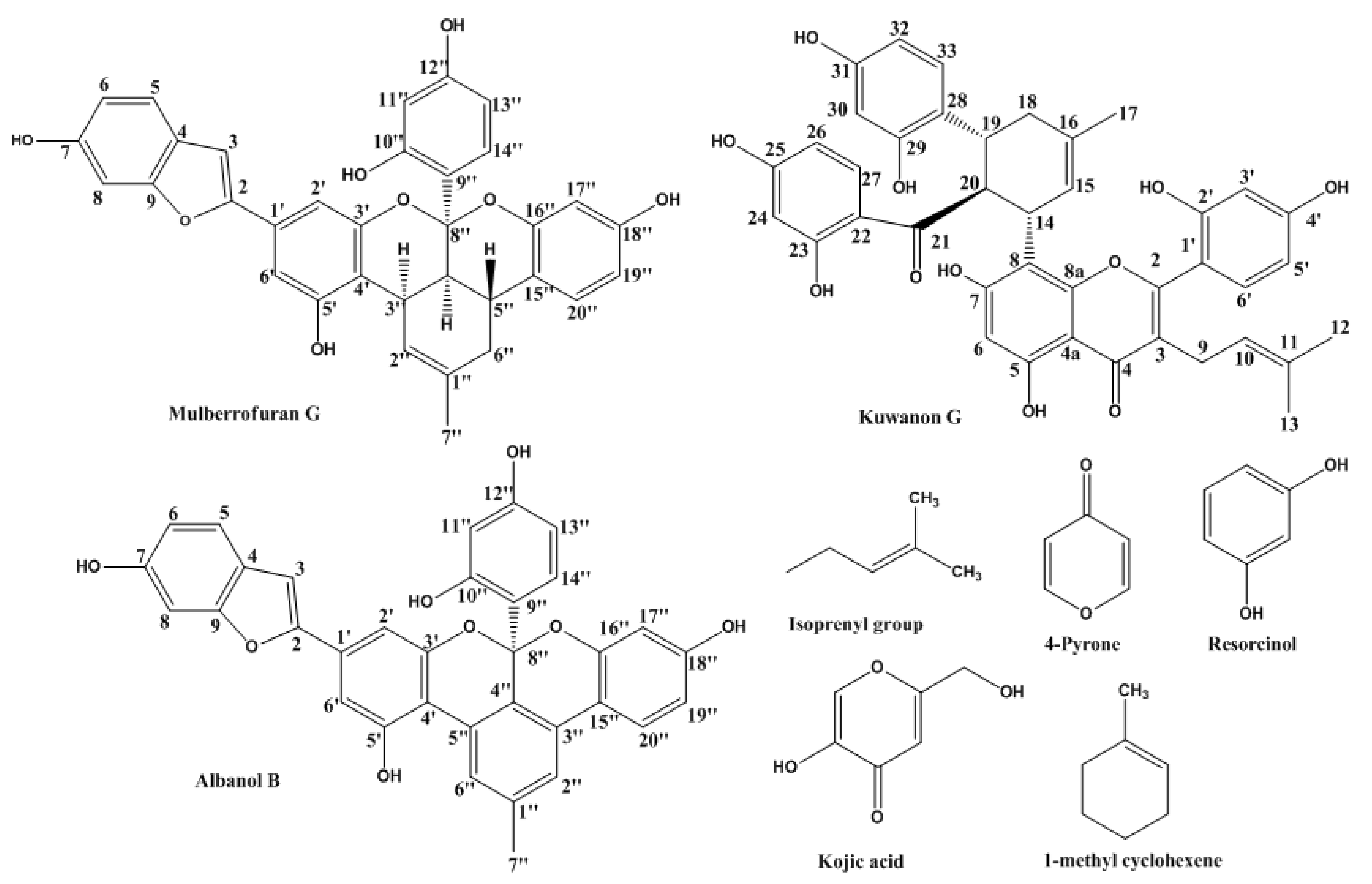
2.1. Inhibitory Activities of KG, MG, AB and 1-Methyl-1-Cyclohexene on Mushroom Tyrosinase (l-Tyrosine and l-DOPA Substrates)
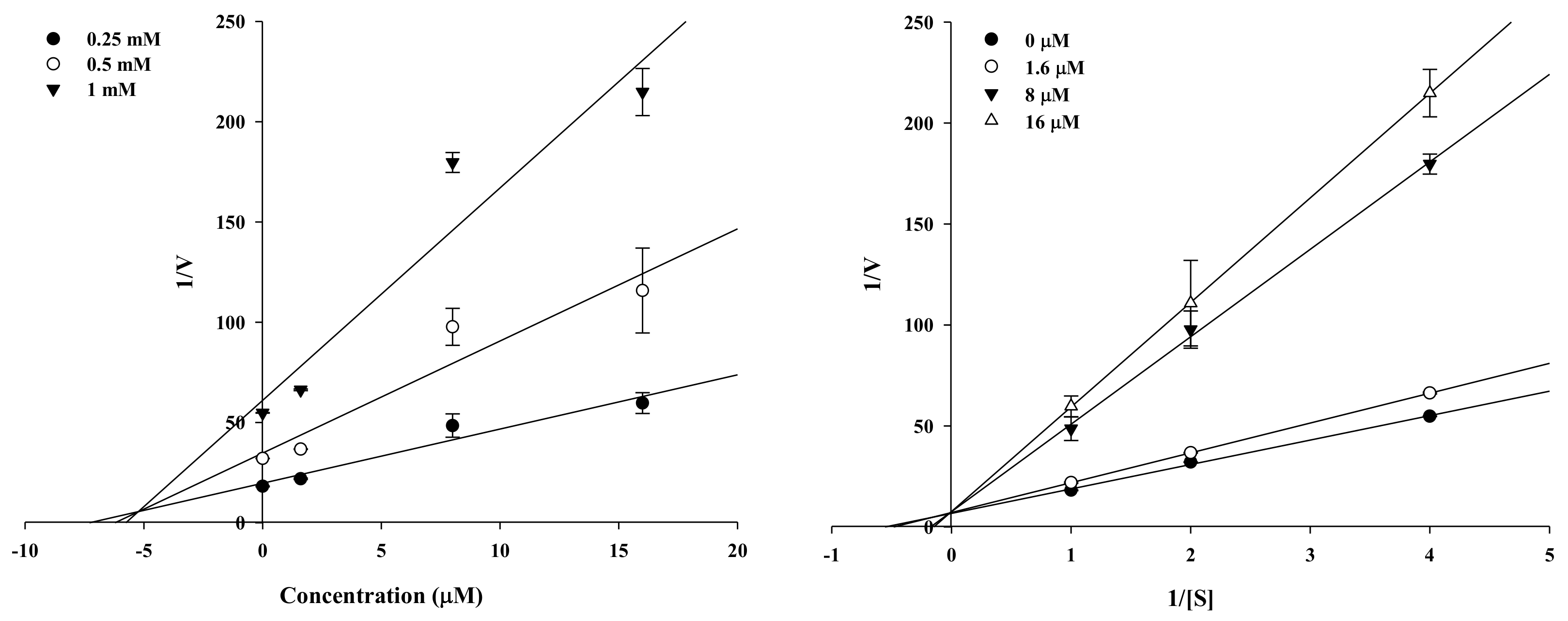
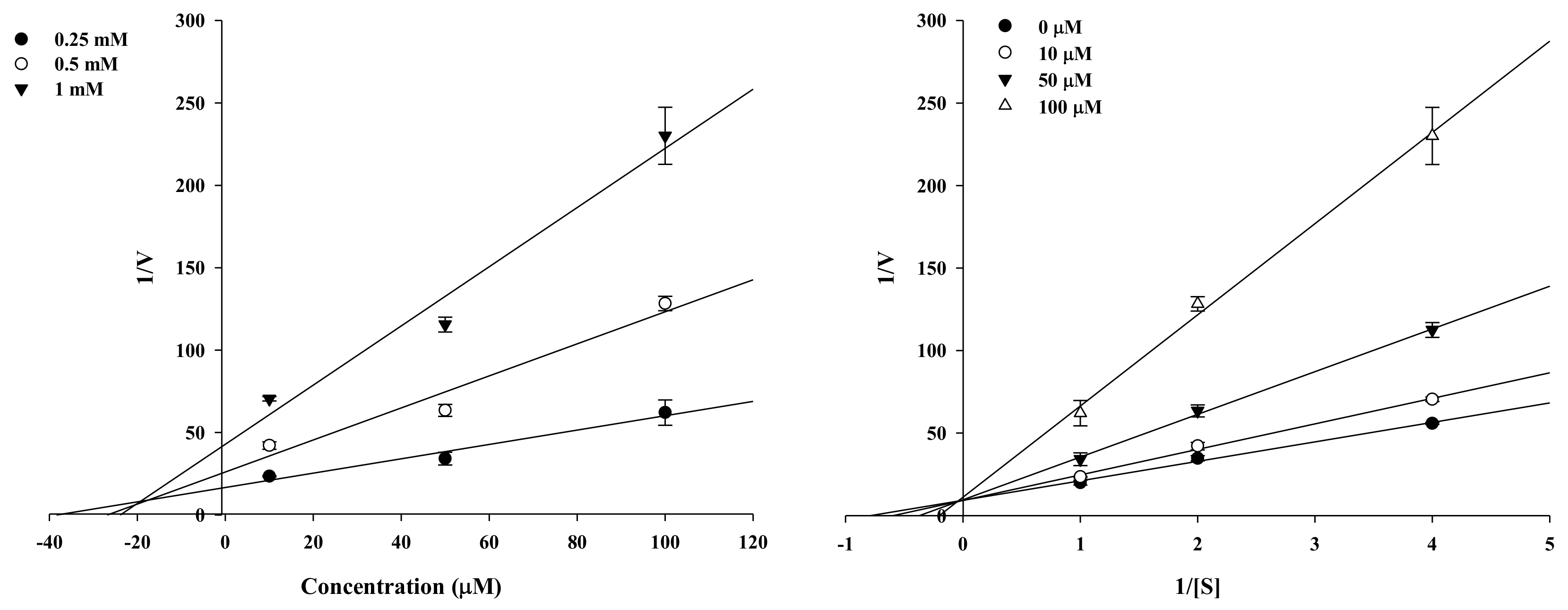
2.2. Enzyme Kinetic Analysis of KG and MG with Monophenolase Activity of Tyrosinase
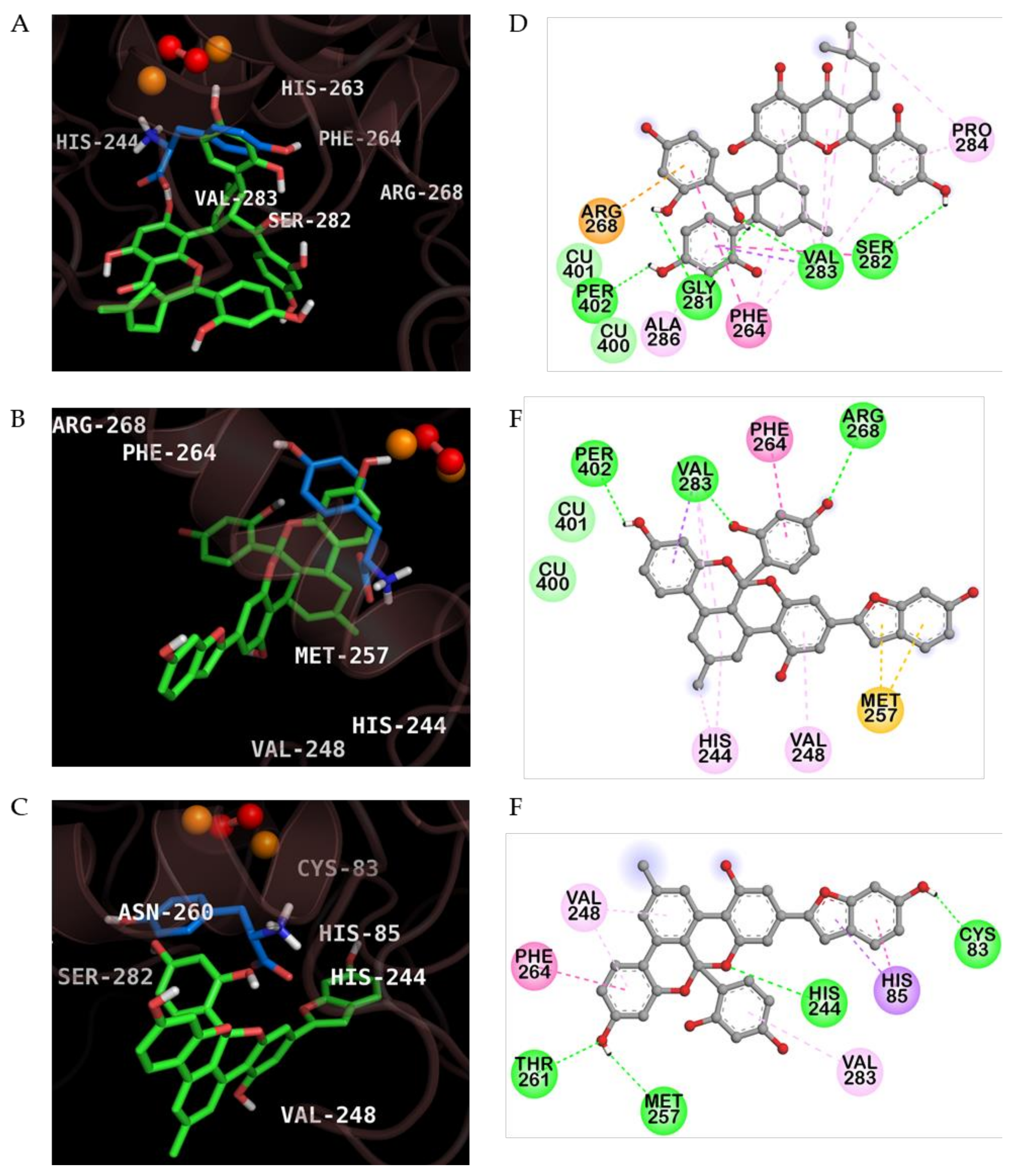
2.3. Molecular Docking Simulation of KG, MG and AB Tyrosinase Inhibition
3. Discussion
4. Materials and Methods
4.1. Chemicals/Reagents and Compounds
4.2. Mushroom Tyrosinase Inhibitory Assay
4.3. Enzyme Kinetic Analysis with Tyrosinase
4.4. Tyrosinase Molecular Docking Simulations
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Bagherzadeh, K.; Shirgahi Talari, F.; Sharifi, A.; Ganjali, M.R.; Saboury, A.A.; Amanlou, M. A new insight into mushroom tyrosinase inhibitors: Docking, pharmacophore-based virtual screening, and molecular modeling studies. J. Biomol. Struct. Dyn. 2015, 33, 487–501. [Google Scholar] [CrossRef] [PubMed]
- Zeng, Q.; Chen, H.; Zhang, C.; Han, M.; Li, T.; Qi, X.; Xiang, Z.; He, N. Definition of eight mulberry species in the genus Morus by internal transcribed spacer-based phylogeny. PLoS ONE 2015, 10, e0135411. [Google Scholar] [CrossRef] [PubMed]
- Kojima, Y.; Kimura, T.; Nakagawa, K.; Asai, A.; Hasumi, K.; Oikawa, S.; Miyazawa, T. Effects of mulberry leaf extract rich in 1-deoxynojirimycin on blood lipid profiles in humans. J. Clin. Biochem. Nutr. 2010, 47, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Hussain, F.; Rana, Z.; Shafique, H.; Malik, A.; Hussain, Z. Phytopharmacological potential of different species of Morus alba and their bioactive phytochemicals: A review. Asian Pac. J. Trop. Biomed. 2017, 7, 950–956. [Google Scholar] [CrossRef]
- Cui, X.Q.; Wang, H.Q.; Liu, C.; Chen, R.Y. Study of anti-oxidant phenolic compounds from stem barks of Morus yunanensis. J. Chin. Mater. Med. 2008, 33, 1569–1572. [Google Scholar]
- Kobayashi, Y.; Miyazawa, M.; Araki, M.; Kamei, A.; Abe, K. Effects of Morus alba L. (Mulberry) leaf extract in hypercholesterolemic mice on suppression of cholesterol synthesis. Pharmacogn. Nat. Prod. 2015, 2, 1000113. [Google Scholar] [CrossRef]
- Lee, J.S.; Kim, Y.R.; Park, J.M.; Kim, Y.E.; Baek, N.I.; Hong, E.K. Cyanidin-3-glucoside isolated from mulberry fruits protects pancreatic β-cells against glucotoxicity-induced apoptosis. Mol. Med. Rep. 2015, 11, 2723–2728. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, I.; Beg, A.Z. Antimicrobial and phytochemical studies on 45 Indian medicinal plants against multi-drug resistant human pathogens. J. Ethnopharmacol. 2001, 74, 113–123. [Google Scholar] [CrossRef]
- Guo, C.; Yang, J.; Wei, J.; Li, Y.; Xu, J.; Jiang, Y. Antioxidant activities of peel, pulp and seed fractions of common fruits as determined by FRAP assay. Nutr. Res. 2003, 23, 1719–1726. [Google Scholar] [CrossRef]
- Mallhi, T.H.; Qadir, M.I.; Khan, Y.H.; Ali, M. Hepatoprotective activity of aqueous methanolic extract of Morus nigra against paracetamol-induced hepatotoxicity in mice. Bangladesh J. Pharmacol. 2014, 9, 60–66. [Google Scholar] [CrossRef]
- Chon, S.U.; Kim, Y.M.; Park, Y.J.; Heo, B.G.; Park, Y.S.; Gorinstein, S. Antioxidant and antiproliferative effects of methanol extracts from raw and fermented parts of mulberry plant (Morus alba L.). Eur. Food Res. Technol. 2009, 230, 231–237. [Google Scholar] [CrossRef]
- Zheng, Z.P.; Cheng, K.W.; Zhu, Q.; Wang, X.C.; Lin, Z.X.; Wang, M. Tyrosinase inhibitory constituents from the roots of Morus nigra: A structure-activity relationship study. J. Agric. Food Chem. 2010, 58, 5368–5373. [Google Scholar] [CrossRef] [PubMed]
- Chaita, E.; Lambrinidis, G.; Cheimonidi, C.; Agalou, A.; Beis, D.; Trougakos, I.; Mikros, E.; Skaltsounis, A.L.; Aligiannis, N. Anti-melanogenic properties of greek plants. A novel depigmenting agent from Morus alba wood. Molecules 2017, 22, 514. [Google Scholar] [CrossRef] [PubMed]
- Abbas, G.M.; Abdel Bar, F.M.; Baraka, H.N.; Gohar, A.A.; Lahloub, M.F. A new antioxidant stilbene and other constituents from the stem bark of Morus nigra L. Nat. Prod. Res. 2014, 28, 952–959. [Google Scholar] [CrossRef] [PubMed]
- Park, K.M.; You, J.S.; Lee, H.Y.; Baek, N.I.; Hwang, J.K. Kuwanon G: An antibacterial agent from the root bark of Morus alba against oral pathogens. J. Ethnopharmacol. 2003, 84, 181. [Google Scholar] [CrossRef]
- Kuk, E.B.; Jo, A.R.; Oh, S.I.; Sohn, H.S.; Seong, S.H.; Roy, A.; Choi, J.S.; Jung, H.A. Anti-Alzheimer’s disease activity of compounds from the root bark of Morus alba L. Arch. Pharm. Res. 2017, 40, 338–349. [Google Scholar] [CrossRef] [PubMed]
- Guo, H.; Xu, Y.; Huang, W.; Zhou, H.; Zheng, Z.; Zhao, Y.; He, B.; Zhu, T.; Tang, S.; Zhu, Q. Kuwanon G preserves LPS-induced disruption of gut epithelial barrier in vitro. Molecules 2016, 21, 1597. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.J.; Ryu, J.; Park, S.H.; Woo, E.R.; Kim, A.R.; Lee, S.K.; Yeong, S.K.; Kim, J.O.; Hong, J.H.; Lee, C.J. Effects of Morus alba L. and natural products including morusin on In vivo secretion and In vitro production of airway MUC5AC mucin. Tuberc. Respir. Dis. 2014, 77, 65–72. [Google Scholar] [CrossRef] [PubMed]
- Jung, H.W.; Kang, S.Y.; Kang, J.S.; Kim, A.R.; Woo, E.R.; Park, Y.K. Effect of kuwanon G isolated from the root bark of Morus alba on ovalbumin-induced allergic response in a mouse model of asthma. Phytother. Res. 2014, 28, 1713–1719. [Google Scholar] [CrossRef] [PubMed]
- Geng, C.A.; Ma, Y.B.; Zhang, X.M.; Yao, S.Y.; Xue, D.Q.; Zhang, R.P.; Chen, J.J. Mulberrofuran G and isomulberrofuran G from Morus alba L.: Anti-hepatitis B virus activity and mass spectrometric fragmentation. J. Agric. Food Chem. 2012, 60, 8197–8202. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Kwon, J.; Kim, D.W.; Lee, H.J.; Lee, D.; Mar, W. Mulberrofuran G protects ischemic injury-induced cell death via inhibition of NOX4-mediated ROS generation and ER stress. Phytother. Res. 2017, 31, 321–329. [Google Scholar] [CrossRef] [PubMed]
- Jung, J.W.; Ko, W.M.; Park, J.H.; Seo, K.H.; Oh, E.J.; Lee, D.Y.; Lee, D.S.; Kim, Y.C.; Lim, D.W.; Han, D.; et al. Isoprenylated flavonoids from the root bark of Morus alba and their hepatoprotective and neuroprotective activities. Arch. Pharm. Res. 2015, 38, 2066–2075. [Google Scholar] [CrossRef] [PubMed]
- Sohn, H.Y.; Son, K.H.; Kwon, C.S.; Kwon, G.S.; Kang, S.S. Antimicrobial and cytotoxic activity of 18 prenylated flavonoids isolated from medicinal plants: Morus alba L., Morus mongolica Schneider, Broussnetia papyrifera (L.) Vent, Sophora flavescens Ait and Echinosophora koreensis Nakai. Phytomedicine 2004, 11, 666–672. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.P.; Tan, H.Y.; Wang, M. Tyrosinase inhibition constituents from the roots of Morus australis. Fitoterapia 2012, 83, 1008–1013. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.; Qi, X.; Liu, Y.; Guo, J.; Zhu, R.; Chen, W.; Zheng, X.; Yu, T. Dietary supplementation with purified mulberry (Morus australis Poir) anthocyanins suppresses body weight gain in high-fat diet fed C57BL/6 mice. Food Chem. 2013, 141, 482–487. [Google Scholar] [CrossRef] [PubMed]
- Hu, S.; Zheng, Z.; Chen, F.; Wang, M. The depigmenting effect of natural resorcinol type polyphenols Kuwanon O and Sanggenon T from the roots of Morus australis. J. Ethnopharmacol. 2017, 195, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Syah, Y.M.; Juliawaty, L.D.; Achmad, S.A.; Hakim, E.H.; Takayama, H.; Said, I.M.; Latip, J. Phenolic constituents from the wood of Morus australis with cytotoxic activity. Z. Naturforsch. C 2008, 63, 35–39. [Google Scholar]
- Liao, Y.R.; Kuo, P.C.; Tsai, W.J.; Huang, G.J.; Lee, K.H.; Wu, T.S. Bioactive chemical constituents from the root bark of Morus australis. Bioorganic Med. Chem. Lett. 2017, 27, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Dalmagro, A.P.; Camargo, A.; Zeni, A.L.B. Morus nigra and its major phenolic, syringic acid, have antidepressant-like and neuroprotective effects in mice. Metab. Brain Dis. 2017, 32, 1963–1973. [Google Scholar] [CrossRef] [PubMed]
- Souza, G.R; Oliveira-Junior, R.G.; Diniz, T.C.; Branco, A.; Lima-Saraiva, S.R.G.; Guimarães, A.L.; Oliveira, A.P.; Pacheco, A.G.M.; Silva, M.G.; Moraes-Filho, M.O.; et al. Assessment of the antibacterial, cytotoxic and antioxidant activities of Morus nigra L. (Moraceae). Braz. J. Biol. 2017. [Google Scholar] [CrossRef] [PubMed]
- Chao, W.W.; Su, C.C.; Peng, H.Y.; Chou, S.T. Melaleuca quinquenervia essential oil inhibits α-melanocyte-stimulating hormone-induced melanin production and oxidative stress in B16 melanoma cells. Phytomedicine. 2017, 34, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Zhao, X.; Tao, G.J.; Chen, J.; Zheng, Z.P. Investigating the inhibitory activity and mechanism differences between norartocarpetin and luteolin for tyrosinase: A combinatory kinetic study and computational simulation analysis. Food Chem. 2017, 223, 40–48. [Google Scholar] [CrossRef] [PubMed]
- Cabanes, J.; Chazarra, S.; Garcia-Carmona, F. Kojic acid, a cosmetic skin whitening agent, is a slow-binding inhibitor of catecholase activity of tyrosinase. J. Pharm. Pharmacol. 1994, 46, 982–985. [Google Scholar] [CrossRef] [PubMed]
- Hassani, S.; Haghbeen, K.; Fazli, M. Non-specific binding sites help to explain mixed inhibition in mushroom tyrosinase activities. Eur. J. Med. Chem. 2016, 122, 138–148. [Google Scholar] [CrossRef] [PubMed]
- Loizzo, M.R.; Tundis, R.; Menichini, F. Natural and synthetic tyrosinase inhibitors as antibrowning agents: An update. Compr. Rev. Food Sci. Food Saf. 2012, 11, 378–398. [Google Scholar] [CrossRef]
- Lee, S.Y.; Baek, N.; Nam, T.G. Natural, semisynthetic and synthetic tyrosinase inhibitors. J. Enzyme Inhib. Med. Chem. 2016, 31, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Slominski, A.; Zmijewski, M.A.; Pawelek, J. L-tyrosine and L-dihydroxyphenylalanine as hormone-like regulators of melanocyte functions. Pigment Cell Melanoma Res. 2012, 25, 14–27. [Google Scholar] [CrossRef] [PubMed]
- Hyun, S.K.; Lee, W.H.; Jeong, D.M.; Kim, Y.; Choi, J.S. Inhibitory effects of kurarinol, kuraridinol, and trifolirhizin from Sophora flavescens on tyrosinase and melanin synthesis. Biol. Pharm. Bull. 2008, 31, 154–158. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.H.; Huang, Y.C.; Tsai, M.L.; Cheng, C.Y.; Liu, L.L.; Yen, Y.W.; Chen, W.L. Inhibition of melanogenesis by β-caryophyllene from lime mint essential oil in mouse B16 melanoma cells. Int. J. Cosmet. Sci. 2015, 37, 550–554. [Google Scholar] [CrossRef] [PubMed]
- Masuda, T.; Odaka, Y.; Ogawa, N.; Nakamoto, K.; Kuninaga, H. Identification of geranic acid, a tyrosinase inhibitor in lemongrass (Cymbopogon citratus). J. Agric. Food Chem. 2007, 56, 597–601. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Z.P.; Chen, S.; Wang, S.; Wang, X.C.; Cheng, K.W.; Wu, J.J.; Yang, D.J.; Wang, M. Chemical components and tyrosinase inhibitors from the twigs of Artocarpus heterophyllus. J. Agric. Food Chem. 2009, 57, 6649–6655. [Google Scholar] [CrossRef] [PubMed]
- Ismaya, W.T.; Rozeboom, H.J.; Weijn, A.; Mes, J.J.; Fusetti, F.; Wichers, H.J.; Dijkstra, B.W. Crystal structure of Agaricus bisporus mushroom tyrosinase: Identity of the tetramer subunits and interaction with tropolone. Biochemistry 2011, 50, 5477–5486. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maria-Solano, M.A.; Ortiz-Ruiz, C.V.; Munoz-Munoz, J.L.; Teruel-Puche, J.A.; Berna, J.; Garcia-Ruiz, P.A.; Garcia-Canovas, F. Further insight into the pH effect on the catalysis of mushroom tyrosinase. J. Mol. Catal. B Enzym. 2016, 125, 6–15. [Google Scholar] [CrossRef]
- Goodsell, D.S.; Morris, G.M.; Olson, A.J. Automated docking of flexible ligands: Applications of AutoDock. J. Mol. Recognit. 1996, 9, 1–5. [Google Scholar] [CrossRef]
- Seong, S.H.; Ali, M.Y.; Kim, H.R.; Jung, H.A.; Choi, J.S. BACE1 inhibitory activity and molecular docking analysis of meroterpenoids from Sargassum serratifolium. Bioorganic Med. Chem. 2017, 25, 3964–3970. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Tested compounds (kuwanon G, mulberrofuran G, and albanol B) are available from the authors. |
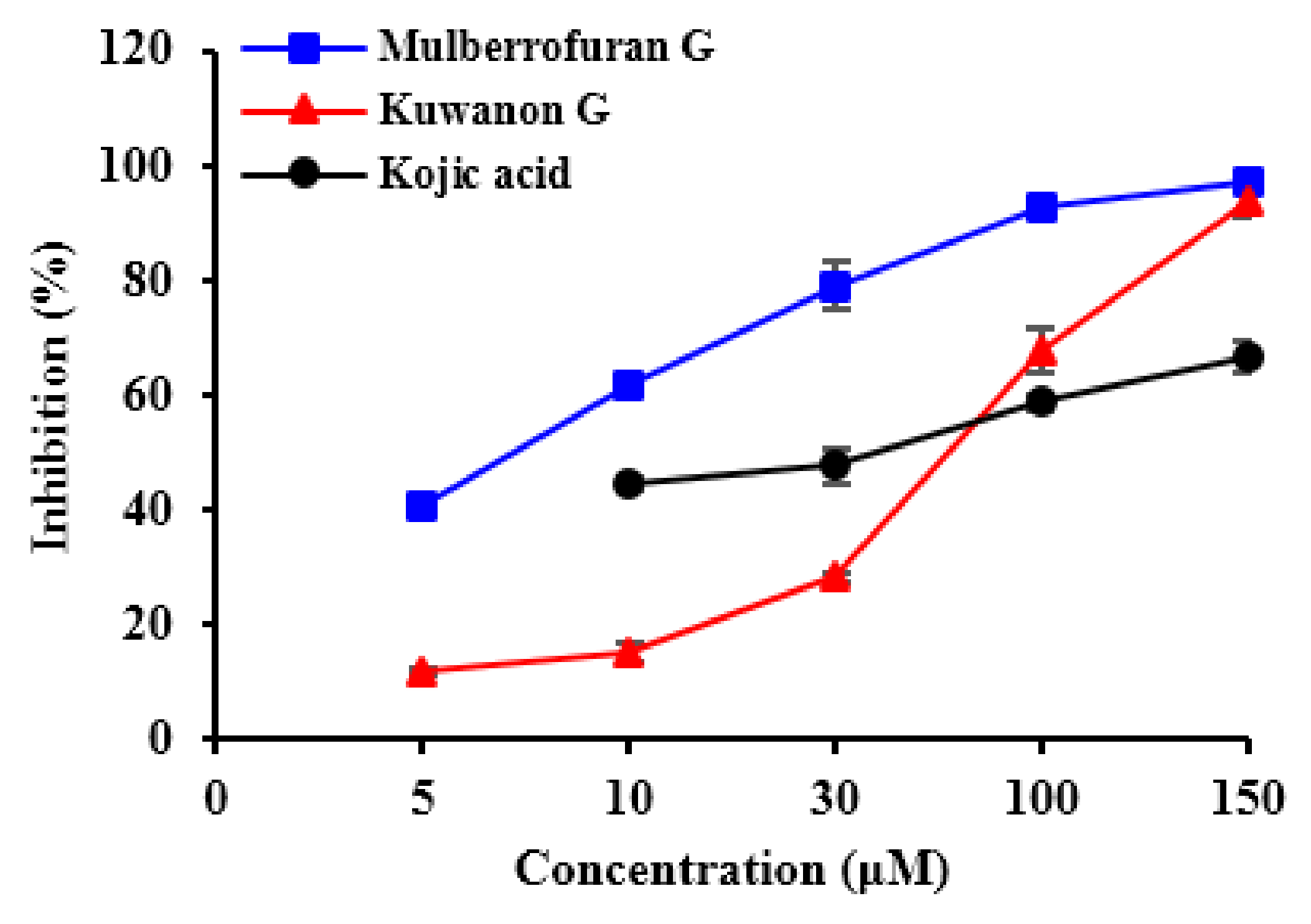
| Compounds | IC50 (μM) Value a | Inhibition Mode b | Inhibition Constant (Ki, μM) c | |
|---|---|---|---|---|
| l-Tyrosine | l-DOPA | |||
| Kuwanon G | 67.6 ± 2.11 | 44.0 ± 3.73 | Competitive | 18.7 |
| Mulberrofuran G | 6.35 ± 0.45 | 105.6 ± 1.85 | Competitive | 5.19 |
| 1-Methyl-1-cyclohexene | >1000 | >1000 | NT e | NT |
| Albanol B | >350 | >350 | NT | NT |
| Kojic acid d | 36.0 ± 0.88 | 79.0 ± 0.06 | NT | NT |
| Compounds | Binding Energy (Kcal/mol) | H-Bond Interaction | Van der Waals Interaction | Hydrophobic Interaction | Others |
|---|---|---|---|---|---|
| Kuwanon G | −6.95 | Per402, Val283, His263, Gly281, Ser282 | Cu400, Cu401 | Pi-sigma: Val283 Pi-Pi Stacked: His263 Pi-Pi T-Shaped: Phe264 Amide-Pi Stacked: Val283, Ser282 Alkyl: Val283, Pro284 Pi-Alkyl: Phe264, Val283, Pro284, Ala286 | Pi-Cation: Arg268 |
| Mulberrofuran G | −7.60 | Per402, Arg268, Val283 | Cu400, Cu401 | Pi-sigma: Val283 Pi-Pi T-Shaped: Phe264 Alkyl: Val283 Pi-Alkyl: His244, Val248 | Pi-Sulfur: Met257 |
| Albanol B | −7.28 | Cys83, His244, Met257, Thr261 | ‒ | Pi-Sigma: His85 Pi-Pi T-shaped: His85, Phe264 Pi-Alkyl: Val248, Val283 | ‒ |
| l-Tyrosine a | −6.31 | His244, Asn260, Met280, Glu256 (Salt-bridge) | Per402, Cu400, Cu401 | Pi-Sigma: Val283 Pi-Pi Stacked: His263 Pi-Alkyl: Ala286 | ‒ |
| Kojic acid b | −5.5 | Met280 | Cu400, Cu401 | Pi-Sigma: Val283 Pi-Pi T-Shaped: His263 Pi-Alkyl: Ala286 | |
| Cinnamic acid b,c | −6.20 | Gln307, Asp312, Glu356 | Tyr314 | Pi-Pi Stacked: Trp358 | ‒ |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koirala, P.; Seong, S.H.; Zhou, Y.; Shrestha, S.; Jung, H.A.; Choi, J.S. Structure–Activity Relationship of the Tyrosinase Inhibitors Kuwanon G, Mulberrofuran G, and Albanol B from Morus Species: A Kinetics and Molecular Docking Study. Molecules 2018, 23, 1413. https://doi.org/10.3390/molecules23061413
Koirala P, Seong SH, Zhou Y, Shrestha S, Jung HA, Choi JS. Structure–Activity Relationship of the Tyrosinase Inhibitors Kuwanon G, Mulberrofuran G, and Albanol B from Morus Species: A Kinetics and Molecular Docking Study. Molecules. 2018; 23(6):1413. https://doi.org/10.3390/molecules23061413
Chicago/Turabian StyleKoirala, Prashamsa, Su Hui Seong, Yajuan Zhou, Srijan Shrestha, Hyun Ah Jung, and Jae Sue Choi. 2018. "Structure–Activity Relationship of the Tyrosinase Inhibitors Kuwanon G, Mulberrofuran G, and Albanol B from Morus Species: A Kinetics and Molecular Docking Study" Molecules 23, no. 6: 1413. https://doi.org/10.3390/molecules23061413





