Binding of Catechins to Staphylococcal Enterotoxin A
Abstract
:1. Introduction
2. Results
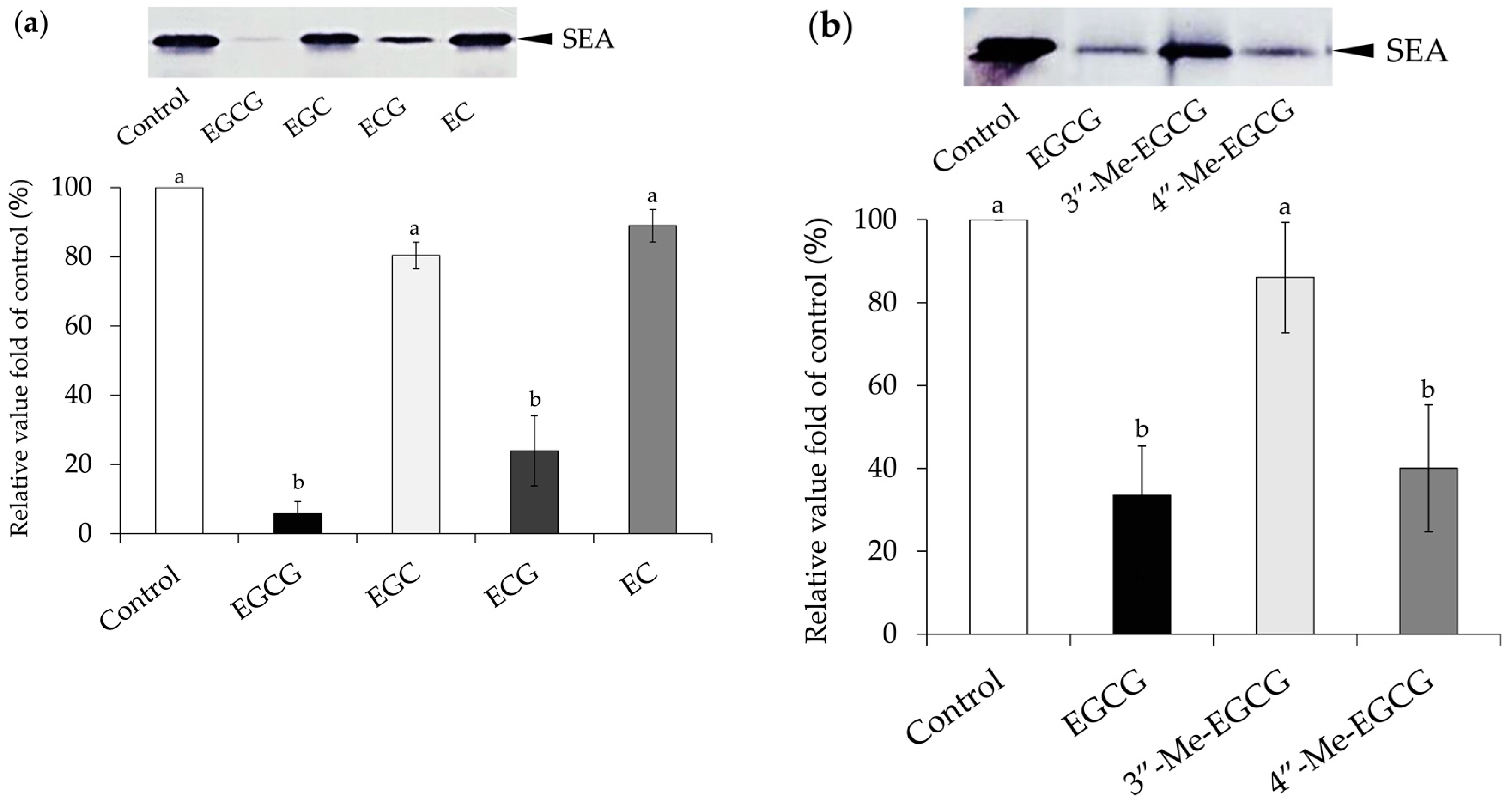
2.1. Interaction between Catechins and SEA
2.2. Interaction between Catechins and SEA Toxin Active Sites
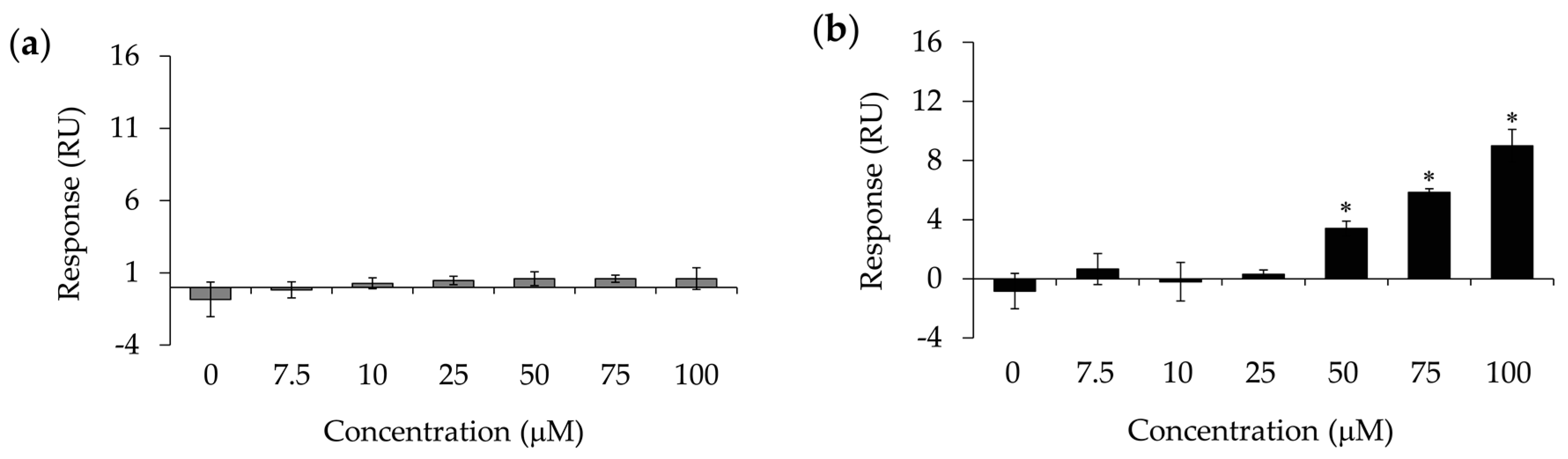
2.3. Surface Plasmon Resonance (SPR) Sensor Measurements
2.4. Fourier Transform Infrared Spectroscopy (FT-IR) Measurements
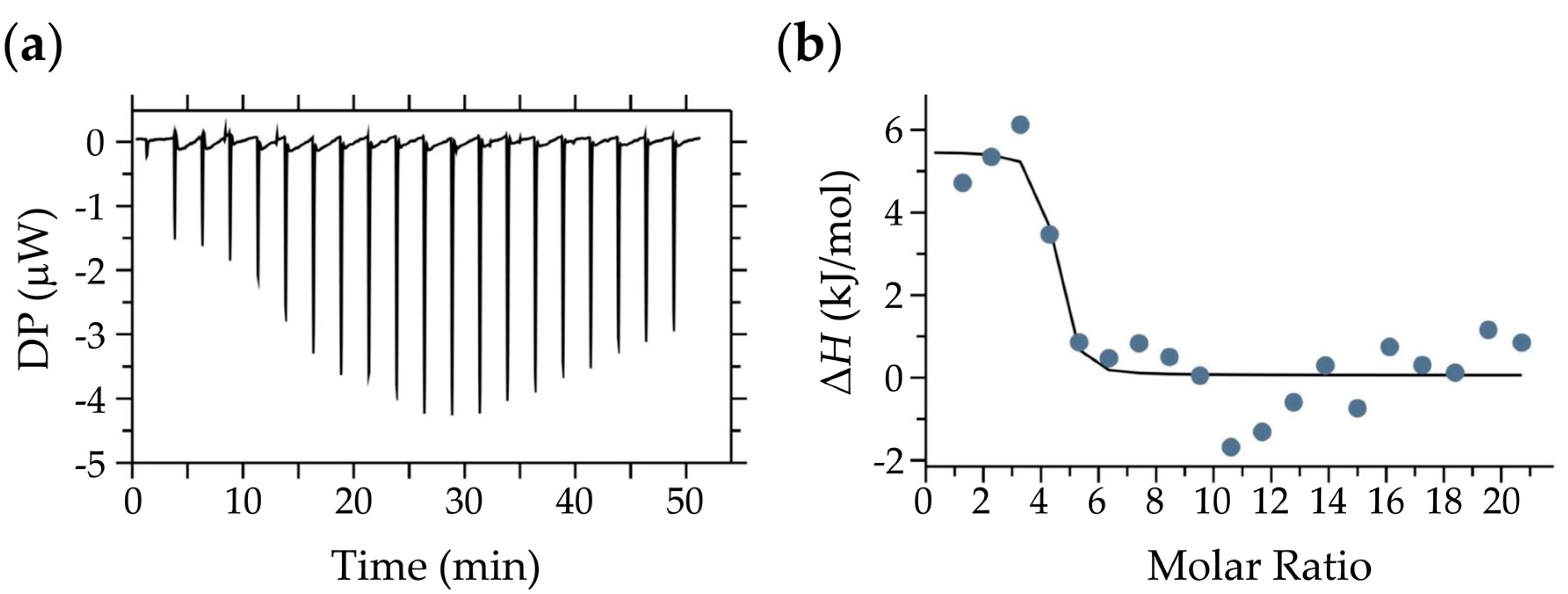
2.5. Isothermal Titration Calorimetry (ITC) Measurements
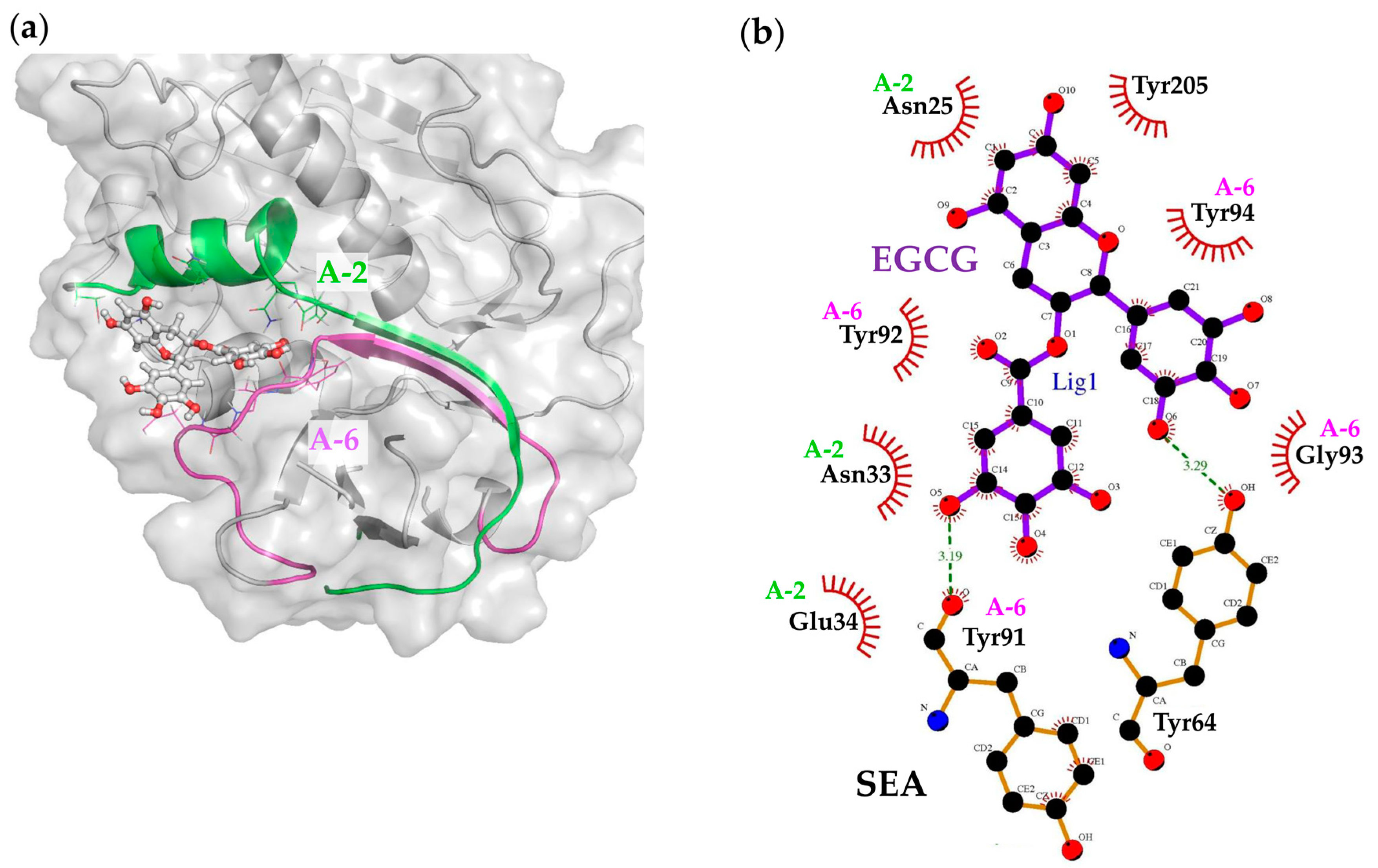
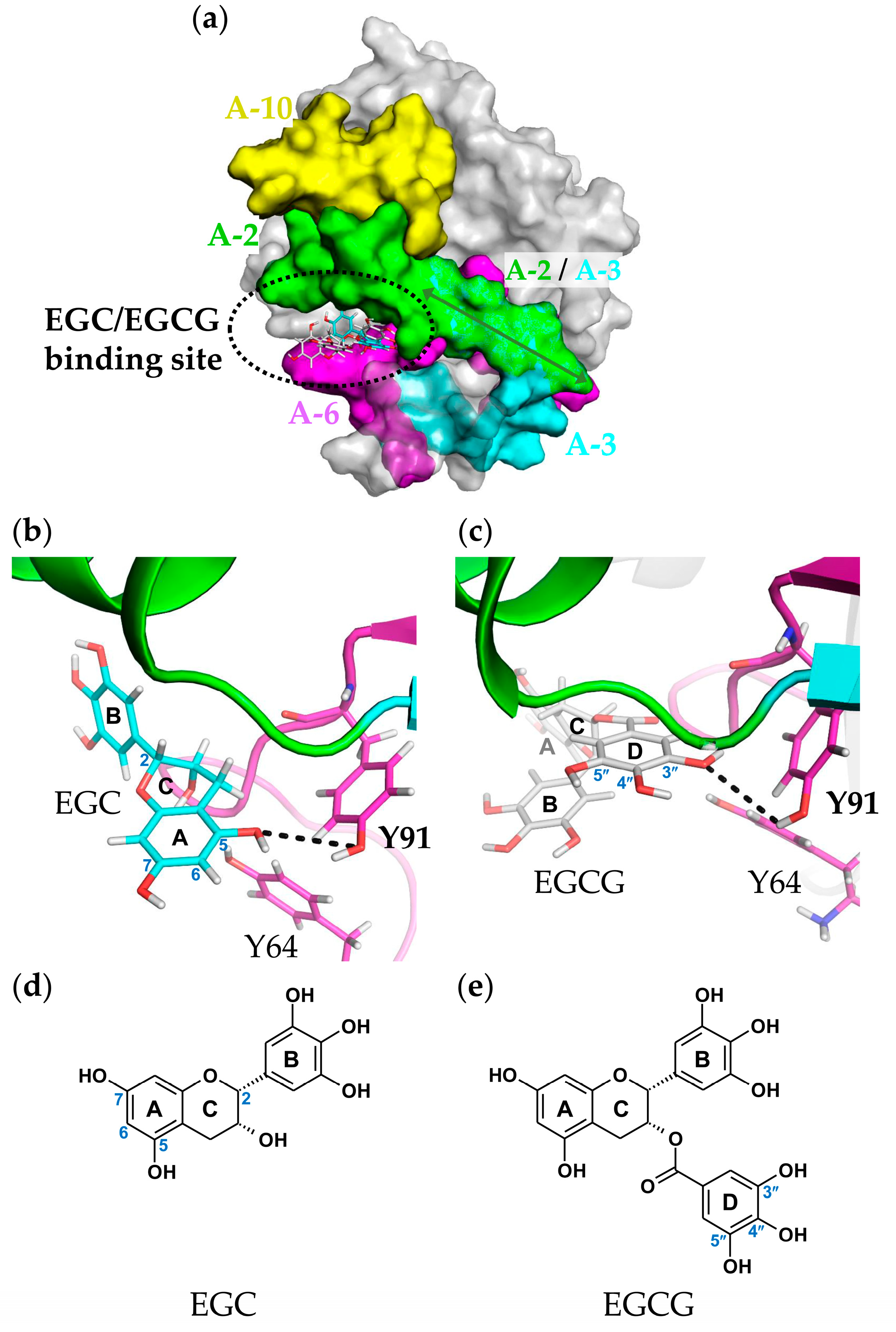
2.6. Molecular Docking and Binding Site Analysis
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Interaction between Catechins and SEA
4.3. Interaction between Catechins and SEA Toxin Active Sites
4.4. Surface Plasmon Resonance (SPR) Sensor Measurements
4.5. FT-IR Measurement
4.6. Isothermal Titration Calorimetry (ITC) Measurements
4.7. Molecular Docking and Binding Site Analysis
4.8. Statistical Analysis
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Argudín, M.Á.; Mendoza, M.C.; Rodicio, M.R. Food poisoning and Staphylococcus aureus enterotoxins. Toxins 2010, 2, 1751–1773. [Google Scholar] [CrossRef] [PubMed]
- Balaban, N.; Rassoly, A. Staphylococcal enterotoxin. Int. J. Food Microbiol. 2000, 61, 1–10. [Google Scholar] [CrossRef]
- Pinchuk, I.V.; Beswick, E.J.; Reyes, V.E. Staphylococcal enterotoxins. Toxins 2010, 2, 2177–2197. [Google Scholar] [CrossRef] [PubMed]
- Maina, E.K.; Hu, D.L.; Asano, K.; Nakane, A. Inhibition of emetic and superantigenic activities of staphylococcal enterotoxin A by synthetic peptides. Peptides 2012, 38, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Mulvey, M.R.; Doupe, M.; Prout, M.; Leong, C.; Hizon, R.; Grossberndt, A.; Klowak, M.; Gupta, A.; Melanson, M.; Gomori, A.; et al. Staphylococcus aureus harbouring Enterotoxin A as a possible risk factor for multiple sclerosis exacerbations. Mult. Scler. 2011, 17, 397–403. [Google Scholar] [CrossRef] [PubMed]
- Schutyser, E.; Struyf, S.; Wuyts, A.; Put, W.; Geboes, K.; Grillet, B.; Opdenakker, G.; Van Damme, J. Selective induction of CCL18/PARC by staphylococcal enterotoxins in mononuclear cells and enhanced levels in septic and rheumatoid arthritis. Eur. J. Immunol. 2001, 31, 3755–3762. [Google Scholar] [CrossRef]
- Tomi, N.S.; Kränke, B.; Aberer, E. Staphylococcal toxins in patients with psoriasis, atopic dermatitis, and erythroderma, and in healthy control subjects. J. Am. Acad. Dermatol. 2005, 53, 67–72. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Gevaert, P.; Van Zele, T.; Perez-Novo, C.; Patou, J.; Holtappels, G.; van Cauwenberge, P.; Bachert, C. An update on the impact of Staphylococcus aureus enterotoxins in chronic sinusitis with nasal polyposis. Rhinology 2005, 43, 162–168. [Google Scholar] [PubMed]
- Zaveri, N.T. Green tea and its polyphenolic catechins: Medicinal uses in cancer and noncancer applications. Life Sci. 2006, 78, 2073–2080. [Google Scholar] [CrossRef] [PubMed]
- Andrushchenko, V.V.; Aarabi, M.H.; Nguyen, L.T.; Prenner, E.J.; Vogel, H.J. Thermodynamics of the interactions of tryptophan-rich cathelicidin antimicrobial peptides with model and natural membranes. Biochim. Biophys. Acta 2008, 1778, 1004–1014. [Google Scholar] [CrossRef] [PubMed]
- Lopez, M.; Edens, L. Effective prevention of chill-haze in beer using an acid proline-specific endoprotease from Aspergillus niger. J. Agric. Food Chem. 2005, 53, 7944–7949. [Google Scholar] [CrossRef] [PubMed]
- Kumazawa, S.; Kajiya, K.; Naito, A.; Saito, H.; Tuzi, S.; Tanio, M.; Suzuki, M.; Nanjo, F.; Suzuki, E.; Nakayama, T. Direct evidence of interaction of a green tea polyphenol, epigallocatechin gallate, with lipid bilayers by solid-state nuclear magnetic resonance. Biosci. Biotechnol. Biochem. 2004, 68, 1743–1747. [Google Scholar] [CrossRef] [PubMed]
- Ishii, T.; Minoda, K.; Bae, M.J.; Mori, T.; Uekusa, Y.; Ichikawa, T.; Aihara, Y.; Furuta, T.; Wakimoto, T.; Kan, T.; et al. Binding affinity of tea catechins for HSA: Characterization by high-performance affinity chromatography with immobilized albumin column. Mol. Nutr. Food Res. 2010, 54, 816–822. [Google Scholar] [CrossRef] [PubMed]
- Minoda, K.; Ichikawa, T.; Katsumata, T.; Onobori, K.I.; Mori, T.; Suzuki, Y.; Ishii, T.; Nakayama, T. Influence of the galloyl moiety in tea catechins on binding affinity for human serum albumin. J. Nutr. Sci. Vitaminol. 2010, 56, 331–334. [Google Scholar] [CrossRef] [PubMed]
- Choi, O.; Yahiro, K.; Morinaga, N.; Miyazaki, M.; Noda, M. Inhibitory effects of various plant polyphenols on the toxicity of staphylococcal alpha-toxin. Microb. Pathog. 2007, 42, 215–224. [Google Scholar] [CrossRef] [PubMed]
- Rasooly, R.; Do, P.M.; Friedman, M. Inhibition of biological activity of staphylococcal enterotoxin A (SEA) by apple juice and apple polyphenols. J. Agric. Food Chem. 2010, 58, 5421–5426. [Google Scholar] [CrossRef] [PubMed]
- Friedman, M.; Rasooly, R.; Do, P.M.; Henika, P.R. The olive compound hydroxytyrosol inactivates Staphylococcus aureus bacteria and Staphylococcal enterotoxin A (SEA). J. Food Sci. 2011, 76, 558–563. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, Y.; Hirai, C.; Sugiyama, Y.; Shibata, M.; Ozaki, J.; Murata, M.; Ohashi, N.; Masuda, S. Inhibitory effects of food additives derived from polyphenols on staphylococcal enterotoxin A production and biofilm formation by Staphylococcus aureus. Biosci. Biotechnol. Biochem. 2017, 81, 2346–2352. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, Y.; Hirai, C.; Sugiyama, Y.; Utsumi, M.; Yanagida, A.; Murata, M.; Ohashi, N.; Masuda, S. Interaction between various apple procyanidin and staphylococcal enterotoxin A and their inhibitory effects on toxin activity. Toxins 2017, 9, 243. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, Y.; Aoki, N.; Sugiyama, Y.; Tanaka, T.; Murata, M.; Masuda, S. Plant-derived polyphenols interact with staphylococcal enterotoxin A and inhibit toxin activity. PLoS ONE 2016, 11, e0157082. [Google Scholar] [CrossRef] [PubMed]
- Shimamura, Y.; Aoki, N.; Sugiyama, Y.; Nakayama, T.; Masuda, S. Screening of tea extract and theaflavins for inhibitory effects on the biological activity and production of staphylococcal enterotoxin A. J. Food Sci. 2014, 79, 2294–2300. [Google Scholar] [CrossRef] [PubMed]
- Benedik, E.; Skrt, M.; Podlipnik, Č.; Ulrih, N.P. Binding of flavonoids to staphylococcal enterotoxin B. Food Chem. Toxicol. 2014, 74, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Yuksel, Z.; Avci, E.; Erdem, Y.K. Characterization of binding interactions between green tea flavanoids and milk proteins. Food Chem. 2010, 121, 450–456. [Google Scholar] [CrossRef]
- Caturla, N.; Vera-Samper, E.; Villalaín, J.; Mateo, C.R.; Micol, V. The relationship between the antioxidant and the antibacterial properties of galloylated catechins and the structure of phospholipid model membranes. Free Radic. Biol. Med. 2003, 34, 648–662. [Google Scholar] [CrossRef]
- Kajiya, K.; Kumazawa, S.; Nakayama, T. Steric effects on the interaction of tea catechins with lipid bilayers. Biosci. Biotechnol. Biochem. 2001, 65, 2638–2643. [Google Scholar] [CrossRef] [PubMed]
- Kajiya, K.; Kumazawa, S.; Nakayama, T. Effects of external factors on the interaction of tea catechins with lipid bilayers. Biosci. Biotechnol. Biochem. 2002, 66, 2330–2335. [Google Scholar] [CrossRef] [PubMed]
- Stapleton, P.D.; Shah, S.; Hara, Y.; Taylor, P.W. Potentiation of catechin gallate-mediated sensitization of Staphylococcus aureus to oxacillin by nongalloylated catechins. Antimicrob. Agents Chemother. 2006, 50, 752–755. [Google Scholar] [CrossRef] [PubMed]
- Palacios, L.; Rosado, H.; Micol, V.; Rosato, A.; Bernal, P.; Arroyo, R.; Grounds, H.; Anderson, J.C.; Stabler, R.A.; Taylor, P.W. Staphylococcal phenotypes induced by naturally occurring and synthetic membrane-interactive galloyl catechin β-lactam-resistance modifiers. PLoS ONE 2014, 9, e93830. [Google Scholar] [CrossRef] [PubMed]
- Hashimoto, T.; Kumazawa, S.; Nanjo, F.; Hara, Y.; Nakayama, T. Interaction of tea catechins with lipid bilayers investigated with liposome systems. Biosci. Biotechnol. Biochem. 1999, 63, 2252–2255. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulou, A.; Green, R.J.; Frazier, R.A. Interaction of flavonoids with bovine serum albumin: A fluorescence quenching study. J. Agric. Food Chem. 2005, 53, 158–163. [Google Scholar] [CrossRef] [PubMed]
- Maiti, T.K.; Ghosh, K.S.; Dasgupta, S. Interaction of (−)-epigallocatechin-3-gallate with human serum albumin: Fluorescence, fourier transform infrared, circular dichroism, and docking studies. Proteins 2006, 64, 355–362. [Google Scholar] [CrossRef] [PubMed]
- Hou, Z.; Sang, S.; You, H.; Lee, M.J.; Hong, J.; Chin, K.V.; Yang, C.S. Mechanism of action of (−)-epigallocatechin-3-gallate: Auto-oxidation–dependent inactivation of epidermal growth factor receptor and direct effects on growth inhibition in human esophageal cancer KYSE 150 cells. Cancer Res. 2005, 65, 8049–8056. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Dashwood, W.M.; Löhr, C.V.; Fischer, K.A.; Pereira, C.B.; Louderback, M.; Nakagama, H.; Bailey, G.S.; Williams, D.E.; Dashwood, R.H. Protective versus promotional effects of white tea and caffeine on PhIP-induced tumorigenesis and β-catenin expression in the rat. Carcinogenesis 2008, 29, 834–839. [Google Scholar] [CrossRef] [PubMed]
- Tachibana, H.; Koga, K.; Fujimura, Y.; Yamada, K. A receptor for green tea polyphenol EGCG. Nat. Struct. Mol. Biol. 2004, 11, 380–381. [Google Scholar] [CrossRef] [PubMed]
- Jakobek, L. Interactions of polyphenols with carbohydrates, lipids and proteins. Food Chem. 2015, 175, 556–567. [Google Scholar] [CrossRef] [PubMed]
- Jelesarov, I.; Bosshard, H.R. Isothermal titration calorimetry and differential scanning calorimetry as complementary tools to investigate the energetics of biomolecular recognition. J. Mol. Recognit. 1999, 12, 3–18. [Google Scholar] [CrossRef]
- Frazier, R.A.; Papadopoulou, A.; Green, R.J. Isothermal titration calorimetry study of epicatechin binding to serum albumin. J. Pharm. Biomed. Anal. 2006, 41, 1602–1605. [Google Scholar] [CrossRef] [PubMed]
- Rödström, K.E.; Regenthal, P.; Bahl, C.; Ford, A.; Baker, D.; Lindkvist-Petersson, K. Two common structural motifs for TCR recognition by staphylococcal enterotoxins. Sci. Rep. 2016, 6, 25796. [Google Scholar] [CrossRef]
- Aihara, Y.; Yoshida, A.; Furuta, T.; Wakimoto, T.; Akizawa, T.; Konishi, M.; Kan, T. Regioselective synthesis of methylated epigallocatechin gallate via nitrobenzenesulfonyl (Ns) protecting group. Bioorg. Med. Chem. Lett. 2009, 19, 4171–4174. [Google Scholar] [CrossRef] [PubMed]
- Grosdidier, A.; Zoete, V.; Michielin, O. SwissDock, a protein-small molecule docking web service based on EADock DSS. Nucl. Acids Res. 2011, 39 (Suppl. 2), W270–W277. [Google Scholar] [CrossRef] [PubMed]
- Irwin, J.J.; Sterling, T.; Mysinger, M.M.; Bolstad, E.S.; Coleman, R.G. ZINC: A free tool to discover chemistry for biology. J. Chem. Inf. Model. 2012, 52, 1757–1768. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Not available. |


| Sample | ΔH (J/mol) | ΔS (J/mol/deg) | ΔG (kJ/mol) | −TΔS (kJ/mol) | |
|---|---|---|---|---|---|
| Cell | Syringe | ||||
| SEA | EC | −61.9 | −162.2 | −11.7 | 50.3 |
| EGCG | 5.41 | 140.9 | −38.3 | −43.7 | |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shimamura, Y.; Utsumi, M.; Hirai, C.; Nakano, S.; Ito, S.; Tsuji, A.; Ishii, T.; Hosoya, T.; Kan, T.; Ohashi, N.; et al. Binding of Catechins to Staphylococcal Enterotoxin A. Molecules 2018, 23, 1125. https://doi.org/10.3390/molecules23051125
Shimamura Y, Utsumi M, Hirai C, Nakano S, Ito S, Tsuji A, Ishii T, Hosoya T, Kan T, Ohashi N, et al. Binding of Catechins to Staphylococcal Enterotoxin A. Molecules. 2018; 23(5):1125. https://doi.org/10.3390/molecules23051125
Chicago/Turabian StyleShimamura, Yuko, Mio Utsumi, Chikako Hirai, Shogo Nakano, Sohei Ito, Ai Tsuji, Takeshi Ishii, Takahiro Hosoya, Toshiyuki Kan, Norio Ohashi, and et al. 2018. "Binding of Catechins to Staphylococcal Enterotoxin A" Molecules 23, no. 5: 1125. https://doi.org/10.3390/molecules23051125





