Occurrence of Functional Molecules in the Flowers of Tea (Camellia sinensis) Plants: Evidence for a Second Resource
Abstract
:1. Introduction
2. Functional Molecules Similar to Those in Tea Leaves
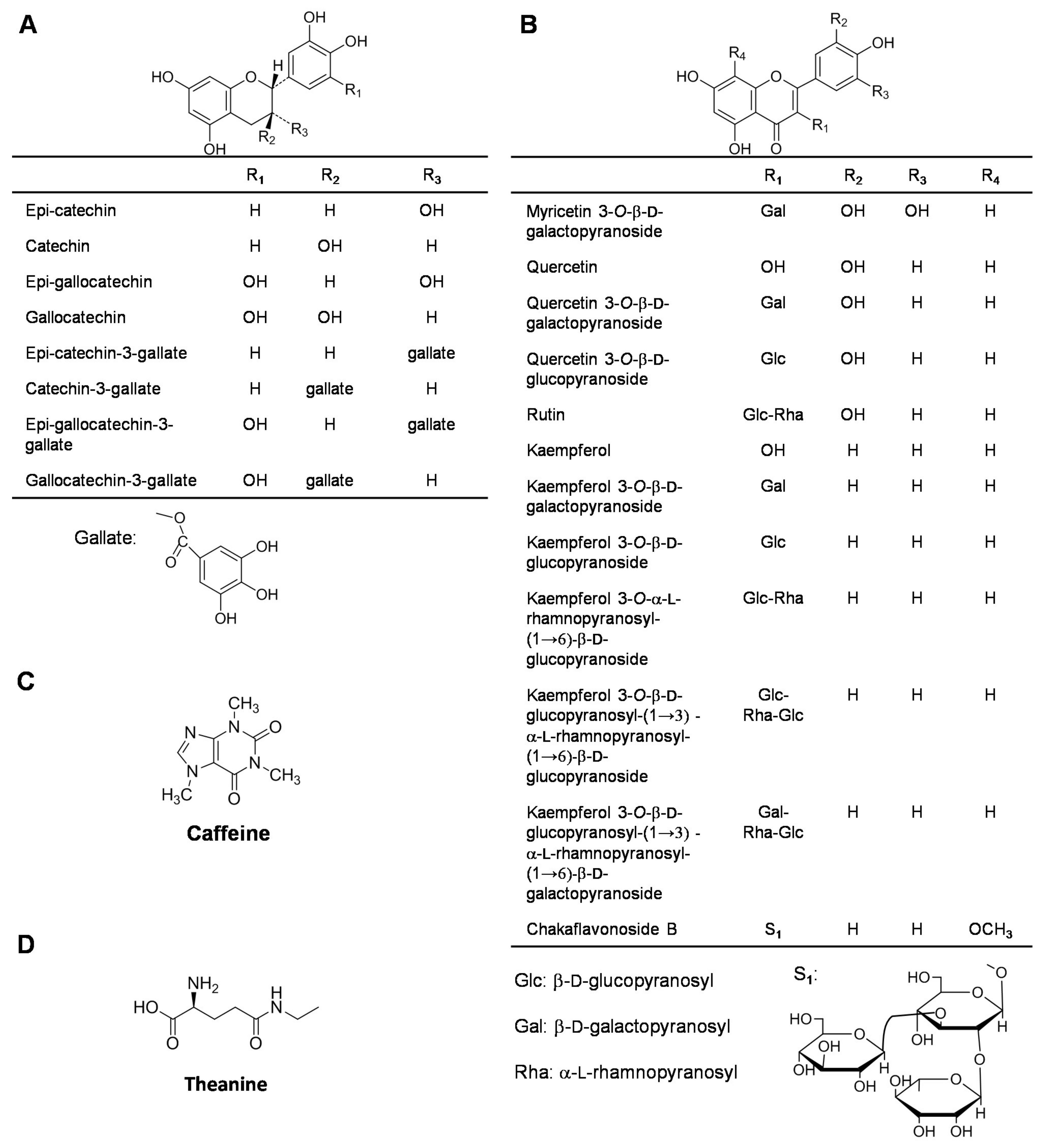
2.1. Catechins
2.2. Flavonols
2.3. Caffeine
2.4. Amino Acids
3. Preponderant Functional Molecules in Tea Flowers
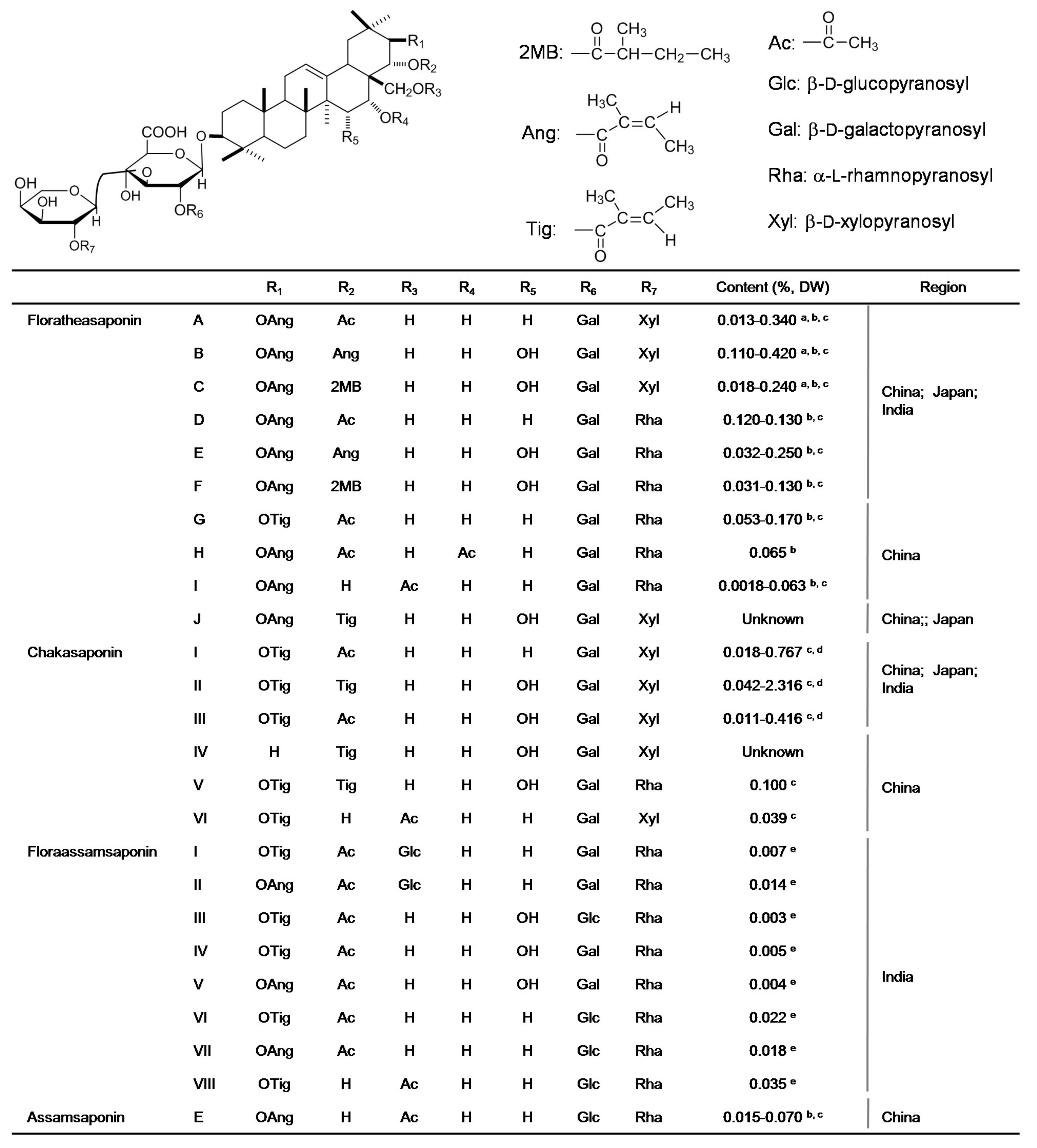
3.1. Saponins
3.2. Polysaccharides
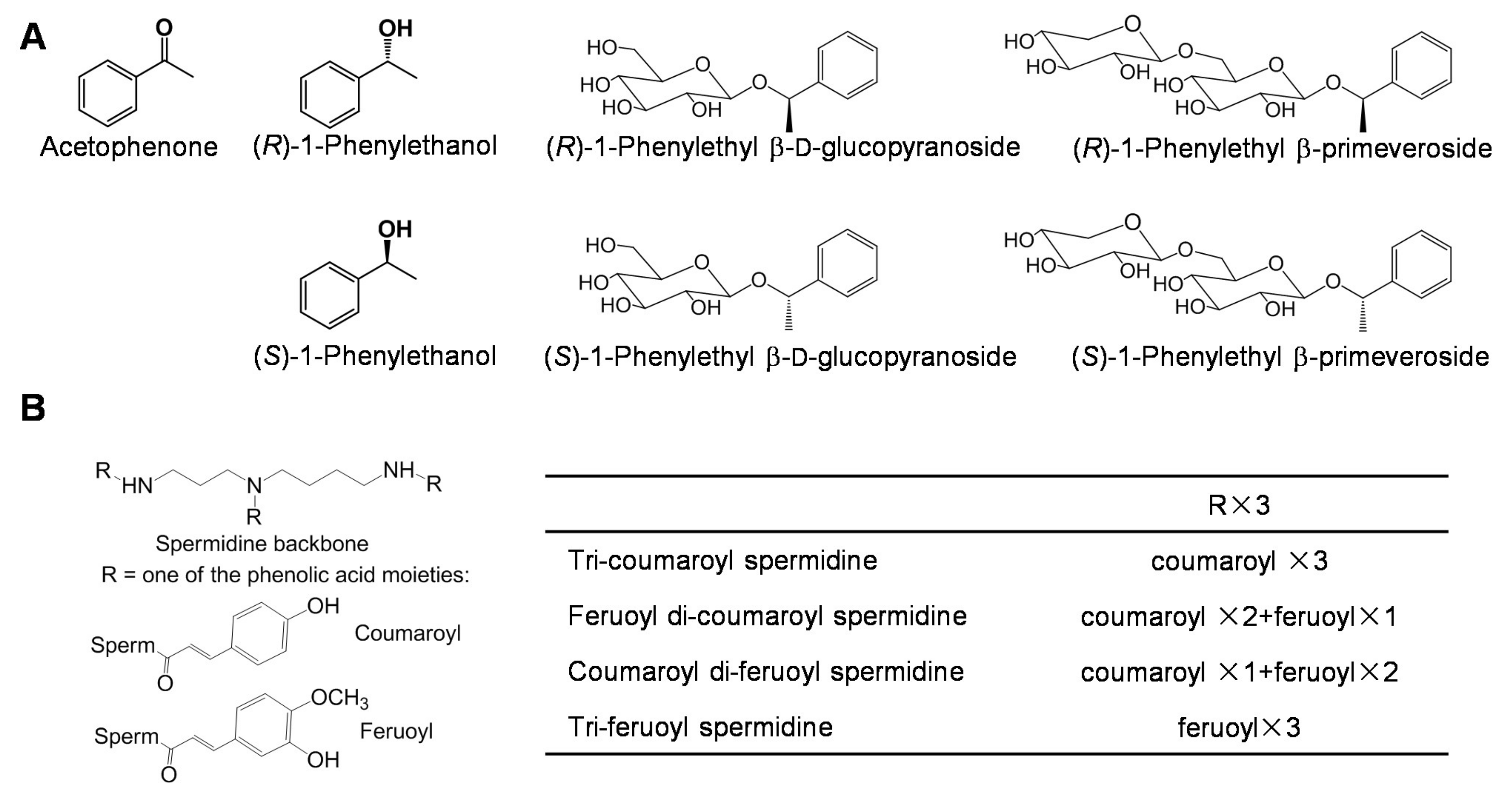
3.3. Aromatic Compounds
3.4. Spermidine Derivatives
3.5. Functional Proteins
4. Safety and Diverse Biological Functions
4.1. Evaluation of Mutagenicity, and Acute and Subchronic Toxicity in Rats
4.2. Biological Functions
4.2.1. Catechin- and Polysaccharide-Derived Antioxidant Abilities
4.2.2. Catechin- and Polysaccharide-Derived Antitumor Abilities
4.2.3. Polysaccharide- and Catechin-Derived Immunostimulating Abilities
4.2.4. Polysaccharide-Derived Antidiabetic Abilities
4.2.5. Saponin-Derived Antiobesity Abilities
4.2.6. Saponin-Derived Antihyperlipidemic and Antihyperglycemic Abilities
4.2.7. Saponin-Derived Antiallergic Abilities
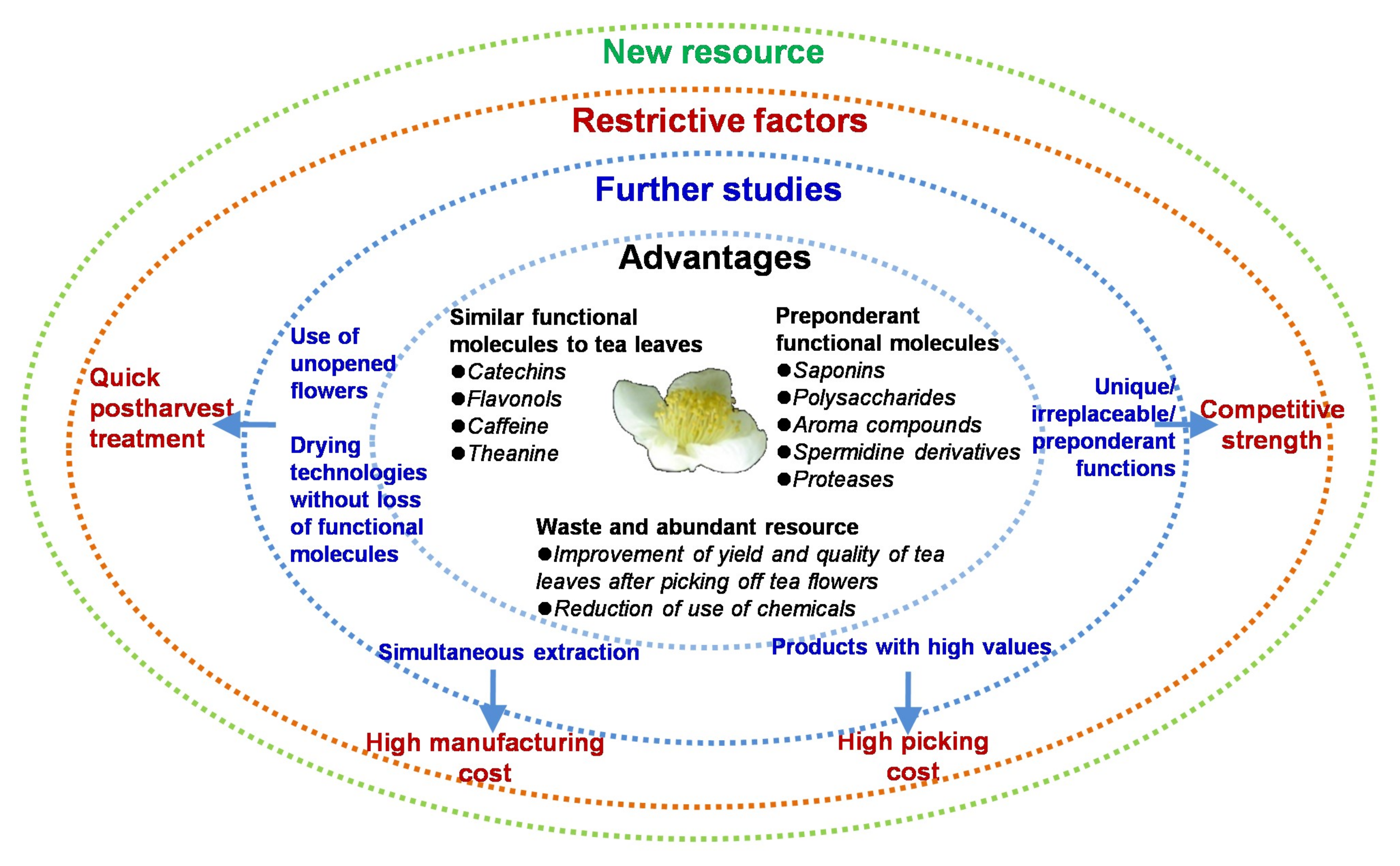
5. Concluding Remarks and Perspectives
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| DPPH | 2,2-Diphenyl-1-picrylhydrazyl |
| EGC | Epigallocatechin |
| EGCG | Epigallocatechin gallate |
| SOD | Superoxide dismutase |
| TFPS | Polysaccharides of tea flowers |
| TLPS | Polysaccharides of tea leaves |
References
- Bushman, J.L. Green tea and cancer in humans: A review of the literature. Nutr. Cancer 1998, 31, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Trevisanato, S.I. Tea and health. Nutr. Rev. 2000, 58, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.C. Tea Biochemistry, 3rd ed.; China Agriculture Press: Beijing, China, 2003; pp. 8–67. (In Chinese) [Google Scholar]
- Lin, Y.S.; Wu, S.S.; Lin, J.K. Determination of tea polyphenols and caffeine in tea flowers (Camellia sinensis) and their hydroxyl radical scavenging and nitric oxide suppressing effects. J. Agric. Food Chem. 2003, 51, 975–978. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, C.; Artacho, R.; Giménez, R. Beneficial effects of green tea—A review. J. Am. Coll. Nutr. 2006, 25, 79–99. [Google Scholar] [CrossRef] [PubMed]
- Wan, X.C.; Xia, T. Secondary Metabolism of Tea Plant, 1st ed.; Science Press: Beijing, China, 2015; pp. 39–64. (In Chinese) [Google Scholar]
- Yang, Z.Y.; Xu, Y.; Jie, G.L.; He, P.M.; Tu, Y.Y. Study on the antioxidant activity of tea flowers (Camellia sinensis). Asia Pac. J. Clin. Nutr. 2007, 16, 148–152. [Google Scholar] [PubMed]
- Yang, Z.Y.; Tu, Y.Y.; Baldermann, S.; Dong, F.; Xu, Y.; Watanabe, N. Isolation and identification of compounds from the ethanolic extract of flowers of the tea (Camellia sinensis) plant and their contribution to the antioxidant capacity. LWT Food Sci. Technol. 2009, 42, 1439–1443. [Google Scholar] [CrossRef]
- Morikawa, T.; Ninomiya, K.; Miyake, S.; Miki, Y.; Okamoto, M.; Yoshikawa, M.; Muraoka, O. Flavonol glycosides with lipid accumulation inhibitory activity and simultaneous quantitative analysis of 15 polyphenols and caffeine in the flower buds of Camellia sinensis from different regions by LCMS. Food Chem. 2013, 140, 353–360. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Liu, Y.; Li, W.; Zhao, L.; Meng, F.; Wang, Y.; Tan, H.; Yang, H.; Wei, C.; Wan, X.; Gao, L.; Xia, T. Tissue-specific, development-dependent phenolic compounds accumulation profile and gene expression pattern in tea plant (Camellia sinensis). PLoS ONE 2013, 8, e62315. [Google Scholar] [CrossRef] [PubMed]
- Forrest, G.I.; Bendall, D.S. The distribution of polyphenols in the tea plant (Camellia sinensis L.). Biochem. J. 1969, 113, 741–755. [Google Scholar] [CrossRef]
- Joshi, R.; Poonam; Gulati, A. Biochemical attributes of tea flowers (Camellia sinensis) at different developmental stages in the Kangra region of India. Sci. Hortic. 2011, 130, 266–274. [Google Scholar] [CrossRef]
- Wang, H.; Provan, G.J.; Helliwell, K. Tea favonoids: Their functions, utilisation and analysis. Trends Food Sci. Tech. 2000, 11, 152–160. [Google Scholar] [CrossRef]
- Balentine, D.A.; Wiseman, S.A.; Bouwens, L.C.M. The chemistry of tea favonoids. Crit. Rev. Food Sci. 1997, 37, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Morikawa, T.; Yamamoto, K.; Kato, Y.; Nagatomo, A.; Matsuda, H. Floratheasaponins A–C, acylated oleanane-type triterpene oligoglycosides with anti-hyperlipidemic activities from flowers of the tea plant (Camellia sinensis). J. Nat. Prod. 2005, 68, 1360–1365. [Google Scholar] [CrossRef] [PubMed]
- Nagata, T.; Sakai, S. Differences in caffeine, flavanols and amino acids contents in leaves of cultivated species of Camellia. Jpn. J. Breed. 1984, 34, 459–467. [Google Scholar] [CrossRef]
- Ashihara, H. Metabolism of alkaloids in coffee plants. Braz. J. Plant Physiol. 2006, 18, 1–8. [Google Scholar] [CrossRef]
- Fujimori, N.; Ashihara, H. Biosynthesis of caffeine in flower buds of Camellia sinensis. Ann. Bot. 1993, 71, 279–284. [Google Scholar] [CrossRef]
- Chou, T.M.; Benowitz, N.L. Caffeine and coffee: Effects on health and cardiovascular disease. Comp. Biochem. Physiol. C Pharmacol. Toxicol. Endocrinol. 1994, 109, 173–189. [Google Scholar] [CrossRef]
- Nurminen, M.L.; Niittynen, L.; Korpela, R.; Vapaatalo, H. Coffee, caffeine and blood pressure: A critical review. Eur. J. Clin. Nutr. 1999, 53, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Kato, M.; Mizuno, K.; Crozier, A.; Fujimura, T.; Ashihara, H. Plant biotechnology: Caffeine synthase gene from tea leaves. Nature 2000, 406, 956–957. [Google Scholar] [CrossRef] [PubMed]
- Mukai, T.; Horie, H.; Goto, T. Differences in free amino acids and total nitrogen contents among various prices of green tea. Tea Res. J. 1992, 76, 45–50. [Google Scholar] [CrossRef]
- Hung, Y.T.; Chen, P.C.; Chen, R.L.C.; Cheng, T.J. Sequential determination of tannin and total amino acid contents in tea for taste assessment by a fluorescent flow-injection analytical system. Food Chem. 2010, 118, 876–881. [Google Scholar] [CrossRef]
- Juneja, L.R.; Chu, D.C.; Okubo, T.; Nagato, Y.; Yokogoshi, H. L-theanine-a unique amino acid of green tea and its relaxation effect in humans. Trends Food Sci. Technol. 1999, 10, 199–204. [Google Scholar] [CrossRef]
- Wang, L.; Xu, R.; Hu, B.; Li, W.; Sun, Y.; Tu, Y.; Zeng, X. Analysis of free amino acids in Chinese teas and flower of tea plant by high performance liquid chromatography combined with solid-phase extraction. Food Chem. 2010, 123, 1259–1266. [Google Scholar] [CrossRef]
- Dong, F.; Yang, Z.Y.; Baldermann, S.; Kajitani, Y.; Ota, S.; Kasuga, H.; Imazeki, Y.; Ohnishi, T.; Watanabe, N. Characterization of l-phenylalanine metabolism to acetophenone and 1-phenylethanol in the flowers of Camellia sinensis using stable isotope labeling. J. Plant Physiol. 2012, 169, 217–225. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Nakamura, S.; Kato, Y.; Matsuhira, K.; Matsuda, H. Medicinal flowers. XIV. New acylated oleanane-type triterpene oligoglycosides with antiallergic activity from flower buds of Chinese tea plant (Camellia sinensis). Chem. Pharm. Bull. 2007, 55, 598–605. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Sugimoto, S.; Nakamura, S.; Matsuda, H. Medicinal flowers. XXII. Structures of chakasaponins V and VI, chakanoside I, and chakaflavonoside A from flower buds of Chinese tea plant (Camellia sinensis). Chem. Pharm. Bull. 2008, 56, 1297–1303. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Sugimoto, S.; Kato, Y.; Nakamura, S.; Wang, T.; Yamashita, C.; Matsuda, H. Acylated oleanane-type triterpene saponins with acceleration of gastrointestinal transit and inhibitory effect on pancreatic lipase from flower buds of Chinese tea plant (Camellia sinensis). Chem. Biodivers. 2009, 6, 903–915. [Google Scholar] [CrossRef] [PubMed]
- Sugimoto, S.; Yoshikawa, M.; Nakamura, S.; Matsuda, H. Medicinal flowers. XXV. Structures of floratheasaponin J and chakanoside II from Japanese tea flower, flower buds of Camellia sinensis. Heterocycles 2009, 78, 1023–1029. [Google Scholar]
- Hamao, M.; Matsuda, H.; Nakamura, S.; Nakashima, S.; Semura, S.; Maekubo, S.; Wakasugi, S.; Yoshikawa, M. Anti-obesity effects of the methanolic extract and chakasaponins from the flower buds of Camellia sinensis in mice. Bioorg. Med. Chem. 2011, 19, 6033–6041. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Hamao, M.; Nakamura, S.; Kon’I, H.; Murata, M.; Yoshikawa, M. Medicinal flowers. XXXIII. Anti-hyperlipidemic and anti-hyperglycemic effects of chakasaponins I-III and structure of chakasaponin IV from flower buds of Chinese tea plant (Camellia sinensis). Chem. Pharm. Bull. 2012, 60, 674–680. [Google Scholar] [CrossRef] [PubMed]
- Matsuda, H.; Nakamura, S.; Morikawa, T.; Muraoka, O.; Yoshikawa, M. New biofunctional effects of the flower buds of Camellia sinensis and its bioactive acylated oleanane-type triterpene oligoglycosides. J. Nat. Med. 2016, 70, 689–701. [Google Scholar] [CrossRef] [PubMed]
- Morikawa, T.; Miyake, S.; Miki, Y.; Ninomiya, K.; Yoshikawa, M.; Muraoka, O. Quantitative analysis of acylated oleananetype triterpene saponins, chakasaponins I–III and floratheasaponins A–F, in the flower buds of Camellia sinensis from different regional origins. J. Nat. Med. 2012, 66, 608–613. [Google Scholar] [CrossRef] [PubMed]
- Ohta, T.; Nakamura, S.; Nakashima, S.; Matsumoto, T.; Ogawa, K.; Fujimoto, K.; Fukaya, M.; Yoshikawa, M.; Matsuda, H. Acylated oleanane-type triterpene oligoglycosides from the flower buds of Camellia sinensis var. assamica. Tetrahedron 2015, 71, 846–851. [Google Scholar] [CrossRef]
- Zhen, Y. Tea-Bioactivity and Therapeutic Potential; Taylor and Francis: New York, NY, USA, 2002; p. 58. [Google Scholar]
- Yoshikawa, M.; Wang, T.; Sugimoto, S.; Nakamura, S.; Nagatomo, A.; Matsuda, H.; Harima, S. Functional saponins in tea flower (flower buds of Camellia sinensis): gastroprotective and hypoglycemic effects of floratheasaponins and qualitative and quantitative analysis using HPLC. Yakugaku Zasshi J. Pharm. Soc. Jpn. 2008, 128, 141–151. [Google Scholar] [CrossRef]
- Weng, W. Study on the Main Bioactive Compounds of Tea (Camellia sinensis) Flower and Its Application Perspectives. Master’s thesis, Zhejiang University, Hangzhou, China, 2004. [Google Scholar]
- Nie, S.P.; Xie, M.Y. A review on the isolation and structure of tea polysaccharides and their bioactivities. Food Hydrocoll. 2011, 25, 144–149. [Google Scholar] [CrossRef]
- Jiang, H.; Xiao, J.B. A review on the structure-function relationship aspect of polysaccharides from tea materials. Crit. Rev. Food Sci. 2015, 55, 930–938. [Google Scholar]
- Wei, X.L.; Chen, M.A.; Xiao, J.B.; Liu, Y.; Yu, L.; Zhang, H.; Wang, Y.F. Composition and bioactivity of tea flower polysaccharides obtained by different methods. Carbohydr. Polym. 2010, 79, 418–422. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, Z.; Wei, X. Sugar compositions, α-glucosidase inhibitory and amylase inhibitory activities of polysaccharides from leaves and flowers of Camellia sinensis obtained by different extraction methods. Int. J. Biol. Macromol. 2010, 47, 534–539. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, L.; Wei, X. Monosaccharide composition and bioactivity of tea flower polysaccharides obtained by ethanol fractional precipitation and stepwise precipitation. CyTA J. Food 2012, 10, 1–4. [Google Scholar] [CrossRef]
- Wang, Y.; Mao, F.; Wei, X. Characterization and antioxidant activities of polysaccharides from leaves, flowers and seeds of green tea. Carbohydr. Polym. 2012, 88, 146–153. [Google Scholar] [CrossRef]
- Xu, R.; Ye, H.; Sun, Y.; Tu, Y.; Zeng, X. Preparation, preliminary characterization, antioxidant, hepatoprotective and antitumor activities of polysaccharides from the flower of tea plant (Camellia sinensis). Food Chem. Toxicol. 2012, 50, 2473–2480. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yu, L.; Zhang, J.; Xiao, J.; Wei, X. Study on the purification and characterization of a polysaccharide conjugate from tea flowers. Int. J. Biol. Macromol. 2010, 47, 266–270. [Google Scholar] [CrossRef] [PubMed]
- Han, Q.; Yu, Q.Y.; Shi, J.; Xiong, C.Y.; Ling, Z.J.; He, P.M. Structural characterization and antioxidant activities of 2 water-soluble polysaccharide fractions purified from tea (Camellia sinensis) flower. J. Food Sci. 2011, 76, C462–C471. [Google Scholar]
- Han, Q.; Yu, Q.Y.; Shi, J.; Xiong, C.Y.; Ling, Z.J.; He, P.M. Molecular characterization and hypoglycemic activity of a novel water-soluble polysaccharide from tea (Camellia sinensis) flower. Carbohydr. Polym. 2011, 86, 797–805. [Google Scholar] [CrossRef]
- Yang, Z.Y.; Baldermann, S.; Watanabe, N. Recent studies of the volatile compounds in tea. Food Res. Int. 2013, 53, 585–599. [Google Scholar] [CrossRef]
- Winterhalter, P.; Skouroumounis, G.K. Glycoconjugated aroma compounds: Occurrence, role and biotechnological transformation. In Advances in Biochemical Engineering/Biotechnology; Berger, R.G., Ed.; Springer: Heidelberger, Berlin, Germany, 1997; pp. 73–105. [Google Scholar]
- Joshi, R.; Poonam; Saini, R.; Guleria, S.; Babu, G.D.; Kumari, M.; Gulati, A. Characterization of volatile components of tea flowers (Camellia sinensis) growing in Kangra by GC/MS. Nat. Prod. Commun. 2011, 6, 1155–1158. [Google Scholar] [PubMed]
- Zhou, Y.; Zhang, L.; Gui, J.D.; Dong, F.; Cheng, S.; Mei, X.; Zhang, L.Y.; Li, Y.Q.; Su, X.G.; Baldermann, S.; et al. Molecular cloning and characterization of a short chain dehydrogenase showing activity with volatile compounds isolated from Camellia sinensis. Plant Mol. Biol. Rep. 2015, 33, 253–263. [Google Scholar] [CrossRef]
- Dong, F.; Zhou, Y.; Zeng, L.T.; Peng, Q.Y.; Chen, Y.Y.; Zhang, L.; Su, X.G.; Watanabe, N.; Yang, Z.Y. Elucidation of differential accumulation of 1-phenylethanol in flowers and leaves of tea (Camellia sinensis) plants. Molecules 2016, 21, 1106. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Dong, F.; Kunimasa, A.; Zhang, Y.; Cheng, S.; Lu, J.; Zhang, L.; Murata, A.; Mayer, F.; Fleischmann, P.; et al. Occurrence of glycosidically conjugated 1-phenylethanol and its hydrolase β-primeverosidase in tea (Camellia sinensis) flowers. J. Agric. Food Chem. 2014, 62, 8042–8050. [Google Scholar] [CrossRef] [PubMed]
- Suan, C.L.; Sarmidi, M.R. Immobilised lipase-catalysed resolution of (R,S)-1-phenylethanol in recirculated packed bed reactor. J. Mol. Catal. B Enzym. 2004, 28, 111–119. [Google Scholar]
- Yang, Z.Y.; Dong, F.; Baldermann, S.; Murata, A.; Tu, Y.Y.; Asai, T.; Watanabe, N. Isolation and identification of spermidine derivatives in flowers of tea (Camellia sinensis) plants and their distributions in floral organs. J. Sci. Food Agric. 2012, 92, 2128–2132. [Google Scholar] [CrossRef] [PubMed]
- Facchini, P.J.; Hagel, J.; Zulak, K.G. Hydroxycinnamic acid amide metabolism: physiology and biochemistry. Can. J. Bot. 2002, 80, 577–589. [Google Scholar] [CrossRef]
- Ma, C.; Nakamura, N.; Hattori, M. Inhibitory effects on HIV-1 protease of tri-p-coumaroylspermidine from Artemisia caruifolia and related amides. Chem. Pharm. Bull. 2001, 49, 915–917. [Google Scholar] [CrossRef] [PubMed]
- Lu, D.; Tong, Q. Studies on extraction and enzymatic activity determination of superoxide dismutase from tea leaves. J. Fujian Tea 1991, 1, 21–24. (In Chinese) [Google Scholar]
- Chen, Z.; Song, W. Study on tea superoxide dismutase. J. Southwest Agric. Univ. 1989, 11, 302–306. (In Chinese) [Google Scholar]
- Leary, D.; Vierros, M.; Hamon, G.; Arico, S.; Monagle, C. Marine genetic resources: A review of scientific and commercial interest. Mar. Policy 2009, 33, 183–194. [Google Scholar] [CrossRef]
- González-Rábade, N.; Badillo-Corona, J.A.; Aranda-Barradas, J.S.; Oliver-Salvador, M.; del Carmen Oliver-Salvador, M. Production of plant proteases in vivo and in vitro—A review. Biotechnol. Adv. 2011, 29, 983–996. [Google Scholar] [CrossRef] [PubMed]
- Sun, S. Effects of Enzyme Treatment on the Quality of Green Tea Extract. Master’s Thesis, Zhejiang University, Hangzhou, China, 2006. (In Chinese). [Google Scholar]
- Guo, H.W. Study of Improving the Flavor of Tea Beverages by Enzymolysis Technology. Master’s Thesis, Zhejiang University, Hangzhou, China, 2013. (In Chinese). [Google Scholar]
- Xu, Y.Q.; Zhong, X.Y.; Chen, S.Q.; Yin, J.F. Hydrolysis of green tea residue protein using proteolytic enzyme derived from Aspergillus oryzae. J. Food Sci. Technol. 2013, 50, 171–175. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.Y.; Fu, X.M.; Mei, X.; Zhou, Y.; Du, B.; Tu, Y.Y.; Yang, Z.Y. Characterization of functional proteases from flowers of tea (Camellia sinensis) plants. J. Funct. Foods 2016, 25, 149–159. [Google Scholar] [CrossRef]
- Dini, I.; Schettino, O.; Simioli, T.; Dini, A. Studies on the constituents of Chenopodium quinoa seeds: isolation and characterization of new triterpene saponins. J. Agric. Food Chem. 2001, 49, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Chow, S.H.H.; Cai, Y.; Hakim, I.A.; Crowell, J.A.; Shahi, F.; Brooks, C.A.; Dorr, R.T.; Hara, Y.; Alberts, D.S. Pharmacokinetics and safety of green tea polyphenols after multiple-dose administration of epigallocatechin gallate and polyphenon E in healthy individuals. Clin. Cancer Res. 2003, 9, 3312–3319. [Google Scholar] [PubMed]
- Knight, C.A.; Knight, I.; Mitchell, D.C.; Zepp, Z.E. Beverage caffeine intake in US consumers and subpopulations of interest: Estimates from the Share of Intake Panel survey. Food Chem. Toxicol. 2004, 42, 1923–1930. [Google Scholar] [CrossRef] [PubMed]
- Sparg, S.G.; Light, M.E.; Staden, J.V. Biological activities and distribution of plant saponins. J. Ethnopharmacol. 2004, 94, 219–243. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Jin, Y.; Xua, Y.; Wu, Y.; Xu, J.; Tu, Y. Safety evaluation of tea (Camellia sinensis (L.) O. Kuntze) flower extract: Assessment of mutagenicity, and acute and subchronic toxicity in rats. J. Ethnopharmacol. 2011, 133, 583–590. [Google Scholar] [CrossRef] [PubMed]
- WHO (World Health Organisation). Principles for the safety assessment of food additives and contaminants in food. In IPCS Environmental Health Criteria; World Health Organisation: Geneva, Switzerland, 1987; pp. 39–59. [Google Scholar]
- Han, Q.; Xiong, C.; Shi, J.; Gao, Y.; Chen, Y.; Ling, Z.; He, P. Isolation, chemical characterization and antioxidant activities of a water-soluble polysaccharide fraction of tea (Camellia sinensis) flower. J. Food Biochem. 2012, 36, 46–55. [Google Scholar] [CrossRef]
- Way, T.D.; Lin, H.Y.; Hua, K.T.; Lee, J.C.; Li, W.H.; Lee, M.R.; Shuang, C.H.; Lin, J.K. Beneficial effects of different tea flowers against human breast cancer MCF-7 cells. Food Chem. 2009, 114, 1231–1236. [Google Scholar] [CrossRef]
- Han, Q.; Ling, Z.J.; He, P.M.; Xiong, C.Y. Immunomodulatory and antitumor activity of polysaccharide isolated from tea plant flower. Prog. Biochem. Biophys. 2010, 37, 646–653. [Google Scholar] [CrossRef]
- Cao, H. Polysaccharides from Chinese tea: Recent advance on bioactivity and function. Int. J. Biol. Macromol. 2013, 62, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Chen, G.; Yuan, Q.; Saeeduddin, M.; Qu, S.; Zeng, X.; Ye, H. Recent advances in tea polysaccharides: Extraction, purification, physicochemical characterization and bioactivities. Carbohydr. Polym. 2016, 153, 663–678. [Google Scholar] [CrossRef] [PubMed]
- Nakamura, Y.; Kawase, I.; Harada, S.; Matsuda, M.; Honma, T.; Tomita, I. Antitumor-promoting effects of tea aqueous nondialysates in mouse epidermal JB6 cells. In Food Factors for Cancer Prevention; Ohigashi, H., Osawa, T., Terao, J., Watanabe, S., Yoshikawa, T., Eds.; Springer: Tokyo, Japan, 1997; pp. 138–141. [Google Scholar]
- Cai, X.; Wang, Y.F.; Mao, F.F.; Yu, L.; Liu, C.X.; Zhu, Q.; Zhang, H.; Wei, X.L. Hypoglycemic and hyperglycemia-prevention effects of crude tea flower polysaccharide. Modern Food Sci. Technol. 2011, 27, 262–266. [Google Scholar]
- Matsuda, H.; Nakamura, S.; Yoshikawa, M. Degranulation inhibitors from medicinal plants in antigen-stimulated rat basophilic leukemia (RBL-2H3) cells. Chem. Pharm. Bull. 2016, 64, 96–103. [Google Scholar] [CrossRef] [PubMed]
| Catechins | Tea Leaves a | Tea Flowers b |
|---|---|---|
| EGCG | 6.81–8.58 | 0.27–1.97 |
| ECG | 1.11–2.07 | 0.21–0.85 |
| EGC | 1.15–1.58 | 0.03–1.96 |
| EC | 0.50–0.69 | 0.04–0.41 |
| C | 0.02–0.11 | 0.00–0.31 |
| MW/kDa | Monosaccharide Composition (mol % or Mole Ratios) | References | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Rhamnose | Arabinose | Galactose | Glucose | Xylose | Mannose | Galacturonic Acid | Glucuronic Acid | Fucose | ||
| 1.1–483.0 | 1.7 | 5.7 | 10.0 | 4.8 | 0.8 | 1.1 | 1.2 | 0.6 | [42] | |
| 1.1–508.0 | 3.4 | 8.7 | 10.4 | 4.2 | 0.8 | 1.0 | 9.2 | 0.5 | ||
| 1.2–465.0 | 10.4 | 22.5 | 24.9 | 7.8 | 1.9 | 1.6 | 16.4 | 0.9 | ||
| Unknown | 5.0 | 11.6 | 11.9 | 4.3 | 1.3 | 2.0 | 8.4 | 1.0 | [44] | |
| Unknown | 14.8 | 18.1 | 45.4 | 12.2 | 6.9 | 2.6 | [45] | |||
| Unknown | 11.2 | 55.2 | 33.7 | |||||||
| Unknown | 21.0 | 53.3 | 25.7 | |||||||
| 2.6–146 | 0.4 | 1.0 | 1.0 | 0.4 | 0.1 | 0.2 | 0.7 | 0.1 | [43] | |
| 500 | 1.0 | 2.9 | 3.3 | 1.3 | 0.5 | [46] | ||||
| 167.5 | 0.8 | 1.0 | 1.0 | 1.2 | [47,48] | |||||
| 10.1 | 2.3 | 2.3 | 1.0 | 0.8 | ||||||
| 31.0 | Unknown | [41] | ||||||||
| 4.4 | Unknown | |||||||||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, Y.; Zhou, Y.; Zeng, L.; Dong, F.; Tu, Y.; Yang, Z. Occurrence of Functional Molecules in the Flowers of Tea (Camellia sinensis) Plants: Evidence for a Second Resource. Molecules 2018, 23, 790. https://doi.org/10.3390/molecules23040790
Chen Y, Zhou Y, Zeng L, Dong F, Tu Y, Yang Z. Occurrence of Functional Molecules in the Flowers of Tea (Camellia sinensis) Plants: Evidence for a Second Resource. Molecules. 2018; 23(4):790. https://doi.org/10.3390/molecules23040790
Chicago/Turabian StyleChen, Yiyong, Ying Zhou, Lanting Zeng, Fang Dong, Youying Tu, and Ziyin Yang. 2018. "Occurrence of Functional Molecules in the Flowers of Tea (Camellia sinensis) Plants: Evidence for a Second Resource" Molecules 23, no. 4: 790. https://doi.org/10.3390/molecules23040790
APA StyleChen, Y., Zhou, Y., Zeng, L., Dong, F., Tu, Y., & Yang, Z. (2018). Occurrence of Functional Molecules in the Flowers of Tea (Camellia sinensis) Plants: Evidence for a Second Resource. Molecules, 23(4), 790. https://doi.org/10.3390/molecules23040790






