Antitumour, Antimicrobial, Antioxidant and Antiacetylcholinesterase Effect of Ganoderma Lucidum Terpenoids and Polysaccharides: A Review
Abstract
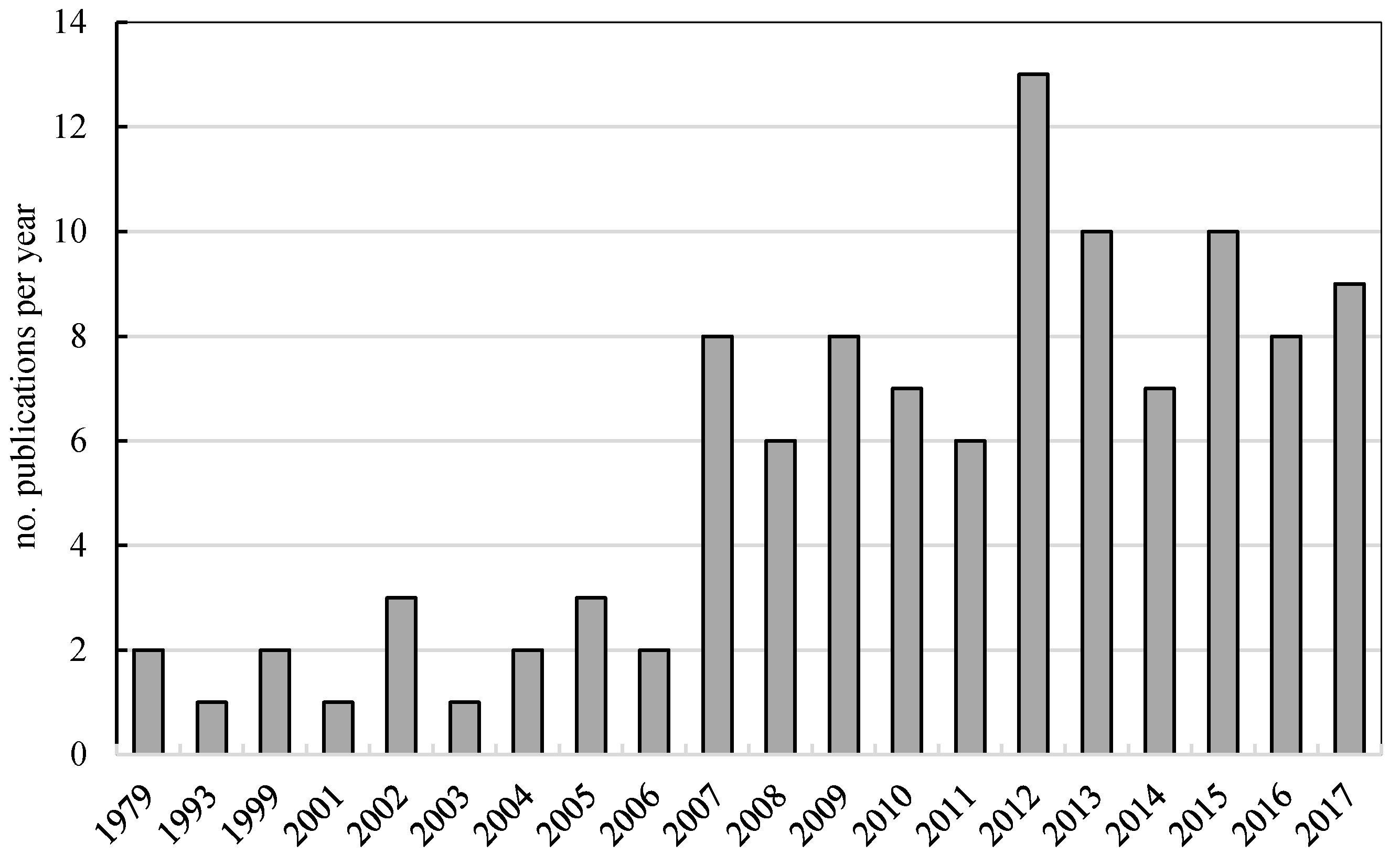
:1. Introduction
2. Composition of G. lucidum
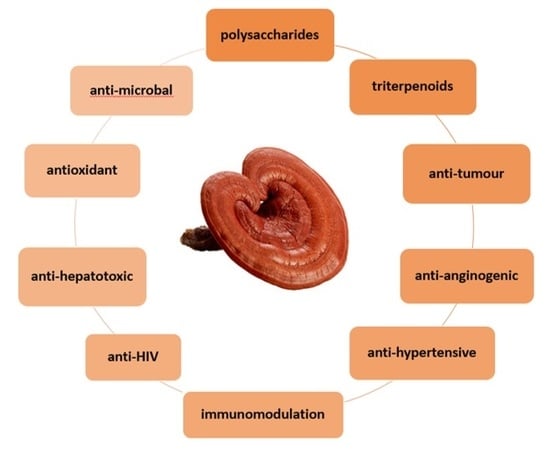
Bioactive Molecules Found in G. lucidum
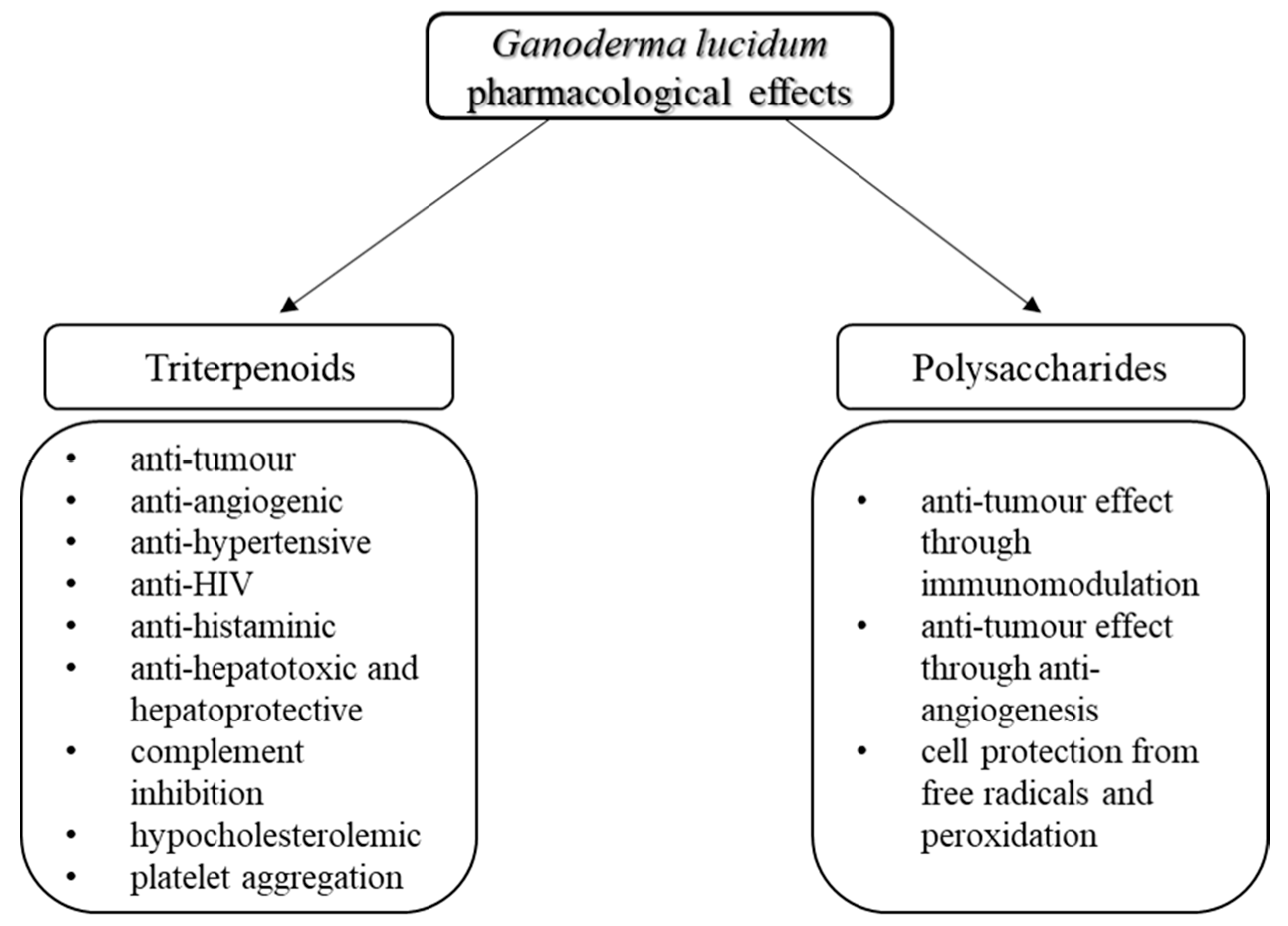
3. Bioactivity of G. lucidum Triterpens
3.1. Cytotoxic and Antitumour Activity
3.2. Anti-Oxidative Effect
3.3. Anti-HIV Activity
3.4. Neuro-Protective Effects
4. Bioactivity of G. lucidum Polysaccharides
4.1. Cytotoxic and Antitumour Activity
4.2. Anti-Oxidative Effect
4.3. Neurological Effects
4.4. Antimicrobial Effects
5. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| 95D | Human lung cancer cell line |
| ABTS | 2,2′-azino-bis(3-ethylbenzothiazoline-6-sulphonic acid |
| AC | Acetylcholine |
| AChE | Acetylcholinesterase |
| AD | Alzheimer’s disease |
| Aha1 | Activator of HsP90 ATPase |
| AIDS | Acquired immunodeficiency syndrome |
| AP | Activator protein |
| Bax | Bcl-2-associated X protein |
| Bcl-2 | B-cell lymphoma |
| BHT | Butylated hydroxytoluene |
| Capase-8 | Capase protein |
| CCL-2 | Carnival cruise lines |
| CD | Cluster of differentiation protein |
| CDC20 | Cell cycle regulatory protein |
| CDK | Cyclin-dependent kinase |
| c-Fos | Protein code by Fos gene |
| C-GLP | Carboxymethylated polysaccharide |
| c-Jun | Protein code by Jun gene |
| c-MMC | Myelocytomatosis viral oncogene homolog |
| CPA | Cyclophosphamide |
| Cy | Cyclosporine |
| Cy775 | Adenocarcinoma |
| CyclinD1 | Protein by the CCND1 gene |
| DCs | Dendric cells |
| DPPH | 2,2-diphenyl-1-picrylhydrazyl |
| EPS | Exopolysaccharide |
| ERK | Extracellular regulated kinase |
| GA | Ganoderic acid |
| GLBR | Germinated brown rice |
| GLIS | Ganoderma lucidum immunomodulation substance |
| GLPG | Ganoderma lucidum proteoglycan |
| GL-PP | Ganoderma lucidum polysaccharide peptide |
| GLPS | Ganoderma lucidum polysaccharides |
| GTM | Ganoderma water soluble polysaccharides |
| HCT-116 | Human colon cancer cell line |
| HCT-26 | Human colon cancer cell line |
| HeLa | Human cervical tumour cell line |
| Hep-G2 | Human liver tumour cell line |
| HL 60 | Human promyelocytic leukaemia cell line |
| HLF | Hepatic leukaemia factor |
| HSV | Herpes simplex virus |
| HTC-116/8 | Human colon tumour cell lime |
| HuH7 | Human hepatocarcinoma cell line |
| IFN | Interferon |
| IL | Interleukin |
| iNOS | Inducible nitrine oxide synthase |
| JNK | Jun nuclear kinase |
| LLC | Human lung cancer cell line |
| LnCaP | Human prostate cancer cell lines |
| MCP-1 | Monocyte chemoattractant protein 1 |
| MDA-MB-231 | Human breast cancer cell line |
| MMP | Matrix metalloproteinase |
| NF-κb | Nuclear factor kappa beta |
| NFκB-α | Nuclear factor kappa beta – alpha |
| P388 | Human leukaemia cell line |
| P53 | Protein 53 |
| PC-3 | Human prostate cancer cell lines |
| PCNA | Proliferating cell nuclear agent |
| PGY | Water-soluble glycopeptide |
| PRDX3 | Peroxiredoxin 3 |
| RAW 264.7 | Leukaemic monocyte macrophage cell line |
| S-180 | Human sarcoma 180 |
| SMMC-7721 | Human hepatoma cell lines |
| T-47D | Human carcinoma cell line |
| TNF | Tumour necrosis factor |
| uPa | Urokinase-type plasminogen activator |
| uPAR | Urokinase-type plasminogen activator receptor |
| VEGF | Vascular endothelial growth factor |
References
- Dinesh Babu, P.D.; Subhasree, R.S. The Sacred Mushroom “Reishi”—A Review. Am. Eurasian J. Bot. 2008, 1, 107–110. [Google Scholar]
- Siwulski, M.; Sobieralski, K.; Golak-Siwulska, I.; Sokół, S.; Sękara, A. Ganoderma lucidum (Curt.: Fr.) Karst.—Health-promoting properties. A review. Herba Pol. 2015, 61, 105–118. [Google Scholar] [CrossRef]
- Yuen, J.W.M.; Gohel, M.D.I. Anticancer effects of Ganoderma lucidum: A review of scientific evidence. Nutr. Cancer Int. J. 2005, 53, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Reishi/Ling Zhi—The Mushroom of Immortality. Available online: https://www.chinesemedicineliving.com/eastern-philosophy/reishi-ling-zhi-the-mushroom-of-immortality/ (accessed on 25 September 2017).
- Cör, D.; Botić, T.; Knez, Ž.; Batista, U.; Gregori, A.; Pohleven, F.; Bončina, T. Two-stage extraction of antitumor, antioxidant and antiacetylcholinesterase compounds from Ganoderma lucidum fruiting body. J. Supercrit. Fluids 2014, 91, 53–60. [Google Scholar] [CrossRef]
- Sanodiya, B.S.; Thakur, G.S.; Baghel, R.K.; Prasad, G.B.K.S.; Bisen, P.S. Ganoderma lucidum: A Potent Pharmacological Macrofungus. Curr. Pharm. Biotechnol. 2009, 10, 717–742. [Google Scholar] [CrossRef] [PubMed]
- Batra, P.; Sharma, A.K.; Khajuria, R. Probing Lingzhi or Reishi Medicinal Mushroom Ganoderma lucidum (Higher Basidiomycetes): A Bitter Mushroom with Amazing Health Benefits. Int. J. Med. Mushrooms 2013, 15, 127–143. [Google Scholar] [CrossRef] [PubMed]
- Kao, C.; Jesuthasan, A.C.; Bishop, K.S.; Glucina, M.P.; Ferguson, L.R. Anti-cancer activities of Ganoderma lucidum: Active ingredients and pathways. Funct. Foods Health Dis. 2013, 3, 48–65. [Google Scholar]
- Zhang, H.-N.; He, J.-H.; Yuan, L.; Lin, Z.-B. In vitro and in vivo protective effect of Ganoderma lucidum polysaccharides on alloxan-induced pancreatic islets damage. Life Sci. 2003, 73, 2307–2319. [Google Scholar] [CrossRef]
- Karwa, A.S.; Rai, M.K. Naturally Occurring Medicinal Mushroom-Derived Antimicrobials: A Case-Study Using Lingzhi or Reishi Ganoderma lucidum (W. Curt.:Fr.) P. Karst. (Higher Basidiomycetes). Int. J. Med. Mushrooms 2012, 14, 481–490. [Google Scholar] [CrossRef] [PubMed]
- Li, R.K.; Vasil’ev, A.V.; Orekhov, A.N.; Tertov, V.V.; Tutel’ian, V.A. [Anti-atherosclerotic properties of higher mushrooms (a clinico-experimental investigation)]. Vopr. Pitan. 1989, 1, 16–19. [Google Scholar]
- Chen, W.Q.; Luo, S.H.; Li, H.Z.; Yang, H. Effects of Ganoderma lucidum polysaccharides on serum lipids and lipoperoxidation in experimental hyperlipidemic rats. J. Chin. Mater. Medica 2005, 30, 1358–1360. [Google Scholar]
- Cherian, E.; Sudheesh, N.P.; Janardhanan, K.K.; Patani, G. Free radical scavenging and mitochondrial antioxidant activities of Reishi- Ganoderma lucidum. J. Basic Clin. Physiol. Pharmacol. 2011, 20, 289–308. [Google Scholar]
- Boh, B.; Berovic, M.; Zhang, J.; Zhi-Bin, L. Ganoderma lucidum and its pharmaceutically active compounds. In Biotechnology Annual Review; El-Gewely, M.R., Ed.; Elsevier: Amsterdam, The Netherlands, 2007; Volume 13, pp. 265–301. [Google Scholar]
- Scopus Data Base. Available online: https://www.scopus.com/search/ (accessed on 8 October 2017).
- Liu, J.; Huang, W.; Lv, M.; Si, J.; Guo, B.; Li, S. Determination of ergosterol in Ganoderma lucidum from different varieties and cultured tree species by HPLC. J. Chin. Med. Mater. 2011, 34, 187–190. [Google Scholar]
- Gao, P.; Hirano, T.; Chen, Z.; Yasuhara, T.; Nakata, Y.; Sugimoto, A. Isolation and identification of C-19 fatty acids with anti-tumor activity from the spores of Ganoderma lucidum (reishi mushroom). Fitoterapia 2012, 83, 490–499. [Google Scholar] [CrossRef] [PubMed]
- Matute, R.G.; Serra, A.; Figlas, D.; Curvetto, N. Copper and zinc bioaccumulation and bioavailability of Ganoderma lucidum. J. Med. Food 2011, 14, 1273–1279. [Google Scholar] [CrossRef] [PubMed]
- Falandysz, J. Selenium in edible mushrooms. J. Environ. Sci. Health Part C Environ. Carcinog. Ecotoxicol. Rev. 2008, 26, 256–299. [Google Scholar] [CrossRef] [PubMed]
- Wachtel-Galor, S.; Yuen, J.; Buswell, J.A.; Benzie, I.F.F. Ganoderma lucidum (Lingzhi or Reishi): A Medicinal Mushroom. In Herbal Medicine: Biomolecular and Clinical Aspects; Benzie, I.F.F., Wachtel-Galor, S., Eds.; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2011. [Google Scholar]
- Frank, K.; Patel, K.; Lopez, G.; Willis, B. Ganoderma lucidum Research Analysis. Available online: https://examine.com/supplements/ganoderma-lucidum/ (accessed on 26 September 2017).
- Ganoderma lucidum: Constituents and Phytochemicals Analysis and Introduction. Available online: https://www.mdidea.com/products/new/new03603.html (accessed on 26 September 2017).
- Yang, M.; Wang, X.; Guan, S.; Xia, J.; Sun, J.; Guo, H.; Guo, D. Analysis of Triterpenoids in Ganoderma lucidum Using Liquid Chromatography Coupled with Electrospray Ionization Mass Spectrometry. J. Am. Soc. Mass Spectrom. 2007, 18, 927–939. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Wang, D. A new class of natural glycopeptides with sugar moiety-dependent antioxidant activities derived from Ganoderma lucidum fruiting bodies. J. Proteome Res. 2009, 8, 436–442. [Google Scholar] [CrossRef] [PubMed]
- Chien, C.M.; Cheng, J.-L.; Chang, W.-T.; Tien, M.-H.; Tsao, C.-M.; Chang, Y.-H.; Chang, H.-Y.; Hsieh, J.-F.; Wong, C.-H.; Chen, S.-T. Polysaccharides of Ganoderma lucidum alter cell immunophenotypic expression and enhance CD56+ NK-cell cytotoxicity in cord blood. Bioorg. Med. Chem. 2004, 12, 5603–5609. [Google Scholar] [CrossRef] [PubMed]
- Ji, Z.; Tang, Q.; Zhang, J.; Yang, Y.; Jia, W.; Pan, Y. Immunomodulation of RAW264.7 macrophages by GLIS, a proteopolysaccharide from Ganoderma lucidum. J. Ethnopharmacol. 2007, 112, 445–450. [Google Scholar] [CrossRef] [PubMed]
- Ho, Y.W.; Yeung, J.S.L.; Chiu, P.K.Y.; Tang, W.M.; Lin, Z.B.; Man, R.Y.K.; Lau, C.S. Ganoderma lucidum polysaccharide peptide reduced the production of proinflammatory cytokines in activated rheumatoid synovial fibroblast. Mol. Cell. Biochem. 2007, 301, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Bao, X.; Liu, C.; Fang, J.; Li, X. Structural and immunological studies of a major polysaccharide from spores of Ganoderma lucidum (Fr.) Karst. Carbohydr. Res. 2001, 332, 67–74. [Google Scholar] [CrossRef]
- Dong, Q.; Wang, Y.; Shi, L.; Yao, J.; Li, J.; Ma, F.; Ding, K. A novel water-soluble β-d-glucan isolated from the spores of Ganoderma lucidum. Carbohydr. Res. 2012, 353, 100–105. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.-M.; Guan, S.-H.; Liu, R.-X.; Sun, J.-H.; Liang, Y.; Yang, M.; Wang, W.; Bi, K.-S.; Guo, D.-A. HPLC determination of four triterpenoids in rat urine after oral administration of total triterpenoids from Ganoderma lucidum. J. Pharm. Biomed. Anal. 2007, 43, 1185–1190. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.-J.; Chau, C.-F.; Chen, K.-D.; Chen, D.-H.; Yen, G.-C. The anti-invasive effect of lucidenic acids isolated from a new Ganoderma lucidum strain. Mol. Nutr. Food Res. 2007, 51, 1472–1477. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, K.; Shuto, T.; Sato, M.; Onuki, K.; Mizunoe, S.; Suzuki, S.; Sato, T.; Koga, T.; Suico, M.A.; Kai, H.; et al. Lucidenic acids-rich extract from antlered form of Ganoderma lucidum enhances TNFα induction in THP-1 monocytic cells possibly via its modulation of MAP kinases p38 and JNK. Biochem. Biophys. Res. Commun. 2011, 408, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.L.; Leung, K.S.Y.; Wang, Y.T.; Lai, C.M.; Li, S.P.; Hu, L.F.; Lu, G.H.; Jiang, Z.H.; Yu, Z.L. Qualitative and quantitative analyses of nucleosides and nucleobases in Ganoderma spp. by HPLC-DAD-MS. J. Pharm. Biomed. Anal. 2007, 44, 807–811. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.; Ren, A.; Mu, D.; Zhao, M. Current progress in the study on biosynthesis and regulation of ganoderic acids. Appl. Microbiol. Biotechnol. 2010, 88, 1243–1251. [Google Scholar] [CrossRef] [PubMed]
- Akihisa, T.; Nakamura, Y.; Tagata, M.; Tokuda, H.; Yasukawa, K.; Uchiyama, E.; Suzuki, T.; Kimura, Y. Anti-Inflammatory and Anti-Tumor-Promoting Effects of Triterpene Acids and Sterols from the Fungus Ganoderma lucidum. Chem. Biodivers. 2007, 4, 224–231. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-B.; Liu, R.-M.; Zhong, J.-J. A new ganoderic acid from Ganoderma lucidum mycelia and its stability. Fitoterapia 2013, 84, 115–122. [Google Scholar] [CrossRef] [PubMed]
- El-Mekkawy, S.; Meselhy, M.R.; Nakamura, N.; Tezuka, Y.; Hattori, M.; Kakiuchi, N.; Shimotohno, K.; Kawahata, T.; Otake, T. Anti-HIV-1 and anti-HIV-1-protease substances from Ganoderma lucidum. Phytochemistry 1998, 49, 1651–1657. [Google Scholar] [CrossRef]
- Yue, Q.-X.; Song, X.-Y.; Ma, C.; Feng, L.-X.; Guan, S.-H.; Wu, W.-Y.; Yang, M.; Jiang, B.-H.; Liu, X.; Cui, Y.-J.; et al. Effects of triterpenes from Ganoderma lucidum on protein expression profile of HeLa cells. Phytomedicine 2010, 17, 606–613. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Kurashiki, K.; Shimizu, K.; Kondo, R. 5α-Reductase Inhibitory Effect of Triterpenoids Isolated from Ganoderma lucidum. Biol. Pharm. Bull. 2006, 29, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Tang, W.; Liu, J.-W.; Zhao, W.-M.; Wei, D.-Z.; Zhong, J.-J. Ganoderic acid T from Ganoderma lucidum mycelia induces mitochondria mediated apoptosis in lung cancer cells. Life Sci. 2006, 80, 205–211. [Google Scholar] [CrossRef] [PubMed]
- Li, C.-H.; Chen, P.-Y.; Chang, U.-M.; Kan, L.-S.; Fang, W.-H.; Tsai, K.-S.; Lin, S.-B. Ganoderic acid X, a lanostanoid triterpene, inhibits topoisomerases and induces apoptosis of cancer cells. Life Sci. 2005, 77, 252–265. [Google Scholar] [CrossRef] [PubMed]
- Hajjaj, H.; Macé, C.; Roberts, M.; Niederberger, P.; Fay, L.B. Effect of 26-Oxygenosterols from Ganoderma lucidum and Their Activity as Cholesterol Synthesis Inhibitors. Appl. Environ. Microbiol. 2005, 71, 3653–3658. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shimizu, K.; Konishi, F.; Kumamoto, S.; Kondo, R. The anti-androgen effect of ganoderol B isolated from the fruiting body of Ganoderma lucidum. Bioorg. Med. Chem. 2007, 15, 4966–4972. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Grieb, B.; Thyagarajan, A.; Silva, D. Ganoderic acids suppress growth and invasive behavior of breast cancer cells by modulating AP-1 and NF-κB signaling. Int. J. Mol. Med. 2008, 21, 577–584. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.-H.; Liu, J.-W.; Zhong, J.-J. Ganoderic acid T inhibits tumor invasion in vitro and in vivo through inhibition of MMP expression. Pharmacol. Rep. 2010, 62, 150–163. [Google Scholar] [CrossRef]
- Chen, N.-H.; Liu, J.-W.; Zhong, J.-J. Ganoderic Acid Me Inhibits Tumor Invasion through Down-Regulating Matrix Metalloproteinases 2/9 Gene Expression. J. Pharmacol. Sci. 2008, 108, 212–216. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shiono, J.; Shimizu, K.; Kukita, A.; Kukita, T.; Kondo, R. Ganoderic acid DM: Anti-androgenic osteoclastogenesis inhibitor. Bioorg. Med. Chem. Lett. 2009, 19, 2154–2157. [Google Scholar] [CrossRef] [PubMed]
- Johnson, B.M.; Doonan, B.P.; Radwan, F.F.; Haque, A. Ganoderic Acid DM: An Alternative Agent for the Treatment of Advanced Prostate Cancer. Open Prostate Cancer J. 2010, 3, 78–85. [Google Scholar] [CrossRef]
- Hsu, C.-L.; Yu, Y.-S.; Yen, G.-C. Lucidenic acid B induces apoptosis in human leukemia cells via a mitochondria-mediated pathway. J. Agric. Food Chem. 2008, 56, 3973–3980. [Google Scholar] [CrossRef] [PubMed]
- Gao, J.J.; Hirakawa, A.; Min, B.S.; Nakamura, N.; Hattori, M. In vivo antitumor effects of bitter principles from the antlered form of fruiting bodies of Ganoderma lucidum. J. Nat. Med. 2006, 60, 42–48. [Google Scholar] [CrossRef]
- Xu, K.; Liang, X.; Gao, F.; Zhong, J.; Liu, J. Antimetastatic effect of ganoderic acid T in vitro through inhibition of cancer cell invasion. Process Biochem. 2010, 45, 1261–1267. [Google Scholar] [CrossRef]
- Jiang, Z.; Jin, T.; Gao, F.; Liu, J.; Zhong, J.; Zhao, H. Effects of Ganoderic acid Me on inhibiting multidrug resistance and inducing apoptosis in multidrug resistant colon cancer cells. Process Biochem. 2011, 46, 1307–1314. [Google Scholar] [CrossRef]
- Zhou, L.; Shi, P.; Chen, N.-H.; Zhong, J.-J. Ganoderic acid Me induces apoptosis through mitochondria dysfunctions in human colon carcinoma cells. Process Biochem. 2011, 46, 219–225. [Google Scholar] [CrossRef]
- Li, F.; Wang, Y.; Wang, X.; Li, J.; Cui, H.; Niu, M. Ganoderic acids suppress growth and angiogenesis by modulating the NF-κB signaling pathway in breast cancer cells. Int. J. Clin. Pharmacol. Ther. 2012, 50, 712–721. [Google Scholar] [CrossRef] [PubMed]
- Yue, Q.-X.; Cao, Z.-W.; Guan, S.-H.; Liu, X.-H.; Tao, L.; Wu, W.-Y.; Li, Y.-X.; Yang, P.-Y.; Liu, X.; Guo, D.-A. Proteomics Characterization of the Cytotoxicity Mechanism of Ganoderic Acid D and Computer-automated Estimation of the Possible Drug Target Network. Mol. Cell. Proteom. 2008, 7, 949–961. [Google Scholar] [CrossRef] [PubMed]
- Wu, T.-S.; Shi, L.-S.; Kuo, S.-C. Cytotoxicity of Ganoderma lucidum Triterpenes. J. Nat. Prod. 2001, 64, 1121–1122. [Google Scholar] [CrossRef] [PubMed]
- Weng, C.-J.; Chau, C.-F.; Hsieh, Y.-S.; Yang, S.-F.; Yen, G.-C. Lucidenic acid inhibits PMA-induced invasion of human hepatoma cells through inactivating MAPK/ERK signal transduction pathway and reducing binding activities of NF-κB and AP-1. Carcinogenesis 2008, 29, 147–156. [Google Scholar] [CrossRef] [PubMed]
- Min, B.-S.; Gao, J.-J.; Nakamura, N.; Hattori, M. Triterpenes from the Spores of Ganoderma lucidum and Their Cytotoxicity against Meth-A and LLC Tumor Cells. Chem. Pharm. Bull. (Tokyo) 2000, 48, 1026–1033. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Jedinak, A.; Sliva, D. Ganodermanontriol (GDNT) exerts its effect on growth and invasiveness of breast cancer cells through the down-regulation of CDC20 and uPA. Biochem. Biophys. Res. Commun. 2011, 415, 325–329. [Google Scholar] [CrossRef] [PubMed]
- Bishop, K.S.; Kao, C.H.J.; Xu, Y.; Glucina, M.P.; Paterson, R.R.M.; Ferguson, L.R. From 2000 years of Ganoderma lucidum to recent developments in nutraceuticals. Phytochemistry 2015, 114, 56–65. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ajith, T.A.; Sudheesh, N.P.; Roshny, D.; Abishek, G.; Janardhanan, K.K. Effect of Ganoderma lucidum on the activities of mitochondrial dehydrogenases and complex I and II of electron transport chain in the brain of aged rats. Exp. Gerontol. 2009, 44, 219–223. [Google Scholar] [CrossRef] [PubMed]
- Smina, T.P.; De, S.; Devasagayam, T.P.A.; Adhikari, S.; Janardhanan, K.K. Ganoderma lucidum total triterpenes prevent radiation-induced DNA damage and apoptosis in splenic lymphocytes in vitro. Mutat. Res. 2011, 726, 188–194. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Chang, Q.; Wong, L.K.; Chong, F.S.; Li, R.C. Triterpene antioxidants from Ganoderma lucidum. Phytother. Res. 1999, 13, 529–531. [Google Scholar] [CrossRef]
- Hasnat, M.A.; Pervin, M.; Lim, B.O. Acetylcholinesterase Inhibition and In Vitro and In Vivo Antioxidant Activities of Ganoderma lucidum Grown on Germinated Brown Rice. Molecules 2013, 18, 6663–6678. [Google Scholar] [CrossRef] [PubMed]
- Paydary, K.; Khaghani, P.; Emamzadeh-Fard, S.; Alinaghi, S.A.S.; Baesi, K. The emergence of drug resistant HIV variants and novel anti-retroviral therapy. Asian Pac. J. Trop. Biomed. 2013, 3, 515–522. [Google Scholar] [CrossRef]
- Min, B.S.; Nakamura, N.; Miyashiro, H.; Bae, K.W.; Hattori, M. Triterpenes from the spores of Ganoderma lucidum and their inhibitory activity against HIV-1 protease. Chem. Pharm. Bull. 1998, 46, 1607–1612. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.-Q.; Ip, F.C.F.; Zhang, D.-M.; Chen, L.-X.; Zhang, W.; Li, Y.-L.; Ip, N.Y.; Ye, W.-C. Triterpenoids with neurotrophic activity from Ganoderma lucidum. Nat. Prod. Res. 2011, 25, 1607–1613. [Google Scholar] [CrossRef] [PubMed]
- Orhan, I.; Kartal, M.; Tosun, F.; Şener, B. Screening of Various Phenolic Acids and Flavonoid Derivatives for their Anticholinesterase Potential. Z. Naturforsch. C 2014, 62, 829–832. [Google Scholar] [CrossRef]
- Chan, W.K.; Law, H.K.W.; Lin, Z.-B.; Lau, Y.L.; Chan, G.C.-F. Response of human dendritic cells to different immunomodulatory polysaccharides derived from mushroom and barley. Int. Immunol. 2007, 19, 891–899. [Google Scholar] [CrossRef] [PubMed]
- Sun, L.-X.; Lin, Z.-B.; Li, X.-J.; Li, M.; Lu, J.; Duan, X.-S.; Ge, Z.-H.; Song, Y.-X.; Xing, E.-H.; Li, W.-D. Promoting Effects of Ganoderma lucidum Polysaccharides on B16F10 Cells to Activate Lymphocytes. Basic Clin. Pharmacol. Toxicol. 2011, 108, 149–154. [Google Scholar] [CrossRef] [PubMed]
- Pan, K.; Jiang, Q.; Liu, G.; Miao, X.; Zhong, D. Optimization extraction of Ganoderma lucidum polysaccharides and its immunity and antioxidant activities. Int. J. Biol. Macromol. 2013, 55, 301–306. [Google Scholar] [CrossRef] [PubMed]
- Paterson, R.R.M. Ganoderma—A therapeutic fungal biofactory. Phytochemistry 2006, 68, 1985–2001. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, M.; Cui, S.W.; Cheung, P.C.K.; Wang, Q. Antitumor polysaccharides from mushrooms: A review on their isolation process, structural characteristics and antitumor activity. Trends Food Sci. Technol. 2007, 18, 4–19. [Google Scholar] [CrossRef]
- Huie, C.W.; Di, X. Chromatographic and electrophoretic methods for Lingzhi pharmacologically active components. J. Chromatogr. B 2004, 812, 241–257. [Google Scholar] [CrossRef]
- Eo, S.K.; Kim, Y.S.; Lee, C.K.; Han, S.S. Antiviral activities of various water and methanol soluble substances isolated from Ganoderma lucidum. J. Ethnopharmacol. 1999, 68, 129–136. [Google Scholar] [CrossRef]
- Kim, Y.S.; Eo, S.K.; Oh, K.W.; Lee, C.; Han, S.S. Antiherpetic activities of acidic protein bound polysacchride isolated from Ganoderma lucidum alone and in combinations with interferons. J. Ethnopharmacol. 2000, 72, 451–458. [Google Scholar] [CrossRef]
- Oh, K.W.; Lee, C.K.; Kim, Y.S.; Eo, S.K.; Han, S.S. Antiherpetic activities of acidic protein bound polysacchride isolated from Ganoderma lucidum alone and in combinations with acyclovir and vidarabine. J. Ethnopharmacol. 2000, 72, 221–227. [Google Scholar] [CrossRef]
- Cao, L.-Z.; Lin, Z.-B. Regulation on maturation and function of dendritic cells by Ganoderma lucidum polysaccharides. Immunol. Lett. 2002, 83, 163–169. [Google Scholar] [CrossRef]
- Peng, Y.; Zhang, L.; Zeng, F.; Kennedy, J.F. Structure and antitumor activities of the water-soluble polysaccharides from Ganoderma tsugae mycelium. Carbohydr. Polym. 2005, 59, 385–392. [Google Scholar] [CrossRef]
- Siwulski, M. Biological study on carboxymethylated (1→3)-α-d-glucans from fruiting bodies of Ganoderma lucidum. Int. J. Biol. Macromol. 2012, 51, 1014–1023. [Google Scholar]
- Xu, Z.; Chen, X.; Zhong, Z.; Chen, L.; Wang, Y. Ganoderma lucidum Polysaccharides: Immunomodulation and Potential Anti-Tumor Activities. Am. J. Chin. Med. 2011, 39, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Tang, Q.; Zimmerman-Kordmann, M.; Reutter, W.; Fan, H. Activation of B lymphocytes by GLIS, a bioactive proteoglycan from Ganoderma lucidum. Life Sci. 2002, 71, 623–638. [Google Scholar] [CrossRef]
- Zhu, X.-L.; Chen, A.-F.; Lin, Z.-B. Ganoderma lucidum polysaccharides enhance the function of immunological effector cells in immunosuppressed mice. J. Ethnopharmacol. 2007, 111, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Li, L.; Lei, L.S.; Yu, C.L. Changes of serum interferon-gamma levels in mice bearing S-180 tumor and the interventional effect of immunomodulators. Nan Fang Yi Ke Da Xue Xue Bao 2008, 28, 65–68. [Google Scholar] [PubMed]
- Gao, Y.; Zhou, S.; Jiang, W.; Huang, M.; Dai, X. Effects of Ganopoly® (A Ganoderma lucidum Polysaccharide Extract) on the Immune Functions in Advanced-Stage Cancer Patients. Immunol. Investig. 2003, 32, 201–215. [Google Scholar] [CrossRef]
- Bukhman, V.M.; Treshchalina, E.M.; Krasnopol’skaia, L.M.; Isakova, E.B.; Sedakova, L.A.; Avtonomova, A.V.; Leont’eva, M.I.; Soboleva, N.; Belitskiĭ, I.V.; Bakanov, A.V. Preparation and biological properties of basidiomycete aqueous extracts and their mycelial compositions. Antibiot. Khimioterapiia Antibiot. Chemoterapy Sic. 2007, 52, 4–9. [Google Scholar]
- Ning, J.; Zhang, W.; Yi, Y.; Yang, G.; Wu, Z.; Yi, J.; Kong, F. Synthesis of β-(1→6)-branched β-(1→3) glucohexaose and its analogues containing an α-(1→3) linked bond with antitumor activity. Bioorg. Med. Chem. 2003, 11, 2193–2203. [Google Scholar] [CrossRef]
- Ferreira, I.C.F.R.; Heleno, S.A.; Reis, F.S.; Stojkovic, D.; Queiroz, M.J.R.P.; Vasconcelos, M.H.; Sokovic, M. Chemical features of Ganoderma polysaccharides with antioxidant, antitumor and antimicrobial activities. Phytochemistry 2015, 114, 38–55. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, J.; Hu, S.; Nie, S.; Yu, Q.; Xie, M. Reviews on Mechanisms of In Vitro Antioxidant Activity of Polysaccharides. Available online: https://www.hindawi.com/journals/omcl/2016/5692852/ (accessed on 22 December 2017).
- Xing, R.; Liu, S.; Guo, Z.; Yu, H.; Wang, P.; Li, C.; Li, Z.; Li, P. Relevance of molecular weight of chitosan and its derivatives and their antioxidant activities in vitro. Bioorg. Med. Chem. 2005, 13, 1573–1577. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.-N.; Luo, X.; Tang, Q.; Liu, Y.; Zhou, S.; Yang, Y.; Zhang, J.-S. Isolation, Purification, and Immunological Activities of a Low-Molecular-Weight Polysaccharide from the Lingzhi or Reishi Medicinal Mushroom Ganoderma lucidum (Higher Basidiomycetes). Int. J. Med. Mushrooms 2013, 15, 407–414. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Chen, Y.; Li, S.B.; Chen, Y.G.; Lan, J.Y.; Liu, L.P. Free radical scavenging of Ganoderma lucidum polysaccharides and its effect on antioxidant enzymes and immunity activities in cervical carcinoma rats. Carbohydr. Polym. 2009, 77, 389–393. [Google Scholar]
- Kao, P.-F.; Wang, S.-H.; Hung, W.-T.; Liao, Y.-H.; Lin, C.-M.; Yang, W.-B. Structural Characterization and Antioxidative Activity of Low-Molecular-Weights Beta-1,3-Glucan from the Residue of Extracted Ganoderma lucidum Fruiting Bodies. J. Biomed. Biotechnol. 2012, 2012, 673764. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Ooi, V.E.C.; Chang, S.T. Free radical scavenging activities of mushroom polysaccharide extracts. Life Sci. 1997, 60, 763–771. [Google Scholar] [CrossRef]
- Jiang, H.; Sun, P.; He, J.; Shao, P. Rapid purification of polysaccharides using novel radial flow ion-exchange by response surface methodology from Ganoderma lucidum. Food Bioprod. Process. 2012, 90, 1–8. [Google Scholar] [CrossRef]
- Sun, J.; He, H.; Xie, B.J. Novel antioxidant peptides from fermented mushroom Ganoderma lucidum. J. Agric. Food Chem. 2004, 52, 6646–6652. [Google Scholar] [CrossRef] [PubMed]
- Xu, J.; Liu, W.; Yao, W.; Pang, X.; Yin, D.; Gao, X. Carboxymethylation of a polysaccharide extracted from Ganoderma lucidum enhances its antioxidant activities in vitro. Carbohydr. Polym. 2009, 78, 227–234. [Google Scholar] [CrossRef]
- Liu, W.; Wang, H.; Yao, W.; Gao, X.; Yu, L. Effects of Sulfation on the Physicochemical and Functional Properties of a Water-Insoluble Polysaccharide Preparation from Ganoderma lucidum. J. Agric. Food Chem. 2010, 58, 3336–3341. [Google Scholar] [CrossRef] [PubMed]
- Matsuzaki, H.; Shimizu, Y.; Iwata, N.; Kamiuchi, S.; Suzuki, F.; Iizuka, H.; Hibino, Y.; Okazaki, M. Antidepressant-like effects of a water-soluble extract from the culture medium of Ganoderma lucidum mycelia in rats. BMC Complement. Altern. Med. 2013, 13, 370. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kamra, A.; Bhatt, A.B. Evaluation of antimicrobial and antioxidant activity of Ganoderma lucidum extracts against human pathogenic bacteria. Int. J. Pharm. Pharm. Sci. 2012, 2, 359–362. [Google Scholar]
- Heleno, S.A.; Ferreira, I.C.F.R.; Esteves, A.P.; Ćirić, A.; Glamočlija, J.; Martins, A.; Soković, M.; Queiroz, M.J.R.P. Antimicrobial and demelanizing activity of Ganoderma lucidum extract, p-hydroxybenzoic and cinnamic acids and their synthetic acetylated glucuronide methyl esters. Food Chem. Toxicol. 2013, 58, 95–100. [Google Scholar] [CrossRef] [PubMed]
- Skalicka-Wozniak, K.; Szypowski, J.; Los, R.; Siwulski, M.; Sobieralski, K.; Glowniak, K.; Malm, A. Evaluation of polysaccharides content in fruit bodies and their antimicrobial activity of four Ganoderma lucidum (W Curt.: Fr.) P. Karst. strains cultivated on different wood type substrates. Acta Soc. Bot. Pol. 2012, 81, 17–21. [Google Scholar] [CrossRef]
- Bai, D.; Chang, N.-T.; Li, D.-H.; Liu, J.-X.; You, X.-Y. Antiblastic Activitiy of Ganoderma lucidum Polysaccharides. Acta Agric. Boreali Sin. 2008, 23, 282–285. [Google Scholar]
- Mahendran, S.; Saravana, S.; Vijayabaskar, P.; Anandapandian, K.T.; Shankar, T. Antibacterial potential of microbial exopolysaccharide from Ganoderma lucidum and Lysinibacillus fusiformis. Int. J. Recent Sci. Res. 2013, 4, 501–505. [Google Scholar]
| Compound | Content [mg/100 g] |
|---|---|
| Calcium | 832 |
| Phosphorus | 4.150 |
| Iron | 82.6 |
| Magnesium | 1.030 |
| Sodium | 375 |
| Potassium | 3.590 |
| Vitamin B1 | 3.49 |
| Vitamin B2 | 17.10 |
| Vitamin B6 | 0.71 |
| Choline | 1.150 |
| Niacin | 61.9 |
| Inositol | 307 |
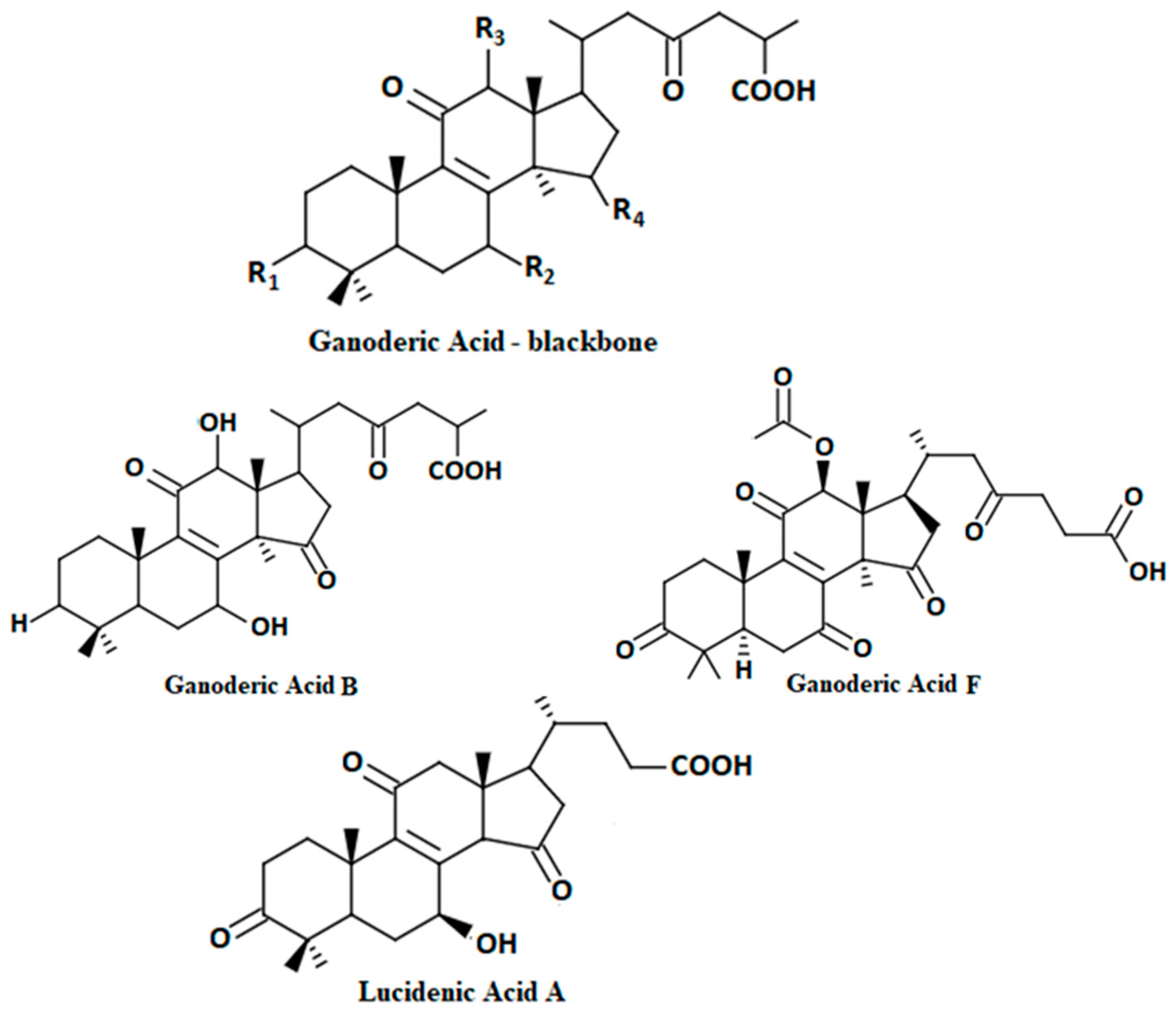
| Triterpene | Tumour cells | Action | Molecular Structure | Target | Ref. | |
|---|---|---|---|---|---|---|
| Ganoderic acid (3α,22β-diacetoxy-7α-hydroxyl-5α-lanost-8,24-E-dien-26-oic acid) | in vitro | Lung:95D Cervical: HeLa | cytotoxic |  | [36] | |
| Ganoderic acid Mk | in vitro | Lung:95D Cervical: HeLa | cytotoxic |  | [36] | |
| Ganoderic acid S | Lung:95D Cervical: HeLa | cytotoxic |  | [36] | ||
| Ganoderic acid Mf | Lung:95D Cervical: HeLa | cytotoxic |  | [36] | ||
| Ganoderic acid R | Lung:95D Cervical: HeLa | Cytotoxic |  | [36] | ||
| Ganoderic acid Mc | Lung:95D Cervical: HeLa | Cytotoxic |  | [36] | ||
| Ganoderic acid A | Breast: MDA-MB-231 | Inhibited growth and invasive behaviour of breast cancer cells |  | AP-1 or NF-κB | [44] | |
| Ganoderic acid F | Breast: MDA-MB-231 | Ineffective |  | AP-1 NF-κB uPA Cdk4 | [44] | |
| Ganoderic acid H | Breast: MDA-MB-231 | Inhibited growth and invasive behaviour of breast cancer cells |  | AP-1 NF-κB uPA Cdk4 | [44] | |
| ganoderic acid X | in vitro | Liver: HuH-7 Colon: HCT-116 | Inhibits topoisomerases and induces apoptosis of cancer cells |  | ERK, JNK | [41] |
| ganoderic acid T | in vitro | lung: 95D liver: SMMC7721 epidermis: KB-A-1 and KB-3-1 cervix: HeLa | Decrease in proliferation of some cancer cells. Strongly inhibits the formation of cell colony of 95-D. |  | p53 Bax caspase-8 | [40] |
| in vitro | colon: HCT-116 | Inhibits proliferation | NFκB-α, MMP-9, uPA, iNOS | [45] | ||
| in vitro | melanoma: A375 colon: Ls174t | Inhibits growth | MMP2/9 NF-κB | [51] | ||
| in vivo | Lung: LLC | Suppresses tumour growth and LLC metastasis | MMP 2/9 | [45] | ||
| ganoderic acid Me | in vitro | Colon: HCT-116 HCT-8 | Possesses cytotoxicity |  | p53 Bax | [52,53] |
| in vitro | Lung: 95-D | inhibits cancer cell invasion | MMP 2/9 | [46] | ||
| in vitro in vivo | Breast: MDA-MB-231 | inhibits proliferation and invasion and induces apoptosis | NF-κB, TNF-α, VEGF, IL-6/8, MMP-9, Bcl-2, c-Myc and CCND1 | [54] | ||
| ganoderic acid D | in vitro | Cervical: HeLa | inhibits proliferation |  | AHA1 Cytokeratin 19 Cytokeratin 1 PRDX3 | [55] |
| ganoderic acid E | in vitro | Liver: Hep G2 Hep G2,2,15 | cytotoxic |  | [56] | |
| ganoderic acid DM | in vitro | Prostate: PC-3, LnCaP | inhibits prostate cancer cell proliferation and metastasis |  | MMP-2 MMP-9 IL-1, IL-6, TNF-α, and CCL-2/MCP-1 | [48] |
| lucidenic acid A | leukaemia: HL 60 | decreases cell population growth, cell cycle arrest |  | Bcl-2 caspase-9 caspase-3 | [49] | |
| lucidenic acid B | leukaemia: HL 60 | Induces apoptosis |  | Bcl-2 caspase-9 caspase-3 | [49,57] | |
| liver: HepG2 Lymphoma: CA46 | MMP-9, NF-κB, ERK1/2, AP-1, c-Jun, c-Fos | |||||
| lucidenic acid C | leukaemia: HL 60 | decreases cell population growth, cell cycle arrest |  | Bcl-2 caspase-9 caspase-3 | [49] | |
| lucidenic acid N | leukaemia: HL 60 | decreases cell population growth, cell cycle arrest |  | Bcl-2 caspase-9 caspase-3 | [49] | |
| Ganoderiol F | in vitro | Lung: LLC Meth A, sarcoma: Sarcoma-180 Carcinoma: T-47D | Cytotoxicity |  | [58] | |
| in vivo | Lung: LLC | inhibitory effect on tumour growth | [50] | |||
| Ganodermanontriol | Colon: HCT-116, HT-29 | Inhibition of cell proliferation |  | β-catenin, cyclin-D1, Cdk4, PCNA, E-cadherin | [59] | |
| Breast: MDA-MB-231 | uPA, uPAR |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cör, D.; Knez, Ž.; Knez Hrnčič, M. Antitumour, Antimicrobial, Antioxidant and Antiacetylcholinesterase Effect of Ganoderma Lucidum Terpenoids and Polysaccharides: A Review. Molecules 2018, 23, 649. https://doi.org/10.3390/molecules23030649
Cör D, Knez Ž, Knez Hrnčič M. Antitumour, Antimicrobial, Antioxidant and Antiacetylcholinesterase Effect of Ganoderma Lucidum Terpenoids and Polysaccharides: A Review. Molecules. 2018; 23(3):649. https://doi.org/10.3390/molecules23030649
Chicago/Turabian StyleCör, Darija, Željko Knez, and Maša Knez Hrnčič. 2018. "Antitumour, Antimicrobial, Antioxidant and Antiacetylcholinesterase Effect of Ganoderma Lucidum Terpenoids and Polysaccharides: A Review" Molecules 23, no. 3: 649. https://doi.org/10.3390/molecules23030649