Contribution of Polyphenol Oxidation, Chlorophyll and Vitamin C Degradation to the Blackening of Piper nigrum L.
Abstract
:1. Introduction
2. Results and Discussion
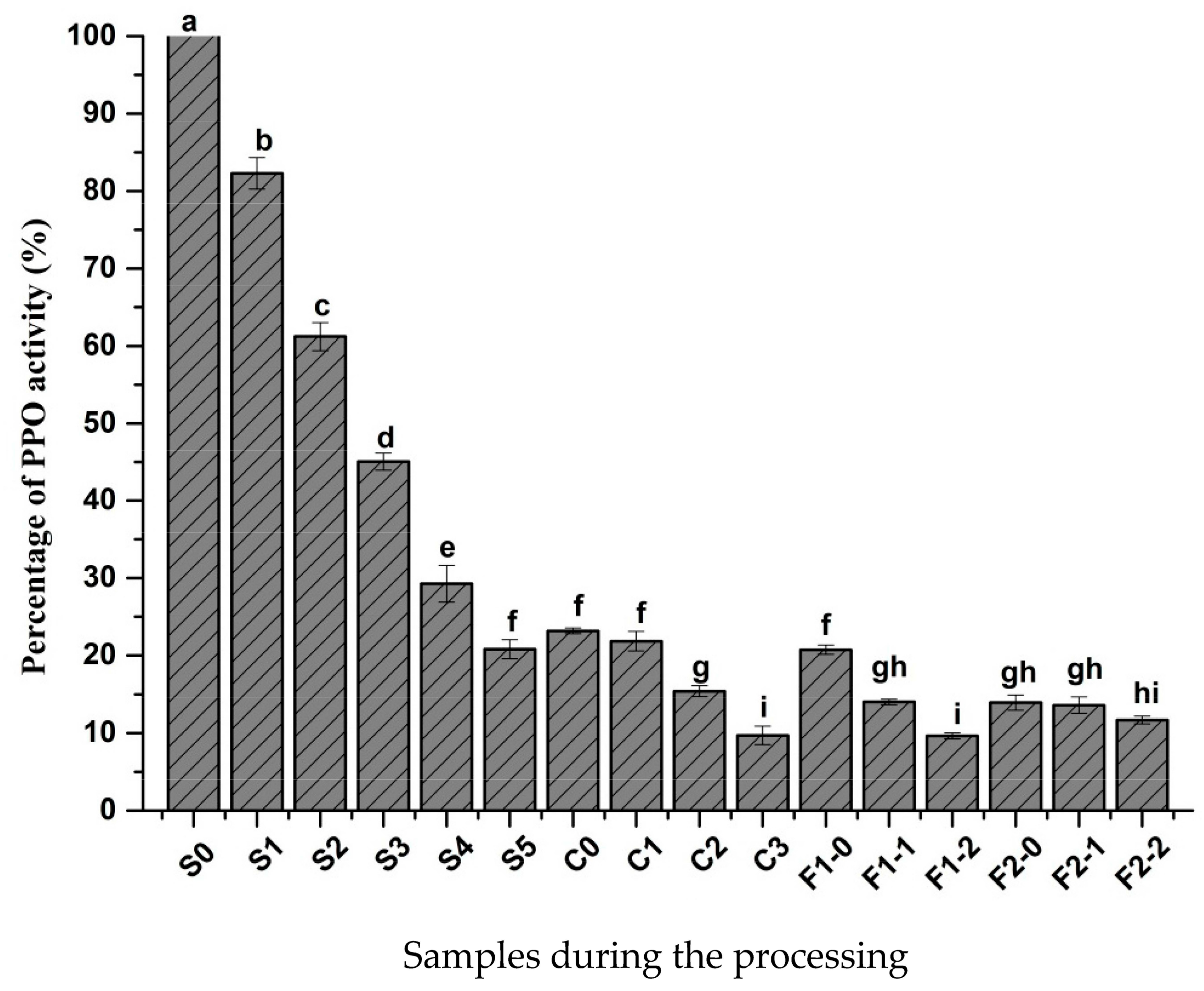
2.1. Changes in PPO Activity
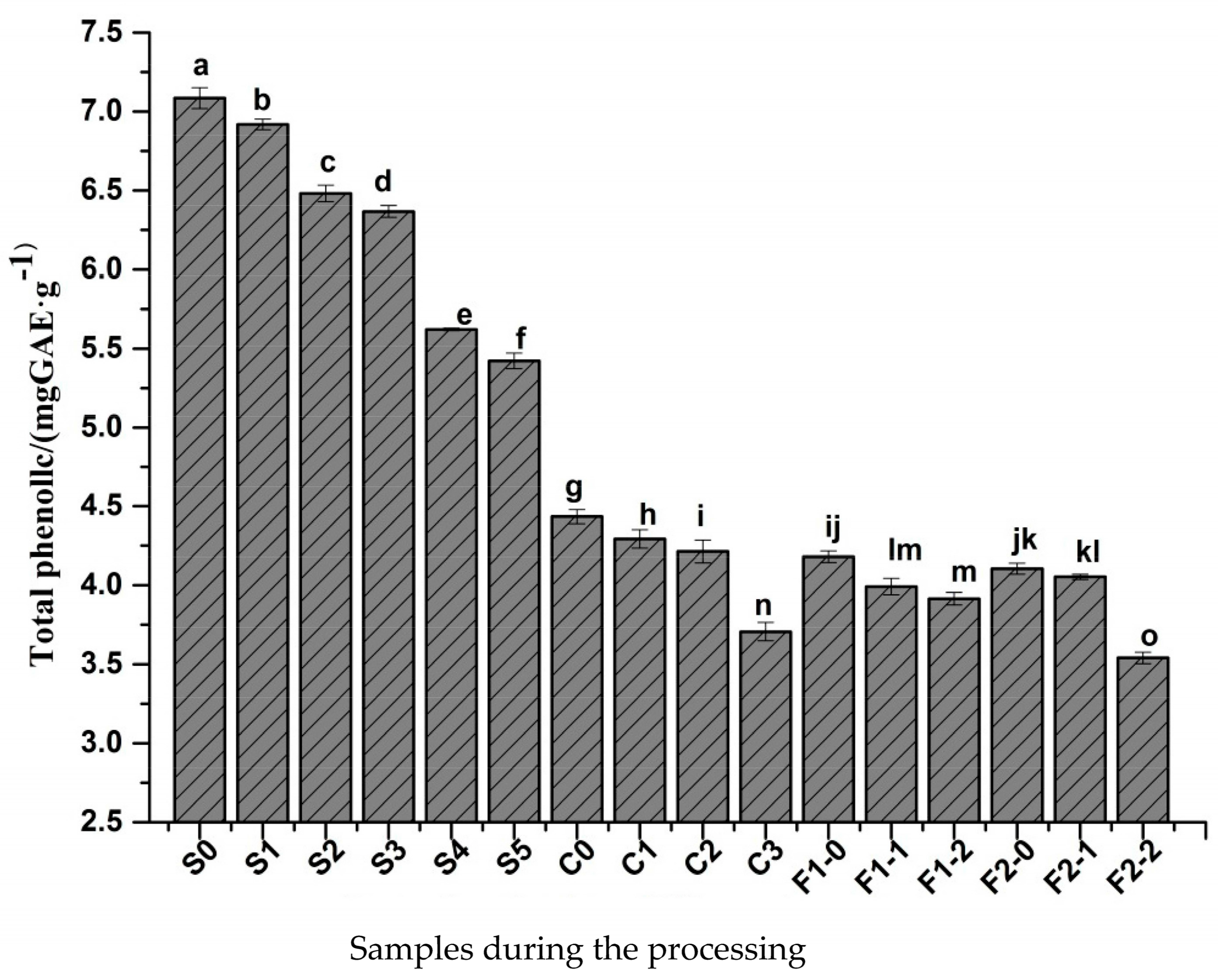
2.2. Changes in Total Phenolic Content
2.3. Separation of Pepper Phenolic Compouds
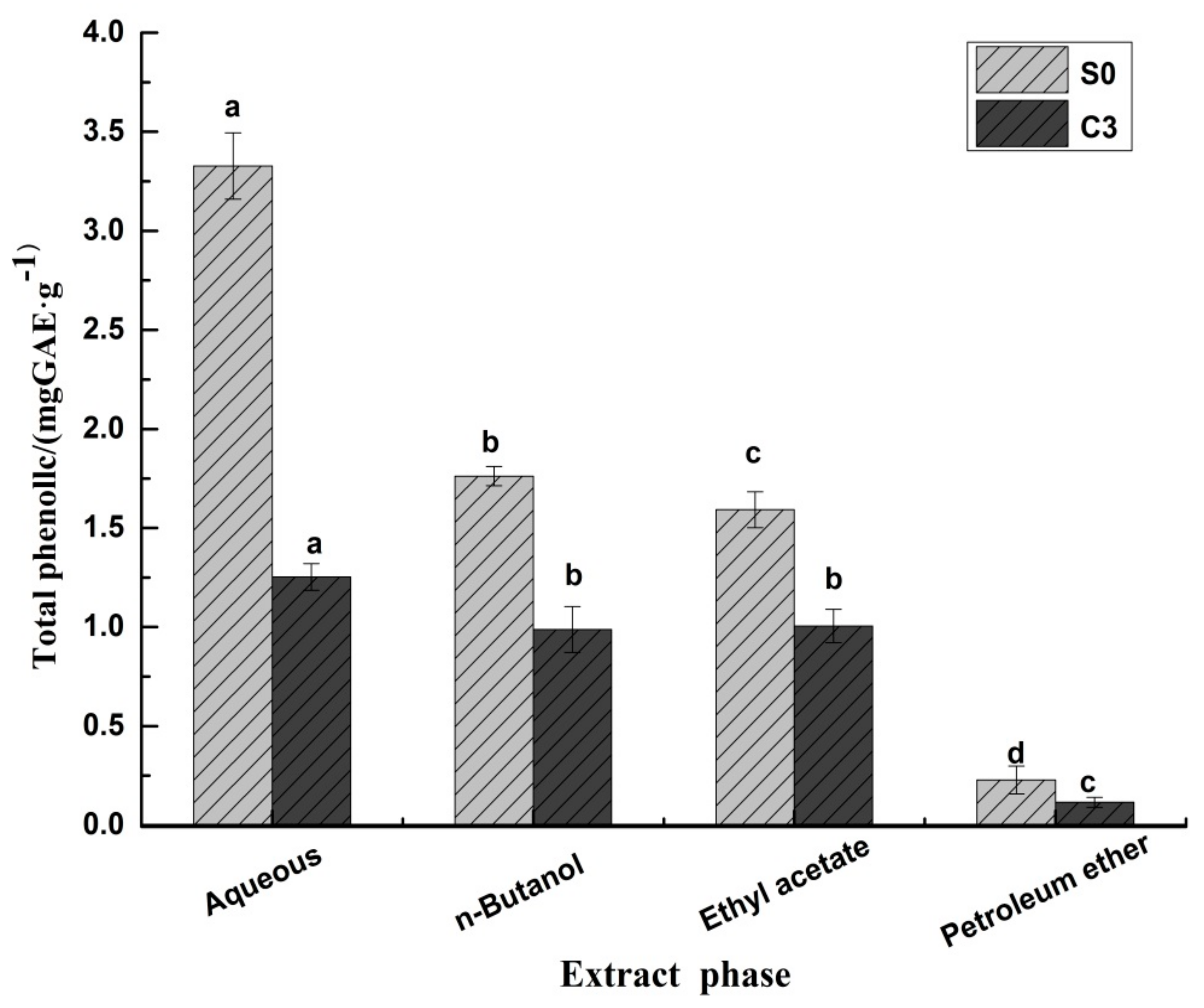
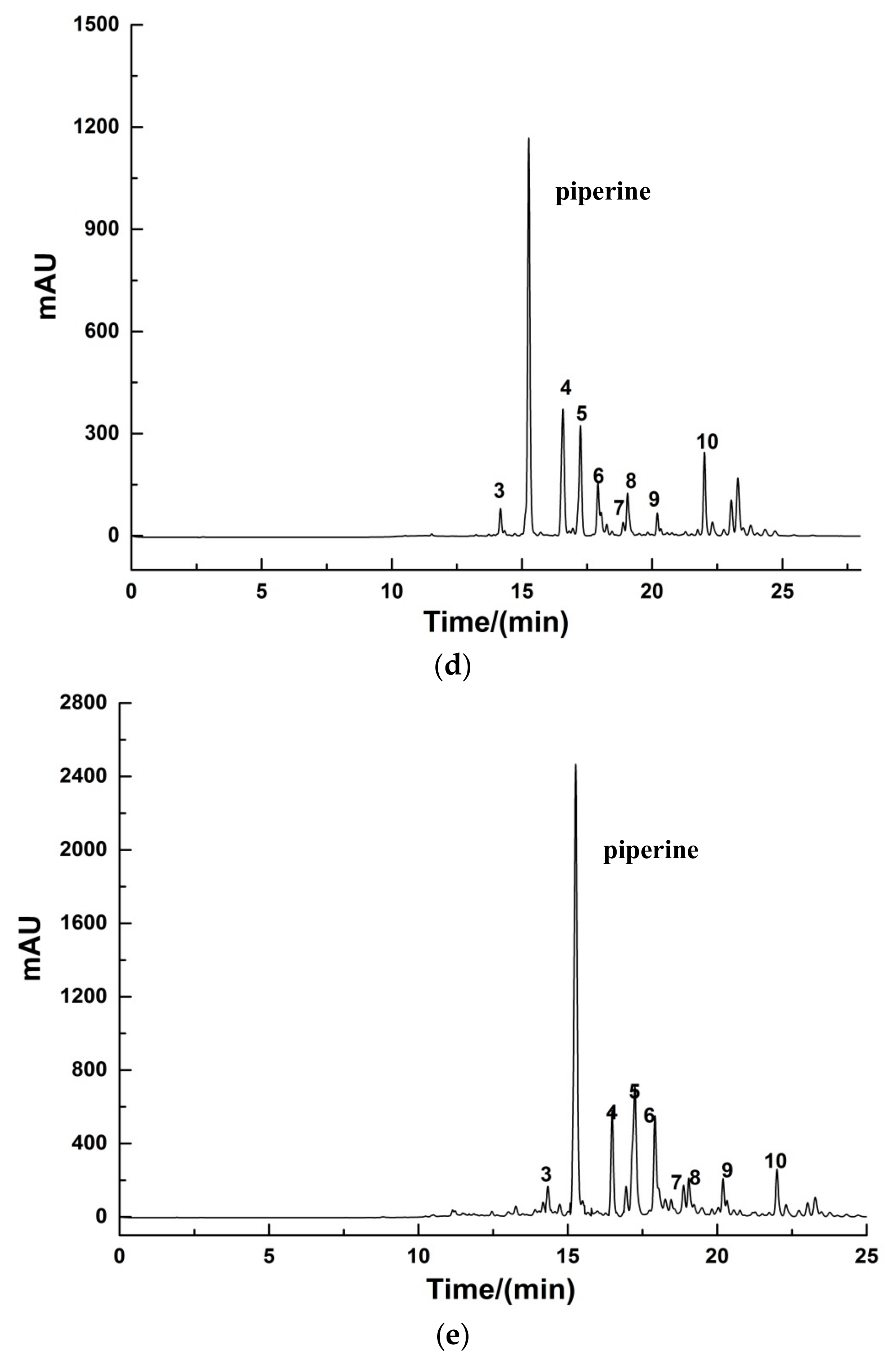
2.3.1. Phenolic Compounds in Black Pepper Extracts
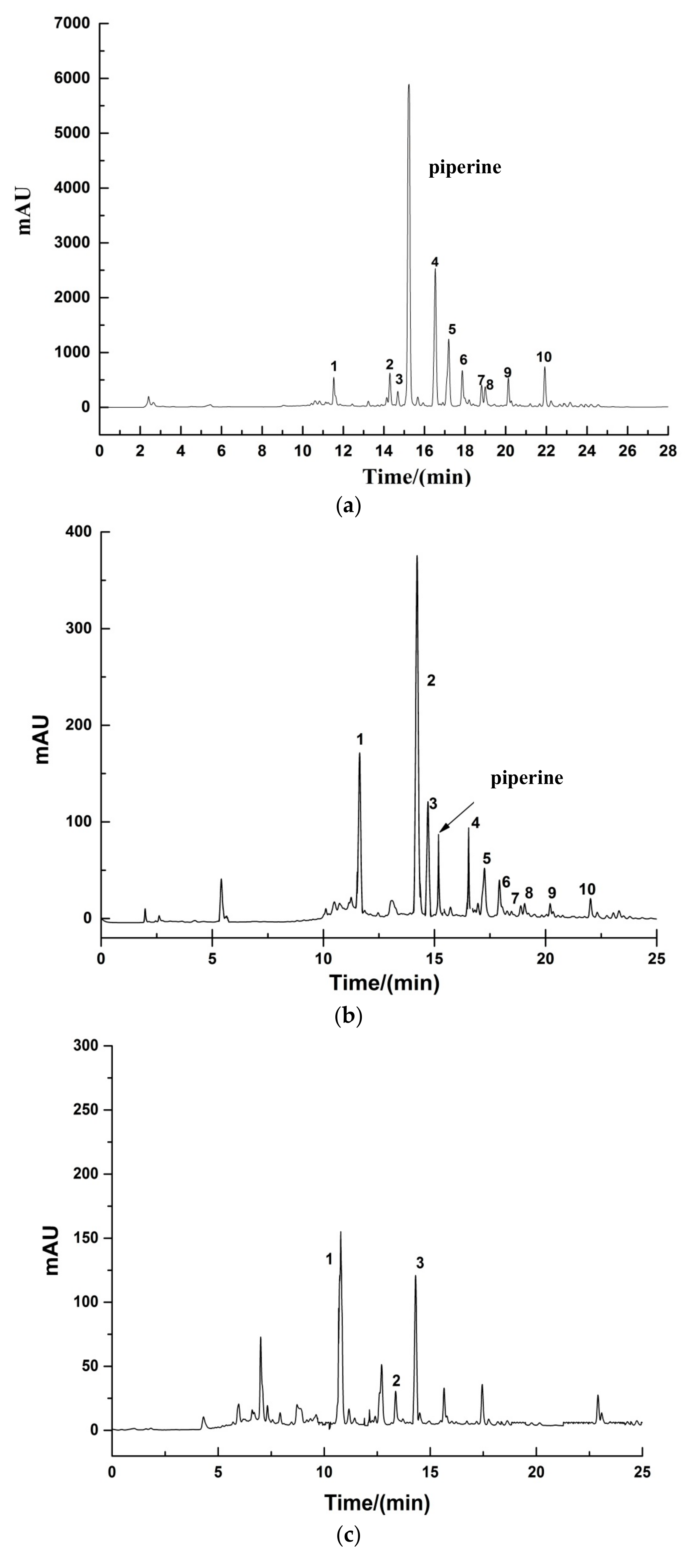
2.3.2. HPLC Analysis
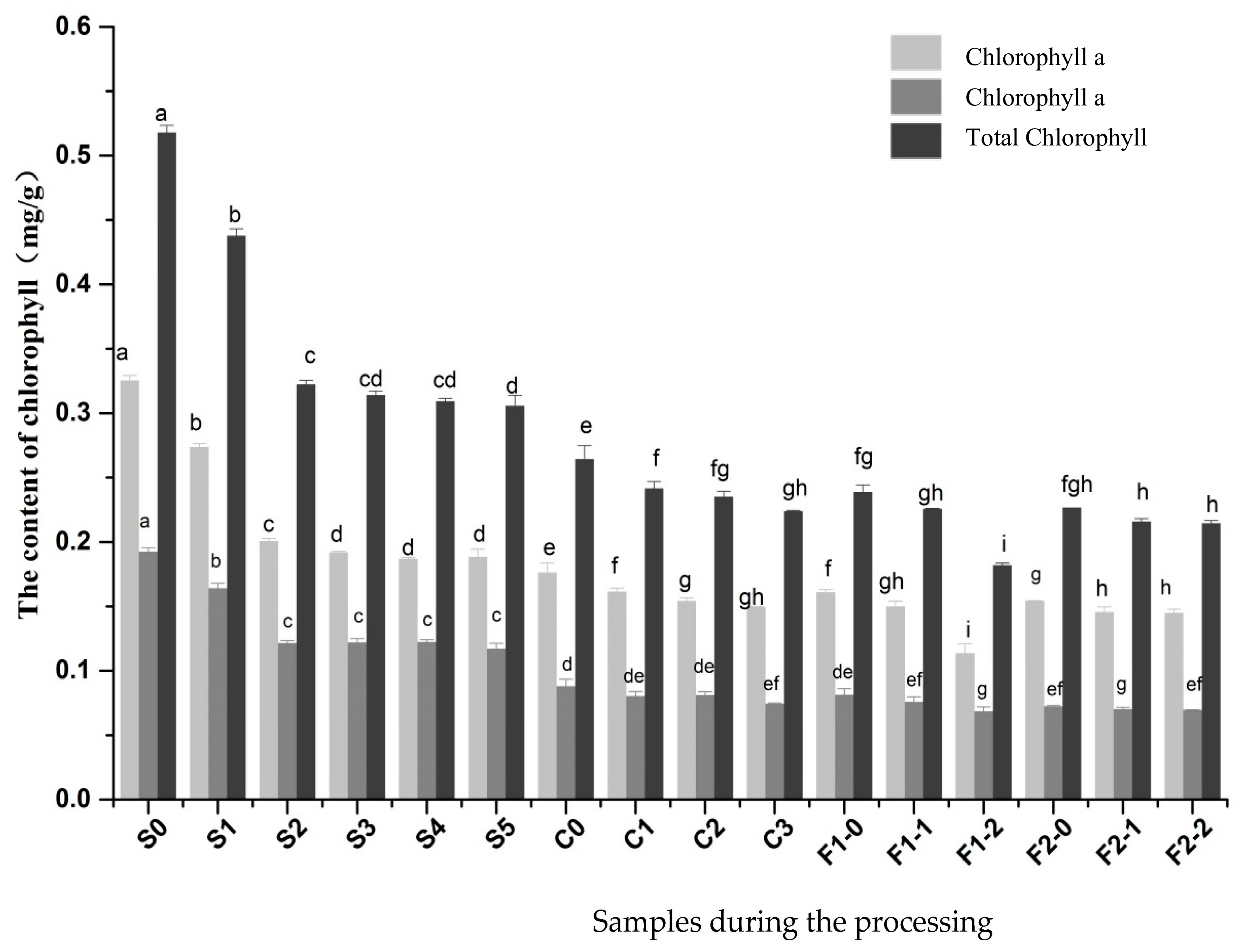
2.4. Changes in Chlorophyll a and Chlorophyll b Content
2.5. Changes in VC Content
3. Experimental Section
3.1. Materials
3.2. Preparation of Black Pepper
3.3. Preparation of Phenolic Extracts
3.4. Determination of Total Phenolic Content
3.5. Solvent Extraction
3.6. High Performance Liquid Chromatography (HPLC)
3.7. PPO Activity Determination
3.8. Determination of Chlorophyll
3.9. Determination of VC
3.10. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Variyar, P.S.; Pendharkar, M.B.; Banerjee, A.; Bandyopadhyay, C. Blackening in green pepper berries. Phytochemistry 1988, 27, 715–717. [Google Scholar] [CrossRef]
- Gu, F.; Tan, L.; Wu, H.; Fang, Y.; Wang, Q. Analysis of the blackening of green pepper (Piper nigrum Linnaeus) berries. Food Chem. 2013, 138, 797–801. [Google Scholar] [CrossRef] [PubMed]
- Vezitskii, A.Y. Chlorophyll a formation in etiolated rye seedlings as dependent on the concentration of infiltrated chlorophyllide b. Russ. J. Plant Physiol. 2000, 47, 499–503. [Google Scholar]
- Koca, N.; Karadeniz, F.; Burdurlu, H.S. Effect of pH on chlorophyll degradation and colour loss in blanched green peas. Food Chem. 2007, 100, 609–615. [Google Scholar] [CrossRef]
- Chen, B.H; Huang, J.H. Degradation and isomerization of chlorophyll a and carotene as affected by various heating and illumination treatments. Food Chem. 1998, 62, 299–307. [Google Scholar] [CrossRef]
- O’Brien, J. The Maillard Reaction in Foods and Medicine; Royal Society of Chemistry: London, UK, 1998. [Google Scholar]
- Mrad, N.D.; Boudhrioua, N.; Kechaou, N. Influence of air drying temperature on kinetics, physicochemical properties, total phenolic content and ascorbic acid of pears. Food Bioprod. Process. 2012, 90, 433–441. [Google Scholar] [CrossRef]
- Pruthi, J.S. Advances in Sun/Solar Drying and Dehydration of Pepper (Piper nigrum L.); International Pepper News Bulletin: Jakarta, Indonesia, 1992; Volume XVI, pp. 6–17. [Google Scholar]
- Acosta-Estrada, B.A.; Gutiérrez-Uribe, J.A.; Serna-Saldívar, S.O. Bound phenolics in foods: A review. Food Chem. 2014, 152, 46–55. [Google Scholar] [CrossRef] [PubMed]
- Schwartz, S.J.; Lorenzo, T.V. Chlorophylls in foods. Crit. Rev. Food. Sci. Nutr. 1990, 29, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Bandyopadhyay, C.; Narayan, V.S.; Variyar, P.S. Phenolics of green pepper berries (Piper nigrum L.). J. Agric. Food Chem. 1990, 38, 1696–1699. [Google Scholar] [CrossRef]
- Chatterjee, S.; Niaz, Z.; Gautam, S.; Adhikari, S.; Variyar, P.S.; Sharma, A. Antioxidant activity of some phenolic constituents from green pepper (Piper nigrum L.) and fresh nutmeg mace (Myristica fragrans). Food Chem. 2007, 101, 515–523. [Google Scholar] [CrossRef]
- Variyar, P.S.; Bandyopadhyay, C. Estimation of phenolic compounds in green pepper berries (Piper nigrum L.) by high-performance liquid chromatography. Chromatographia 1994, 39, 743–746. [Google Scholar] [CrossRef]
- Herbig, A.L.; Renard, C.M. Factors that impact the stability of vitamin C at intermediate temperatures in a food matrix. Food Chem. 2016, 220, 444–451. [Google Scholar] [CrossRef] [PubMed]
- Aguilar, K.; Garvín, A.; Ibarz, A.; Augusto, P.E. Ascorbic acid stability in fruit juices during thermosonication. Ultrason. Sonochem. 2017, 37, 375–381. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.W.; Kim, Y.J.; Lee, H.J.; Lee, C.Y. Cocoa Has More Phenolic Phytochemicals and a Higher Antioxidant Capacity than Teas and Red Wine. J. Agr. Food Chem. 2003, 51, 7292–7295. [Google Scholar] [CrossRef] [PubMed]
- Vranac, A.; Begić-Akagić, A.; Gaši, F.; Spaho, N.; Drkenda, P. Polyphenolic profiles of traditional and international apple and pear cultivars using high performance liquid chromatography (HPLC). In Proceedings of the 2nd International Congress on Food Technology, Kuşadasi, Turkey, 5–7 November 2014. [Google Scholar]
- Arnon, D.I. Copper enzymes in isolated chloroplasts. Polyphenoloxidase in beta vulgaris. Plant Physiol. 1949, 24, 1–15. [Google Scholar] [PubMed]
- Tiwari, B.K.; Donnell, C.P.; Patras, A. Effect of ozone processing on anthocyanins and ascorbic acid degradation of strawberry juice. Food Chem. 2009, 113, 1119–1126. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds are available from the authors. |

| Methods | Processes |
|---|---|
| Direct sun drying | The berries were processed by sun-drying for five days. Sample S0 represents fresh pepper berries, Samples S1–S5 represent berries that underwent sun-drying for one to five days, respectively. |
| Blanching before sun drying | Fresh pepper berries were subjected to heat treatment with hot water for 2 min at 80 °C, and then the berries were processed by sun-drying for three days. Sample S0 represents fresh pepper berries. Sample C0 refers to berries subjected to heat treatment. Samples C1–C3 represent berries that underwent sun-drying for one to three days after heat treatment, respectively. |
| One-day sun drying before mild fermentation | Fresh pepper berries were subjected to heat treatment with hot water for 2 min at 80 °C. The berries were processed by sun-drying for one day. Then, they were fermented for one day before sun-drying. Samples F1-0 refer to berries that underwent sun-drying for one day after heat treatment and fermentation for one day. Samples F1-1 and F1-2 represent berries that underwent sun-drying for one or two days after fermentation, respectively. |
| Two-day sun drying before mild fermentation | Fresh pepper berries were subjected to heat treatment with hot water for 2 min at 80 °C, and then the berries were processed by sun-drying for two days. Then, they were fermented for one day before sun-drying. Samples F2-0 represent berries that underwent sun-drying for two days after heat treatment and fermentation for one day. Samples F2-1 and F2-2 refer to berries that underwent sun-drying for one or two days after fermentation, respectively. |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gu, F.; Huang, F.; Wu, G.; Zhu, H. Contribution of Polyphenol Oxidation, Chlorophyll and Vitamin C Degradation to the Blackening of Piper nigrum L. Molecules 2018, 23, 370. https://doi.org/10.3390/molecules23020370
Gu F, Huang F, Wu G, Zhu H. Contribution of Polyphenol Oxidation, Chlorophyll and Vitamin C Degradation to the Blackening of Piper nigrum L. Molecules. 2018; 23(2):370. https://doi.org/10.3390/molecules23020370
Chicago/Turabian StyleGu, Fenglin, Feifei Huang, Guiping Wu, and Hongying Zhu. 2018. "Contribution of Polyphenol Oxidation, Chlorophyll and Vitamin C Degradation to the Blackening of Piper nigrum L." Molecules 23, no. 2: 370. https://doi.org/10.3390/molecules23020370




