Understanding of MYB Transcription Factors Involved in Glucosinolate Biosynthesis in Brassicaceae
Abstract
1. Introduction
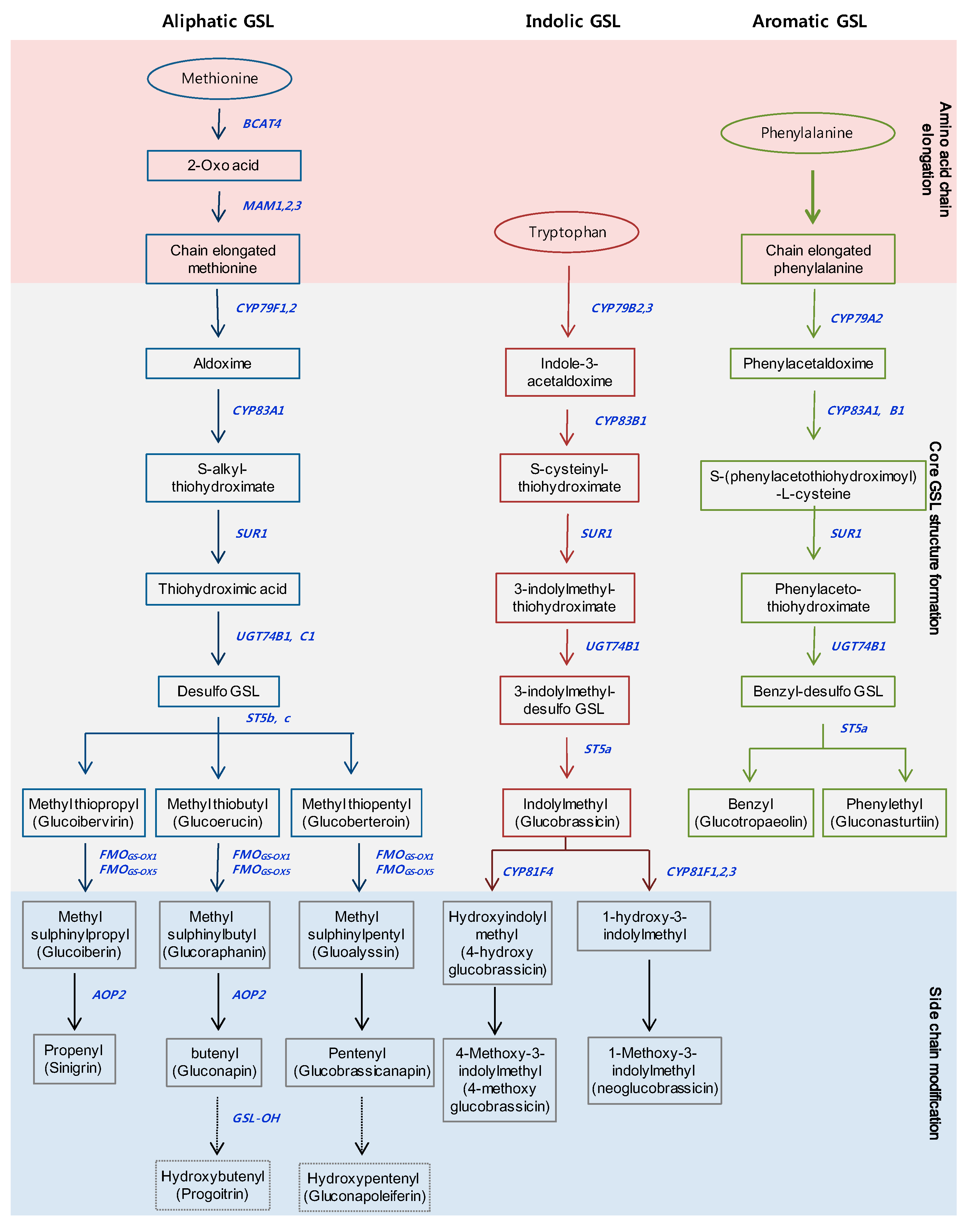
2. Metabolic and Regulatory Pathways of GSL Biosynthesis
2.1. Metabolic Pathway of GSLs
2.2. Regulators of the GSL Biosynthesis Pathway in the Model Plant Arabidopsis
3. MYB TFs Involved in GSL Biosynthesis in Genome-Sequenced Brassica Species
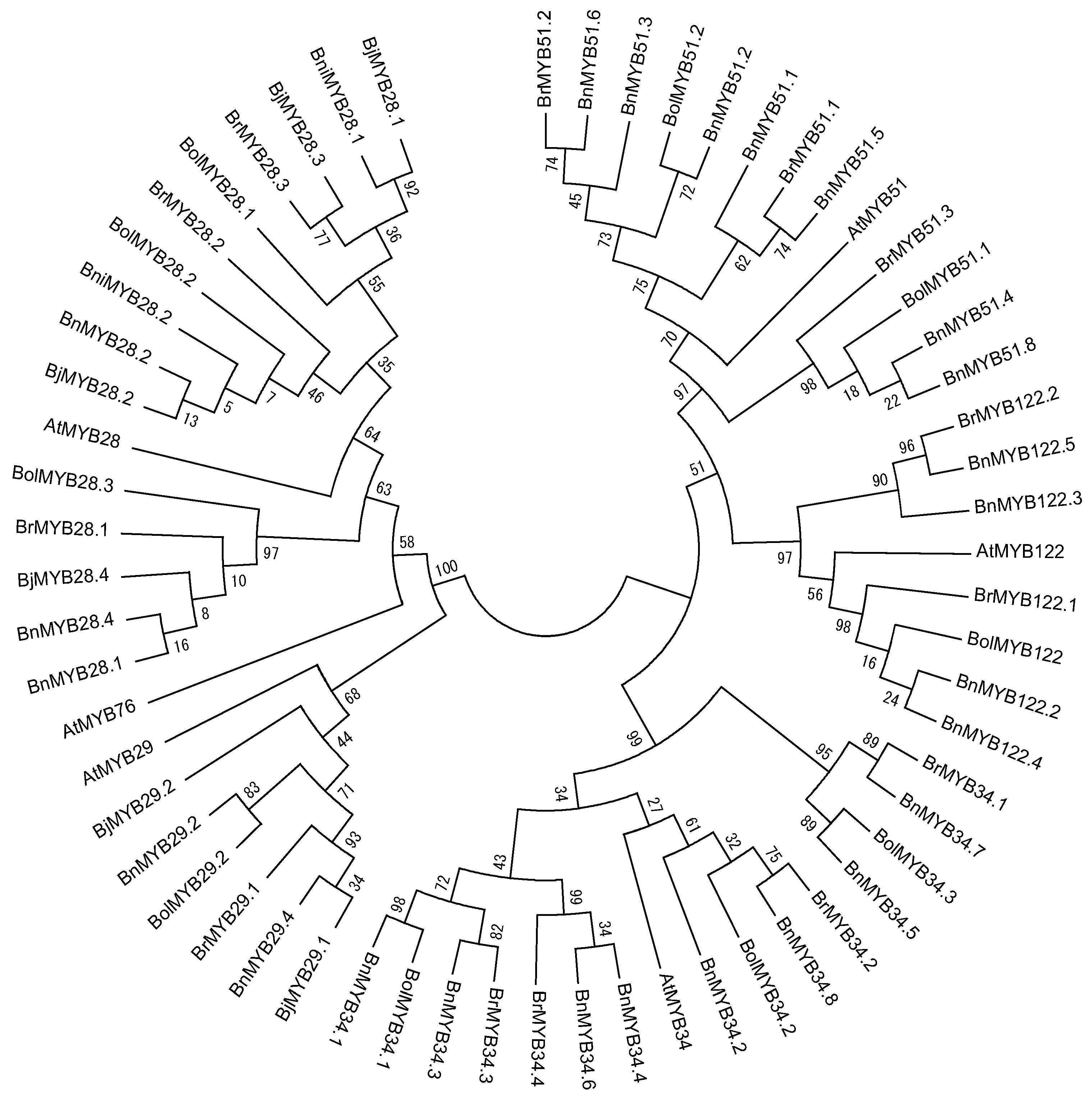
3.1. Characterization of MYB TFs in Brassica Species
3.2. Functional Description of MYB TFs Related to GSL Biosynthesis in Brassica Species
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Verpoorte, R.; Memelink, J. Engineering secondary metabolite production in plants. Curr. Opin. Biotechnol. 2002, 13, 181–187. [Google Scholar] [CrossRef]
- Yonekura-Sakakibara, K.; Saito, K. Functional genomics for plant natural product biosynthesis. Nat. Prod. Rep. 2009, 26, 1466–1487. [Google Scholar] [CrossRef] [PubMed]
- Cartea, M.E.; Francisco, M.; Soengas, P.; Velasco, P. Phenolic compounds in brassica vegetables. Molecules 2010, 16, 251–280. [Google Scholar] [CrossRef] [PubMed]
- Hayes, J.D.; Kelleher, M.O.; Eggleston, I.M. The cancer chemopreventive actions of phytochemicals derived from glucosinolates. Eur. J. Nutr. 2008, 47, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Beilstein, M.A.; Al-Shehbaz, I.A.; Kellogg, E.A. Brassicaceae phylogeny and trichome evolution. Am. J. Bot. 2006, 93, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Lysak, M.A.; Koch, M.A.; Pecinka, A.; Schubert, I. Chromosome triplication found across the tribe brassiceae. Genome Res. 2005, 15, 516–525. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, H.; Wang, J.; Sun, R.; Wu, J.; Liu, S.; Bai, Y.; Mun, J.H.; Bancroft, I.; Cheng, F.; et al. The genome of the mesopolyploid crop species Brassica rapa. Nat. Genet. 2011, 43, 1035–1039. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Liu, Y.; Yang, X.; Tong, C.; Edwards, D.; Parkin, I.A.; Zhao, M.; Ma, J.; Yu, J.; Huang, S.; et al. The Brassica oleracea genome reveals the asymmetrical evolution of polyploid genomes. Nat. Commun. 2014, 5, 3930. [Google Scholar] [CrossRef] [PubMed]
- Chalhoub, B.; Denoeud, F.; Liu, S.; Parkin, I.A.; Tang, H.; Wang, X.; Chiquet, J.; Belcram, H.; Tong, C.; Samans, B.; et al. Early allopolyploid evolution in the post-neolithic Brassica napus oilseed genome. Science 2014, 345, 950–953. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Liu, D.; Wang, X.; Ji, C.; Cheng, F.; Liu, B.; Hu, Z.; Chen, S.; Pental, D.; Ju, Y.; et al. The genome sequence of allopolyploid Brassica juncea and analysis of differential homoeolog gene expression influencing selection. Nat. Genet. 2016, 48, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Adams, K.L.; Wendel, J.F. Polyploidy and genome evolution in plants. Curr. Opin. Plant Biol. 2005, 8, 135–141. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.S.; Chung, T.Y.; King, G.J.; Jin, M.; Yang, T.J.; Jin, Y.M.; Kim, H.I.; Park, B.S. A sequence-tagged linkage map of Brassica rapa. Genetics 2006, 174, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Park, B.S.; Kwon, S.J.; Kim, J.S.; Lim, M.H.; Park, Y.D.; Kim, D.Y.; Suh, S.C.; Jin, Y.M.; Ahn, J.H.; et al. Delayed flowering time in Arabidopsis and Brassica rapa by the overexpression of FLOWERING LOCUS C (FLC) homologs isolated from Chinese cabbage (Brassica rapa L. ssp. pekinensis). Plant Cell Rep. 2007, 26, 327–336. [Google Scholar] [CrossRef] [PubMed]
- Zang, Y.X.; Kim, H.U.; Kim, J.A.; Lim, M.H.; Jin, M.; Lee, S.C.; Kwon, S.J.; Lee, S.I.; Hong, J.K.; Park, T.H.; et al. Genome-wide identification of glucosinolate synthesis genes in Brassica rapa. FEBS J. 2009, 276, 3559–3574. [Google Scholar] [CrossRef] [PubMed]
- Guo, R.; Huang, Z.; Deng, Y.; Chen, X.; XuHan, X.; Lai, Z. Comparative transcriptome analyses reveal a special glucosinolate metabolism mechanism in Brassica alboglabra sprouts. Front. Plant Sci. 2016, 7, 1497. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.S.; Jin, M.; Chun, J.H.; Kim, S.J.; Park, B.S.; Shon, S.H.; Kim, J.S. Functional analysis of three BrMYB28 transcription factors controlling the biosynthesis of glucosinolates in Brassica rapa. Plant Mol. Biol. 2016, 90, 503–516. [Google Scholar] [CrossRef] [PubMed]
- Borpatragohain, P.; Rose, T.J.; King, G.J. Fire and brimstone: Molecular interactions between sulfur and glucosinolate biosynthesis in model and crop brassicaceae. Front. Plant Sci. 2016, 7, 1735. [Google Scholar] [CrossRef] [PubMed]
- Sonderby, I.E.; Burow, M.; Rowe, H.C.; Kliebenstein, D.J.; Halkier, B.A. A complex interplay of three R2R3 MYB transcription factors determines the profile of aliphatic glucosinolates in Arabidopsis. Plant Physiol. 2010, 153, 348–363. [Google Scholar] [CrossRef] [PubMed]
- Hirai, M.Y.; Sugiyama, K.; Sawada, Y.; Tohge, T.; Obayashi, T.; Suzuki, A.; Araki, R.; Sakurai, N.; Suzuki, H.; Aoki, K.; et al. Omics-based identification of Arabidopsis MYB transcription factors regulating aliphatic glucosinolate biosynthesis. Proc. Natl. Acad. Sci. USA 2007, 104, 6478–6483. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Majee, M.; Gershenzon, J.; Bisht, N.C. Four genes encoding MYB28, a major transcriptional regulator of the aliphatic glucosinolate pathway, are differentially expressed in the allopolyploid Brassica juncea. J. Exp. Bot. 2013, 64, 4907–4921. [Google Scholar] [CrossRef] [PubMed]
- Frerigmann, H.; Gigolashvili, T. MYB34, MYB51, and MYB122 distinctly regulate indolic glucosinolate biosynthesis in Arabidopsis thaliana. Mol. Plant 2014, 7, 814–828. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, R.J.; van Dam, N.M.; van Loon, J.J. Role of glucosinolates in insect-plant relationships and multitrophic interactions. Annu. Rev. Entomol. 2009, 54, 57–83. [Google Scholar] [CrossRef] [PubMed]
- Kliebenstein, D.J.; Kroymann, J.; Brown, P.; Figuth, A.; Pedersen, D.; Gershenzon, J.; Mitchell-Olds, T. Genetic control of natural variation in Arabidopsis glucosinolate accumulation. Plant Physiol. 2001, 126, 811–825. [Google Scholar] [CrossRef] [PubMed]
- Seo, M.S.; Jin, M.; Sohn, S.H.; Kim, J.S. Expression profiles of BrMYB transcription factors related to glucosinolate biosynthesis and stress response in eight subspecies of Brassica rapa. FEBS Open Biol. 2017. [Google Scholar] [CrossRef]
- Padilla, G.; Cartea, M.E.; Velasco, P.; de Haro, A.; Ordas, A. Variation of glucosinolates in vegetable crops of Brassica rapa. Phytochemistry 2007, 68, 536–545. [Google Scholar] [CrossRef] [PubMed]
- Cartea, M.E.; Velasco, P.; Obregon, S.; Padilla, G.; de Haro, A. Seasonal variation in glucosinolate content in Brassica oleracea crops grown in northwestern spain. Phytochemistry 2008, 69, 403–410. [Google Scholar] [CrossRef] [PubMed]
- Verkerk, R.; Schreiner, M.; Krumbein, A.; Ciska, E.; Holst, B.; Rowland, I.; De Schrijver, R.; Hansen, M.; Gerhauser, C.; Mithen, R.; et al. Glucosinolates in Brassica vegetables: The influence of the food supply chain on intake, bioavailability and human health. Mol. Nutr. Food Res. 2009, 53. [Google Scholar] [CrossRef] [PubMed]
- Brooks, J.D.; Paton, V.G.; Vidanes, G. Potent induction of phase 2 enzymes in human prostate cells by sulforaphane. Cancer Epidemiol. Biomark. Prev. 2001, 10, 949–954. [Google Scholar]
- Mazumder, A.; Dwivedi, A.; du Plessis, J. Sinigrin and its therapeutic benefits. Molecules 2016, 21, 416. [Google Scholar] [CrossRef] [PubMed]
- Powolny, A.; Takahashi, K.; Hopkins, R.G.; Loo, G. Induction of gadd gene expression by phenethylisothiocyanate in human colon adenocarcinoma cells. J. Cell. Biochem. 2003, 90, 1128–1139. [Google Scholar] [CrossRef] [PubMed]
- Pearson, A.W.; Greenwood, N.M.; Butler, E.J.; Fenwick, G.R. Biochemical changes in layer and broiler chickens when fed on a high-glucosinolate rapeseed meal. Br. Poult. Sci. 1983, 24, 417–427. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.H.; Lee, B.W.; Schroeder, F.C.; Jander, G. Identification of indole glucosinolate breakdown products with antifeedant effects on Myzus persicae (green peach aphid). Plant J. 2008, 54, 1015–1026. [Google Scholar] [CrossRef] [PubMed]
- Pfalz, M.; Vogel, H.; Kroymann, J. The gene controlling the indole glucosinolate modifier1 quantitative trait locus alters indole glucosinolate structures and aphid resistance in Arabidopsis. Plant Cell 2009, 21, 985–999. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y.; Hull, A.K.; Gupta, N.R.; Goss, K.A.; Alonso, J.; Ecker, J.R.; Normanly, J.; Chory, J.; Celenza, J.L. Trp-dependent auxin biosynthesis in Arabidopsis: Involvement of cytochrome P450s CYP79b3 and CYP79b3. Gene. Dev. 2002, 16, 3100–3112. [Google Scholar] [CrossRef] [PubMed]
- Glawischnig, E.; Hansen, B.G.; Olsen, C.E.; Halkier, B.A. Camalexin is synthesized from indole-3-acetaldoxime, a key branching point between primary and secondary metabolism in Arabidopsis. Proc. Natl. Acad. Sci. USA 2004, 101, 8245–8250. [Google Scholar] [CrossRef] [PubMed]
- Marino, D.; Ariz, I.; Lasa, B.; Santamaria, E.; Fernandez-Irigoyen, J.; Gonzalez-Murua, C.; Aparicio Tejo, P.M. Quantitative proteomics reveals the importance of nitrogen source to control glucosinolate metabolism in Arabidopsis thaliana and Brassica oleracea. J. Exp. Bot. 2016, 67, 3313–3323. [Google Scholar] [CrossRef] [PubMed]
- Huseby, S.; Koprivova, A.; Lee, B.R.; Saha, S.; Mithen, R.; Wold, A.B.; Bengtsson, G.B.; Kopriva, S. Diurnal and light regulation of sulphur assimilation and glucosinolate biosynthesis in Arabidopsis. J. Exper. Bot. 2013, 64, 1039–1048. [Google Scholar] [CrossRef] [PubMed]
- Schonhof, I.; Klaring, H.P.; Krumbein, A.; Schreiner, M. Interaction between atmospheric CO2 and glucosinolates in broccoli. J. Chem. Ecol. 2007, 33, 105–114. [Google Scholar] [CrossRef] [PubMed]
- Gigolashvili, T.; Kopriva, S. Transporters in plant sulfur metabolism. Front. Plant Sci. 2014, 5, 442. [Google Scholar] [CrossRef] [PubMed]
- Koroleva, O.A.; Cramer, R. Single-cell proteomic analysis of glucosinolate-rich S-cells in Arabidopsis thaliana. Methods 2011, 54, 413–423. [Google Scholar] [CrossRef] [PubMed]
- Skirycz, A.; Reichelt, M.; Burow, M.; Birkemeyer, C.; Rolcik, J.; Kopka, J.; Zanor, M.I.; Gershenzon, J.; Strnad, M.; Szopa, J.; et al. Dof transcription factor Atdof1.1 (OBP2) is part of a regulatory network controlling glucosinolate biosynthesis in Arabidopsis. Plant J. 2006, 47, 10–24. [Google Scholar] [CrossRef] [PubMed]
- Levy, M.; Wang, Q.; Kaspi, R.; Parrella, M.P.; Abel, S. Arabidopsis IQD1, a novel calmodulin-binding nuclear protein, stimulates glucosinolate accumulation and plant defense. Plant J. 2005, 43, 79–96. [Google Scholar] [CrossRef] [PubMed]
- Gigolashvili, T.; Yatusevich, R.; Berger, B.; Muller, C.; Flugge, U.I. The R2R3-MYB transcription factor HAG1/MYB28 is a regulator of methionine-derived glucosinolate biosynthesis in Arabidopsis thaliana. Plant J. 2007, 51, 247–261. [Google Scholar] [CrossRef] [PubMed]
- Gigolashvili, T.; Engqvist, M.; Yatusevich, R.; Muller, C.; Flugge, U.I. HAG2/MYB76 and HAG3/MYB29 exert a specific and coordinated control on the regulation of aliphatic glucosinolate biosynthesis in Arabidopsis thaliana. New Phytol. 2008, 177, 627–642. [Google Scholar] [CrossRef] [PubMed]
- Frerigmann, H. Glucosinolate regulation in a complex relationship—MYC and MYB—No one can act without each other. Adv. Bot. Res. 2016, 80, 57–97. [Google Scholar]
- Sonderby, I.E.; Hansen, B.G.; Bjarnholt, N.; Ticconi, C.; Halkier, B.A.; Kliebenstein, D.J. A systems biology approach identifies a R2R3 MYB gene subfamily with distinct and overlapping functions in regulation of aliphatic glucosinolates. PLoS ONE 2007, 2, e1322. [Google Scholar] [CrossRef] [PubMed]
- Burow, M.; Atwell, S.; Francisco, M.; Kerwin, R.E.; Halkier, B.A.; Kliebenstein, D.J. The glucosinolate biosynthetic gene AOP2 mediates feed-back regulation of jasmonic acid signaling in Arabidopsis. Mol. Plant 2015, 8, 1201–1212. [Google Scholar] [CrossRef] [PubMed]
- Gigolashvili, T.; Berger, B.; Mock, H.P.; Muller, C.; Weisshaar, B.; Flugge, U.I. The transcription factor HIG1/MYB51 regulates indolic glucosinolate biosynthesis in Arabidopsis thaliana. Plant J. 2007, 50, 886–901. [Google Scholar] [CrossRef] [PubMed]
- Yatusevich, R.; Mugford, S.G.; Matthewman, C.; Gigolashvili, T.; Frerigmann, H.; Delaney, S.; Koprivova, A.; Flugge, U.I.; Kopriva, S. Genes of primary sulfate assimilation are part of the glucosinolate biosynthetic network in Arabidopsis thaliana. Plant J. 2010, 62, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Stracke, R.; Werber, M.; Weisshaar, B. The R2R3-MYB gene family in Arabidopsis thaliana. Curr. Opin. Plant Biol. 2001, 4, 447–456. [Google Scholar] [CrossRef]
- Prouse, M.B.; Campbell, M.M. The interaction between MYB proteins and their target DNA binding sites. Biochim. Biophys. Acta 2012, 1819, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Malitsky, S.; Blum, E.; Less, H.; Venger, I.; Elbaz, M.; Morin, S.; Eshed, Y.; Aharoni, A. The transcript and metabolite networks affected by the two clades of Arabidopsis glucosinolate biosynthesis regulators. Plant Physiol. 2008, 148, 2021–2049. [Google Scholar] [CrossRef] [PubMed]
- Yi, G.E.; Robin, A.H.; Yang, K.; Park, J.I.; Kang, J.G.; Yang, T.J.; Nou, I.S. Identification and expression analysis of glucosinolate biosynthetic genes and estimation of glucosinolate contents in edible organs of Brassica oleracea subspecies. Molecules 2015, 20, 13089–13111. [Google Scholar] [CrossRef] [PubMed]
- Cheng, F.; Liang, J.; Cai, C.; Cai, X.; Wu, J.; Wang, X. Genome sequencing supports a multi-vertex model for Brassiceae species. Curr. Opin. Plant Biol. 2017, 36, 79–87. [Google Scholar] [CrossRef] [PubMed]
- Udall, J.A.; Swanson, J.M.; Nettletion, D.; Percifield, R.J.; Wendel, J.F. A novel approach for characterizing expression levels of genes dupicated by polyploidy. Genetics 2006, 173, 1823–1827. [Google Scholar] [CrossRef] [PubMed]
- Lukens, L.N.; Pires, J.C.; Leon, E.; Vogelzang, R.; Oslach, L.; Osborn, T. Patterns of sequence loss and cytosine methylation within a population of newly resynthesized Brassica napus allopolyploids. Plant Physiol. 2006, 140, 336–348. [Google Scholar] [CrossRef] [PubMed]
- Small, R.I.; Ryburn, J.A.; Wendel, J.F. Low levels of nucleotide diversity at homeologous Adh loci in allotetraploid cotton (Gossypium L.). Mol. Biol. Evolut. 1999, 16, 491–501. [Google Scholar] [CrossRef]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evolut. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed]
- Augustine, R.; Mukhopadhyay, A.; Bisht, N.C. Targeted silencing of BjMYB28 transcription factor gene directs development of low glucosinolate lines in oilseed Brassica juncea. Plant Biotechnol. J. 2013, 11, 855–866. [Google Scholar] [CrossRef] [PubMed]
- Robin, A.H.; Yi, G.E.; Laila, R.; Yang, K.; Park, J.I.; Kim, H.R.; Nou, I.S. Expression profiling of glucosinolate biosynthetic genes in Brassica oleracea l. Var. Capitata inbred lines reveals their association with glucosinolate content. Molecules 2016, 21, 787. [Google Scholar] [CrossRef] [PubMed]
- Bisht, N.C.; Gupta, V.; Ramchiary, N.; Sodhi, Y.S.; Mukhopadhyay, A.; Arumugam, N.; Pental, D.; Pradhan, A.K. Fine mapping of loci involved with glucosinolate biosynthesis in oilseed mustard (Brassica juncea) using genomic information from allied species. Theor. Appl. Genet. 2009, 118, 413–421. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Long, Y.; Shi, L.; Shi, J.; Barker, G.; Meng, J. Characterization of metabolite quantitative trait loci and metabolic networks that control glucosinolate concentration in the seeds and leaves of Brassica napus. New Phytol. 2012, 193, 96–108. [Google Scholar] [CrossRef] [PubMed]
- Harper, A.L.; Trick, M.; Higgins, J.; Fraser, F.; Clissold, L.; Wells, R.; Hattori, C.; Werner, P.; Bancroft, I. Associative transcriptomics of traits in the polyploid crop species Brassica napus. Nat. Biotechnol. 2012, 30, 798–802. [Google Scholar] [CrossRef] [PubMed]
- Qu, C.M.; Li, S.M.; Duan, X.J.; Fan, J.H.; Jia, L.D.; Zhao, H.Y.; Lu, K.; Li, J.N.; Xu, X.F.; Wang, R. Identification of candidate genes for seed glucosinolate content using association mapping in Brassica napus L. Genes 2015, 6, 1215–1229. [Google Scholar] [CrossRef] [PubMed]
- Rout, K.; Sharma, M.; Gupta, V.; Mukhopadhyay, A.; Sodhi, Y.S.; Pental, D.; Pradhan, A.K. Deciphering allelic variations: For seed glucosinolate traits in oilseed mustard (Brassica juncea) using two bi-parental mapping populations. Theor. Appl. Genet. 2015, 128, 657–666. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are available from the authors. |

| Species | Gene Name | Gene ID | CDS Length (bp) |
|---|---|---|---|
| A. thaliana (diploid) | AtMYB28 | AT5G61420 | 1101 |
| AtMYB29 | AT5G07690 | 1011 | |
| AtMYB34 | AT5G60890 | 888 | |
| AtMYB51 | AT1G18570 | 1059 | |
| AtMYB76 | AT5G07700 | 1017 | |
| AtMYB122 | AT1G74080 | 1002 | |
| B. rapa (diploid) | BrMYB28.1 | Bra012961 | 1065 |
| BrMYB28.2 | Bra035929 | 1074 | |
| BrMYB28.3 | Bra029311 | 1119 | |
| BrMYB29.1 | Bra005949 | 993 | |
| BrMYB29.2 | Bra009245 | 267 * | |
| BrMYB34.1 | Bra035954 | 909 | |
| BrMYB34.2 | Bra013000 | 951 | |
| BrMYB34.3 | Bra029349 | 843 | |
| BrMYB34.4 | Bra029350 | 930 | |
| BrMYB51.1 | Bra031035 | 963 | |
| BrMYB51.2 | Bra016553 | 1002 | |
| BrMYB51.3 | Bra025666 | 1026 | |
| BrMYB122.1 | Bra015939 | 981 | |
| BrMYB122.2 | Bra008131 | 1005 | |
| B. oleracea (diploid) | BolMYB28.1 | XM_013766140 | 1059 |
| BolMYB28.2 | XM_013750838 | 1083 | |
| BolMYB28.3 | XM_013738078 | 1068 | |
| BolMYB28.4 | Bol036743 | 426 * | |
| BolMYB29.1 | Bol043899 | 324 * | |
| BolMYB29.2 | XM_013771134 | 993 | |
| BolMYB34.1 | Bol007760 | 843 | |
| BolMYB34.2 | Bol017062 | 951 | |
| BolMYB34.3 | XM_013754685 | 882 | |
| BolMYB51.1 | Bol030761 | 990 | |
| BolMYB51.2 | Bol013207 | 1002 | |
| BolMYB122 | Bol026204 | 981 | |
| B. nigra (diploid) | BniMYB28.1 | JX947841 | 1053 |
| BniMYB28.2 | JX947842 | 1095 | |
| B. juncea (allopolyploid) | BjMYB28.1 | JQ666166 | 1053 |
| BjMYB28.2 | JQ666167 | 1095 | |
| BjMYB28.3 | JQ666168 | 1065 | |
| BjMYB28.4 | JQ666169 | 1065 | |
| BjMYB29.1 | JX316031 | 993 | |
| BjMYB29.2 | JX316032 | 1020 | |
| B. napus (allopolyploid) | BnMYB28.1 | GSBRNA2T00040913001 | 1011 |
| BnMYB28.2 | GSBRNA2T00146148001 | 420 *,1 | |
| BnMYB28.3 | GSBRNA2T00146147001 | 555 * | |
| BnMYB28.4 | GSBRNA2T00113547001 | 987 | |
| BnMYB29.1 | GSBRNA2T00092496001 | 867 * | |
| BnMYB29.2 | GSBRNA2T00134508001 | 966 | |
| BnMYB29.3 | GSBRNA2T00136149001 | 1005 * | |
| BnMYB29.4 | GSBRNA2T00129660001 | 948 | |
| BnMYB34.1 | GSBRNA2T00052239001 | 843 | |
| BnMYB34.2 | GSBRNA2T00003879001 | 948 | |
| BnMYB34.3 | GSBRNA2T00075530001 | 843 | |
| BnMYB34.4 | GSBRNA2T00075529001 | 918 | |
| BnMYB34.5 | GSBRNA2T00146117001 | 882 | |
| BnMYB34.6 | GSBRNA2T00071741001 | 867 | |
| BnMYB34.7 | GSBRNA2T00094719001 | 909 | |
| BnMYB34.8 | GSBRNA2T00113599001 | 951 | |
| BnMYB51.1 | GSBRNA2T00136837001 | 963 | |
| BnMYB51.2 | GSBRNA2T00139596001 | 1002 | |
| BnMYB51.3 | GSBRNA2T00139597001 | 480 *,1 | |
| BnMYB51.4 | GSBRNA2T00006526001 | 990 | |
| BnMYB51.5 | GSBRNA2T00016192001 | 963 | |
| BnMYB51.6 | GSBRNA2T00102930001 | 1002 | |
| BnMYB51.7 | GSBRNA2T00035671001 | 309 * | |
| BnMYB51.8 | GSBRNA2T00035672001 | 477 *,1 | |
| BnMYB122.1 | GSBRNA2T00088839001 | 870 * | |
| BnMYB122.2 | GSBRNA2T00103485001 | 1113 | |
| BnMYB122.3 | GSBRNA2T00082812001 | 960 | |
| BnMYB122.4 | GSBRNA2T00102693001 | 981 | |
| BnMYB122.5 | GSBRNA2T00092372001 | 957 |
| Allopolyploid | Diploid | ||||||
|---|---|---|---|---|---|---|---|
| BrMYB28.1 | BrMYB28.2 | BrMYB28.3 | BniMYB28.1 | BniMYB28.2 | |||
| BjMYB28.1 | 92.16 | 97.06 | 97.06 | 100 | 97.06 | ||
| BjMYB28.2 | 95.1 | 100 | 98.04 | 97.06 | 100 | ||
| BjMYB28.3 | 93.14 | 98.04 | 100 | 97.06 | 98.04 | ||
| BjMYB28.4 | 99.02 | 96.08 | 94.12 | 93.14 | 96.08 | ||
| BrMYB28.1 | BrMYB28.2 | BrMYB28.3 | BolMYB28.2 | BolMYB28.3 | |||
| BnMYB28.1 | 99.02 | 96.08 | 94.12 | 96.08 | 100 | ||
| BnMYB28.2 | 95.1 | 100 | 98.04 | 100 | 96.08 | ||
| BnMYB28.4 | 99.02 | 96.08 | 94.12 | 96.08 | 100 | ||
| BrMYB29.1 | BolMYB29.2 | ||||||
| BnMYB29.2 | 97.06 | 99.02 | |||||
| BnMYB29.4 | 100 | 96.08 | |||||
| BrMYB34.1 | BrMYB34.2 | BrMYB34.3 | BrMYB34.4 | BolMYB34.1 | BolMYB34.2 | BolMYB34.3 | |
| BnMYB34.1 | 90.2 | 94.12 | 96.08 | 91.18 | 100 | 92.16 | 91.18 |
| BnMYB34.2 | 93.14 | 98.04 | 93.14 | 90.2 | 92.16 | 98.04 | 94.12 |
| BnMYB34.3 | 90.2 | 94.12 | 98.04 | 90.2 | 96.08 | 92.16 | 91.18 |
| BnMYB34.4 | 89.22 | 92.16 | 91.18 | 98.04 | 91.18 | 90.2 | 90.2 |
| BnMYB34.5 | 98.04 | 96.08 | 92.16 | 90.2 | 91.18 | 94.12 | 100 |
| BnMYB34.6 | 90.2 | 93.14 | 92.16 | 99.02 | 92.16 | 91.18 | 91.18 |
| BnMYB34.7 | 100 | 95.1 | 91.18 | 89.22 | 90.2 | 93.14 | 98.04 |
| BnMYB34.8 | 95.1 | 100 | 95.1 | 92.16 | 94.12 | 98.04 | 96.08 |
| BrMYB51.1 | BrMYB51.2 | BrMYB51.3 | BolMYB51.1 | BolMYB51.2 | |||
| BnMYB51.1 | 99.02 | 97.06 | 94.12 | 94.12 | 97.06 | ||
| BnMYB51.2 | 97.06 | 98.04 | 93.14 | 93.14 | 100 | ||
| BnMYB51.3 | 97.06 | 99.02 | 93.14 | 93.14 | 99.02 | ||
| BnMYB51.4 | 95.1 | 93.14 | 100 | 100 | 93.14 | ||
| BnMYB51.5 | 100 | 96.08 | 95.1 | 95.1 | 97.06 | ||
| BnMYB51.6 | 96.08 | 100 | 93.14 | 93.14 | 98.04 | ||
| BnMYB51.8 | 95.1 | 93.14 | 100 | 100 | 93.14 | ||
| BrMYB122.1 | BrMYB122.2 | BolMYB122 | |||||
| BnMYB122.2 | 100 | 94.12 | 100 | ||||
| BnMYB122.3 | 95.1 | 97.06 | 95.1 | ||||
| BnMYB122.4 | 100 | 94.12 | 100 | ||||
| BnMYB122.5 | 94.12 | 100 | 94.12 | ||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Seo, M.-S.; Kim, J.S. Understanding of MYB Transcription Factors Involved in Glucosinolate Biosynthesis in Brassicaceae. Molecules 2017, 22, 1549. https://doi.org/10.3390/molecules22091549
Seo M-S, Kim JS. Understanding of MYB Transcription Factors Involved in Glucosinolate Biosynthesis in Brassicaceae. Molecules. 2017; 22(9):1549. https://doi.org/10.3390/molecules22091549
Chicago/Turabian StyleSeo, Mi-Suk, and Jung Sun Kim. 2017. "Understanding of MYB Transcription Factors Involved in Glucosinolate Biosynthesis in Brassicaceae" Molecules 22, no. 9: 1549. https://doi.org/10.3390/molecules22091549
APA StyleSeo, M.-S., & Kim, J. S. (2017). Understanding of MYB Transcription Factors Involved in Glucosinolate Biosynthesis in Brassicaceae. Molecules, 22(9), 1549. https://doi.org/10.3390/molecules22091549






