Synthesis and Biological Activity of Novel O-Alkyl Derivatives of Naringenin and Their Oximes
Abstract
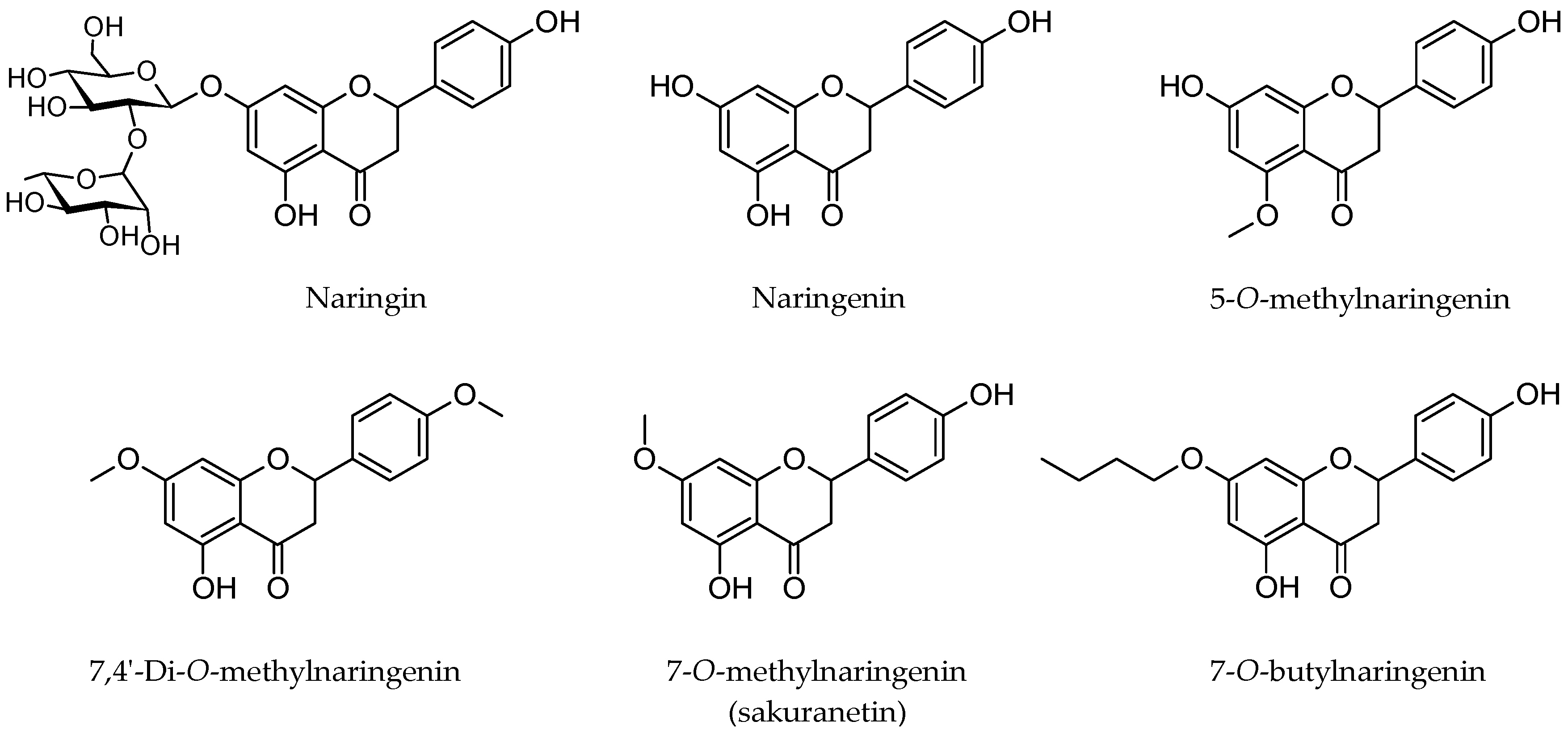
:1. Introduction
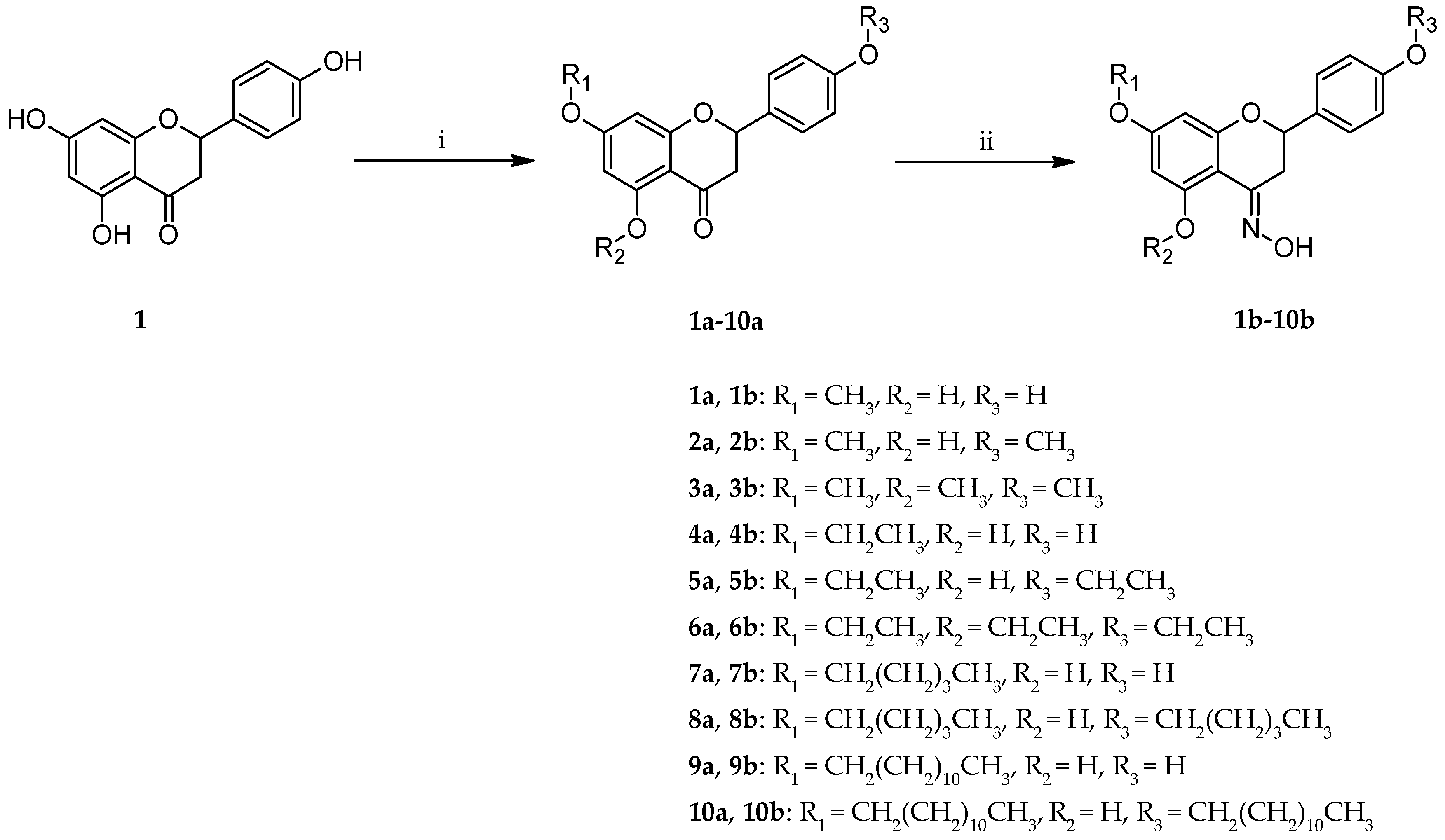
2. Results and Discussion
3. Materials and Methods
3.1. Chemicals
3.2. Analysis
3.3. Synthesis of O-Alkyl Derivatives of Naringenin
3.3.1. Synthesis of Mono- (1a, 4a, 7a, 9a) and Di-O-alkyl Derivatives of Naringenin (2a, 5a, 8a, 10a)
3.3.2. Synthesis of Tri-O-alkyl Derivatives of Naringenin (3a, 6a)
3.4. Synthesis of Oximes (1b–10b)
3.5. Biological Activity
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Tripoli, E.; Guardia, M.; Giammanco, S.; Majo, D.; Giammanco, M. Citrus flavonoids: Molecular structure, biological activity and nutritional properties: A review. Food Chem. 2007, 104, 466–479. [Google Scholar] [CrossRef]
- Walle, T. Absorption and metabolism of flavonoids. Free Radic. Biol. Med. 2004, 36, 829–837. [Google Scholar] [CrossRef] [PubMed]
- Erlund, I. Review of the flavonoids quercetin, hesperetin, and naringenin. Dietary sources, bioactivities, bioavailability, and epidemiology. Nutr. Res. 2004, 24, 851–874. [Google Scholar] [CrossRef]
- Fuhr, U.; Klittich, K.; Stain, A.H. Inhibitory effect of grapefruit juice and its bitter principal, naringenin, on CYP1A2 dependent metabolism of caffeine in man. Br. J. Clin. Pharma. 1993, 35, 431–436. [Google Scholar] [CrossRef]
- Woo, Y.; Shin, S.Y.; Hyun, J.; Lee, S.D.; Lee, Y.H.; Lim, Y. Flavanones inhibit the clonogenicity of HCT116 cololectal cancer cells. Int. J. Mol. Med. 2012, 29, 403–408. [Google Scholar] [CrossRef] [PubMed]
- Hoang, T.K.D.; Huynh, T.K.C.; Nguyen, R.D. Synthesis, characterization, anti-inflammatory and anti-proliferative activity against MCF-7 cells of O-alkyl and O-acyl flavonoid derivatives. Bioorg. Chem. 2015, 63, 45–52. [Google Scholar] [CrossRef] [PubMed]
- Silva, S.A.S.; Fatima Agra, M.; Tavares, J.F.; Cunha, E.V.L.; Barbosa-Filho, J.M.; Silva, M.S. Flavanones from aerial parts of Cordia globosa (Jacq.) Kunth, Boraginaceae. Rev. Bras. Farmacogn. Braz. J. Pharmacogn. 2010, 20, 682–685. [Google Scholar] [CrossRef]
- Nobakht, M.; Grkovic, T.; Trueman, S.J.; Wallace, H.M.; Katouli, M.; Quinn, R.J.; Brooks, P.R. Chemical Constituents of Kino Extract from Corymbia torelliana. Molecules 2014, 19, 17862–17871. [Google Scholar] [CrossRef] [PubMed]
- Drira, R.; Sakamoto, K. Sakuranetin Induces Melanogenesis in B16BL6 Melanoma Cells through Inhibition of ERK and PI3K/AKT Signaling Pathways. Phytother. Res. 2016, 30, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Sakoda, C.P.P.; Toledo, C.A.; Perini, A.; Pinheiro, N.M.; Hiyane, M.I.; Santos Grecco, S.; Fátima Lopes Calvo Tibério, I.; Câmara, N.O.S.; Arruda Martins, M.; Lago, J.H.G.; et al. Sakuranetin reverses vascular peribronchial and lung parenchyma remodeling in a murine model of chronic allergic pulmonary inflammation. Acta Histochem. 2016, 118, 615–624. [Google Scholar] [CrossRef] [PubMed]
- Lee, E.R.; Kang, Y.J.; Kim, H.J.; Choi, H.Y.; Kang, G.H.; Kim, J.H.; Kim, B.W.; Jeong, H.S.; Park, Y.S.; Cho, S.G. Regulation of Apoptosis by Modified Naringenin Derivatives in Human Colorectal Carcinoma RKO Cells. J. Cell. Biochem. 2008, 104, 259–273. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.A.; Moon, S.H.; Lee, J.Y.; Kim, K.T.; Park, Y.S.; Paik, H.D. Antibacterial Activity of a Novel Flavonoid, 7-O-Butyl Naringenin, against Methicillin-Resistant Staphylococcus aureus (MRSA). Food Sci. Biotechnol. 2013, 22, 1725–1728. [Google Scholar] [CrossRef]
- Emami, S.; Falahati, M.; Banifatemi, A.; Amanlou, M.; Shafiee, A. (E)- and (Z)-1,2,4-Triazolylchromanone oxime ethers as conformationally constrained antifungals. Bioorg. Med. Chem. 2004, 12, 3971–3976. [Google Scholar] [CrossRef] [PubMed]
- Emami, S.; Falahati, M.; Banifatemi, A.; Moshiri, K.; Shafiee, A. Stereoselective Synthesis and in Vitro Antifungal Evaluation of (E)- and (Z)-Imidazolylchromanone Oxime Ethers. Arch. Pharm. Pharm. Med. Chem. 2002, 7, 318–324. [Google Scholar] [CrossRef]
- Türkkan, B.; Özyürek, M.; Bener, M.; Güclü, K.; Apak, R. Synthesis, characterization and antioxidant capacity of naringenin-oxime. Spectrochim. Acta A 2012, 85, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Özyurek, M.; Akpınar, D.; Bener, M.; Türkkan, B.; Güclü, K.; Apak, R. Novel oxime based flavanone, naringin-oxime: Synthesis, characterization and screening for antioxidant activity. Chem.-Biol. Interact. 2014, 212, 40–46. [Google Scholar] [CrossRef] [PubMed]
- Potaniec, B.; Grabarczyk, M.; Stompor, M.; Szumny, A.; Zieliński, P.; Żołnierczyk, A.K.; Anioł, M. Antioxidant activity and spectroscopic data of isoxanthohumol oxime and related compounds. Spectrochim. Acta A 2014, 118, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Kocyigit, A.; Koyuncu, I.; Dikilitas, M.; Bahadori, F.; Turkkan, B. Cytotoxic, genotoxic and apoptotic effects of naringenin-oxime relative to naringenin on normal and cancer cell lines. Asian Pac. J. Trop. Biomed. 2016, 6, 872–880. [Google Scholar] [CrossRef]
- Yoon, H.; Kim, T.W.; Shin, S.Y.; Park, M.J.; Yong, Y.; Kim, D.W.; Islam, T.; Lee, Y.H.; Jung, K.Y.; Lim, Y. Design, synthesis and inhibitory activities of naringenin derivatives on human colon cancer cells. Bioorg. Med. Chem. Lett. 2013, 23, 232–238. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.Y.; Wang, T.C.; Lee, C.H.; Chen, C.S.; Wang, S.H.; Lin, Y.C.; Juang, S.H. WTC-01, a novel synthetic oxime-flavone compound, destabilizes microtubules in human nasopharyngeal carcinoma cells in vitro and in vivo. Br. J. Pharmacol. 2015, 172, 4671–4683. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.C.; Chen, I.L.; Lu, C.M.; Kuo, D.H.; Liao, C.H. Synthesis, and Cytotoxic and Antiplatelet of Oxime- and Methyloxime-Containing Flavone, Isoflavone, and Xanthone Derivatives. Chem. Biodivers. 2005, 2, 253–263. [Google Scholar] [CrossRef] [PubMed]
- Yenjai, C.; Wanich, S. Cytotoxicity against KB and NCI-H187 cell lines of modified flavonoids from Kaempferia parviflora. Bioorg. Med. Chem. Lett. 2010, 20, 2821–2823. [Google Scholar] [CrossRef] [PubMed]
- Cushnie, T.P.T.; Lamb, A.J. Antimicrobial activity of flavonoids. Int. J. Antimicrob. Ag. 2005, 26, 343–356. [Google Scholar] [CrossRef]
- Deka, N.; Mariotte, A.M.; Boumendje, A. Microwave mediated solvent-free acetylation of deactivated and hindered phenols. Green Chem. 2001, 3, 263–264. [Google Scholar] [CrossRef]
- Feng, L.; Maddox, M.M.; Alam, M.Z.; Tsutsumi, L.S.; Narula, G.; Bruhn, D.F.; Wu, X.; Sandhaus, S.; Lee, R.B.; Simmons, C.J.; et al. Synthesis, structure-activity relationship studies, and antibacterial evaluation of 4-chromanones and chalcones, as well as olympicin A and derivatives. J. Med. Chem. 2014, 57, 8398–8420. [Google Scholar] [CrossRef] [PubMed]
- Yenjai, C.; Wanich, S.; Pitchuanchom, S.; Sripanidkulchai, B. Structural Modification of 5,7-Dimethoxyflavone from Kaempferia parviflora and biological activities. Arch. Pharm. Res. 2009, 32, 1179–1184. [Google Scholar] [CrossRef] [PubMed]
- Suksamrarn, A.; Chotipong, A.; Suavansri, T.; Boongird, S.; Timsuksai, P.; Vimuttipong, S.; Chuaynugul, A. Antimycobacterial activity and cytotoxicity of flavonoids from the flowers of Chromolaena odorata. Arch. Pharm. Res. 2004, 27, 507–511. [Google Scholar] [CrossRef] [PubMed]
- Bae, E.A.; Han, M.J.; Kim, D.H. In Vitro Anti-Helicobacter Pylori Activity of Some Flavonoids and Their Metabolites. Planta Med. 1999, 65, 442–443. [Google Scholar] [CrossRef] [PubMed]
- Ilboudo, O.; Bonzi, S.; Tapsoba, I.; Somda, I.; Bonzi-Coulibaly, Y.L. In vitro antifungal activity of flavonoid diglycosides of Mentha piperita and their oxime derivatives against two cereals fungi. CR Chim. 2016, 19, 857–862. [Google Scholar] [CrossRef]
- Oyama, K.; Kondo, T. Total Synthesis of Flavocommelin, a Component of the Blue Supramolecular Pigment from Commelina communis, on the Basis of Direct 6-C-Gycosylation of Flavan. J. Org. Chem. 2004, 69, 5240–5246. [Google Scholar] [CrossRef] [PubMed]
- Righi, G.; Silvestri, I.P.; Barontini, M.; Crisante, F.; Di Manno, A.; Pelagalli, R.; Bovicelli, P. Efficient synthesis of scutellarein. Nat. Prod. Res. 2012, 26, 1278–1284. [Google Scholar] [CrossRef] [PubMed]
- Aritomi, M.; Kawasaki, T. Dehydro-para-asebotin, a New Chalcone Glucoside in the Flowers of Gnaphalium affine D. DON. Chem. Pharm. Bull. 1974, 22, 1800–1805. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds naringenin, naringenin oxime, 1a–10a and 1b–10b are available from the authors. |
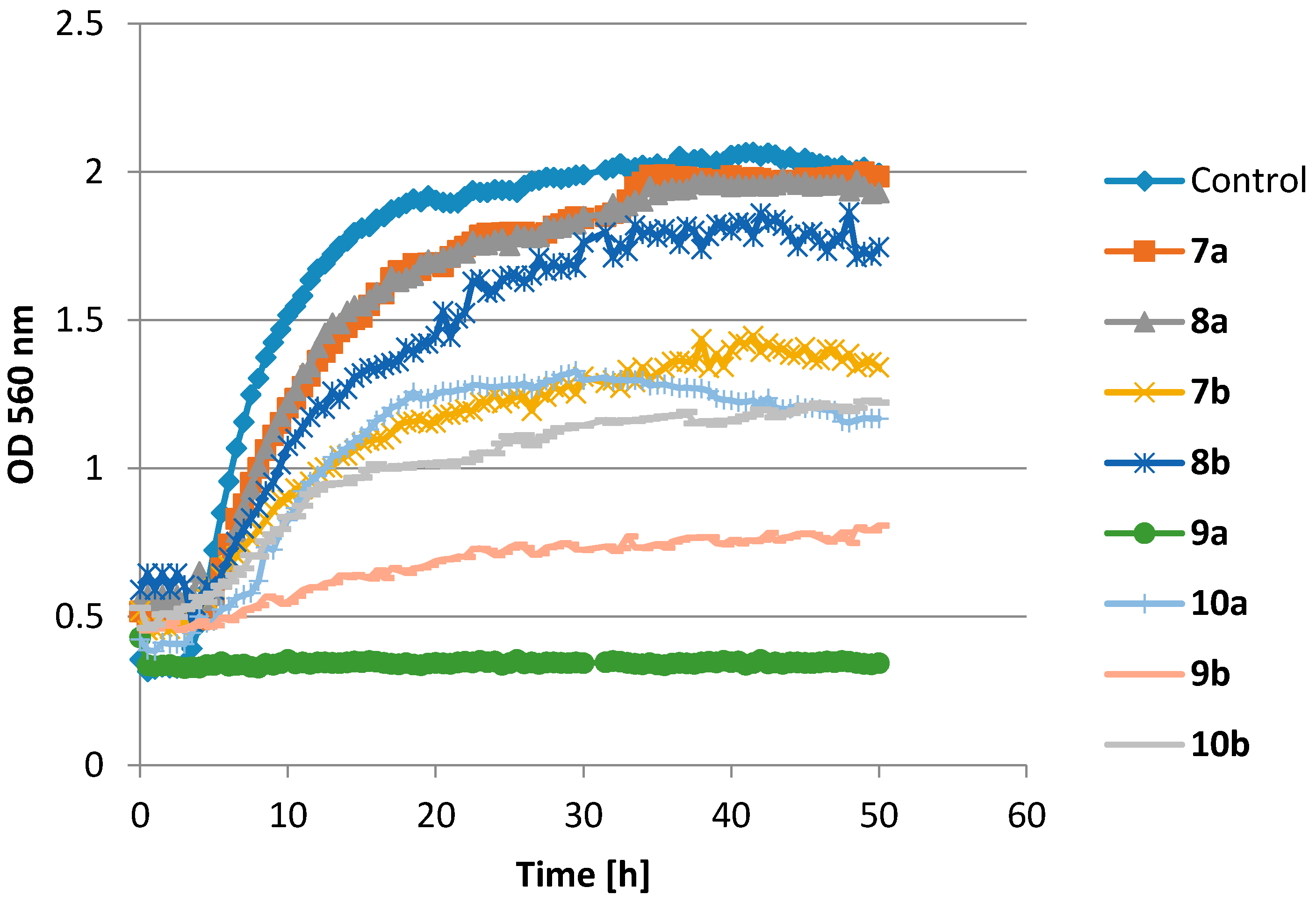

| Strain | E. coli | S. aureus | C. albicans | A. alternata | F. linii | A. niger | |
|---|---|---|---|---|---|---|---|
| Control | Lag-phase (h) | 4.0 | 2.5 | 3.0 | 16.5 | 14.5 | 11.0 |
| ∆OD | 1.65 | 1.74 | 1.60 | 1.87 | 1.96 | 2.14 | |
| NG | Lag-phase (h) | 15.0 | 4.5 | 5.0 | 20.0 | - | 5.5 |
| ∆OD | 1.30 | 1.49 | 1.50 | 1.34 | 0 | 1.74 | |
| 1a | Lag-phase (h) | 5.5 | 3.5 | 5.0 | 21.5 | 26.0 | 9.0 |
| ∆OD | 0.75 | 1.59 | 1.45 | 0.72 | 1.17 | 1.55 | |
| 2a | Lag-phase (h) | 4.0 | 4.0 | 4.0 | 16.0 | 9.5 | 12.5 |
| ∆OD | 0.63 | 1.73 | 1.09 | 0.49 | 0.72 | 1.49 | |
| 3a | Lag-phase (h) | 4.5 | 4.0 | 5.0 | 14.0 | 19.5 | 15.0 |
| ∆OD | 0.53 | 1.73 | 1.57 | 1.31 | 1.81 | 1.21 | |
| 4a | Lag-phase (h) | 4.0 | 2.5 | 5.5 | 25.0 | 26.5 | 6.5 |
| ∆OD | 0.52 | 1.65 | 1.35 | 0.46 | 1.18 | 1.36 | |
| 5a | Lag-phase (h) | 4.5 | 5.0 | 5.5 | 18.0 | 24.0 | 7.5 |
| ∆OD | 0.51 | 1.64 | 0.93 | 0.48 | 0.88 | 1.03 | |
| 6a | Lag-phase (h) | 5.0 | 2.5 | 5.5 | 19.5 | 14.0 | 13.0 |
| ∆OD | 0.24 | 1.07 | 0.50 | 0.47 | 0.52 | 0.54 | |
| 7a | Lag-phase (h) | 4.0 | 4.0 | 7.0 | 19.0 | 25.5 | 32.5 |
| ∆OD | 0.51 | 1.67 | 1.19 | 1.00 | 1.44 | 1.00 | |
| 8a | Lag-phase (h) | - | 4.5 | 6.5 | 9.5 | 34.0 | - |
| ∆OD | 0 | 1.46 | 1.00 | 0.47 | 0.33 | 0 | |
| 9a | Lag-phase (h) | - | 26.0 | 5.5 | 23.0 | 3.5 | 38.5 |
| ∆OD | 0 | 0.83 | 0.97 | 0.42 | 0.98 | 0.96 | |
| 10a | Lag-phase (h) | 3.0 | 3.0 | 0.5 | 33.0 | - | 6.0 |
| ∆OD | 0.23 | 0.91 | 1.19 | 1.63 | 0 | 1.58 | |
| Strain | E. coli | S. aureus | C. albicans | A. alternata | F. linii | A. niger | |
|---|---|---|---|---|---|---|---|
| Control | Lag-phase (h) | 4.0 | 2.5 | 3.0 | 16.5 | 14.5 | 11.0 |
| ∆OD | 1.65 | 1.74 | 1.60 | 1.87 | 1.96 | 2.14 | |
| NG-OX | Lag-phase (h) | - | 3.5 | 4.0 | 21.5 | 29.0 | - |
| ∆OD | 0 | 1.66 | 1.69 | 0.96 | 1.20 | 0 | |
| 1b | Lag-phase (h) | 5.0 | 4.0 | 5.0 | 37.5 | 26.5 | 45.5 |
| ∆OD | 0.73 | 1.46 | 1.30 | 0.69 | 1.41 | 0.49 | |
| 2b | Lag-phase (h) | 3.5 | 2.0 | 4.0 | 16.0 | 13.0 | 4.0 |
| ∆OD | 0.82 | 1.46 | 0.68 | 0.77 | 0.62 | 0.59 | |
| 3b | Lag-phase (h) | 4.0 | 2.5 | 10.0 | 11.5 | 11.0 | 10.0 |
| ∆OD | 0.74 | 1.90 | 1.29 | 1.10 | 1.51 | 1.10 | |
| 4b | Lag-phase (h) | 4.0 | 2.5 | 3.0 | 16.0 | 12.0 | 11.0 |
| ∆OD | 0.80 | 1.20 | 0.81 | 1.03 | 0.92 | 0.88 | |
| 5b | Lag-phase (h) | 4.5 | 2.0 | 4.0 | 19.0 | 13.0 | 5.0 |
| ∆OD | 0.29 | 0.95 | 0.41 | 0.51 | 0.55 | 0.40 | |
| 6b | Lag-phase (h) | 6.0 | 4.5 | 11.0 | 11.5 | 10.5 | 9.5 |
| ∆OD | 0.30 | 1.29 | 1.30 | 0.69 | 1.47 | 0.76 | |
| 7b | Lag-phase (h) | 5.0 | 2.0 | 3.0 | 16.0 | 13.0 | 11.0 |
| ∆OD | 0.45 | 0.97 | 0.77 | 1.02 | 0.82 | 0.83 | |
| 8b | Lag-phase (h) | - | 3.5 | 6.0 | 18.5 | 22.0 | 37.5 |
| ∆OD | 0 | 1.27 | 0.87 | 1.24 | 0.53 | 1.05 | |
| 9b | Lag-phase (h) | - | 4.5 | 3.0 | 34.5 | - | 10.5 |
| ∆OD | 0 | 0.35 | 0.22 | 0.54 | 0 | 0.58 | |
| 10b | Lag-phase (h) | 0.5 | 1.0 | 1.0 | 31.5 | - | 7.0 |
| ∆OD | 0.27 | 0.80 | 1.03 | 1.24 | 0 | 1.71 | |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kozłowska, J.; Potaniec, B.; Żarowska, B.; Anioł, M. Synthesis and Biological Activity of Novel O-Alkyl Derivatives of Naringenin and Their Oximes. Molecules 2017, 22, 1485. https://doi.org/10.3390/molecules22091485
Kozłowska J, Potaniec B, Żarowska B, Anioł M. Synthesis and Biological Activity of Novel O-Alkyl Derivatives of Naringenin and Their Oximes. Molecules. 2017; 22(9):1485. https://doi.org/10.3390/molecules22091485
Chicago/Turabian StyleKozłowska, Joanna, Bartłomiej Potaniec, Barbara Żarowska, and Mirosław Anioł. 2017. "Synthesis and Biological Activity of Novel O-Alkyl Derivatives of Naringenin and Their Oximes" Molecules 22, no. 9: 1485. https://doi.org/10.3390/molecules22091485
APA StyleKozłowska, J., Potaniec, B., Żarowska, B., & Anioł, M. (2017). Synthesis and Biological Activity of Novel O-Alkyl Derivatives of Naringenin and Their Oximes. Molecules, 22(9), 1485. https://doi.org/10.3390/molecules22091485






