Non-Polar Natural Products from Bromelia laciniosa, Neoglaziovia variegata and Encholirium spectabile (Bromeliaceae)
Abstract
:1. Introduction
1.1. Bromeliaceae
1.2. B. laciniosa
1.3. N. variegata
1.4. E. spectabile
2. Results and Discussion
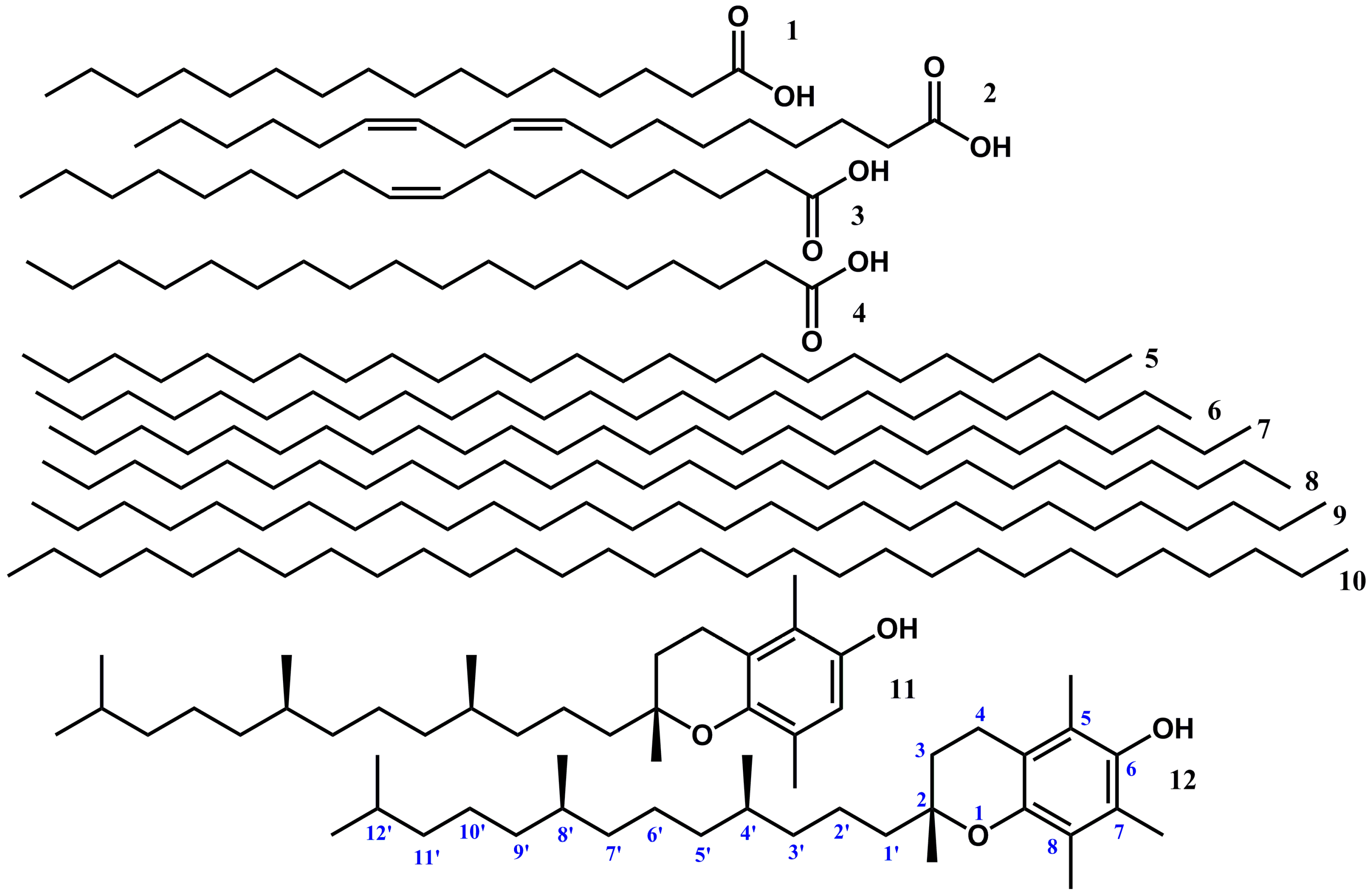
2.1. Fatty Acids and Their Derivatives
2.2. Very Long-Chain Alkanes (VLCA)
2.3. Vitamins
2.4. Other Compounds
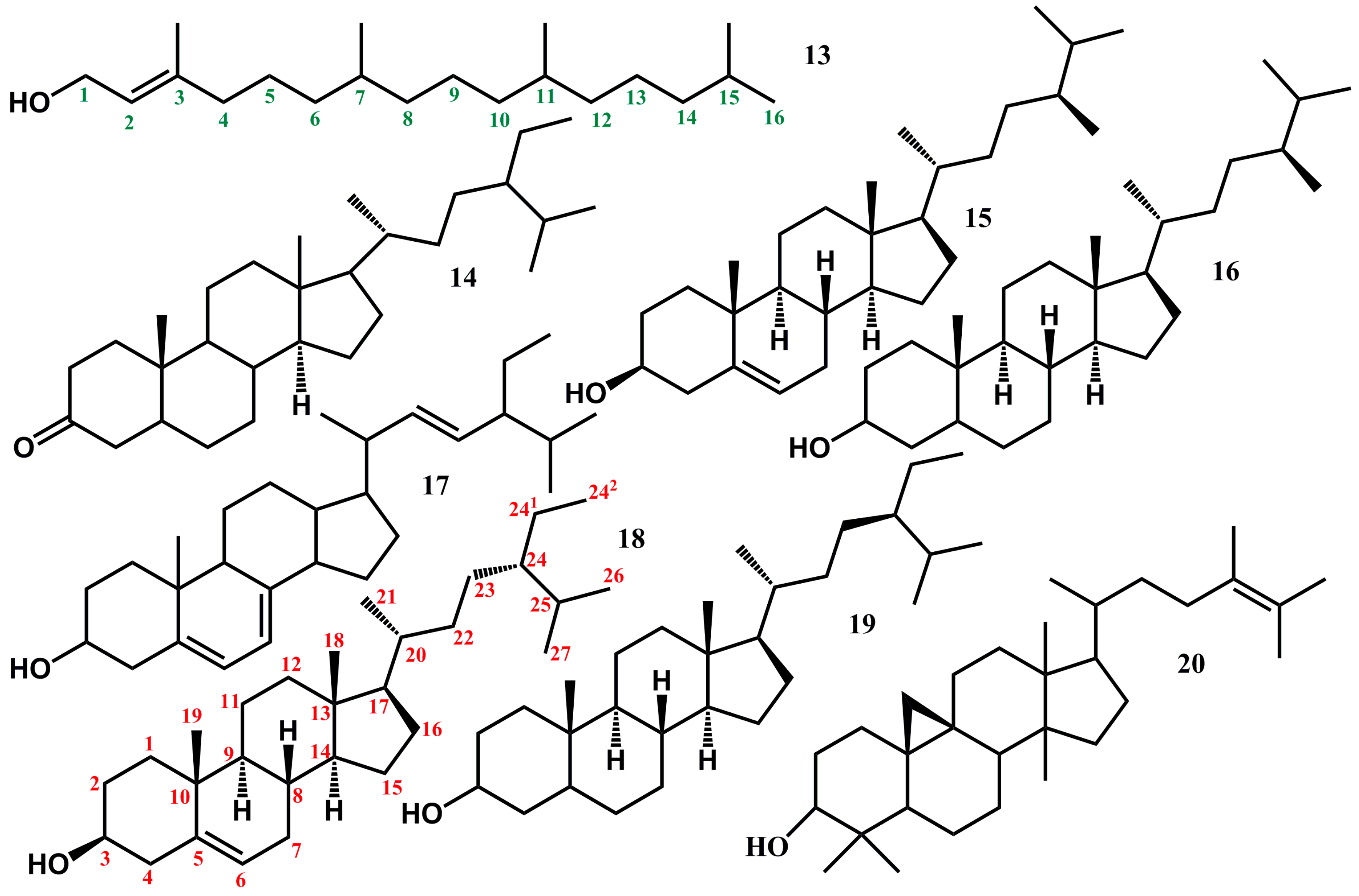
2.5. Triterpenoids and Derivatives Therefrom
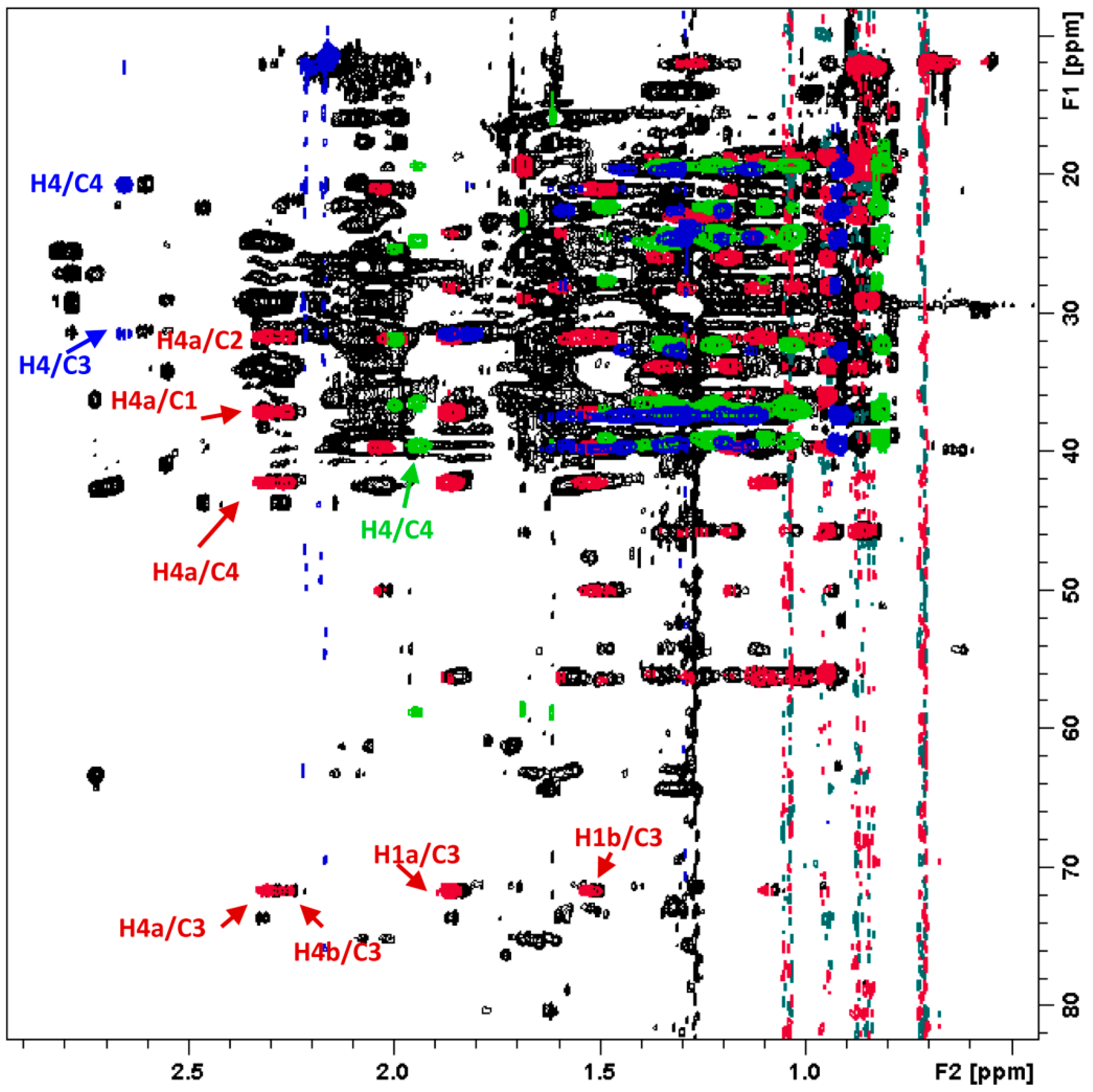
2.6. NMR Plant Metabolomics
3. Materials and Methods
3.1. Plant Material
3.2. Extraction and Concentration
3.3. GC-FID
3.4. GC-MS (TOF)
3.5. NMR Spectroscopy
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Givnish, T.J.; Barfuss, M.H.J.; Van Ee, B.; Riina, R.; Schulte, K.; Horres, R.; Gonsiska, P.A.; Jabaily, R.S.; Crayn, D.M.; Smith, J.A.C.; et al. Phylogeny, adaptive radiation, and historical biogeography in Bromeliaceae: Insights from an eight-locus plastid phylogeny. Am. J. Bot. 2011, 98, 872–895. [Google Scholar] [CrossRef] [PubMed]
- Manera, G.; Nunes, W. Convivendo com a Seca: Plantas Forrageiras. Ed. UEFS: Feira de Santana, Brazil, 2001; pp. 7–8. [Google Scholar]
- Angelim, A.E.S.; De Moraes, J.P.S.; Da Silva, J.A.B.; Gervásio, R.d.C.R.G. Germinação e aspectos morfológicos de plantas de macambira (Bromelia laciniosa), encontradas na região do vale do são francisco [Germination and morphological aspects of macambira (Bromelia laciniosa), found in the region of Vale do São Francisco]. Rev. Bras. Biocienc. 2007, 5, 1065–1067. [Google Scholar]
- Dutra, A.S.; Teófilo, E.M.; Filho, S.M. Germinação de sementes de macambira (Bromelia laciniosa mart. Ex schult) [Germination of macambira (Bromelia laciniosa mart. Ex schult. & schult. F.) seeds]. Rev. Caatinga 2010, 23, 12–17. [Google Scholar]
- Santo, F.D.S.D.E.; Maciel, J.R.; de Siqueira Filho, J.A. Impacto da herbivoria por caprinos sobre as populacos naturais de Bromelia laciniosa mart. Ex schult. F. (bromeliaceae) [Impact of herbivory by goats on natural populations of Bromelia laciniosa mart. Ex. Schult & schult. F.]. Rev. Arvore 2012, 36, 143–149. [Google Scholar]
- De Lima, J.L.S. Plantas Forrageiras das Caatingas, Usos e Potencialidades; Rocha, P., Ed.; EMBRAPA-CPATSA/PNE/RBG-KEW: Petrolina, Brazil, 1996; pp. 1–45. [Google Scholar]
- Agra, M.F.; Baracho, G.S.; Nurit, K.; Basilio, I.J.; Coelho, V.P. Medicinal and poisonous diversity of the flora of “cariri paraibano”, Brazil. J. Ethnopharmacol. 2007, 111, 383–395. [Google Scholar] [CrossRef] [PubMed]
- De Albuquerque, U.P.; De Medeiros, P.M.; De Almeida, A.L.S.; Monteiro, J.M.; Neto, E.M.D.F.L.; De Melo, J.G.; Dos Santos, J.P. Medicinal plants of the Caatinga (semi-arid) vegetation of ne Brazil: A quantitative approach. J. Ethnopharmacol. 2007, 114, 325–354. [Google Scholar] [CrossRef] [PubMed]
- Bongiorno de Pfirter, G.M.; Buttazzoni de Cozzarin, M.S.; Caffini, N.O. Otros exudados gomosos en especies argentinas del genero bromelia. I. Las gomas de Bromelia serra gris. Y Bromelia laciniosa mart. (Bromeliaceae). Rev. Farm. 1973, 115, 98–99. [Google Scholar]
- De Oliveira-Júnior, R.G.; De Oliveira, A.P.; Guimarães, A.L.; Araújo, E.C.C.; Braz-Filho, R.; Øvstedal, D.O.; Fossen, T.; da Silva Almeida, J.R.G. The first flavonoid isolated from Bromelia laciniosa (Bromeliaceae). J. Med. Plants Res. 2014, 8, 558–563. [Google Scholar]
- Mayo, S. Neoglaziovia variegata, Bromeliaceae. Curtis’s Bot. Mag. 1992, 9, 124–127. [Google Scholar] [CrossRef]
- D’Almeida, J.R.M.; d’Almeida, A.L.F.S.; de Carvalho, L.H. Mechanical, morphological, and structural characteristics of caroa (Neoglaziovia variegata) fibers. Polym. Polym. Compos. 2008, 16, 589–595. [Google Scholar]
- De Oliveira-Júnior, R.G.; Araújo, C.D.S.; Santana, C.R.R.; Souza, G.R.; de Lima-Saraiva, S.R.G.; Guimarães, A.L.; de Oliveira, A.P.; de Siqueira Filho, J.A.; Pacheco, A.G.M.; da Silva Almeida, J.R.G. Phytochemical screening, antioxidant and antibacterial activity of extracts from the flowers of Neoglaziovia variegata (Bromeliaceae). J. Chem. Pharm. Res. 2012, 4, 4489–4494. [Google Scholar]
- Silveira, D.G.; Santana, J.R.F.; Souza, F.V.D.; Ledo, C.A.S.; Cunha, E.C. Development of micropropagated shoots and plants of caroá in different substrates. Acta Hortic. 2010, 865, 305–313. [Google Scholar] [CrossRef]
- Silveira, D.G.; Souza, F.V.D.; Pelacani, C.R.; Souza, A.D.S.; Ledo, C.A.d.S.; Ferreira de Santana, J.R. Micropropagation and in vitro conservation of Neoglaziovia variegata (arr. Cam.) mez, a fiber producing bromeliad from Brazil. Braz. Arch. Biol. Technol. 2009, 52, 923–932. [Google Scholar] [CrossRef]
- De Lima-Saraiva, S.R.G.; Guimarães, A.L.; de Oliveira, A.P.; Saraiva, H.C.C.; de Oliveira-Junior, R.G.; de Barros, V.R.P.; Menezes, V.G.; de Oliveira, R.A.; Silva, F.S.; de Lima, R.S.; et al. Antioxidant activity and acute toxicity of Neoglaziovia variegata (Bromeliaceae). Afr. J. Biotechnol. 2012, 11, 13998–14006. [Google Scholar] [CrossRef]
- Machado, F.D.F.; Silva, F.V.; Fernandes, H.B.; Freitas, F.F.B.P.; Arcanjo, D.D.R.; Lima, J.T.; da Silva Almeida, J.R.G.; Oliveira, F.A.; Oliveira, R.C.M. Gastroprotective effect of an ethanolic extract from Neoglaziovia variegata (arruda) mez (Bromeliaceae) in rats and mice. Z. Naturforsch. (C) 2013, 68, 97–107. [Google Scholar] [CrossRef]
- Da Silva, V.F.; Franco, I.; Damasceno, T.E.F.; da Silva Almeida, J.R.G.; da Costa, M.M. Antimicrobial potential of ethanol extracts of plants against Gram-negative bacilli isolated from cervicovaginal mucosa of sheep bred in the region of Petrolina-pe. Semin. Cienc. Agrar. 2014, 35, 883–890. [Google Scholar]
- Do Nascimento, V.T.; Vasconcelos, M.A.D.S.; Maciel, M.I.S.; Albuquerque, U.P. Famine foods of Brazil’s seasonal dry forests: Ethnobotanical and nutritional aspects. Econ. Bot. 2012, 66, 22–34. [Google Scholar] [CrossRef]
- De Lima-Saraiva, S.R.G.; Silva, J.C.; Branco, C.R.C.; Branco, A.; Amorim, E.L.C.; da Silva Almeida, J.R.G. Antinociceptive effect of Encholirium spectabile: A Bromeliaceae from the Brazilian Caatinga biome. Pharmacogn. Mag. 2014, 10, 655–660. [Google Scholar]
- Santana, C.R.R.; De Oliveira-Junior, R.G.; Araújo, C.D.S.; Souza, G.R.; De Lima-Saraiva, S.R.G.; Guimarães, A.L.; De Oliveira, A.P.; Filho, J.A.D.S.; Pacheco, A.G.M.; Da Silva Almeida, J.R.G. Phytochemical screening, antioxidant and antibacterial activity of Encholirium spectabile (Bromeliaceae). Int. J. Sci. 2012, 1, 1–19. [Google Scholar]
- De Oliveira-Junior, R.G.; Souza, G.R.; Guimarães, A.L.; de Oliveira, A.P.; Silva Morais, A.C.; Araújo, E.C.D.C.; Nunes, X.P.; da Silva Almeida, J.R.G. Dried extracts of Encholirium spectabile (Bromeliaceae) present antioxidant and photoprotective activities in vitro. J. Young Pharm. 2013, 5, 102–105. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho, K.I.M.; Fernandes, H.B.; Machado, F.D.F.; Oliveira, I.S.; Oliveira, F.A.; Nunes, P.H.M.; Lima, J.T.; da Silva Almeida, J.R.G.; Oliveira, R.C.M. Antiulcer activity of ethanolic extract of Encholirium spectabile mart. Ex schult & schult f. (Bromeliaceae) in rodents. Biol. Res. 2010, 43, 459–465. [Google Scholar] [PubMed]
- Prinsen, P.; Gutiérrez, A.; Faulds, C.B.; del Rio, J.C. Comprehensive study of valuable lipophilic phytochemicals in wheat bran. J. Agric. Food. Chem. 2014, 62, 1664–1673. [Google Scholar] [CrossRef] [PubMed]
- Hargrove, J.L.; Greenspan, P.; Hartle, D.K. Nutritional significance and metabolism of very long chain fatty alcohols and acids from dietary waxes. Exp. Biol. Med. 2004, 229, 215–226. [Google Scholar] [CrossRef]
- Morohashi, K.-I.; Sadano, H.; Okada, Y.; Omura, T. Position specificity in n-hexane hydroxylation by two forms of cytochrome P-450 in rat liver microsomes. J. Biochem. 1983, 93, 413–419. [Google Scholar] [CrossRef] [PubMed]
- Parada, F.; Duque, C. Studies on the aroma of piñuela fruit pulp (Bromelia plumieri): Free and bound volatile composition and characterization of some glucoconjugates as aroma precursors. J. High Resol. Chromatogr. 1998, 21, 577–581. [Google Scholar] [CrossRef]
- Marques, G.; Gutierrez, A.; del Rio, J.C. Chemical characterization of lignin and lipophilic fractions from leaf fibers of curaua (Ananas erectifolius). J. Agric. Food Chem. 2007, 55, 1327–1336. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vetter, W.; Schröder, M.; Lehnert, K. Differentiation of refined and virgin edible oils by means of the trans- and cis-phytol isomer distribution. J. Agric. Food Chem. 2012, 60, 6103–6107. [Google Scholar] [CrossRef] [PubMed]
- Coppack, S.W.; Evans, R.; Gibberd, F.B.; Clemens, M.E.; Billimoria, J.D. Can patients with refsum’s disease safely eat green vegetables? Br. Med. J. (Clin. Res. Ed.) 1988, 296, 828. [Google Scholar] [CrossRef]
- Pakrashi, S.C.; Achari, B.; Majumdar, P.C. Studies on indian medicinal-plants: Part XXXII. constituents of Ananas comosus (linn) merr leaves. Indian J. Chem. 1975, 13, 755–756. [Google Scholar] [PubMed]
- Cantillo-Ciau, Z.; Brito-Loeza, W.; Quijano, L. Triterpenoids from Tillandsia fasciculata. J. Nat. Prod. 2001, 64, 953–955. [Google Scholar] [CrossRef] [PubMed]
- Manetti, L.M.; Delaporte, R.H.; Laverde-Junior, A. Metabólitos secundários da família Bromeliaceae [Secondary metabolites from Bromeliaceae family]. Quim. Nova 2009, 32, 1885–1897. [Google Scholar] [CrossRef]
- Delaporte, R.H.; Sarragiotto, M.H.; Takemura, O.S.; Sanchez, G.M.; Filho, B.P.D.; Nakamura, C.V. Evaluation of the antioedematogenic, free radical scavenging and antimicrobial activities of aerial parts of Tillandsia streptocarpa baker—Bromeliaceae. J. Ethnopharmacol. 2004, 95, 229–233. [Google Scholar] [CrossRef] [PubMed]
- Cabrera, G.M.; Gallo, M.; Seldes, A.M. Cycloartane derivatives from Tillandsia usneoides. J. Nat. Prod. 1996, 59, 343–347. [Google Scholar] [CrossRef]
- Katan, M.B.; Grundy, S.M.; Jones, P.; Law, M.; Miettinen, T.; Paoletti, R. Efficacy and safety of plant stanols and sterols in the management of blood cholesterol levels. Mayo Clin. Proc. 2003, 78, 965–978. [Google Scholar] [CrossRef]
- Farquhar, J.W.; Sokolow, M. Response of serum lipids and lipoproteins of man to beta-sitosterol and safflower oil—A long term study. Circulation 1958, 17, 890–899. [Google Scholar] [CrossRef] [PubMed]
- AbuMweis, S.S.; Marinangeli, C.P.F.; Frohlich, J.; Jones, P.J. Implementing phytosterols into medical practice as a cholesterol—Lowering strategy: Overview of efficacy, effectiveness, and safety. Can. J. Cardiol. 2014, 30, 1225–1232. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Ding, Y.; Xing, D.-M.; Wang, J.-P.; Du, L.-J. Studies on phenolic constituents from leaves of pineapple (Ananas comosus). China J. Chin. Mater. Med. 2006, 31, 1242–1244. [Google Scholar]
- Marker, R.E.; Wagner, R.B.; Ulshafer, P.R.; Wittbecker, E.L.; Goldsmith, D.P.J.; Ruof, C.H. Sterols. CLVII. Sapogenins. LXIX. Isolation and structures of thirteen new steroidal sapogenins. New sources for known sapogenins. J. Am. Chem. Soc. 1943, 65, 1199–1209. [Google Scholar] [CrossRef]
- Djerassi, C.; McCrindle, R. 789. Terpenoids. Part LI. The isolation of some new cyclopropane-containing triterpenes from spanish moss (Tillandsia usneoides, L.). J. Chem. Soc. 1962, 4034–4039. [Google Scholar] [CrossRef]
- Cabrera, G.M.; Seldes, A.M. Hydroperoxycycloartanes from Tillandsia recurvata. J. Nat. Prod. 1995, 58, 1920–1924. [Google Scholar] [CrossRef]
- Palomino-Schätzlein, M.; Escrig, P.V.; Boira, H.; Primo, J.; Pineda-Lucena, A.; Cabedo, N. Evaluation of nonpolar metabolites in plant extracts by 13C NMR spectroscopy. J. Agric. Food Chem. 2011, 59, 11407–11416. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.K.; Choi, Y.H.; Verpoorte, R. NMR-based plant metabolomics: Where do we stand, where do we go? Trends Biotechnol. 2011, 29, 267–275. [Google Scholar] [CrossRef] [PubMed]
- Kontogianni, V.G.; Exarchou, V.; Troganis, A.; Gerothanassis, I.P. Rapid and novel discrimination and quantification of oleanolic and ursolic acids in complex plant extracts using two-dimensional nuclear magnetic resonance spectroscopy-Comparison with HPLC methods. Anal. Chim. Acta 2009, 635, 188–195. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds phytol, β-sitosterol and α-tocopherol are available from the authors. |

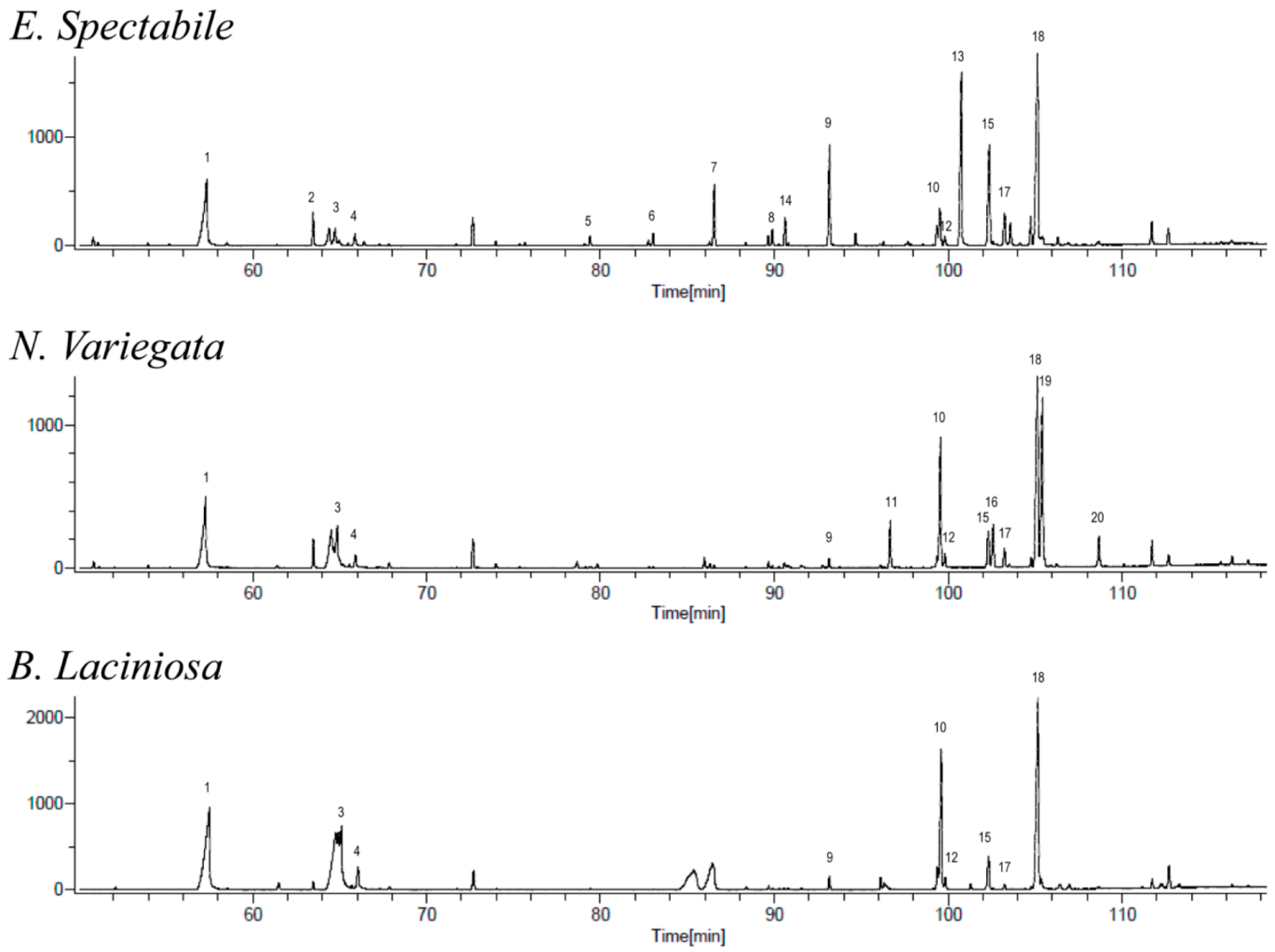
| Nr. | Compounds According to Group | MF | Exact Mass * | Retention Time (min) | Content (%) | |||||
|---|---|---|---|---|---|---|---|---|---|---|
| Observed | Calculated | B. L. | N. V. | E.S. | B.L. | N.V. | E.S. | |||
| Fatty acids and their derivatives | ||||||||||
| 1 | n-Hexadecanoic acid (Palmitic acid) | C16H32O2 | 256.24110 | 256.24023 | 57.52 | 57.30 | 57.38 | 11.3 | 7.4 | 6.1 |
| 2 | Octadeca-(9,12)dienoic acid | C18H32O2 | 280.24398 | 280.24023 | 64.38 | 3.1 | ||||
| 3 | (9Z)-Octadec-9-enoic acid (Oleic acid) | C18H34O2 C18H32O a | n.a. 264.2434 | 264.24532 | 65.10 | 64.86 | 64.75 | 8.8 | 4.5 | 1.7 |
| 4 | Octadecanoic acid (Stearic acid) | C18H36O2 | 284.27187 | 284.27153 | 66.05 | 65.88 | 65.88 | 3.2 | 1.4 | 1.1 |
| Alkanes | ||||||||||
| 5 | n-Pentacosane | C25H52 | 79.41 | 0.8 | ||||||
| 6 | n-Hexacosane | C26H54 | 83.03 | 1.1 | ||||||
| 7 | n-Heptacosane | C27H56 | 86.54 | 5.7 | ||||||
| 8 | n-Octacosane | C28H58 | 89.90 | 1.4 | ||||||
| 9 | n-Nonacosane | C29H60 | 93.16 | 93.14 | 93.18 | 2.0 | 1.0 | 9.3 | ||
| 10 | n-Triacontane | C30H62 | 99.36 | 99.34 | 99.35 | 18.9 | 13.6 | 3.5 | ||
| Vitamins | ||||||||||
| 11 | β-Tocopherol | C28H48O2 | 416.36392 | 416.36543 | 96.67 | 4.9 | ||||
| 12 | α-Tocopherol | C29H50O2 | 430.38089 | 430.38108 | 99.60 | 99.56 | 99.52 | 1.8 | 1.5 | 0.9 |
| Phytol | ||||||||||
| 13 | (2E,7R,11R)-3,7,11,15-Tetramethyl-2-hexadecen-1-ol (Phytol) | C20H40O1 | 296.30982 | 296.30791 | 100.76 | 16.1 | ||||
| Triterpenoids and derivatives | ||||||||||
| 14 | Stigmastan-3-one | C29H50O1 | 414.38688 | 414.38616 | 90.62 | 2.6 | ||||
| 15 | Campesterol ((3β,24R)-Ergost-5-en-3-ol) | C27H44O2 | 400.33890 | 400.33413 | 102.33 | 102.33 | 102.37 | 4.5 | 3.9 | 9.4 |
| 16 | Ergostanol | C28H50O1 | 402.38723 | 402.38616 | 102.60 | 4.5 | ||||
| 17 | Stigmasta-4,22-dien-3-β-ol | C29H48O1 | 412.37123 | 412.37051 | 103.24 | 103.24 | 103.25 | 0.7 | 2.0 | 3.1 |
| 18 | β-Sitosterol | C29H50O1 | 414.38655 | 414.38616 | 105.18 | 105.14 | 105.16 | 25.8 | 19.8 | 17.8 |
| 19 | Stigmastanol | C29H52O1 | 416.40276 | 416.40181 | 105.42 | 17.6 | ||||
| 20 | 24-Methyl-3-β-9,19-cyclolanost-24-en-3-ol | C31H52O1 | 440.40234 | 440.40181 | 108.67 | 3.2 | ||||
| Unidentified | 23.0 | 14.7 | 16.3 | |||||||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Juvik, O.J.; Holmelid, B.; Francis, G.W.; Lie Andersen, H.; De Oliveira, A.P.; Gonçalves de Oliveira Júnior, R.; Guedes da Silva Almeida, J.R.; Fossen, T. Non-Polar Natural Products from Bromelia laciniosa, Neoglaziovia variegata and Encholirium spectabile (Bromeliaceae). Molecules 2017, 22, 1478. https://doi.org/10.3390/molecules22091478
Juvik OJ, Holmelid B, Francis GW, Lie Andersen H, De Oliveira AP, Gonçalves de Oliveira Júnior R, Guedes da Silva Almeida JR, Fossen T. Non-Polar Natural Products from Bromelia laciniosa, Neoglaziovia variegata and Encholirium spectabile (Bromeliaceae). Molecules. 2017; 22(9):1478. https://doi.org/10.3390/molecules22091478
Chicago/Turabian StyleJuvik, Ole Johan, Bjarte Holmelid, George W. Francis, Heidi Lie Andersen, Ana Paula De Oliveira, Raimundo Gonçalves de Oliveira Júnior, Jackson Roberto Guedes da Silva Almeida, and Torgils Fossen. 2017. "Non-Polar Natural Products from Bromelia laciniosa, Neoglaziovia variegata and Encholirium spectabile (Bromeliaceae)" Molecules 22, no. 9: 1478. https://doi.org/10.3390/molecules22091478