Molecular Cloning and Characterization of Carotenoid Pathway Genes and Carotenoid Content in Ixeris dentata var. albiflora
Abstract
:1. Introduction
2. Results and Discussion
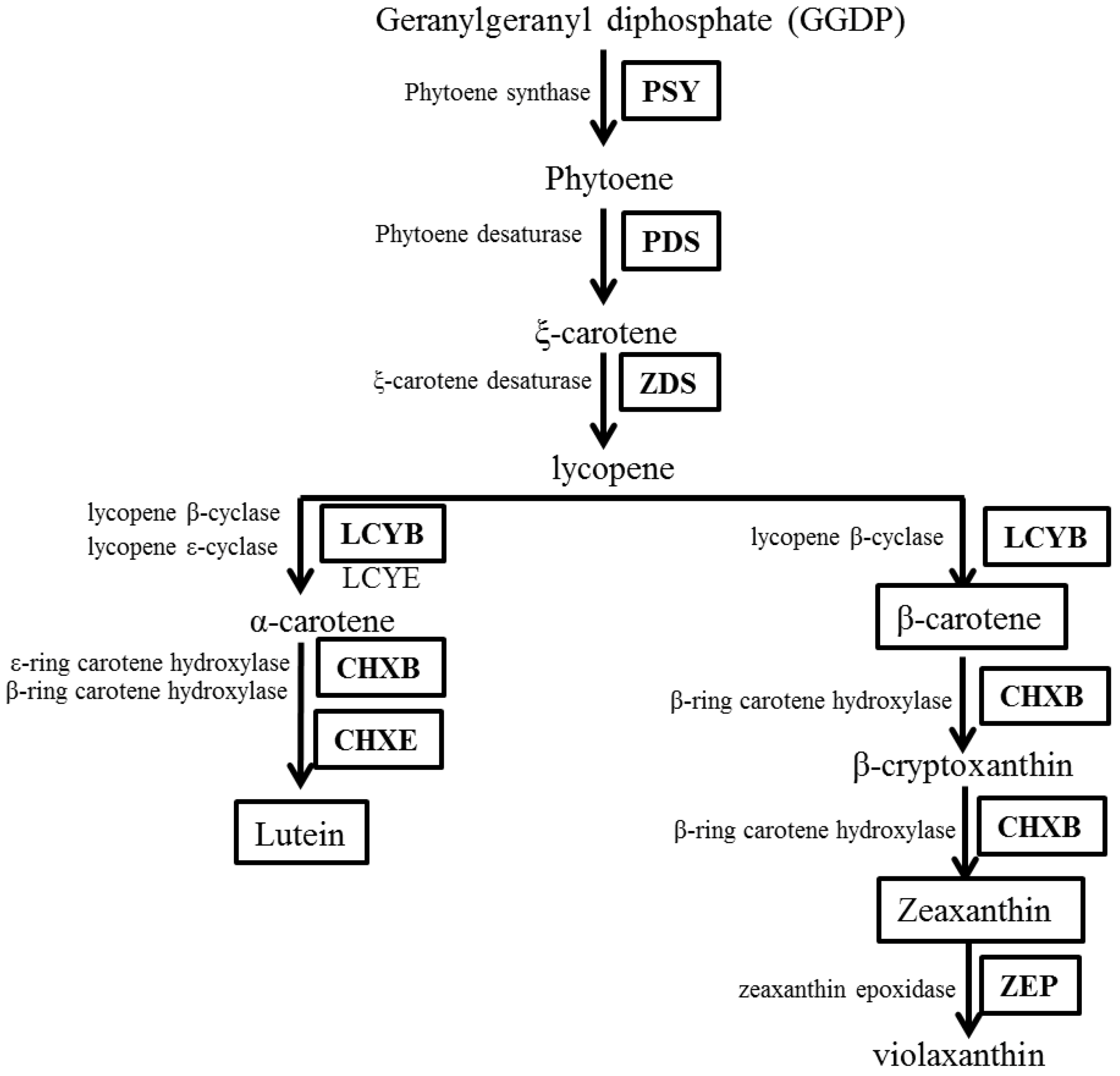
2.1. In Silico Analyses of Carotenoid Biosynthetic Genes from I. dentata
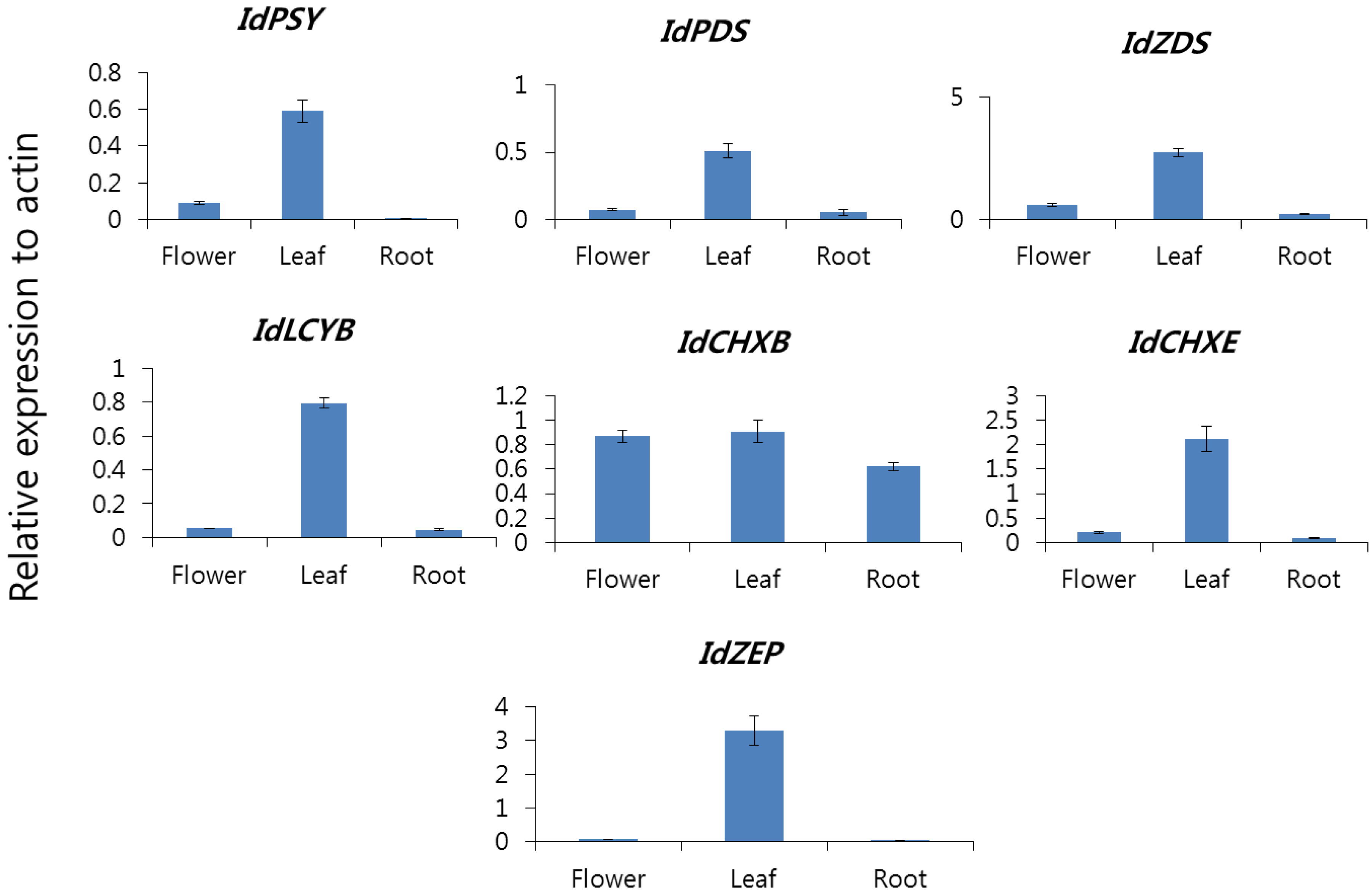
2.2. Carotenoid Biosynthetic Genes Spatial Expression Profiles in of I. dentata
2.3. Carotenoids Analysis in Different Organs of I. dentata by HPLC
3. Experimental Section
3.1. Plant Materials
3.2. Total RNA Isolation and Quantitative Real Time-PCR (qRT-PCR)
3.3. Sequence Analysis
3.4. Isolation and HPLC Analysis of I. Dentata Carotenoids
3.5. Statistical Analysis
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Ahn, E.M.; Bang, M.H.; Song, M.C.; Park, M.H.; Kim, H.Y.; Kwon, B.M.; Baek, N.I. Cytotoxic and ACAT-inhibitory sesquiterpene lactones from the root of Ixeris dentata forma albiflora. Arch. Pharm. Res. 2006, 29, 937–941. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.A.; Lee, H.N.; Choo, G.S.; Kim, H.J.; Che, J.H.; Jung, J.Y. Ixeris dentata (Thunb. Ex Thunb.) Nakai extract inhibits proliferation and induces apoptosis in breast cancer cells through Akt/NF-κB pathways. Int. J. Mol. Sci. 2017, 18, 275. [Google Scholar] [CrossRef] [PubMed]
- Jung, Y.Y.; Hong, J.T.; Han, S.B.; Park, Y.H.; Son, D.J. Effect of Ixeris dentata Nakai Extract on Nitric Oxide Production and Prostaglandin E2 Generation in LPS-stimulated RAW264. 7 Cells. Immune Netw. 2015, 15, 325–330. [Google Scholar] [CrossRef] [PubMed]
- Park, E.K.; Sung, J.H.; Trinh, H.T.; Bea, E.A.; Yun, H.K.; Hong, S.S.; Kim, D.H. Lactic acid bacterial fermentation increases the antiallergic effects of Ixeris dentata. J. Microbiol. Biotechnol. 2008, 18, 308–313. [Google Scholar] [PubMed]
- Lim, S.S.; Lee, J.H. Effect of Aster scaber and Ixeris dentate on contractility and vasodilation of cardiovascula and endothelial cell in hyperlipidemic rat. J. Korean Soc. Food Sci. Nutr. 1997, 26, 300–307. [Google Scholar]
- Karppinen, K.; Zoratti, L.; Sarala, M.; Carvalho, E.; Hirsimäki, J.; Mentula, H.; Jaakola, L. Carotenoid metabolism during bilberry (Vaccinium myrtillus L.) fruit development under different light conditions is regulated by biosynthesis and degradation. BMC Plant Biol. 2016, 16, 95. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Zhang, J.; Nageswaran, D.; Li, L. Carotenoid metabolism and regulation in horticultural crops. Hortic. Res. 2015, 2. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Sola, M.Á.; Rodríguez-Concepción, M. Carotenoid biosynthesis in Arabidopsis: A colorful pathway. Arabidopsis Book 2012, 10, e0158. [Google Scholar] [CrossRef] [PubMed]
- Howitt, C.A.; Pogson, B.J. Carotenoid accumulation and function in seeds and non-green tissues. Plant Cell Environ. 2006, 29, 435–445. [Google Scholar] [CrossRef] [PubMed]
- Tuan, P.A.; Kim, Y.B.; Kim, J.K.; Arasu, M.V.; Al-Dhabi, N.A.; Park, S.U. Molecular characterization of carotenoid biosynthetic genes and carotenoid accumulation in Scutellaria baicalensis Georgi. EXCLI J. 2015, 14, 146–157. [Google Scholar] [PubMed]
- Giuliano, G.; Al-Babili, S.; von Lintig, J. Carotenoid oxygenases: Cleave it or leave it. Trends Plant Sci. 2003, 8, 145–149. [Google Scholar] [CrossRef]
- Sommer, A.; Vyas, K.S. A global clinical view on vitamin A and carotenoids. Am. J. Clin. Nutr. 2012, 96, 1204S–1206S. [Google Scholar] [CrossRef] [PubMed]
- Mayne, S.T. Beta-carotene, carotenoids, and disease prevention in humans. FASEB J. 1996, 10, 690–701. [Google Scholar] [PubMed]
- Li, X.; Park, N.I.; Xu, H.; Woo, S.H.; Park, C.H.; Park, S.U. Differential expression of flavonoid biosynthesis genes and accumulation of phenolic compounds in common buckwheat (Fagopyrum esculentum). J. Agric. Food Chem. 2010, 58, 12176–12181. [Google Scholar] [CrossRef] [PubMed]
- Zhao, S.; Park, C.H.; Li, X.; Kim, Y.B.; Yang, J.; Sung, G.B.; Park, S.U. Accumulation of rutin and betulinic acid and expression of phenylpropanoid and triterpenoid biosynthetic genes in Mulberry (Morus alba L.). J. Agric. Food Chem. 2015, 63, 8622–8630. [Google Scholar] [CrossRef] [PubMed]
- Hadjipieri, M.; Georgiadou, E.C.; Marin, A.; Diaz-Mula, H.M.; Goulas, V.; Fotopoulos, V.; Manganaris, G.A. Metabolic and transcriptional elucidation of the carotenoid biosynthesis pathway in peel and flesh tissue of loquat fruit during on-tree development. BMC Plant Biol. 2017, 17, 102. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Tomatoes, tomato-based products, lycopene, and cancer: Review of the epidemiologic literature. J. Natl. Cancer Inst. 1999, 91, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Shao, Z.; Zhang, M.; Wang, Q. Regulation of carotenoid metabolism in tomato. Mol. Plant 2015, 8, 28–39. [Google Scholar] [CrossRef] [PubMed]
- Ohnuma, S.I.; Nakazawa, T.; Hemmi, H.; Hallberg, A.M.; Koyama, T.; Ogura, K.; Nishino, T. Conversion from farnesyl diphosphate synthase to geranylgeranyl diphosphate synthase by random chemical mutagenesis. J. Biol. Chem. 1996, 271, 10087–10095. [Google Scholar] [CrossRef] [PubMed]
- Rasid, O.A.; Wan Nur Syuhada, W.S.; Nor Hanin, A.; Masura, S.S.; Zulqarnain, M.; Ho, C.L.; Sambanthamurthi, R.; Suhaimi, N. RT-PCR Amplification and Cloning of Partial DNA Sequence Coding for Oil Palm (Elaeis oleifera) Phytoene Synthase Gene. Asia-Pac. J. Mol. Biol. Biotechnol. 2008, 16, 17–24. [Google Scholar]
- Yan, P.; Gao, X.Z.; Shen, W.T.; Zhou, P. Cloning and expression analysis of phytoene desaturase and ζ-carotene desaturase genes in Carica papaya. Mol. Biol. Rep. 2011, 38, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.H.; Jiang, J.G.; Yan, Y.; Chen, X.W. Isolation and characterization of phytoene desaturase cDNA involved in the β-carotene biosynthetic pathway in Dunaliella salina. J. Agric. Food Chem. 2005, 53, 5593–5597. [Google Scholar] [CrossRef] [PubMed]
- Zeng, J.; Wang, C.; Chen, X.; Zang, M.; Yuan, C.; Wang, X.; Li, K. The lycopene β-cyclase plays a significant role in provitamin A biosynthesis in wheat endosperm. BMC Plant Biol. 2015, 15, 112. [Google Scholar] [CrossRef] [PubMed]
- Bouvier, F.; Keller, Y.; d’Harlingue, A.; Camara, B. Xanthophyll biosynthesis: Molecular and functional characterization of carotenoid hydroxylases from pepper fruits (Capsicum annuum L.). Biochim. Biophys. Acta Lipids Lipid Metab. 1998, 1391, 320–328. [Google Scholar] [CrossRef]
- Graham-Lorence, S.; Peterson, J.A.; Amarneh, B.; Simpson, E.R.; White, R.E. A three-dimensional model of aromatase cytochrome P450. Protein Sci. 1995, 4, 1065–1080. [Google Scholar] [CrossRef] [PubMed]
- Durocher, D.; Jackson, S.P. The FHA domain. FEBS Lett. 2002, 513, 58–66. [Google Scholar] [CrossRef]
- Hieber, A.D.; Bugos, R.C.; Yamamoto, H.Y. Plant lipocalins: Violaxanthin de-epoxidase and zeaxanthin epoxidase. Biochim. Biophys. Acta Protein Struct. Mol. Enzymol. 2000, 1482, 84–91. [Google Scholar] [CrossRef]
- Meier, S.; Tzfadia, O.; Vallabhaneni, R.; Gehring, C.; Wurtzel, E.T. A transcriptional analysis of carotenoid, chlorophyll and plastidial isoprenoid biosynthesis genes during development and osmotic stress responses in Arabidopsis thaliana. BMC Syst. Biol. 2011, 5, 77. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, F.X.; Gantt, E. Genes and enzymes of carotenoid biosynthesis in plants. Annu. Rev. Plant Physiol. Plant Mol. Biol. 1998, 49, 557–583. [Google Scholar] [CrossRef] [PubMed]
- Cazzonelli, C.I.; Pogson, B.J. Source to sink: Regulation of carotenoid biosynthesis in plants. Trends Plant Sci. 2010, 15, 266–274. [Google Scholar] [CrossRef] [PubMed]
- Bouvier, F.; d’Harlingue, A.; Hugueney, P.; Marin, E.; Marion-Poll, A.; Camara, B. Xanthophyll biosynthesis cloning, expression, functional reconstitution, and regulation of β-cyclohexenyl carotenoid epoxidase from pepper (Capsicum annuum). J. Biol. Chem. 1996, 271, 28861–28867. [Google Scholar] [CrossRef] [PubMed]
- Tuan, P.A.; Kim, J.K.; Kim, H.H.; Lee, S.Y.; Park, N.I.; Park, S.U. Carotenoid accumulation and characterization of cDNAs encoding phytoene synthase and phytoene desaturase in garlic (Allium sativum). J. Agric. Food Chem. 2011, 59, 5412–5417. [Google Scholar] [CrossRef] [PubMed]
- Tuan, P.A.; Kim, J.K.; Park, N.I.; Lee, S.Y.; Park, S.U. Carotenoid content and expression of phytoene synthase and phytoene desaturase genes in bitter melon (Momordica charantia). Food Chem. 2011, 126, 1686–1692. [Google Scholar] [CrossRef] [PubMed]
- Voutilainen, S.; Nurmi, T.; Mursu, J.; Rissanen, T.H. Carotenoids and cardiovascular health. Am. J. Clin. Nutr. 2006, 83, 1265–1271. [Google Scholar] [PubMed]
- Polidori, M.C.; Cherubini, A.; Stahl, W.; Senin, U.; Sies, H.; Mecocci, P. Plasma carotenoid and malondialdehyde levels in ischemic stroke patients: Relationship to early outcome. Free Radic. Res. 2002, 36, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Gallicchio, L.; Boyd, K.; Matanoski, G.; Tao, X.G.; Chen, L.; Lam, T.K.; Herman, J.G. Carotenoids and the risk of developing lung cancer: A systematic review. Am. J. Clin. Nutr. 2008, 88, 372–383. [Google Scholar] [PubMed]
- Zhao, D.; Zhou, C.; Tao, J. Carotenoid accumulation and carotenogenic genes expression during two types of persimmon fruit (Diospyros kaki L.) development. Plant Mol. Biol. Rep. 2011, 29, 646–654. [Google Scholar] [CrossRef]
- Li, S.Y.; Fung, F.K.; Fu, Z.J.; Wong, D.; Chan, H.H.; Lo, A.C. Anti-Inflammatory Effects of Lutein in Retinal Ischemic/Hypoxic Injury: In vivo and in vitro studies anti-inflammatory effects of lutein. Invest. Ophthalmol. Vis. Sci. 2012, 53, 5976–5984. [Google Scholar] [CrossRef] [PubMed]
- He, R.R.; Tsoi, B.; Lan, F.; Yao, N.; Yao, X.S.; Kurihara, H. Antioxidant properties of lutein contribute to the protection against lipopolysaccharide-induced uveitis in mice. Chin. Med. 2011, 6, 38. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.H.; Park, Y.J.; Park, S.U.; Lee, S.W.; Kim, S.C.; Jung, C.S.; Kim, Y.B. Expression of genes related to phenylpropanoid biosynthesis in different organs of Ixeris dentata var. albiflora. Molecules 2017, 22, 901. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: The sample compounds used in this study are not available with authors due to the limited scale extraction. |
| Name of the Carotenoid |  |  |  |
|---|---|---|---|
(1)  | 415.32 ± 32.62a | N/D | 197.28 ± 36.22b |
(2)  | 21.59 ± 0.94a | N/D | 24.03 ± 3.41a |
(3)  | 140.88 ± 16.79a | N/D | 21.6 ± 3.95b |
(4)  | 803.43 ± 61.5a | N/D | 97.26 ± 16.91b |
(5)  | 117.03 ± 4.2a | N/D | 13.41 ± 2.33b |
| Total carotenoid | 1498.25 | N/D | 353.59 |
| Primer Name | Sequence (5′ → 3′) | Size (bp) | GenBank Access. No |
|---|---|---|---|
| IdPSY (F) | CCGCAAGATGAGCTGGCGAA | 179 | MF611777 |
| IdPSY (R) | GATGCCCAAACCGGCCATCT | ||
| IdPDS (F) | GGAAAGGTGGCGGCTTGGA | 136 | MF611774 |
| IdPDS (R) | GCTCCTTCCATTGCAATCGGTCAT | ||
| IdZDS (F) | TGCTGTGGCTCTTGCTTTAAGTCC | 160 | MF611772 |
| IdZDS (R) | TGCAACCGGGTCCCACATTC | ||
| IdLCYB (F) | GGTCGCGGAGGTTGTGTCAA | 174 | MF611775 |
| IdLCYB (R) | GACTTGTTGGGCGACTGCGA | ||
| IdCHXB (F) | TGGCAAATGGAGGGTGGAGAGG | 103 | MF614116 |
| IdCHXB (R) | GCGCCCATCTCGCCCAATAC | ||
| IdCHXE (F) | TTCACTGTGGACGGCAAGGC | 142 | MF611776 |
| IdCHXE (R) | AGCGGTGTCATTGCGTGCAT | ||
| IdZEP (F) | GGAGGTGGGATCGGAGGGTT | 155 | MF611773 |
| IdZEP (R) | GCTTCCAAAGCAGCCAACGC | ||
| IdActin (F) | CAATGGAACCGGAATGGTCA | 100 | KY419710 |
| IdActin (R) | CATTACGCCCGTGTGTCGAG |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Reddy, C.S.; Lee, S.-H.; Yoon, J.S.; Kim, J.K.; Lee, S.W.; Hur, M.; Koo, S.C.; Meilan, J.; Lee, W.M.; Jang, J.K.; et al. Molecular Cloning and Characterization of Carotenoid Pathway Genes and Carotenoid Content in Ixeris dentata var. albiflora. Molecules 2017, 22, 1449. https://doi.org/10.3390/molecules22091449
Reddy CS, Lee S-H, Yoon JS, Kim JK, Lee SW, Hur M, Koo SC, Meilan J, Lee WM, Jang JK, et al. Molecular Cloning and Characterization of Carotenoid Pathway Genes and Carotenoid Content in Ixeris dentata var. albiflora. Molecules. 2017; 22(9):1449. https://doi.org/10.3390/molecules22091449
Chicago/Turabian StyleReddy, Chinreddy Subramanyam, Sang-Hoon Lee, Jeong Su Yoon, Jae Kwang Kim, Sang Won Lee, Mok Hur, Sung Cheol Koo, Jin Meilan, Woo Moon Lee, Jae Ki Jang, and et al. 2017. "Molecular Cloning and Characterization of Carotenoid Pathway Genes and Carotenoid Content in Ixeris dentata var. albiflora" Molecules 22, no. 9: 1449. https://doi.org/10.3390/molecules22091449





