Bioactive Constituents Obtained from the Seeds of Lepidium apetalum Willd
Abstract
:1. Introduction
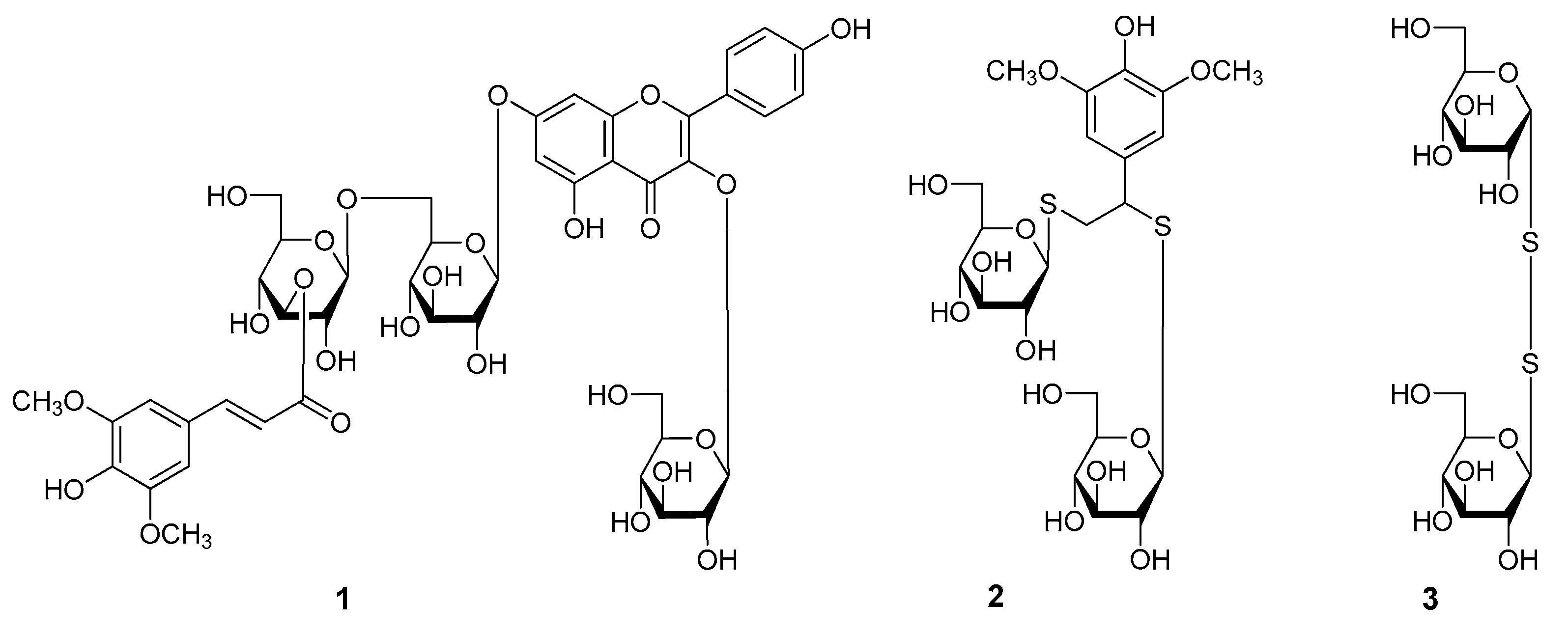
2. Results and Discussion
3. Experimental
3.1. General
3.2. Plant Material
3.3. Extraction and Isolation
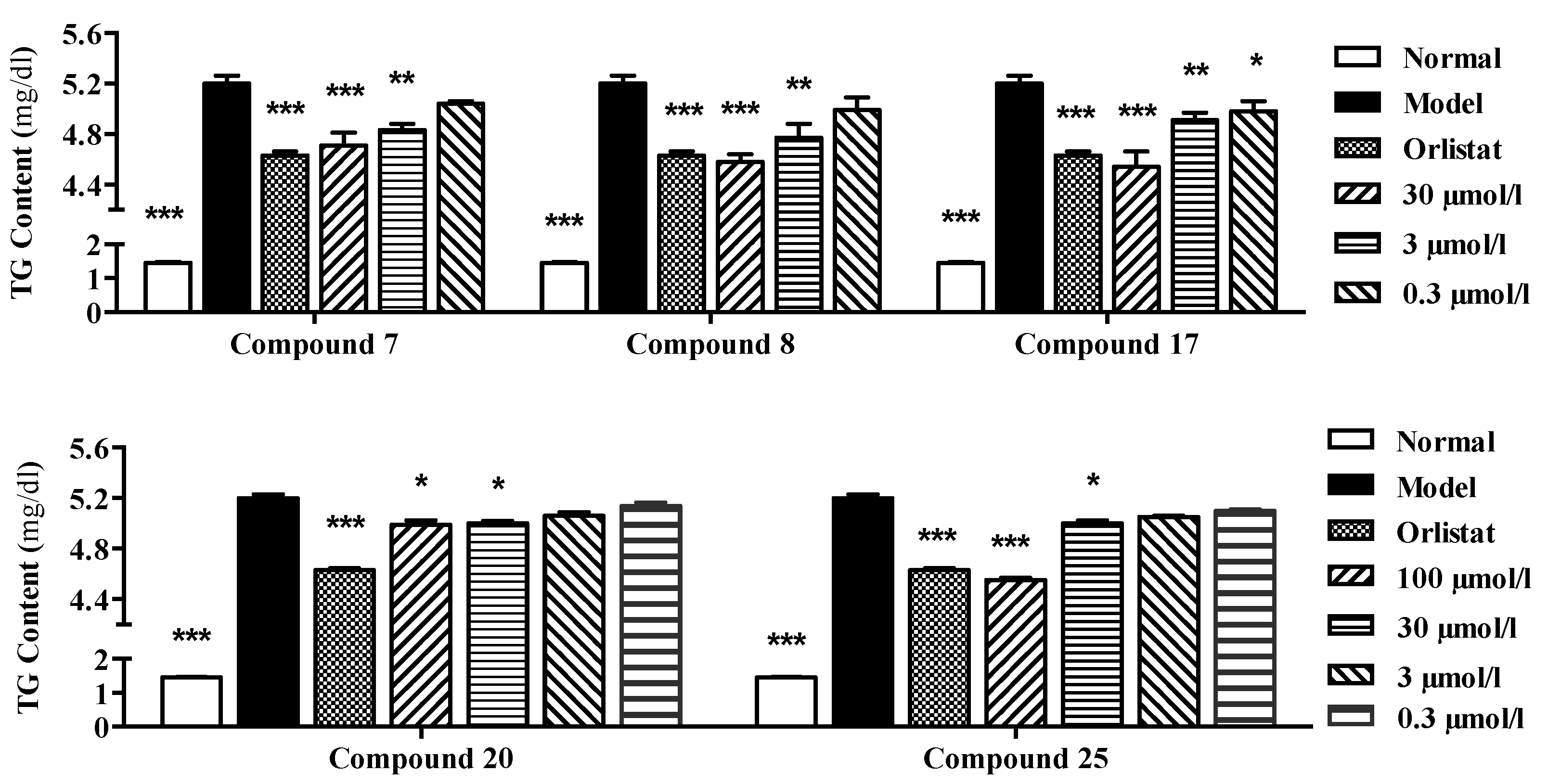
3.4. Evaluation of Effects on Sodium Oleate-Induced TG Overloading in HepG2 Cells
3.5. Statistical Analysis
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Han, L.; Shi, P.; Dong, Y.; Wang, T.; Li, X.; Hao, J.; Zhang, Y.; Wang, T. New rare sinapoyl acylated flavonoid glycosides obtained from the seeds of Lepidium apetalum Willd. Molecules 2015, 20, 13982–13996. [Google Scholar] [PubMed]
- Shi, P.; Chao, L.; Wang, T.; Liu, E.; Han, L.; Zong, Q.; Li, X.; Zhang, Y.; Wang, T. New bioactive flavonoid glycosides isolated from the seeds of Lepidium apetalum Willd. Fitoterapia 2015, 103, 197–205. [Google Scholar] [PubMed]
- Wei, Y.; Xie, Q.; Fisher, D.; Sutherland, I.A. Separation of patuletin-3-O-glucoside, astragalin, quercetin, kaempferol and isorhamnetin from Flaveria bidentis (L.) Kuntze by elution-pump-out high-performance counter-current chromatography. J. Chromatogr. A 2011, 1218, 6206–6211. [Google Scholar] [PubMed]
- Bacon, J.D.; Mabry, T.J. Flavonol glycosides of Nerisyrenia (Cruciferae). Phytochemistry 1976, 15, 1087–1088. [Google Scholar] [CrossRef]
- Moon, S.S.; Rahman, M.A.; Manir, M.M.; Jamal Ahamed, V.S. Kaempferol glycosides and cardenolide glycosides, cytotoxic constituents from the seeds of Draba nemorosa (Brassicaceae). Arch. Pharm. Res. 2010, 33, 1169–1173. [Google Scholar] [CrossRef] [PubMed]
- Halabalaki, M.; Urbain, A.; Paschali, A.; Mitakou, S.; Tillequin, F.; Skaltsounis, A.L. Quercetin and kaempferol 3-O-[α-l-rhamnopyranosyl-(1→2)-α-l-arabinopyranoside]-7-O-α-l-rhamnopyranosides from Anthyllis hermanniae: Structure determination and conformational studies. J. Nat. Prod. 2011, 74, 1939–1945. [Google Scholar] [CrossRef] [PubMed]
- Atay, I.; Kirmizibekmez, H.; Goren, A.C.; Yesilada, E. Secondary metabolites from Sambucus ebulus. Turk. J. Chem. 2015, 39, 34–41. [Google Scholar] [CrossRef]
- Park, S.H.; Kim, H.; Rhyu, D.Y. Flavonoids from the stems of eastern picklypear Opuntia humifusa, Cactaceae. J. Appl. Biol. Chem. 2007, 50, 254–258. [Google Scholar]
- Zhang, Y.B.; Wu, P.; Zhang, X.L.; Xia, C.; Li, G.Q.; Ye, W.C.; Wang, G.C.; Li, Y.L. Phenolic compounds from the flowers of Bombax malabaricum and their antioxidant and antiviral activities. Molecules 2015, 20, 19947–19957. [Google Scholar] [CrossRef] [PubMed]
- Nakayama, R.; Kikuzaki, H.; Nakatani, N.; Hisashi, H. Antioxidative activity of constituents from fennel seeds. J. Home Econ. Jpn. 1996, 47, 1193–1200. [Google Scholar]
- Hashimoto, K.; Katsuhara, T.; Niitsu, K.; Ikeya, Y.; Okada, M.; Mitsuhashi, H. Two glycosides from roots of Asiasarum sieboldi. Phytochemistry 1992, 31, 2477–2480. [Google Scholar] [CrossRef]
- Pauli, G.F.; Junior, P. Phenolic glycosides from Adonis aleppica. Phytochemistry 1995, 38, 1245–1250. [Google Scholar] [CrossRef]
- Strack, D.; Dahlbender, B.; Grotjahn, L.; Wray, V. 1,2-disinapolylglucose accumulated in cotyledons of dark-grown raphanus sativus seedlings. Phytochemistry 1984, 23, 657–659. [Google Scholar] [CrossRef]
- Price, K.R.; Casuscelli, F.; Colquhoun, I.J.; Rhodes, M.J.C. Hydroxycinnamic acid esters from Broccoli florets. Phytochemistry 1997, 45, 1683–1687. [Google Scholar] [CrossRef]
- Li, T.; Zhang, W.; Gu, Z.; Liu, W.; Zhang, C.; Liu, R. Lignans from Patrinia scabra. Zhongcaoyao 2005, 36, 338–340. [Google Scholar]
- Yang, B.H.; Zhang, W.D.; Liu, R.H.; Li, T.Z.; Zhang, C.; Zhou, Y.; Su, J. Lignans from bark of Larix olgensis var. koreana. J. Nat. Prod. 2005, 68, 1175–1179. [Google Scholar] [CrossRef] [PubMed]
- Kizu, H.; Shimana, H.; Tomimori, T. Studies on the constituents of Clematis species. VI. The constituents of Clematis stans Sieb. et Zucc. Chem. Pharm. Bull. 1995, 43, 2187–2194. [Google Scholar] [CrossRef] [PubMed]
- Yuan, X.; Gao, H.; Chen, L.; Zhang, Q.; Wang, Z. A new lignan from stems of Sargentodoxa cuneata. Zhongguo Zhongyao Zazhi 2013, 38, 2118–2124. [Google Scholar] [PubMed]
- Jia, L.; Zhong, L.; Li, H.; Jing, L. Chemical constituents in water fraction of Abelmoschus esculentus. Zhongcaoyao 2011, 42, 2186–2188. [Google Scholar]
- Youssef, D.T.A.; Badr, J.M.; Shaala, L.A.; Mohamed, G.A.; Bamanie, F.H. Ehrenasterol and biemnic acid; new bioactive compounds from the Red Sea sponge Biemna ehrenbergi. Phytochem. Lett. 2015, 12, 296–301. [Google Scholar] [CrossRef]
- Abou-Hussein, D.R.; Badr, J.M.; Youssef, D.T.A. Nucleoside constituents of the Egyptian tunicate Eudistoma laysani. Nat. Prod. Sci. 2007, 13, 229–233. [Google Scholar]
- Montilla, A.; Corzo, N.; Olano, A.; Jimeno, M.L. Identification of oligosaccharides formed during stachyose hydrolysis by Pectinex Ultra SP-L. J. Agric. Food Chem. 2009, 57, 5007–5013. [Google Scholar] [CrossRef] [PubMed]
- Shaw, C.F., III; Eldridge, J.; Cancro, M.P. 13C-NMR studies of aurothioglucose: Ligand exchange and redox disproportionation reactions. J. Inorg. Biochem. 1981, 14, 267–274. [Google Scholar] [CrossRef]
- Ngane, A.N.; Lavault, M.; Séraphin, D.; Landreau, A.; Richomme, P. Three 1-thio-β-d-glucopyranosides from the seeds of Afrostyrax lepidophyllus Mildbr. Carbohydr. Res. 2006, 341, 2799–2802. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Adelakun, T.A.; Wang, S.; Ruan, J.; Yang, S.; Li, X.; Zhang, Y.; Wang, T. Inhibitory effects of constituents from the aerial parts of Rosmarinus officinalis L. on triglyceride accumulation. Molecules 2017, 22, 110. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of all compounds are available from the authors. |



| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 2 | 156.7 | — | 2′′′ | 73.0 | 3.28 (dd, 7.0, 9.5) |
| 3 | 133.4 | — | 3′′′ | 76.2 | 3.32 (dd, 9.5, 9.5) |
| 4 | 177.6 | — | 4′′′ | 69.2 | 3.26 (m, overlapped) |
| 5 | 160.8 | — | 5′′′ | 75.3 | 3.75 (m) |
| 6 | 99.4 | 6.51 (br. s) | 6′′′ | 68.9 | 3.71 (dd, 5.5, 11.5) |
| 7 | 162.7 | — | 3.99 (br. d, ca. 12) | ||
| 8 | 94.4 | 6.85 (br. s) | 1′′′′ | 103.5 | 4.35 (d, 8.0) |
| 9 | 155.9 | — | 2′′′′ | 71.5 | 3.22 (dd, 7.5, 8.0) |
| 10 | 105.6 | — | 3′′′′ | 77.5 | 4.90 (dd, 7.5, 9.0) |
| 1′ | 120.7 | — | 4′′′′ | 68.1 | 3.34 (dd, 9.0, 9.0) |
| 2′,6′ | 130.9 | 8.09 (d, 9.0) | 5′′′′ | 77.4 | 3.08 (m) |
| 3′,5′ | 115.2 | 6.92 (d, 9.0) | 6′′′′ | 60.7 | 3.56 (br. d, ca. 12) |
| 4′ | 160.1 | — | 3.70 (dd, 5.5, 11.5) | ||
| 5-OH | — | 12.65 (br. s) | 1′′′′′ | 124.5 | — |
| 1′′ | 100.6 | 5.50 (d, 8.0) | 2′′′′′,6′′′′′ | 105.9 | 7.00 (s) |
| 2′′ | 74.2 | 3.21 (dd, 7.5, 8.0) | 3′′′′′,5′′′′′ | 147.9 | — |
| 3′′ | 76.8 | 3.26 (m, overlapped) | 4′′′′′ | 138.0 | — |
| 4′′ | 69.8 | 3.08 (m, overlapped) | 7′′′′′ | 144.9 | 7.51 (d, 16.0) |
| 5′′ | 76.3 | 3.21 (m) | 8′′′′′ | 115.5 | 6.53 (d, 16.0) |
| 6′′ | 60.8 | 3.30 (br. d, ca. 11) | 9′′′′′ | 166.2 | — |
| 3.50 (dd, 5.5, 10.5) | 3′′′′′,5′′′′′-OCH3 | 56.0 | 3.81 (s) | ||
| 1′′′ | 99.7 | 5.12 (d, 7.0) |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 130.2 | — | 4′ | 70.0 a | 3.07 (m, overlapped) |
| 2,6 | 105.6 | 6.58 (s) | 5′ | 78.1 | 3.12 (m, overlapped) |
| 3,5 | 147.5 | — | 6′ | 61.2 b | 3.46 (dd, 5.0, 12.5) |
| 4 | 134.5 | — | 3.70 (br. d, ca. 13) | ||
| 7 | 46.5 | 4.28 (br. d, ca. 5) | 1′′ | 84.7 | 4.27 (d, 10.0) |
| 8 | 34.8 | 3.18 (br. d, ca. 11) | 2′′ | 80.8 | 3.13 (m, overlapped) |
| 3.39 (dd, 5.0, 11.0) | 3′′ | 78.0 | 3.12 (m, overlapped) | ||
| 3,5-OCH3 | 55.9 | 3.75 (s) | 4′′ | 69.9 a | 3.07 (m, overlapped) |
| 1′ | 83.7 | 4.31 (d, 9.5) | 5′′ | 72.9 | 3.01 (dd, 9.5, 10.0) |
| 2′ | 72.9 | 3.01 (dd, 8.0, 9.5) | 6′′ | 61.1 b | 3.46 (dd, 5.0, 12.5) |
| 3′ | 80.8 | 3.13 (m, overlapped) | 3.70 (br. d, ca. 13) |
| No. | δC | δH (J in Hz) | No. | δC | δH (J in Hz) |
|---|---|---|---|---|---|
| 1 | 96.1 | 5.57 (d, 5.5) | 1′ | 92.4 | 4.68 (d, 9.0) |
| 2 | 74.3 | 3.87 (dd, 5.5, 9.5) | 2′ | 74.4 | 3.49 (dd, 9.0, 9.5) |
| 3 | 76.2 | 3.58 (dd, 9.5, 9.5) | 3′ | 80.0 | 3.52 (dd, 9.5, 9.5) |
| 4 | 72.3 | 3.44 (dd, 9.5, 9.5) | 4′ | 72.1 | 3.42 (dd, 9.5, 9.5) |
| 5 | 76.4 | 3.94 (m) | 5′ | 83.1 | 3.50 (m) |
| 6 | 63.4 | 3.80 (dd, 5.5, 12.5) | 6′ | 63.7 | 3.72 (dd, 5.5, 12.5) |
| 3.88 (dd, 1.5, 12.5) | 3.91 (dd, 1.5, 12.5) |
| Sample (µmol/L) | 7 | 8 | 17 | ||||||
|---|---|---|---|---|---|---|---|---|---|
| 30 | 3 | 0.3 | 30 | 3 | 0.3 | 30 | 3 | 0.3 | |
| TG clearance (%) | 9.46 ± 1.89 | 7.16 ± 0.87 | 3.24 ± 0.35 | 12.00 ± 1.17 | 8.27 ± 2.14 | 4.15 ± 1.97 | 12.70 ± 2.39 | 5.68 ± 1.15 | 4.32 ± 1.57 |
| Sample (µmol/L) | 20 | 25 | ||||||
|---|---|---|---|---|---|---|---|---|
| 100 | 30 | 3 | 0.3 | 100 | 30 | 3 | 0.3 | |
| TG clearance (%) | 4.14 ± 1.40 | 3.91 ± 1.79 | 2.82 ± 1.06 | 1.31 ± 1.02 | 12.66 ± 0.77 | 3.91 ± 0.90 | 2.96 ± 0.45 | 2.00 ± 0.34 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, S.; Shi, P.; Qu, L.; Ruan, J.; Yang, S.; Yu, H.; Zhang, Y.; Wang, T. Bioactive Constituents Obtained from the Seeds of Lepidium apetalum Willd. Molecules 2017, 22, 540. https://doi.org/10.3390/molecules22040540
Wang S, Shi P, Qu L, Ruan J, Yang S, Yu H, Zhang Y, Wang T. Bioactive Constituents Obtained from the Seeds of Lepidium apetalum Willd. Molecules. 2017; 22(4):540. https://doi.org/10.3390/molecules22040540
Chicago/Turabian StyleWang, Sijian, Pingping Shi, Lu Qu, Jingya Ruan, Shengcai Yang, Haiyang Yu, Yi Zhang, and Tao Wang. 2017. "Bioactive Constituents Obtained from the Seeds of Lepidium apetalum Willd" Molecules 22, no. 4: 540. https://doi.org/10.3390/molecules22040540





