Phenylpropionamides, Piperidine, and Phenolic Derivatives from the Fruit of Ailanthus altissima
Abstract
:1. Introduction
2. Results
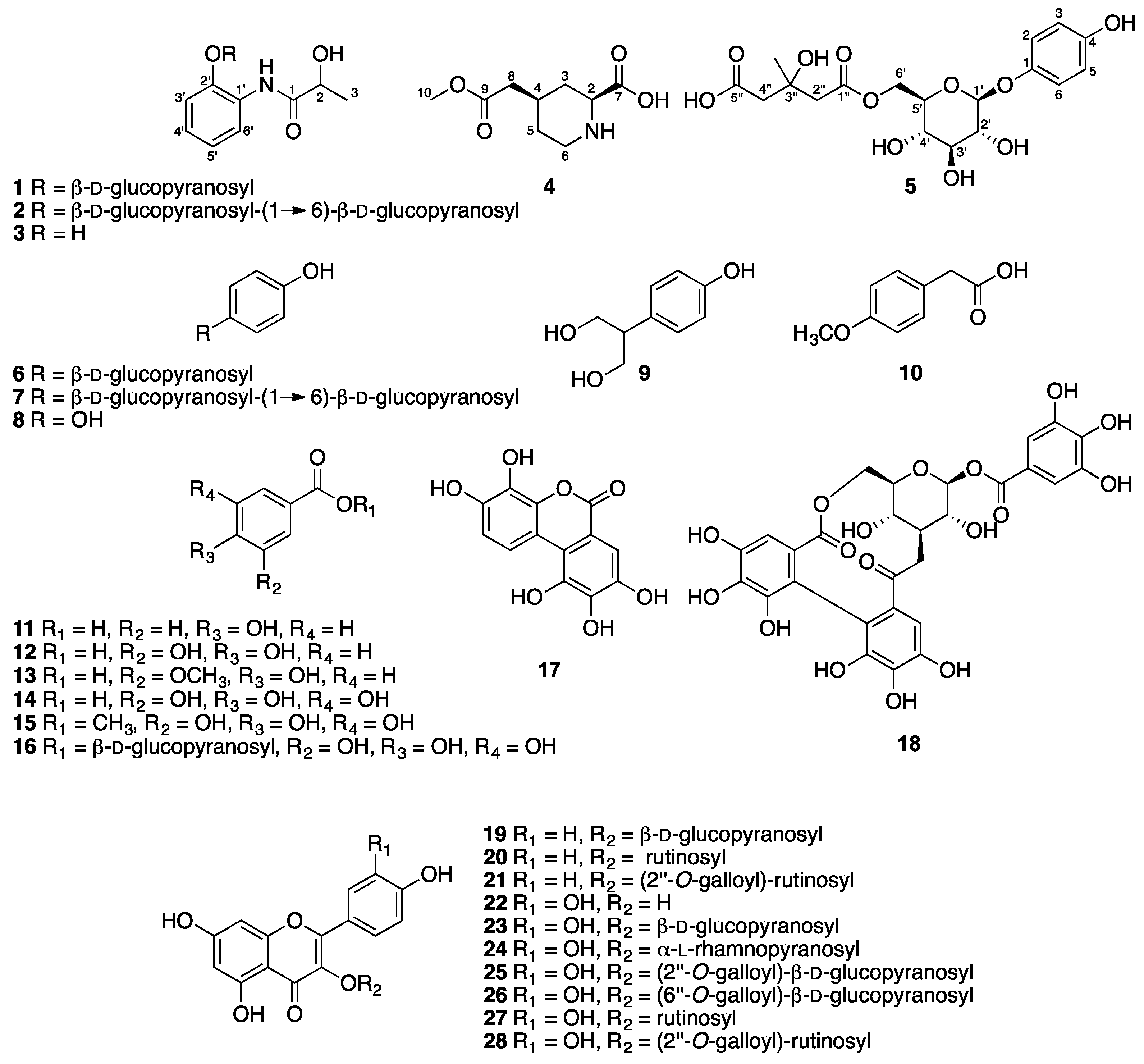
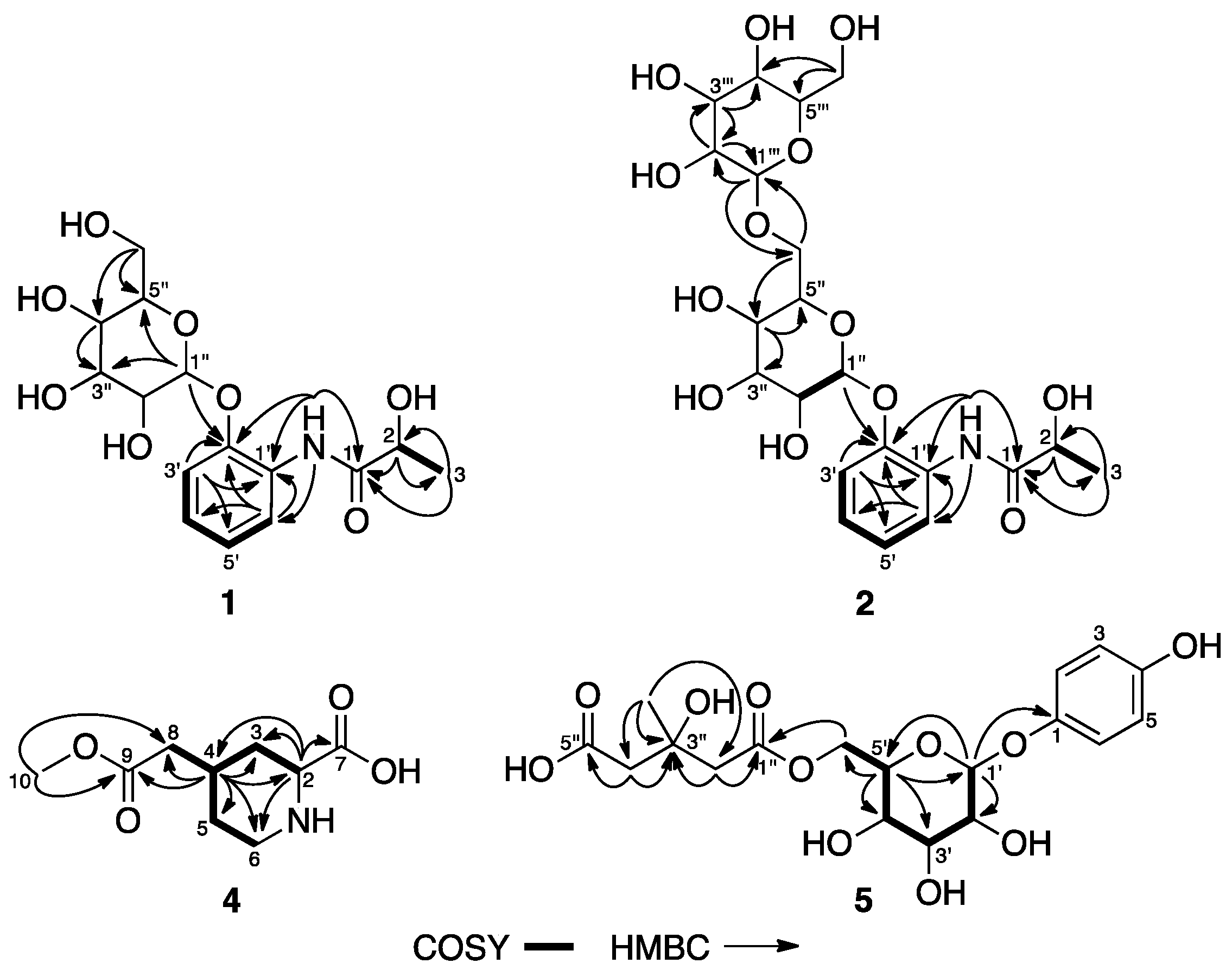
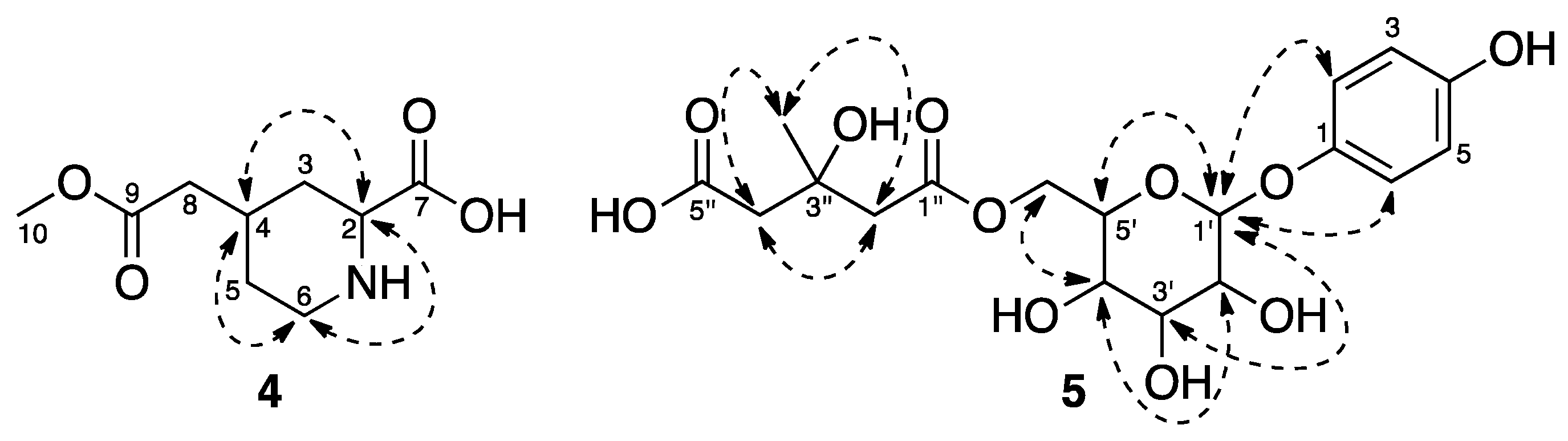
2.1. Extraction, Isolation, and Sructure Elucidation
2.2. Anti-Tobacco Mosaic Virus Activity
3. Discussion
4. Materials and Methods
4.1. General Experimental Procedures
4.2. Plant Material
4.3. Extraction, Fraction, and Isolation
4.4. Acid Hydrolysis
4.5. Anti-TMV Assay
4.5.1. Virus and Host Plant
4.5.2. The Leaf-Disc Method
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Kowarik, I. Clonal growth in Ailanthus altissima on a natural site in West Virginia. J. Veg. Sci. 1995, 6, 853–856. [Google Scholar] [CrossRef]
- Dao, T.T.; Tran, T.L.; Kim, J.; Nguyen, P.H.; Lee, E.H.; Park, J.; Jang, I.S.; Oh, W.K. Terpenylated coumarins as SIRT1 activators isolated from Ailanthus altissima. J. Nat. Prod. 2012, 75, 1332–1338. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Wang, W.J.; Su, C.; Zhang, D.M.; Xu, L.P.; He, R.R.; Wang, L.; Zhang, J.; Zhang, X.Q.; Ye, W.C. Cytotoxic quassinoids from Ailathus altissima. Bioorg. Med. Chem. Lett. 2013, 23, 654–657. [Google Scholar] [CrossRef] [PubMed]
- De Feo, V.; De Martino, L.; Quaranta, E.; Pizza, C. Isolation of phytotoxic compounds from tree-of-heaven (Ailanthus altissima Swingle). J. Agric. Food Chem. 2003, 51, 1177–1180. [Google Scholar] [CrossRef] [PubMed]
- Tamura, S.; Fukamiya, N.; Okano, M.; Koyama, J.; Koike, K.; Tokuda, H.; Aoi, W.; Takayasu, J.; Kuchide, M.; Nishino, H. Three new quassinoids, ailantinol E, F, and G, from Ailanthus altissima. Chem. Pharm. Bull. 2003, 51, 385–389. [Google Scholar] [CrossRef] [PubMed]
- Niimi, Y.; Tsuyuki, T.; Takahashi, T.; Matsushita, K. Structure determination of shinjulactones M and N, new bitter principles from Ailanthus altissima Swingle. Bull. Chem. Soc. Jpn. 1986, 59, 1638–1640. [Google Scholar] [CrossRef]
- Kim, H.M.; Kim, S.J.; Kim, H.Y.; Ryu, B.; Kwak, H.; Hur, J.; Choi, J.H.; Jang, D.S. Constituents of the stem barks of Ailanthus altissima and their potential to inhibit LPS-induced nitric oxide production. Bioorg. Med. Chem. Lett. 2015, 25, 1017–1020. [Google Scholar] [CrossRef] [PubMed]
- Ohmoto, T.; Koike, K. Studies on the constituents of Ailanthus altissima Swingle. III. The alkaloidal constituents. Chem. Pharm. Bull. 1984, 32, 170–173. [Google Scholar] [CrossRef]
- Hong, Z.L.; Xiong, J.; Wu, S.B.; Zhu, J.J.; Hong, J.L.; Zhao, Y.; Xia, G.; Hu, J.F. Tetracyclic triterpenoids and terpenylated coumarins from the bark of Ailanthus altissima (“Tree of heaven”). Phytochemistry 2013, 86, 159–167. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.J.; Xu, M.; Li, X.S.; Wang, Y.H.; Gao, Y.; Cai, R.; Cheng, Y.X. Triterpenoids and sterones from the stem bark of Ailanthus altissima. Bull. Korean Chem. Soc. 2011, 32, 127–130. [Google Scholar] [CrossRef]
- Tan, Q.W.; Ouyang, M.A.; Wu, Z.J. A new seco-neolignan glycoside from the root bark of Ailanthus altissima. Nat. Prod. Res. 2012, 26, 1375–1380. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.W.; Wu, Z.J.; Ouyang, M.A. Research progress in chemical constituents and bioactivities of Ailanthus ailanthus. Nat. Prod. Res. Dev. 2008, 20, 748–755. [Google Scholar]
- Shin, Y.; Masami, I.; Takakiko, T.; Takahashi, T.; Matsushita, K. Constituents of Ailanthus altissima Swingle. Isolation and structures of shinjuglycosides A, B, C, and D. Bull. Chem. Soc. Jpn. 1984, 57, 2496–2501. [Google Scholar]
- Zhao, C.C.; Wang, J.H.; Li, W.; Sha, Y.; Li, X. Studies on the chemical constituents of fructus Ailanthus altissima. Chin. J. Med. Chem. 2003, 13, 211–214. [Google Scholar]
- Zhao, C.C.; Shao, J.H.; Li, X.; Xu, J.; Zhang, P. Antimicrobial constituents from fruits of Ailanthus altissima Swingle. Arch. Pharm. Res. 2005, 28, 1147–1151. [Google Scholar] [CrossRef] [PubMed]
- Xie, H.; Liang, Y.; Xue, J.; Xu, Q.; Jiang, Y.; Wei, X. Secondary metabolites of the phytopathogen Peronophythora litchii. Nat. Prod. Commun. 2010, 5, 245–248. [Google Scholar] [PubMed]
- Pawlowska, A.M.; De Leo, M.; Braca, A. Phenolics of Arbutus unedo L. (Ericaceae) fruits: Identification of anthocyanins and gallic acid derivatives. J. Agric. Food Chem. 2006, 54, 10234–10238. [Google Scholar] [CrossRef] [PubMed]
- Park, T.H.; Choi, K.W.; Park, C.S.; Lee, S.B.; Kang, H.Y.; Shon, K.J.; Park, J.S.; Cha, J. Substrate specificity and transglycosylation catalyzed by a thermostable beta-glucosidase from marine hyperthermophile Thermotoga neapolitana. Appl. Microbiol. Biotechnol. 2005, 69, 411–422. [Google Scholar] [CrossRef] [PubMed]
- Joval, E.; Kroeger, P.; Towers, N. Hydroquinone: The toxic compound of Agaricus hondensis. Planta Med. 1996, 62, 185. [Google Scholar] [CrossRef] [PubMed]
- Tyman, J.H.P.; Payne, P.B. The synthesis of phenolic propane-1,2- and 1,3-diols as intermediates in immobilised chelatants for the borate anion. J. Chem. Res. 2006, 2006, 691–695. [Google Scholar] [CrossRef]
- Sadeghian, H.; Attaran, N.; Jafari, Z.; Saberi, M.R.; Seyedi, S.M.; Eshghi, H.; Pordel, M.; Riazi, M.M. Design and synthesis of 4-methoxyphenylacetic acid esters as 15-lipoxygenase inhibitors and SAR comparative studies of them. Bioorg. Med. Chem. 2009, 17, 2327–2335. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.; Liao, L.; Moore, J.; Wu, T.; Wang, Z. Antioxidant phenolic compounds from walnut kernels (Juglans regia L.). Food Chem. 2009, 113, 160–165. [Google Scholar] [CrossRef]
- Kamatham, S.; Kumar, N.; Gudipalli, P. Isolation and characterization of gallic acid and methyl gallate from the seed coats of Givotia rottleriformis Griff. and their anti-proliferative effect on human epidermoid carcinoma A431 cells. Toxicol. Rep. 2015, 2, 520–529. [Google Scholar] [CrossRef] [PubMed]
- Ma, X.; Wu, L.; Ito, Y.; Tian, W. Application of preparative high-speed counter-current chromatography for separation of methyl gallate from Acer truncatum Bunge. J. Chromatogr. A 2005, 1076, 212–215. [Google Scholar] [CrossRef] [PubMed]
- Saijo, R.; Nonaka, G.I.; Nishioka, I. Gallic acid esters of bergenin and norbergenin from Mallotus japonicus. Phytochemistry 1990, 29, 267–270. [Google Scholar] [CrossRef]
- Pfundstein, B.; El Desouky, S.K.; Hull, W.E.; Haubner, R.; Erben, G.; Owen, R.W. Polyphenolic compounds in the fruits of Egyptian medicinal plants (Terminalia bellerica, Terminalia chebula and Terminalia horrida): Characterization, quantitation and determination of antioxidant capacities. Phytochemistry 2010, 71, 1132–1148. [Google Scholar] [CrossRef] [PubMed]
- Sudjaroen, Y.; Hull, W.E.; Erben, G.; Würtele, G.; Changbumrung, S.; Ulrich, C.M.; Owen, R.W. Isolation and characterization of ellagitannins as the major polyphenolic components of Longan (Dimocarpus longan Lour) seeds. Phytochemistry 2012, 77, 226–237. [Google Scholar] [CrossRef] [PubMed]
- Wei, Y.; Xie, Q.Q.; Fisher, D.; Sutherland, I.A. Separation of patuletin-3-O-glucoside, astragalin, quercetin, kaempferol and isorhamnetin from Flaveria bidentis (L.) Kuntze by elution-pump-out high-performanoe counter-current chromatography. J. Chromatogr. A 2011, 1218, 6206–6211. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.Y.; Gao, J.J.; Lee, W.C.; Ryu, K.S.; Lee, K.R.; Kim, Y.C. Antioxidative flavonoids from the leaves of Morus alba. Arch. Pharm. Res. 1999, 22, 81–85. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.N.; Wang, W.; Ding, Y.; Xie, W.D.; Ma, C.; Du, L.J. A new flavonol glycoside and activity of compounds from the flower of Nymphaea candida. J. Asian Nat. Prod. Res. 2007, 9, 333–338. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.H.; Ku, C.H.; Baek, N.I.; Kim, S.H.; Park, H.W.; Kim, D.K. Phytochemical constituents from Diodia teres. Arch. Pharm. Res. 2004, 27, 40–43. [Google Scholar] [CrossRef] [PubMed]
- Fossen, T.; Larsen, Å.; Kiremire, B.T.; Andersen, Ø.M. Flavonoids from blue flowers of Nymphaèa caerulea. Phytochemistry 1999, 51, 1133–1137. [Google Scholar] [CrossRef]
- Hwang, E.I.; Ahn, B.T.; Lee, H.B.; Kim, Y.K.; Lee, K.S.; Bok, S.H.; Kim, Y.T.; Kim, S.U. Inhibitory activity for chitin synthase II from Saccharomyces cerevisiae by tannins and related compounds. Planta Med. 2001, 67, 501–504. [Google Scholar] [CrossRef] [PubMed]
- Tran, T.T.T.; Nguyen, T.Y.; Tran, M.H.; Weon, K.Y.; Woo, M.H.; Min, B.S. Compounds from the aerial parts of aceriphyllum rossii and their cytotoxic activity. Nat. Prod. Sci. 2014, 20, 146–151. [Google Scholar]
- Okano, M.; Fukamiya, N.; Lee, K.H. Studies in Natural Products Chemistry, Vol. 7, Biologically Active Compounds from Simaroubaceous Plants; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 1990; pp. 369–404. [Google Scholar]
- Ohmoto, T.; Koike, K.; Sakamoto, Y. Studies on the constituents of Ailanthus altissima Swingle. II. Alkaloidal constituents. Chem. Pharm. Bull. 1981, 20, 390–395. [Google Scholar] [CrossRef]
- Crespi-Perellino, N.; Guicciardi, A.; Malyszko, G. Occurrence of indole alkaloids in Ailanthus altissima cell cultures. J. Nat. Prod. 1986, 49, 1010–1014. [Google Scholar] [CrossRef]
- Souleles, C.; Kokkalou, E. A new β-carboline alkaloid from Ailanthus altissima. Planta Med. 1989, 55, 286–287. [Google Scholar] [CrossRef] [PubMed]
- Westerbeek, A.; Szymanski, W.; Feringa, B.L.; Janssen, D.B. Dynamic kinetic resolution process employing haloalkane dehalogenase. ACS Catal. 2011, 1, 1654–1660. [Google Scholar] [CrossRef]
- Freyer, A.J.; Patil, A.D.; Killmer, L.; Troupe, N.; Mentzer, M.; Carte, B.; Faucette, L.; Johnson, R.K. Threee new pseudodistomins, piperidine alkaloids from the ascidian Pseudodistoma megalarva. J. Nat. Prod. 1997, 60, 986–990. [Google Scholar] [CrossRef] [PubMed]
- Reina, L.; Bennadji, Z.; Vinciguerra, V.; Ferreira, F.; Moyna, G.; Menendez, P. Isolation and structural characterization of new piperidine alkaloids from Prosopis affinis. Phytochem. Lett. 2015, 14, 265–269. [Google Scholar] [CrossRef]
- Rahman, A.A.; Samoylenko, V.; Jacob, M.R.; Sahu, R.; Jain, S.K.; Khan, S.I.; Tekwani, B.L.; Muhammad, I. Antiparasitic and antimicrobial indolizidines from the leaves of Proscopsis glandulosa var. glandulosa. Planta Med. 2011, 77, 1639–1643. [Google Scholar] [CrossRef] [PubMed]
- Fyhrquist, P.; Virjamo, V.; Hiltunen, E.; Julkunen-Tiitto, R. Epidihydropinidine, the main piperidine alkaloid compound of Norway spruce (Picea abies) shows promising antibacterial and anti-Candida activity. Fitoterapia 2017, 117, 138–146. [Google Scholar] [CrossRef] [PubMed]
- Du, P.; Ma, Q.; Zhu, Z.D.; Li, G.; Wang, Y.; Li, Q.Q.; Chen, Y.F.; Shang, Z.Z.; Zhang, J.; Zhao, L. Mechanism of Corilagin interference with IL-13/STAT6 signaling pathways in hepatic alternative activation macrophages in schistosomiasis-induced liver fibrosis in mouse model. Eur. J. Pharmacol. 2016, 793, 119–126. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.J.; Luo, T.; Wu, F.; Liu, H.; Li, H.R.; Mei, Y.W.; Zhang, S.L.; Tao, J.Y.; Dong, J.H.; Fang, Y.; et al. Corilagin protects against HSV1 encephalitis through inhibiting the TLR2 signaling pathways in vivo and in vitro. Mol. Neurobiol. 2015, 52, 1547–1560. [Google Scholar] [CrossRef] [PubMed]
- Guo, Y.J.; Zhao, L.; Li, X.F.; Mei, Y.W.; Zhang, S.L.; Tao, J.Y.; Zhou, Y.; Dong, J.H. Effect of corilagin on anti-inflammation in HSV-1 encephalitis and HSV-1 infected microglias. Eur. J. Pharmacol. 2010, 635, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Tan, Q.W.; Ni, J.C.; Fang, P.H.; Chen, Q.J. A new erythrinan alkaloid glycoside from the seeds of Erythrina crista-galli. Molecules 2017, 22, 1558. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.G.; Zhang, Z.K.; Wu, Z.J.; Ouyang, M.A.; Xie, L.H.; Lin, Q.Y. Antiphytoviral activity of bruceine-D from Brucea javanica seeds. Pest Manag. Sci. 2008, 64, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.J.; Ouyang, M.A.; Wang, C.Z.; Zhang, Z.K.; Shen, J.G. Anti-Tobacco mosaic virus (TMV) triterpenoid saponins from the leaves of Ilex oblonga. J. Agric. Food Chem. 2007, 55, 1712–1717. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds 1–28 are available from the authors. |
| Position | 1 | 2 | ||
|---|---|---|---|---|
| δC (ppm) | δH (ppm, J in Hz) | δC (ppm) | δH (ppm, J in Hz) | |
| 1 | 173.1 | 173.2 | ||
| 2 | 67.8 | 4.15 (1H, qd, 6.8, 5.1) | 67.8 | 4.16 (1H, qd, 6.8, 5.2) |
| 3 | 20.8 | 1.33 (3H, d, 6.8) | 20.8 | 1.32 (1H, d, 6.8) |
| 1′ | 128.6 | 128.0 | ||
| 2′ | 146.5 | 146.4 | ||
| 3′ | 116.4 | 7.20 (1H, dd, 7.5, 2.1) | 116.0 | 7.28 (1H, dd, 8.2, 1.1) |
| 4′ | 123.8 | 7.05 (1H, td, 7.5, 2.0) | 124.1 | 7.05 (1H, td, 7.8, 1.8) |
| 5′ | 122.6 | 7.02 (1H, td, 7.5, 1.8) | 122.2 | 6.99 (1H, td, 7.7, 1.5) |
| 6′ | 119.6 | 8.23 (1H, dd, 7.4, 2.3) | 119.6 | 8.19 (1H, dd, 8.0, 1.8) |
| Glc-1″ | 102.1 | 4.85 (1H, d, 7.5) | 101.5 | 4.86 (1H, d, 7.4) |
| 2″ | 73.3 | 3.27–3.32 (1H, overlap) | 73.3 | 3.25–3.34 (1H, overlap) |
| 3″ | 76.5 | 3.27–3.32 (1H, overlap) | 76.4 | 3.55–3.68 (1H, overlap) |
| 4″ | 69.6 | 3.18 (1H, td, 9.2, 5.4) | 69.7 | 3.18 (1H, td, 8.7, 5.3) |
| 5″ | 77.1 | 3.27–3.32 (1H, overlap) | 76.0 | 3.25–3.34 (1H, overlap) |
| 6″ | 60.7 | 3.69 (1H, ddd, 11.8, 5.4, 2.2) | 68.3 | 4.00 (1H, d, 10.3) |
| 3.49 (1H, dt, 11.8, 5.9) | 3.55–3.68 (1H, overlap) | |||
| Glc-1‴ | 103.2 | 4.22 (1H, d, 7.8) | ||
| 2‴ | 73.6 | 2.96 (1H, td, 8.3, 4.5) | ||
| 3‴ | 76.6 | 3.10 (1H, td, 8.4, 4.5) | ||
| 4‴ | 70.1 | 2.98–3.06 (1H, overlap) | ||
| 5‴ | 76.8 | 2.98–3.06 (1H, overlap) | ||
| 6‴ | 61.0 | 3.65 (ddd, 1H, 11.4, 5.8, 1.9) | ||
| 3.41 (1H, dt, 11.4, 5.8) | ||||
| NH | 9.42 (1H, s) | 9.36 (1H, s) | ||
| OH-2 | 5.99 (1H, d, 5.1) | 5.89 (1H, d, 5.2) | ||
| OH-6″ | 4.58 (1H, t, 5.9) | |||
| OH-6‴ | 4.46 (1H, t, 5.8) | |||
| Compounds | Inhibitory Rate (%, Mean Value ± SD) | Compounds | Inhibitory Rate (%, Mean Value ± SD) |
|---|---|---|---|
| 1 | 12.2 ± 2.2 | 15 | – |
| 2 | 10.8 ± 2.5 | 16 | – |
| 3 | 24.6 ± 3.5 | 17 | 14.0 ± 4.3 |
| 4 | 42.0 ± 4.5 | 18 | 57.5 ± 4.4 |
| 5 | – | 19 | – |
| 6 | 50.5 ± 4.3 | 20 | 18.0 ± 4.8 |
| 7 | 49.8 ± 4.9 | 21 | 38.0 ± 6.5 |
| 8 | 47.0 ± 2.6 | 22 | 31.9 ± 6.3 |
| 9 | 12.7 ± 2.4 | 23 | – |
| 10 | 72.3 ± 4.9 | 24 | 13.2 ± 6.9 |
| 11 | – | 25 | 32.3 ± 5.3 |
| 12 | 43.2 ± 5.7 | 26 | 37.4 ± 5.5 |
| 13 | 16.3 ± 4.9 | 27 | 28.4 ± 1.9 |
| 14 | 37.0 ± 5.8 | 28 | 45.0 ± 6.3 |
| Ningnanmycin | 86.9 ± 3.6 | Ribavirin | 73.9 ± 4.5 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ni, J.-C.; Shi, J.-T.; Tan, Q.-W.; Chen, Q.-J. Phenylpropionamides, Piperidine, and Phenolic Derivatives from the Fruit of Ailanthus altissima. Molecules 2017, 22, 2107. https://doi.org/10.3390/molecules22122107
Ni J-C, Shi J-T, Tan Q-W, Chen Q-J. Phenylpropionamides, Piperidine, and Phenolic Derivatives from the Fruit of Ailanthus altissima. Molecules. 2017; 22(12):2107. https://doi.org/10.3390/molecules22122107
Chicago/Turabian StyleNi, Jian-Cheng, Jian-Ting Shi, Qing-Wei Tan, and Qi-Jian Chen. 2017. "Phenylpropionamides, Piperidine, and Phenolic Derivatives from the Fruit of Ailanthus altissima" Molecules 22, no. 12: 2107. https://doi.org/10.3390/molecules22122107





