RETRACTED: Fluorinated Analogs of Organosulfur Compounds from Garlic (Allium sativum): Synthesis, Chemistry and Anti-Angiogenesis and Antithrombotic Studies
Abstract
:1. Introduction
2. Results and Discussion
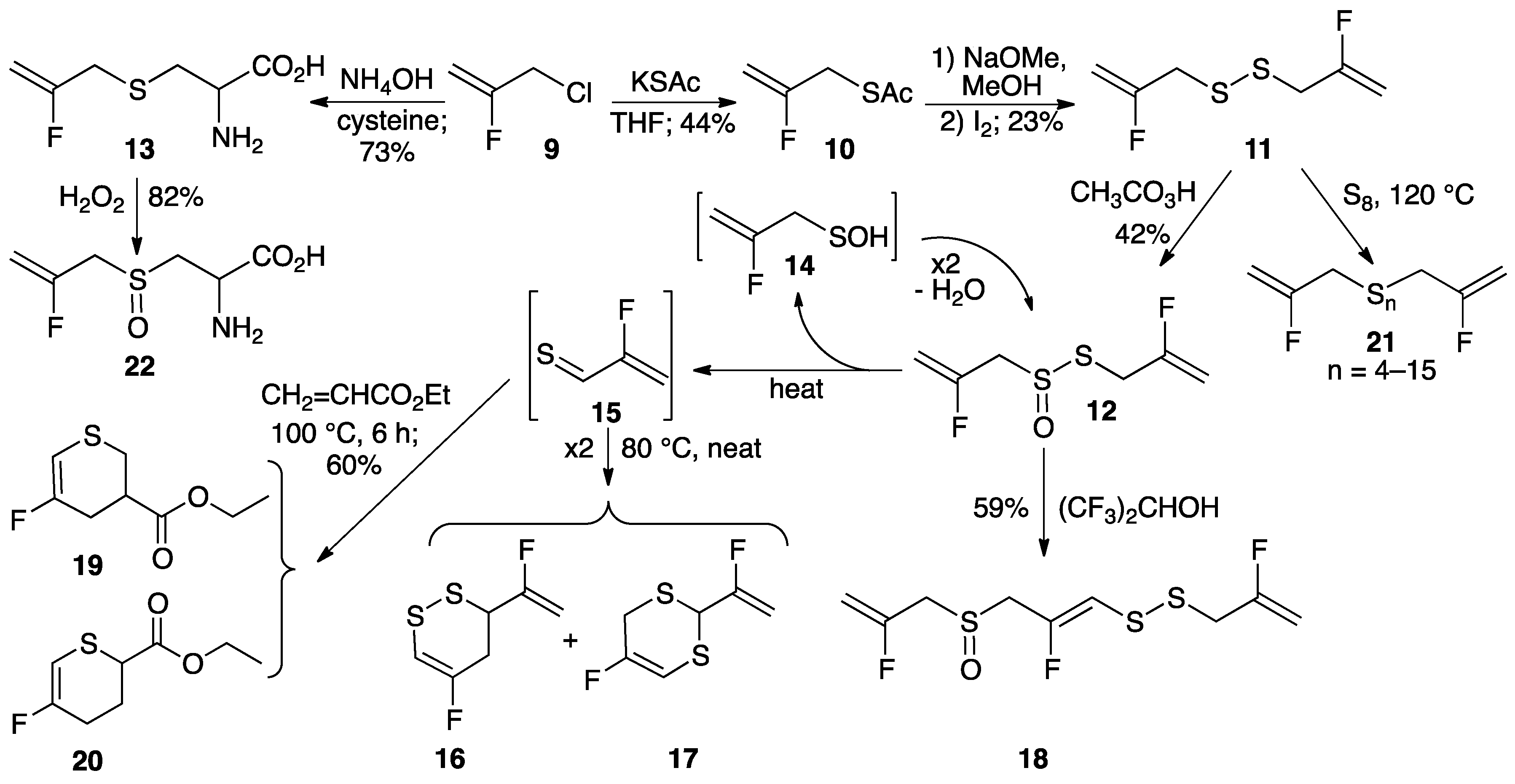
2.1. Synthesis of Fluorinated Analogues of Garlic-Derived Organosulfur Compounds
2.2. Biological Studies of Fluorinated Analogues of Garlic-Derived Organosulfur Compounds
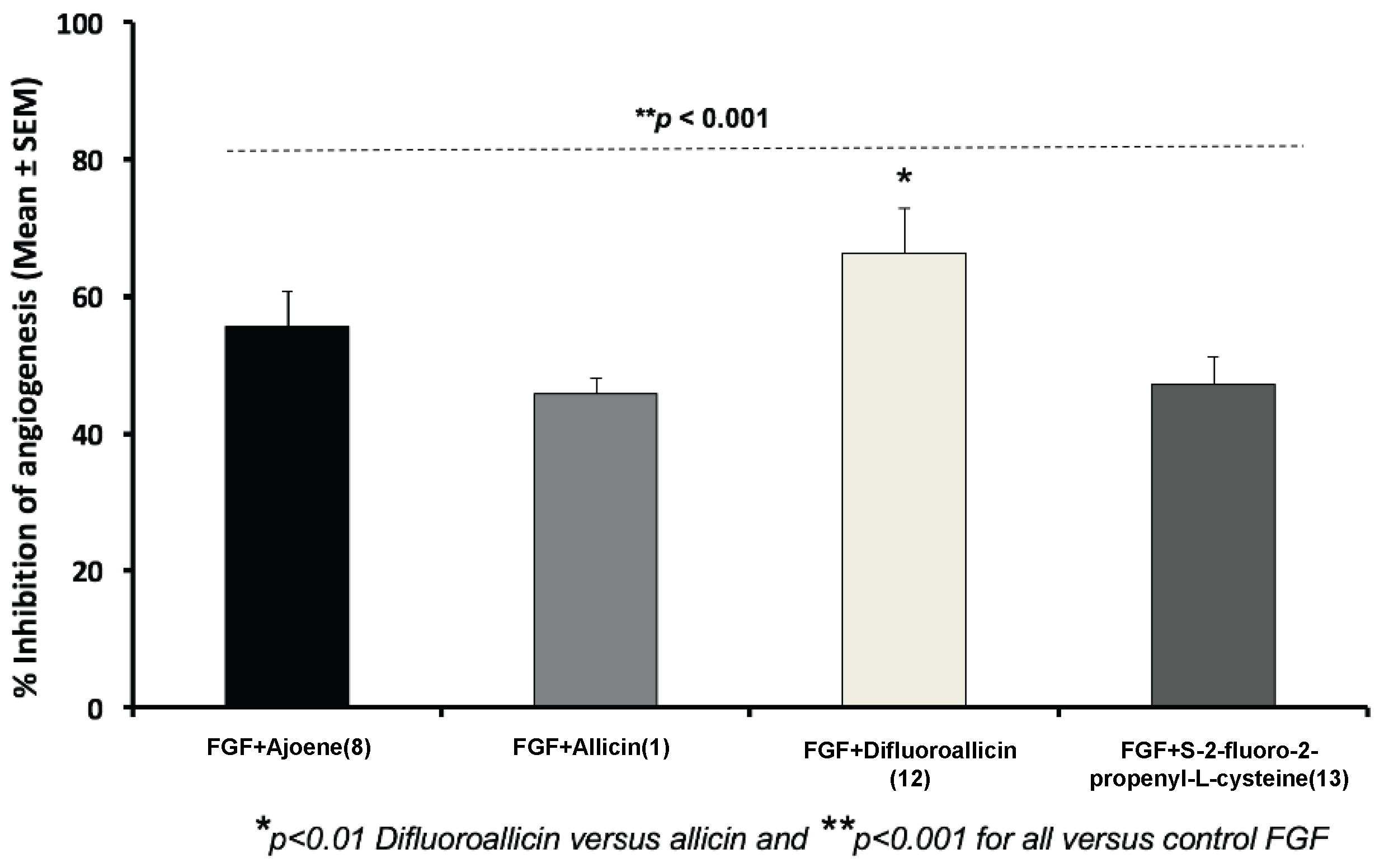
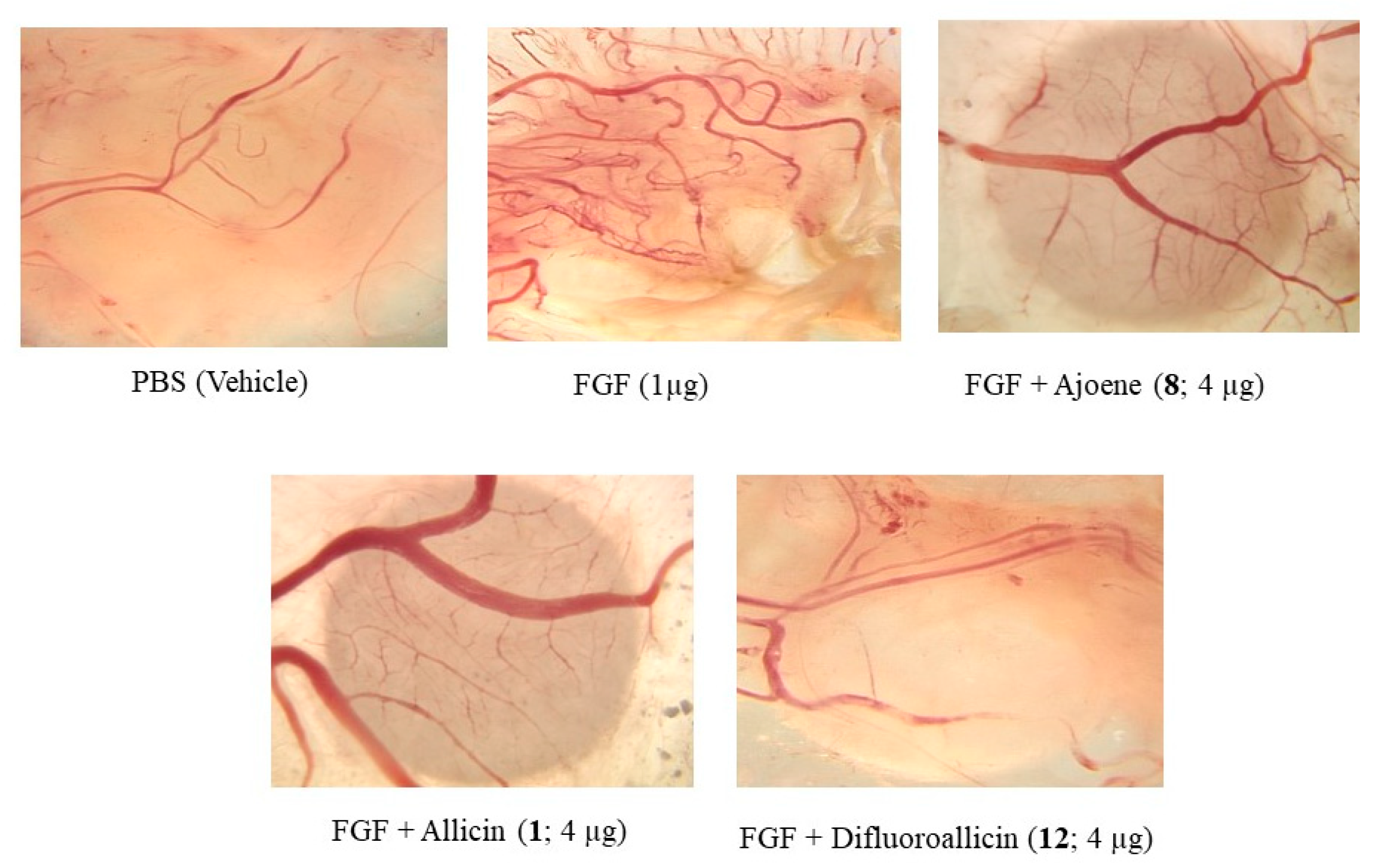
2.2.1. Anti-Angiogenesis Efficacy of Fluorinated Compounds in the Chick Chorioallantoic Membrane (CAM) Model
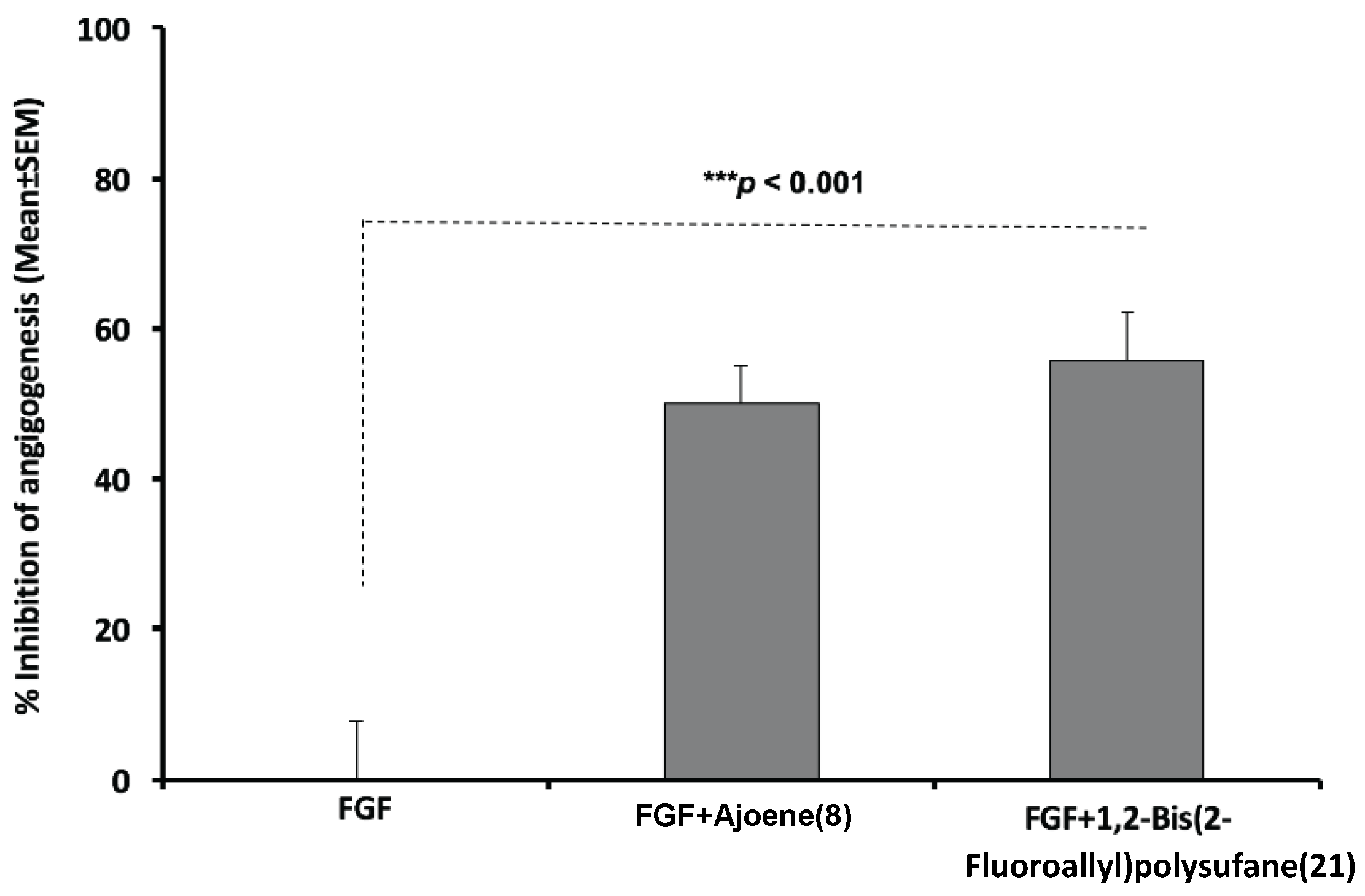
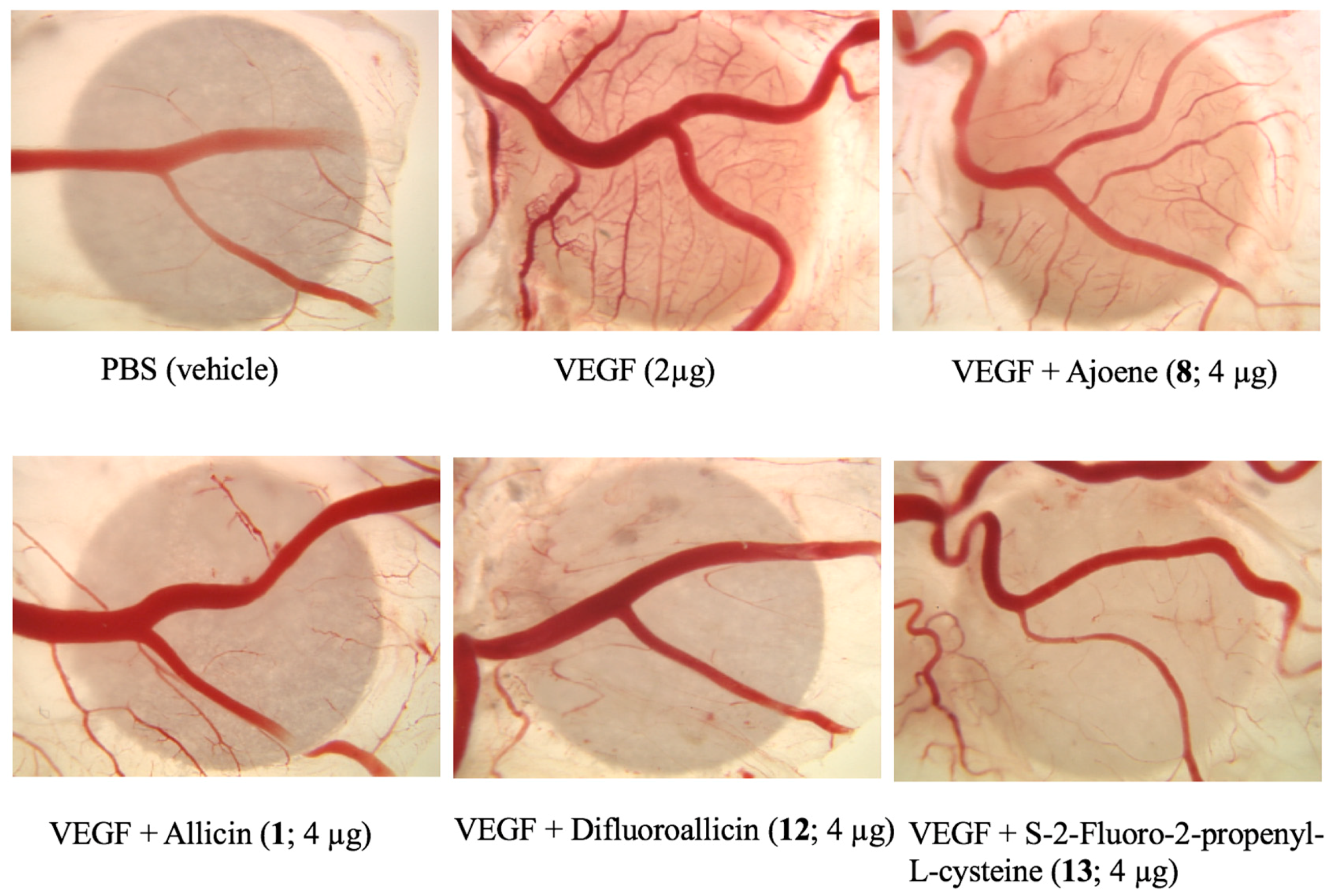
2.2.2. Anti-Angiogenesis Efficacy of Organosulfur Compounds in the Mouse Matrigel® Model
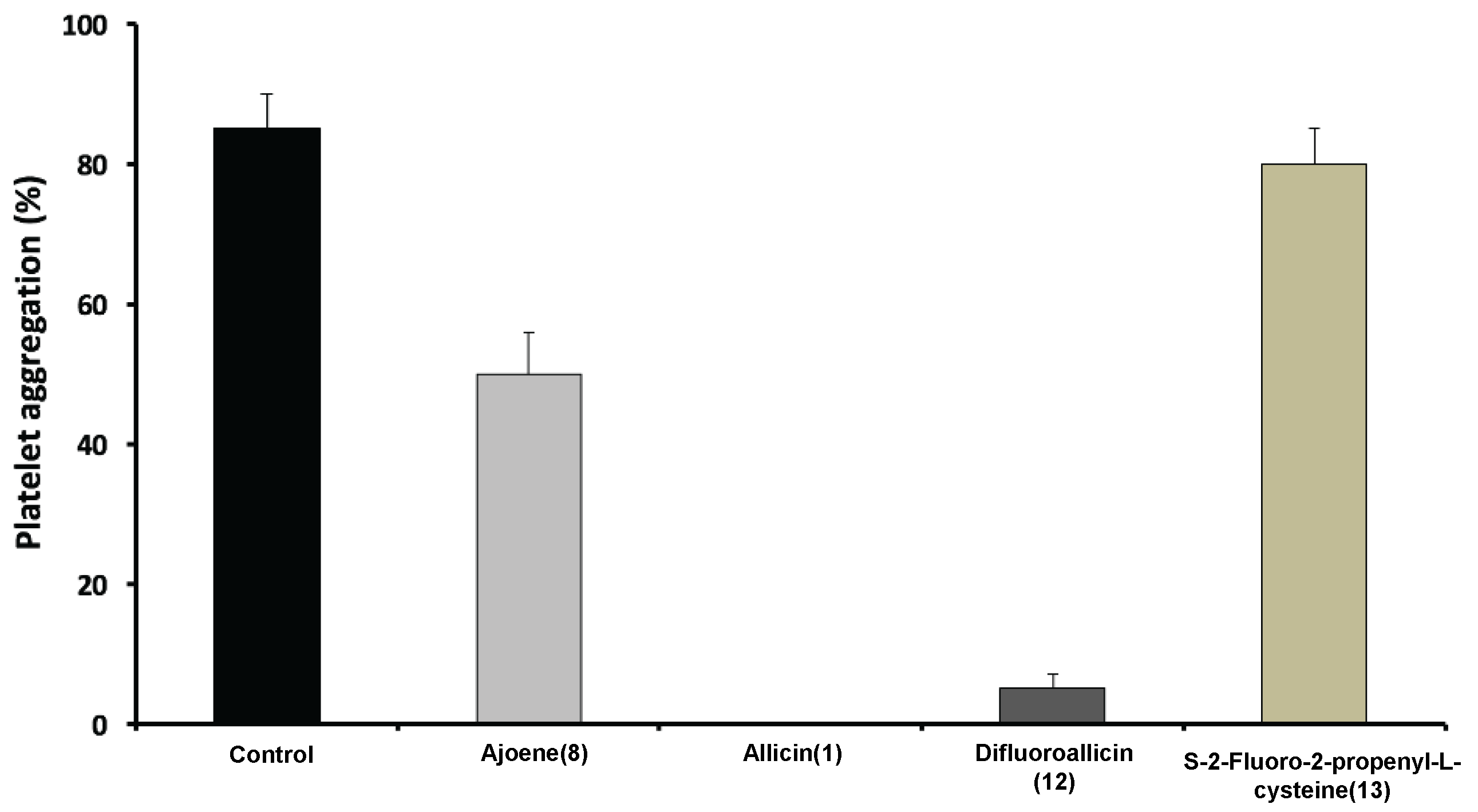
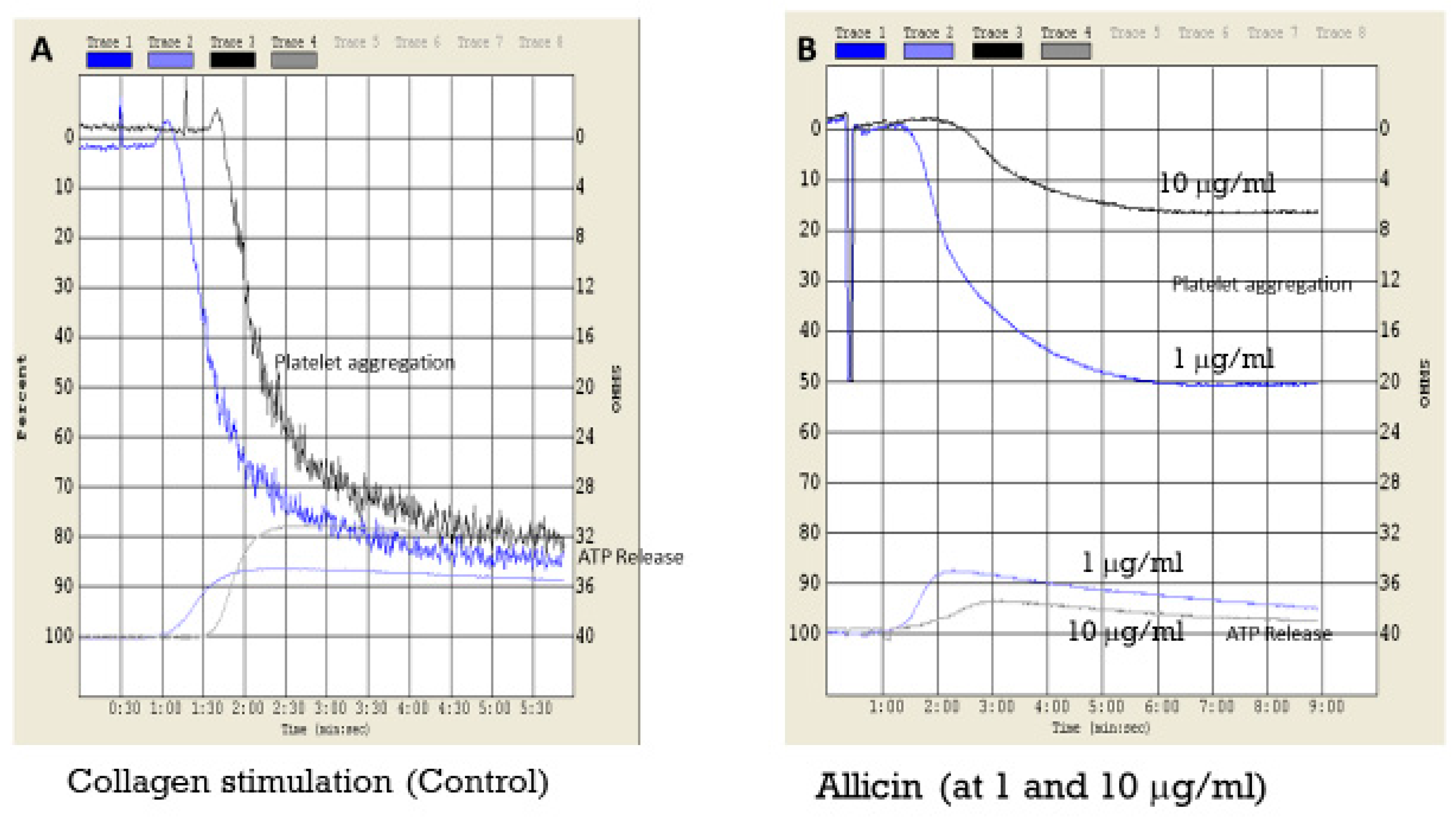
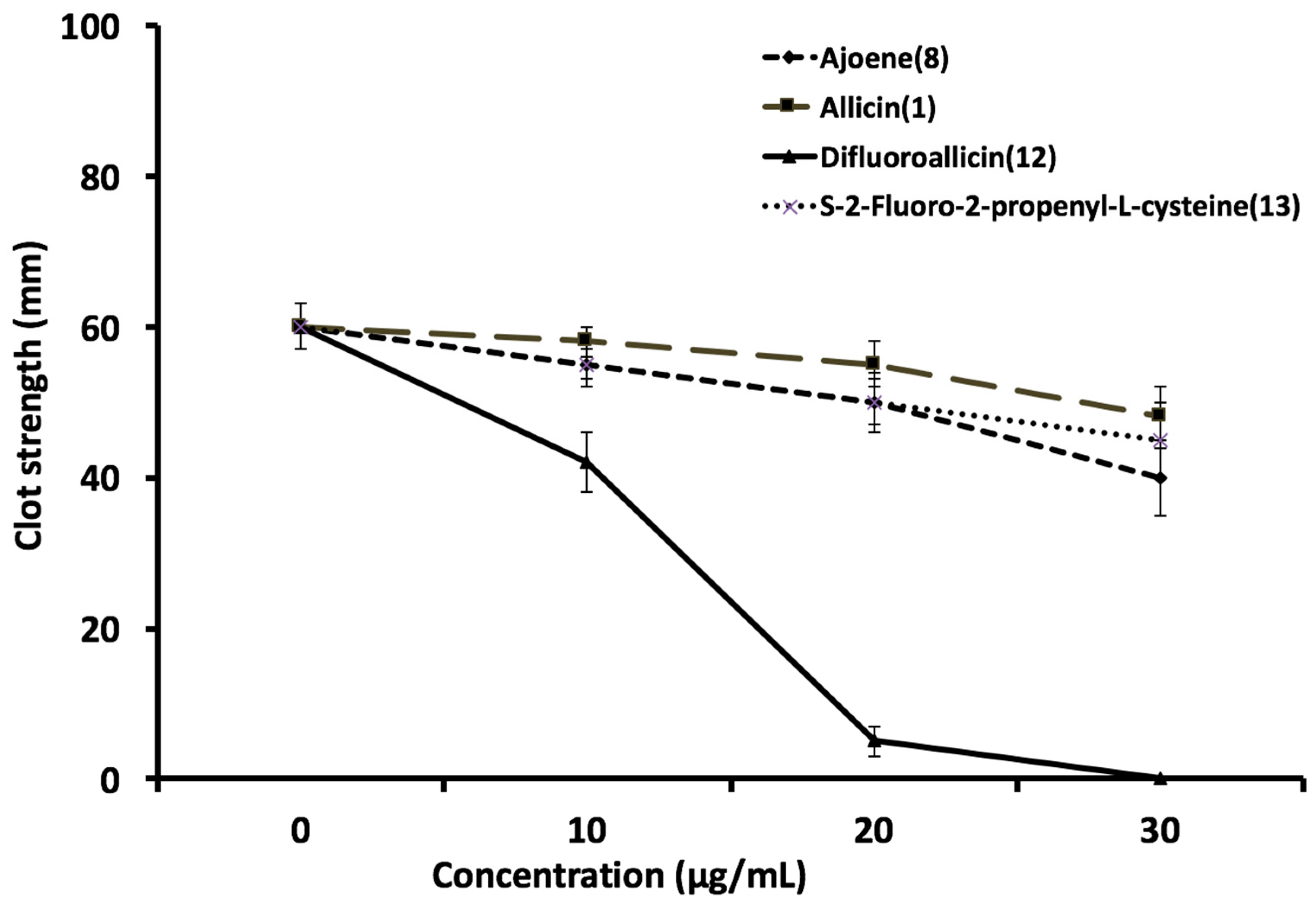
2.2.3. Effect of Fluorinated and Normal Organosulfur Compounds on Platelet Aggregation and Platelet/Fibrin Clot Dynamics
2.2.4. Effect of Fluorine Substitution on Biological Activity
3. Materials and Methods
3.1. General Procedures
3.2. Synthesis of Fluorinated Garlic Organosulfur Compounds
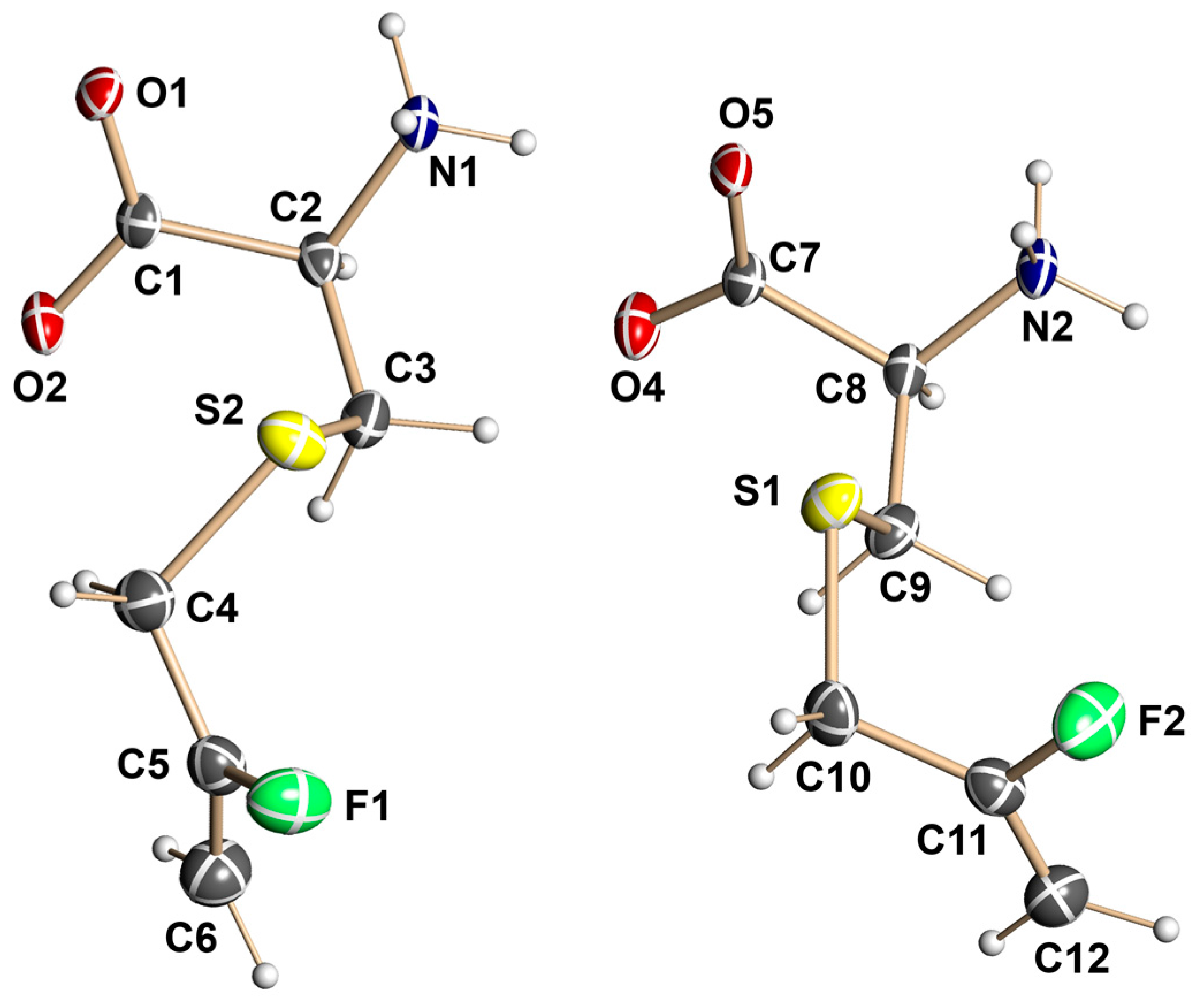
3.3. X-ray Crystallographic Structural Determination for 13
3.4. Chick Chorioallantoic Membrane (CAM) Protocols
3.4.1. Growth Factor-Mediated Angiogenesis
Chick Chorioallantoic Membrane (CAM) Model of Angiogenesis
Microscopic Analysis of CAM Sections
3.4.2. Mouse Matrigel®-Growth Factors Implant Angiogenesis Model
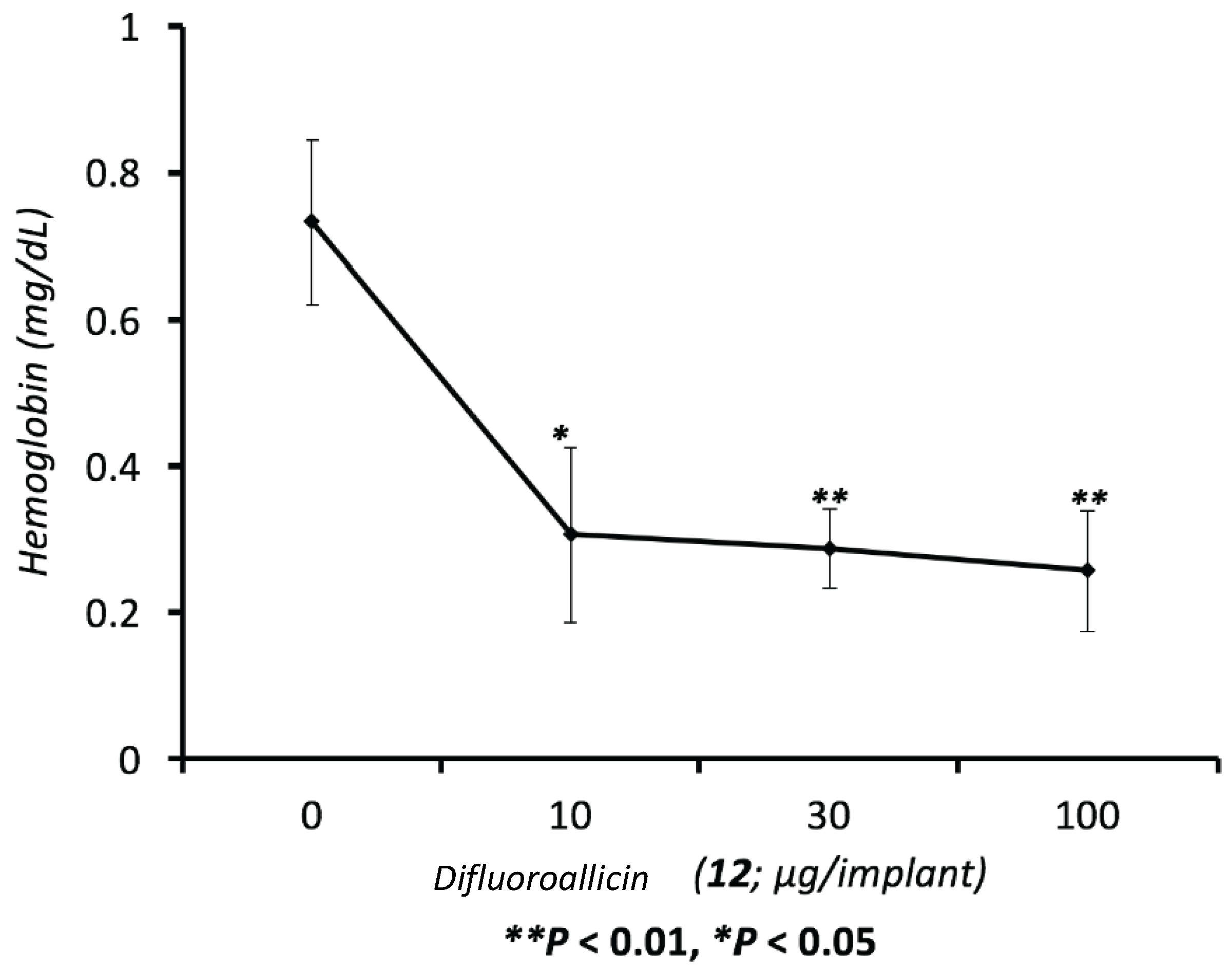
3.4.3. Determination of Hemoglobin (Hb) Levels (Measure of Angiogenesis Index)
3.4.4. Platelet Aggregation in Whole Blood (Impedance Method)
3.4.5. Global Coagulation Assay (Thrombelastography)
Blood Sampling
Thrombelastography (TEG)
4. Conclusions
X-Ray Crystallographic Data
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Block, E. Garlic and Other Alliums: The Lore and the Science; Royal Society of Chemistry: Cambridge, UK, 2010. [Google Scholar]
- Block, E. Garlic and Other Alliums: The Lore and the Science; Chemistry Industry Press: Beijing, China, 2017. (In Chinese) [Google Scholar]
- Reiter, J.; Levina, N.; van der Linden, M.; Gruhlke, M.; Martin, C.; Slusarenko, A. Diallylthiosulfinate (Allicin), a Volatile Antimicrobial from Garlic (Allium Sativum), Kills Human Lung Pathogenic Bacteria, Including MDR Strains, as a Vapor. Molecules 2017, 22, 1711. [Google Scholar] [CrossRef] [PubMed]
- Borlinghaus, J.; Albrecht, F.; Gruhlke, M.; Nwachukwu, I.; Slusarenko, A. Allicin: Chemistry and Biological Properties. Molecules 2014, 19, 12591–12618. [Google Scholar] [CrossRef] [PubMed]
- Mousa, A.S.; Mousa, S.A. Anti-Angiogenesis Efficacy of the Garlic Ingredient Alliin and Antioxidants: Role of Nitric Oxide and p53. Nutr. Cancer 2005, 53, 104–110. [Google Scholar] [CrossRef] [PubMed]
- Sela, U.; Brill, A.; Kalchenko, V.; Dashevsky, O.; Hershkoviz, R. Allicin Inhibits Blood Vessel Growth and Downregulates Akt Phosphorylation and Actin Polymerization. Nutr. Cancer 2008, 60, 412–420. [Google Scholar] [CrossRef] [PubMed]
- Mathan, S.V.; Singh, S.V.; Singh, R.P. Fighting Cancer with Phytochemicals from Allium Vegetables. Mol. Cancer Biol. 2017, 1, e1–e23. [Google Scholar]
- Beretta, H.V.; Bannoud, F.; Insani, M.; Berli, F.; Hirschegger, P.; Galmarini, C.R.; Cavagnaro, P.F. Relationships among Bioactive Compounds Content and the Antiplatelet and Antioxidant Activities of Six Allium Vegetable Species. Food Technol. Biotechnol. 2017, 55, 266–275. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Sánchez-Roselló, M.; Aceña, J.L.; del Pozo, C.; Sorochinsky, A.E.; Fustero, S.; Soloshonok, V.A.; Liu, H. Fluorine in Pharmaceutical Industry: Fluorine-Containing Drugs Introduced to the Market in the Last Decade (2001–2011). Chem. Rev. 2014, 114, 2432–2506. [Google Scholar] [CrossRef] [PubMed]
- Hiyama, T.; Yamamoto, H. Biologically active organofluorine compounds. In Organofluorine Compounds: Chemistry and Applications; Yamamoto, H., Ed.; Springer: Berlin/Heidelberg, Germany, 2000; pp. 137–182. [Google Scholar]
- Han, Z.; Czap, G.; Chiang, C.; Xu, C.; Wagner, P.J.; Wei, X.; Zhang, Y.; Wu, R.; Ho, W. Imaging the Halogen Bond in Self-Assembled Halogenbenzenes on Silver. Science 2017, 358, 206–210. [Google Scholar] [CrossRef] [PubMed]
- Block, E.; Booker, S.J.; Flores-Penalba, S.; George, G.N.; Gundala, S.; Landgraf, B.J.; Liu, J.; Lodge, S.N.; Pushie, M.J.; Rozovsky, S.; et al. Trifluoroselenomethionine: A New Unnatural Amino Acid. ChemBioChem 2016, 17, 1738–1751. [Google Scholar] [CrossRef] [PubMed]
- Wallock-Richards, D.; Doherty, C.J.; Doherty, L.; Clarke, D.J.; Place, M.; Govan, J.R.W.; Campopiano, D.J. Garlic Revisited: Antimicrobial Activity of Allicin-Containing Garlic Extracts against Burkholderia Cepacia Complex. PLoS ONE 2014, 9, e112726. [Google Scholar] [CrossRef] [PubMed]
- Müller, A.; Eller, J.; Albrecht, F.; Prochnow, P.; Kuhlmann, K.; Bandow, J.E.; Slusarenko, A.J.; Leichert, L.I.O. Allicin Induces Thiol Stress in Bacteria through S-Allylmercapto Modification of Protein Cysteines. J. Biol. Chem. 2016, 291, 11477–11490. [Google Scholar] [CrossRef] [PubMed]
- Gruhlke, M.C.H.; Schlembach, I.; Leontiev, R.; Uebachs, A.; Gollwitzer, P.U.G.; Weiss, A.; Delaunay, A.; Toledano, M.; Slusarenko, A.J. Yap1p, the Central Regulator of the S. cerevisiae Oxidative Stress Response, Is Activated by Allicin, a Natural Oxidant and Defence Substance of Garlic. Free Radic. Biol. Med. 2017, 108, 793–802. [Google Scholar] [CrossRef] [PubMed]
- Guiot, J.; Ameduri, B.; Boutevin, B. Synthesis and Polymerization of Fluorinated Monomers Bearing a Reactive Lateral Group. XII. Copolymerization of Vinylidene Fluoride with 2,3,3-Trifluoroprop-2-Enol. J. Polym. Sci. Part A Polym. Chem. 2002, 40, 3634–3643. [Google Scholar] [CrossRef]
- Wlassics, I.; Tortelli, V.; Carella, S.; Monzani, C.; Marchionni, G. Perfluoro Allyl Fluorosulfate (FAFS): A Versatile Building Block for New Fluoroallylic Compounds. Molecules 2011, 16, 6512–6540. [Google Scholar] [CrossRef] [PubMed]
- Daniele, G.; Corral, J.; Molife, L.R.; de Bono, J.S. FGF Receptor Inhibitors: Role in Cancer Therapy. Curr. Oncol. Rep. 2012, 14, 111–119. [Google Scholar] [CrossRef] [PubMed]
- Falanga, A.; Russo, L.; Milesi, V.; Vignoli, A. Mechanisms and Risk Factors of Thrombosis in Cancer. Crit. Rev. Oncol. Hematol. 2017, 118, 79–83. [Google Scholar] [CrossRef] [PubMed]
- Meikle, C.K.S.; Kelly, C.A.; Garg, P.; Wuescher, L.M.; Ali, R.A.; Worth, R.G. Cancer and Thrombosis: The Platelet Perspective. Front. Cell Dev. Biol. 2017, 4, 147. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.A. Antithrombotic Effects of Naturally Derived Products on Coagulation and Platelet Function. Methods Mol. Biol. 2010, 663, 229–240. [Google Scholar] [PubMed]
- Chan, J.Y.-Y.; Yuen, A.C.-Y.; Chan, R.Y.-K.; Chan, S.-W. A Review of the Cardiovascular Benefits and Antioxidant Properties of Allicin. Phyther. Res. 2013, 27, 637–646. [Google Scholar] [CrossRef] [PubMed]
- Wang, K.; Groom, M.; Sheridan, R.; Zhang, S.; Block, E. Liquid Sulfur as a Reagent: Synthesis of Polysulfanes with 20 or More Sulfur Atoms with Characterization by UPLC-(Ag+)-Coordination Ion Spray-MS. J. Sulfur Chem. 2013, 34, 55–66. [Google Scholar] [CrossRef]
- Li, Z.; Wang, C.; Fu, Y.; Guo, Q.-X.; Liu, L. Substituent Effect on the Efficiency of Desulfurizative Rearrangement of Allylic Disulfides. J. Org. Chem. 2008, 73, 6127–6136. [Google Scholar] [CrossRef] [PubMed]
- Folkman, J.; Shing, Y. Angiogenesis. J. Biol. Chem. 1992, 267, 10931–10934. [Google Scholar] [PubMed]
- Laschke, M.; Menger, M. The Dorsal Skinfold Chamber: A Versatile Tool for Preclinical Research in Tissue Engineering and Regenerative Medicine. Eur. Cells Mater. 2016, 32, 202–215. [Google Scholar] [CrossRef] [PubMed]
- Mayeux, P.R.; Agrawal, K.C.; Tou, J.S.; King, B.T.; Lippton, H.L.; Hyman, A.L.; Kadowitz, P.J.; McNamara, D.B. The Pharmacological Effects of Allicin, a Constituent of Garlic Oil. Agents Actions 1988, 25, 182–190. [Google Scholar] [CrossRef] [PubMed]
- Gruhlke, M.; Nicco, C.; Batteux, F.; Slusarenko, A. The Effects of Allicin, a Reactive Sulfur Species from Garlic, on a Selection of Mammalian Cell Lines. Antioxidants 2016, 6, 1. [Google Scholar] [CrossRef] [PubMed]
- Tsuchiya, H.; Nagayama, M. Garlic Allyl Derivatives Interact with Membrane Lipids to Modify the Membrane Fluidity. J. Biomed. Sci. 2008, 15, 653–660. [Google Scholar] [CrossRef] [PubMed]
- Sheldrick, G.M. Crystal Structure Refinement with SHELXL. Acta Crystallogr. Sect. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Marcinkiewicz, C.; Weinreb, P.H.; Calvete, J.J.; Kisiel, D.G.; Mousa, S.A.; Tuszynski, G.P.; Lobb, R.R. Obtustatin. Cancer Res. 2003, 63, 2020–2023. [Google Scholar] [PubMed]
- Deryugina, E.I.; Quigley, J.P. Chick embryo chorioallantoic membrane models to quantify angiogenesis induced by inflammatory and tumor cells or purified effector molecules. Methods Enzymol. 2008, 444, 21–41. [Google Scholar] [PubMed]
- Bridoux, A.; Cui, H.; Dyskin, E.; Yalcin, M.; Mousa, S.A. Semisynthesis and Pharmacological Activities of Tetrac Analogs: Angiogenesis Modulators. Bioorg. Med. Chem. Lett. 2009, 19, 3259–3263. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.S.; Davis, F.B.; Davis, P.J.; Mousa, S.A. Human Platelet Aggregation and Degranulation Is Induced In Vitro by L-Thyroxine, but Not by 3,5,3′-Triiodo-l-Thyronine or Diiodothyropropionic Acid (DITPA). Clin. Appl. Thromb. 2010, 16, 288–293. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.A. In Vitro Efficacy of Different Platelet Glycoprotein IIb/IIIa Antagonists and Thrombolytics on Platelet/fibrin-Mediated Clot Dynamics in Human Whole Blood Using Thrombelastography. Blood Coagul. Fibrinolysis 2007, 18, 55–60. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.A.; Forsythe, M.S. Comparison of the Effect of Different Platelet GPIIb/IIa Antagonists on the Dynamics of Platelet/fibrin-Mediated Clot Strength Induced Using Thromboelastography. Thromb. Res. 2001, 104, 49–56. [Google Scholar] [CrossRef]
- Mousa, S.A. Comparative Efficacy of Different Low-Molecular-Weight Heparins (LMWHs) and Drug Interactions with LMWH: Implications for Management of Vascular Disorders. Semin. Thromb. Hemost. 2000, 26 (Suppl. 1), 39–46. [Google Scholar] [CrossRef] [PubMed]
- Mousa, S.A.; Khurana, S.; Forsythe, M.S. Comparative in Vitro Efficacy of Different Platelet Glycoprotein IIb/IIIa Antagonists on Platelet-Mediated Clot Strength Induced by Tissue Factor with Use of Thromboelastography: Differentiation among Glycoprotein IIb/IIIa Antagonists. Arterioscler. Thromb. Vasc. Biol. 2000, 20, 1162–1167. [Google Scholar] [CrossRef] [PubMed]
- Laue, K.W.; Haufe, G. 3-Bromo-2-Fluoropropene—A Fluorinated Building Block. 2-Fluoroallylation of Glycine and Alanine Ester Imines. Synthesis 1998, 1998, 1453–1456. [Google Scholar] [CrossRef]
- Moore, L.O.; Henry, J.P.; Clark, J.W. Chlorination of 2-Fluoropropene. 3-Chloro-2-Fluoropropene and Some of Its Derivatives. J. Org. Chem. 1970, 35, 4201–4204. [Google Scholar] [CrossRef]
- Tkachenko, A.N.; Radchenko, D.S.; Mykhailiuk, P.K.; Grygorenko, O.O.; Komarov, I.V. 4-Fluoro-2,4-Methanoproline. Org. Lett. 2009, 11, 5674–5676. [Google Scholar] [CrossRef] [PubMed]
- Smyth, H.F.; Carpenter, C.P.; Well, C.S.; Pozzani, U.C.; Striegel, J.A. Range-Finding Toxicity Data: List VI. Am. Ind. Hyg. Assoc. J. 1962, 23, 95–107. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds described in the text are not available from the authors. |




| Bond Lengths | |||||
| S(1)-C(10) | 1.806(3) | C(10)-C(11) | 1.472(5) | F(1)-C(5) | 1.360(4) |
| S(1)-C(9) | 1.812(3) | C(9)-C(8) | 1.512(5) | C(1)-C(2) | 1.524(4) |
| O(5)-C(7) | 1.242(4) | C(11)-C(12) | 1.287(5) | C(5)-C(6) | 1.286(5) |
| O(4)-C(7) | 1.238(4) | S(2)-C(4) | 1.803(4) | C(5)-C(4) | 1.477(5) |
| N(2)-C(8) | 1.479(4) | S(2)-C(3) | 1.815(3) | C(2)-N(1) | 1.488(4) |
| F(2)-C(11) | 1.362(4) | O(1)-C(1) | 1.242(4) | C(2)-C(3) | 1.510(4) |
| C(7)-C(8) | 1.525(4) | O(2)-C(1) | 1.247(4) | ||
| Angles | |||||
| C(4)-S(2)-C(3) | 100.4(2) | C(6)-C(5)-F(1) | 119.4(3) | N(1)-C(2)-C(1) | 109.6(3) |
| O(1)-C(1)-O(2) | 125.8(3) | C(6)-C(5)-C(4) | 129.0(4) | C(3)-C(2)-C(1) | 112.4(3) |
| O(1)-C(1)-C(2) | 118.5(3) | F(1)-C(5)-C(4) | 111.5(3) | C(2)-C(3)-S(2) | 113.4(2) |
| O(2)-C(1)-C(2) | 115.7(3) | N(1)-C(2)-C(3) | 110.8(3) | C(5)-C(4)-S(2) | 113.4(3) |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Block, E.; Bechand, B.; Gundala, S.; Vattekkatte, A.; Wang, K.; Mousa, S.S.; Godugu, K.; Yalcin, M.; Mousa, S.A. RETRACTED: Fluorinated Analogs of Organosulfur Compounds from Garlic (Allium sativum): Synthesis, Chemistry and Anti-Angiogenesis and Antithrombotic Studies. Molecules 2017, 22, 2081. https://doi.org/10.3390/molecules22122081
Block E, Bechand B, Gundala S, Vattekkatte A, Wang K, Mousa SS, Godugu K, Yalcin M, Mousa SA. RETRACTED: Fluorinated Analogs of Organosulfur Compounds from Garlic (Allium sativum): Synthesis, Chemistry and Anti-Angiogenesis and Antithrombotic Studies. Molecules. 2017; 22(12):2081. https://doi.org/10.3390/molecules22122081
Chicago/Turabian StyleBlock, Eric, Benjamin Bechand, Sivaji Gundala, Abith Vattekkatte, Kai Wang, Shaymaa S. Mousa, Kavitha Godugu, Murat Yalcin, and Shaker A. Mousa. 2017. "RETRACTED: Fluorinated Analogs of Organosulfur Compounds from Garlic (Allium sativum): Synthesis, Chemistry and Anti-Angiogenesis and Antithrombotic Studies" Molecules 22, no. 12: 2081. https://doi.org/10.3390/molecules22122081
APA StyleBlock, E., Bechand, B., Gundala, S., Vattekkatte, A., Wang, K., Mousa, S. S., Godugu, K., Yalcin, M., & Mousa, S. A. (2017). RETRACTED: Fluorinated Analogs of Organosulfur Compounds from Garlic (Allium sativum): Synthesis, Chemistry and Anti-Angiogenesis and Antithrombotic Studies. Molecules, 22(12), 2081. https://doi.org/10.3390/molecules22122081







