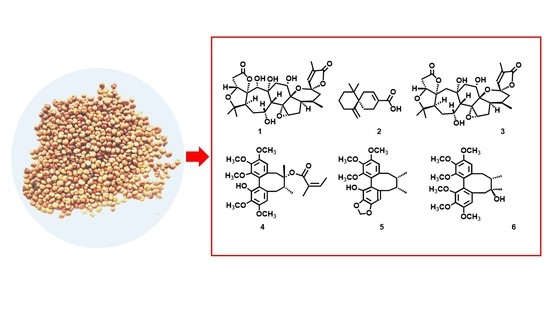
A New Nortriterpenoid, a Sesquiterpene and Hepatoprotective Lignans Isolated from the Fruit of Schisandra chinensis
Abstract
:1. Introduction
2. Results and Discussion
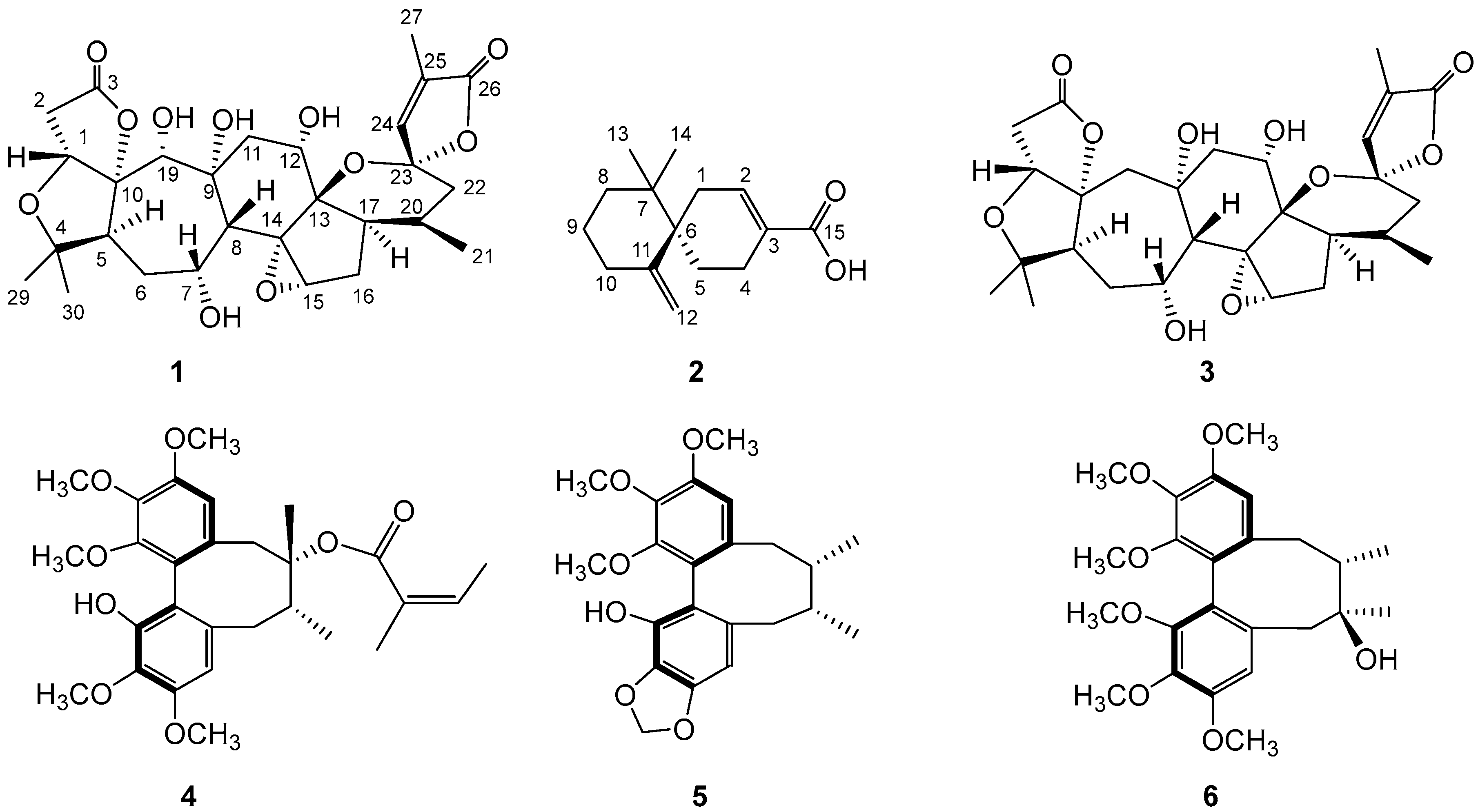
2.1. Structural Analysis
2.2. Hepatoprotective Activities of 4–6
3. Materials and Methods
3.1. Plant Material
3.2. Chemicals and Instruments
3.3. Extraction and Isolation
3.4. Characterization of Compounds 1–2
3.4.1. 19(R)-Hydroxyl-Wuweizidilactone H (1)
3.4.2. (6R)-β-Chamigrenic Acid (2)
3.5. Hepatoprotective Effects of Compounds on Damaged HepG2 Cells Induced by APAP
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Alexander, P.; Wikman, G. Pharmacology of Schisandra chinensis Bail.: An overview of Russian research and uses in medicine. J. Ethnopharmacol. 2008, 118, 183–212. [Google Scholar] [CrossRef]
- Liu, H.; Lai, H.; Jia, X.; Liu, J.; Zhang, Z.; Qi, Y.; Zhang, J.; Song, J.; Wu, C.; Zhang, B.; et al. Comprehensive chemical analysis of Schisandra chinensis by HPLC-DAD-MS combined with chemometrics. Phytomedicine 2013, 20, 1135–1143. [Google Scholar] [CrossRef] [PubMed]
- Smejkal, K.; Slapetova, T.; Krmencik, P.; Babula, P.; Dall’Acqua, S.; Innocenti, G.; Vanco, J.; Casarin, E.; Carrara, M.; Kalvarova, K.; et al. Evaluation of cytotoxic activity of Schisandra chinensis lignans. Planta Med. 2010, 76, 1672–1677. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.C.; Liaw, C.C.; Cheng, Y.B.; Lin, Y.C.; Chen, C.H.; Huang, Y.T.; Liou, S.S.; Chen, S.Y.; Chien, C.T.; Lee, G.C.; et al. Anti-liver fibrotic lignans from the fruits of Schisandra arisanensis and Schisandra sphenanthera. Bioorg. Med. Chem. Lett. 2013, 23, 880–885. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Li, B.; Liu, X.; Huang, G.; Meng, X. Purification of six lignans from the stems of Schisandra chinensis by using high-speed counter-current chromatography combined with preparative high-performance liquid chromatography. Food Chem. 2015, 186, 146–152. [Google Scholar] [CrossRef] [PubMed]
- Xue, Y.; Zhang, Y.; Yang, J.; Du, X.; Pu, J.; Zhao, W.; Li, X.; Xiao, W.; Sun, H. Nortriterpenoids and lignans from the fruit of Schisandra chinensis. Chem. Pharm. Bull. 2010, 58, 1606–1611. [Google Scholar] [CrossRef] [PubMed]
- Szopa, A.; Ekiert, R.; Ekiert, H. Current knowledge of Schisandra chinensis (Turcz.) Baill. (Chinese magnolia vine) as a medicinal plant species: A review on the bioactive components, pharmacological properties, analytical and biotechnological studies. Phytochem. Rev. 2017, 16, 195–218. [Google Scholar] [CrossRef] [PubMed]
- Szopa, A.; Ekiert, H. Production of schisantherin A and gomisin G in in vitro cultures of Schisandra chinensis. Phytochem. Lett. 2015, 11, 440–444. [Google Scholar] [CrossRef]
- Li, L.; Liu, H.; Zhang, R.; Zeng, J.; Wu, L. Two new schisdilactone-type compounds from Schisandra chinensis. J. Asian Nat. Prod. Res. 2013, 15, 1168–1172. [Google Scholar] [CrossRef] [PubMed]
- Wang, O.; Cheng, Q.; Liu, J.; Wang, Y.; Zhao, L.; Zhou, F.; Ji, B. Hepatoprotective effect of Schisandra chinensis (Turcz.) Baill. lignans and its formula with Rubus idaeus on chronic alcohol-induced liver injury in mice. Food Funct. 2014, 5, 3018–3025. [Google Scholar] [CrossRef] [PubMed]
- Liu, G. From the research of Schisandra chinensis to the discovery of Bifendate. Yao Xue Xue Bao 1983, 18, 714–720. [Google Scholar] [PubMed]
- Zhang, C.; Wang, J. The clinic application of Bifendate. Shi Yong Yi Xue Za Zhi 1986, 2, 31–32. [Google Scholar]
- Song, Q.; Gao, K.; Nan, Z. Highly oxygenated triterpenoids from the roots of Schisandra chinensis and their anti-inflammatory activities. J. Asian Nat. Prod. Res. 2016, 18, 189–194. [Google Scholar] [CrossRef] [PubMed]
- Slanina, J.; Pachnikova, G.; Carnecka, M.; Porubova, L.; Adamkova, L.; Humpa, O.; Smejkal, K.; Slaninova, I. Identification of key structural characteristics of Schisandra chinensis lignans involved in P-glycoprotein inhibition. J. Nat. Prod. 2014, 77, 2255–2263. [Google Scholar] [CrossRef] [PubMed]
- Xiao, W.; Huang, S.; Wang, R.; Zhong, J.; Gao, X.; He, F.; Pu, J.; Lu, Y.; Zheng, Y.; Zheng, Q.; et al. Nortriterpenoids and lignans from Schisandra sphenanthera. Phytochemistry 2008, 69, 2862–2866. [Google Scholar] [CrossRef] [PubMed]
- Huang, S.; Han, Q.; Lei, C.; Pu, J.; Xiao, W.; Yu, J.; Yang, L.; Xu, H.; Zheng, Y.; Sun, H. Isolation and characterization of miscellaneous terpenoids of Schisandra chinensis. Tetrahedron 2008, 64, 4260–4267. [Google Scholar] [CrossRef]
- Li, R.; Han, Q.; Zhen, Y.; Wang, R.; Yang, L.; Peng, L.; Xiao, W.; Sun, H. Anti-HIV lignans from Schisandra chinensis. Chin. J. Nat. Med. 2005, 3, 208–212. [Google Scholar]
- Ikeya, Y.; Taguchi, H.; Yosioka, I. The constituents of Schisandra chinensis Baill. X. The structures of γ-schizandrin and four new lignans, (−)-gomisin L1 and L2, (±)-gomisin M1 and (+)-gomisin M2. Chem. Pharm. Bull. 1982, 30, 132–139. [Google Scholar] [CrossRef]
- Ikeya, Y.; Taguchi, H.; Yosioka, I.; Kobayashi, H. The constituents of Schisandra chinensis Baill. I. Isolation and structure determination of five new lignans, gomisin A, B, C, F and G, and the absolute structure of Schizandrin. Chem. Pharm. Bull. 1979, 27, 1383–1394. [Google Scholar] [CrossRef] [PubMed]
- Kuo, Y.; Lin, Y. Two new sesquiterpenes 3β-hydroxycedrol and widdringtonia acid II-A co-crystal of β-chamigrenic acid and hinokiic acid. J. Chin. Chem. Soc. 1980, 27, 15–18. [Google Scholar] [CrossRef]
- Hikino, H.; Hosaka, K.; Ogawa, Y.; Iketani, Y.; Kubota, K.; Taguchi, H. Chamigrenol Derivatives for Treatment of Liver Failure. Kokai Tokkyo Koho JP Patent 1984-113121 A, 20 December 1985. [Google Scholar]
- Pei, J.; Lv, Q.; Han, J.; Li, X.; Jin, S.; Huang, Y.; Jin, S.; Yuan, H. Schisandra lignans-loaded enteric nanoparticles: Preparation, characterization, and in vitro-in vivo evaluation. J. Drug Target. 2013, 21, 180–187. [Google Scholar] [CrossRef] [PubMed]
- Liang, J.; Hou, H.; Lan, X.; Sun, Y.; Chen, Y. Advance in study on pharmacological effect and molecular mechanism of schisandrin B. Chin. J. Mod. Appl. Pharm. 2014, 31, 506–510. [Google Scholar]
- He, T.; Wang, Q.Y.; Shi, J.Y.; Fan, T.Y.; Yan, C.; Huang, L.J.; Liu, S.; Hao, X.J.; Mu, S.Z. Synthesis and the hepatoprotective activity of dibenzocyclooctadiene lignin derivatives. Bioorg. Med. Chem. Lett. 2014, 24, 1808–1811. [Google Scholar] [CrossRef] [PubMed]
- Zhang, T.; Zhang, Q.; Guo, J.; Yuan, H.; Peng, H.; Cui, L.; Yin, J.; Zhang, L.; Zhao, J.; Li, J. Non-cytotoxic concentrations of acetaminophen induced mitochondrial biogenesis and antioxidant response in HepG2 cells. Environ. Toxiol. Pharmacol. 2016, 46, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Shao, S.; Feng, Z.; Yang, Y.; Jiang, J.; Zhang, P. Eight new phenylethanoid glycoside derivatives possessing potential hepatoprotective activities from the fruits of Forsythia suspense. Fitoterapia 2017, 122, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Li, C.; Yang, J.; Sun, H.; Zhang, D. New phenylpropanoid and coumarin glycosides from the stems of Hydrangea paniculata sieb. Molecules 2017, 22, 133. [Google Scholar] [CrossRef] [PubMed]
- Jeong, M.; Kim, H.; Kim, H.; Choi, J.; Jang, D. Kudsuphilactone B, a nortriterpenoid isolated from Schisandra chinensis fruit, induces caspase-dependent apoptosis in human ovarian cancer A2780 cells. Arch. Pharm. Res. 2017, 40, 500–508. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, H.; Ikeya, Y. The constituents of Schizandra chinensis Baill. I. The structures of gomisin A, B, and C. Chem. Pharm. Bull. 1975, 23, 3296–3298. [Google Scholar] [CrossRef]
- Ikeya, Y.; Taguchi, H.; Iitaka, Y. The constituents of Schizandra chinensis Baill. The structure of a new lignan, gomisin D. Tetrahedron Lett. 1976, 27, 1359–1362. [Google Scholar] [CrossRef]
- Taguchi, H.; Ikeya, Y. The constituents of Schizandra chinensis Baill. The structures of two new lignans, gomisin F and G, and the absolute structures of gomisin A, B, and C. Chem. Pharm. Bull. 1977, 25, 364–366. [Google Scholar] [CrossRef]
- Xue, Y.; Li, X.; Du, X.; Li, X.; Wang, W.; Yang, J.; Chen, J.; Pu, J.; Sun, H. Isolation and anti-hepatitis B virus activity of dibenzocyclooctadiene lignans from the fruits of Schisandra chinensis. Phytochemistry 2015, 116, 253–261. [Google Scholar] [CrossRef] [PubMed]
- Li, W.; Liu, Y.; Wang, Z.; Yu, T.; Lu, Q.; Chen, H. Suppression of MAPK and NF-κB pathways by schisandrin B contributes to attenuation of DSS-induced mice model of inflammatory bowel disease. Pharmazie 2015, 70, 598–603. [Google Scholar]
Sample Availability: Samples of the compounds 1–6 are available from the authors. |
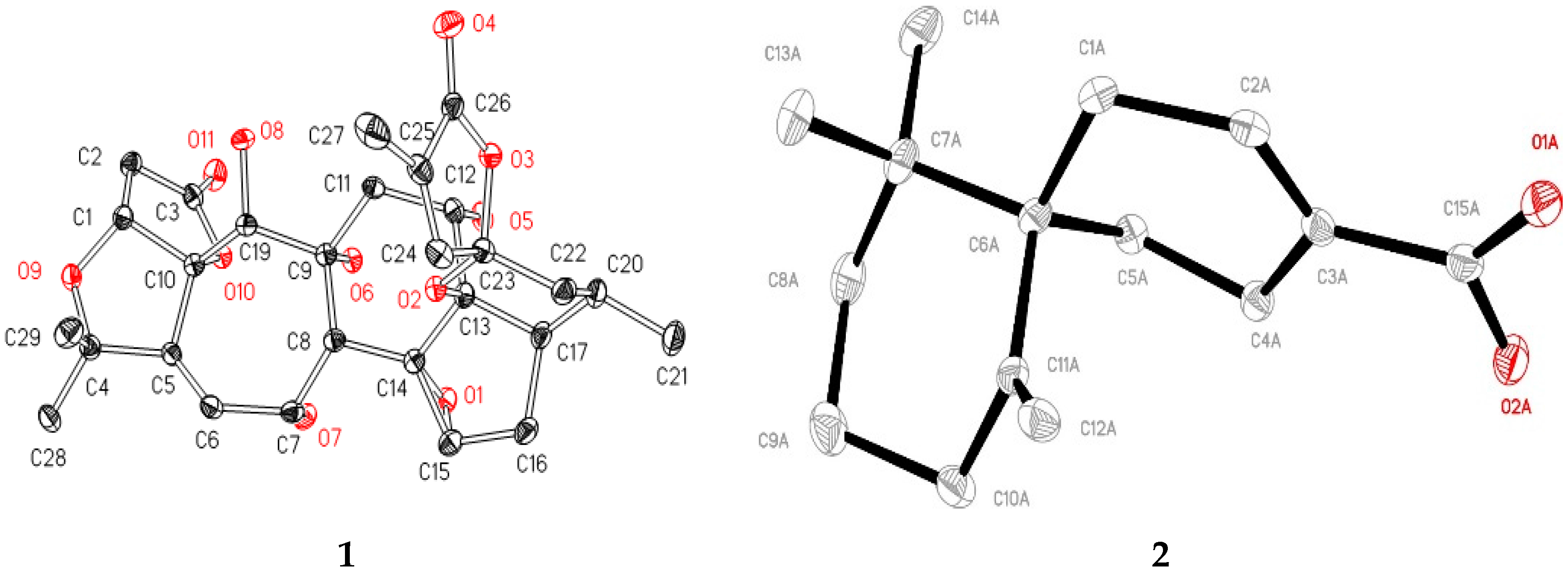
 ) and NOE (
) and NOE (  ) correlations of compounds 1 and 2.
) correlations of compounds 1 and 2.
| Compounds | OD Value | Survival Rate (%) |
|---|---|---|
| Blank control | 1.957 ± 0.116 | 100.0 |
| Model (APAP 8 mM) | 0.759 ± 0.012 *** | 38.7 |
| Bicyclol (positive control) | 1.061 ± 0.053 ## | 50.6 |
| 4 | 1.061 ± 0.053 ## | 44.5 |
| 5 | 1.061 ± 0.053 ## | 43.5 |
| 6 | 1.061 ± 0.053 ## | 44.6 |
| Position | Compound 1 a | Compound 2 b | ||
|---|---|---|---|---|
| 13C | 1H- (J Hz) | 13C | 1H- (J Hz) | |
| 1 | 80.9 | 4.34 m | 31.5 | 2.30 m (2 H) |
| 2 | 37.0 | 2.18 d (17.6) 2.82 dd (17.6, 6.0) | 141.5 | 6.96 s |
| 3 | 175.9 | --- | 131.5 | --- |
| 4 | 83.1 | --- | 38.5 | 1.86 m (2 H) |
| 5 | 50.9 | 2.42 dd (13.6, 1.2) | 27.3 | 1.44 m, 2.14 m |
| 6 | 31.7 | 1.86 m, 1.55 m | 46.8 | --- |
| 7 | 67.4 | 3.99 m | 38.7 | --- |
| 7-OH | --- | 4.61 m | --- | --- |
| 8 | 42.2 | 2.26 m | 25.4 | 1.52 m, 1.61 m |
| 9 | 82.4 | --- | 23.8 | 1.71 m, 2.30 m |
| 9-OH | --- | 4.62 m | --- | --- |
| 10 | 101.5 | --- | 33.7 | 2.11 m, 2.30 m |
| 11 | 35.2 | 2.21 m, 1.55 m | 150.7 | --- |
| 12 | 75.4 | 3.65 m | 111.9 | 4.89 s, 4.43 s |
| 12-OH | --- | 4.41 m | --- | --- |
| 13 | 83.2 | --- | 25.9 | 0.88 s (3 H) |
| 14 | 69.3 | --- | 21.4 | 0.92 s (3 H) |
| 15 | 53.2 | 3.75 brs | 170.5 | --- |
| 16 | 24.2 | 1.86 m, 1.65 m | ||
| 17 | 33.1 | 2.42 m | ||
| 19 | 67.1 | 4.22 m | ||
| 19-OH | --- | 5.56 m | ||
| 20 | 21.6 | 2.10 m | ||
| 21 | 18.7 | 0.91 d (6.4, 3 H) | ||
| 22 | 33.7 | 1.40 m, 1.41 dd (14, 3.6) | ||
| 23 | 105.4 | --- | ||
| 24 | 148.9 | 7.11 s | ||
| 25 | 130.3 | --- | ||
| 26 | 171.2 | --- | ||
| 27 | 10.2 | 1.80 s (3H) | ||
| 29 | 22.1 | 1.04 s (3H) | ||
| 30 | 28.1 | 1.19 s (3H) | ||
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Li, F.; Zhang, T.; Sun, H.; Gu, H.; Wang, H.; Su, X.; Li, C.; Li, B.; Chen, R.; Kang, J. A New Nortriterpenoid, a Sesquiterpene and Hepatoprotective Lignans Isolated from the Fruit of Schisandra chinensis. Molecules 2017, 22, 1931. https://doi.org/10.3390/molecules22111931
Li F, Zhang T, Sun H, Gu H, Wang H, Su X, Li C, Li B, Chen R, Kang J. A New Nortriterpenoid, a Sesquiterpene and Hepatoprotective Lignans Isolated from the Fruit of Schisandra chinensis. Molecules. 2017; 22(11):1931. https://doi.org/10.3390/molecules22111931
Chicago/Turabian StyleLi, Fenghua, Ting Zhang, Hua Sun, Haifeng Gu, Hongqing Wang, Xianming Su, Changkang Li, Baoming Li, Ruoyun Chen, and Jie Kang. 2017. "A New Nortriterpenoid, a Sesquiterpene and Hepatoprotective Lignans Isolated from the Fruit of Schisandra chinensis" Molecules 22, no. 11: 1931. https://doi.org/10.3390/molecules22111931