Evaluating the Potential Value of Natural Product Cuminic Acid against Plant Pathogenic Fungi in Cucumber
Abstract
:1. Introduction
2. Results
2.1. Sensitivity to Cuminic Acid
2.2. Protective and Curative Activity of Cuminic Acid
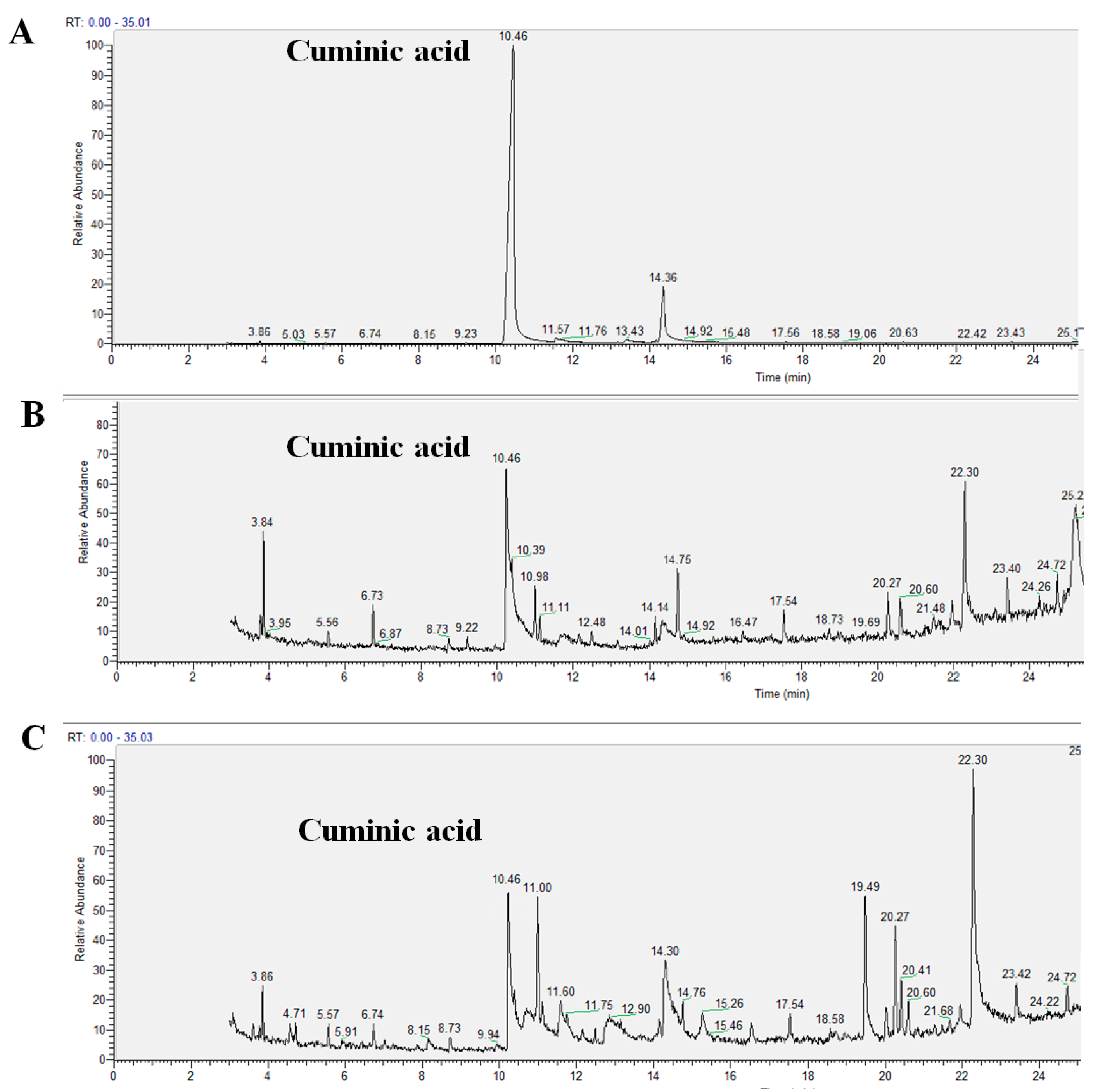
2.3. Translocation Behavior of Cuminic Acid
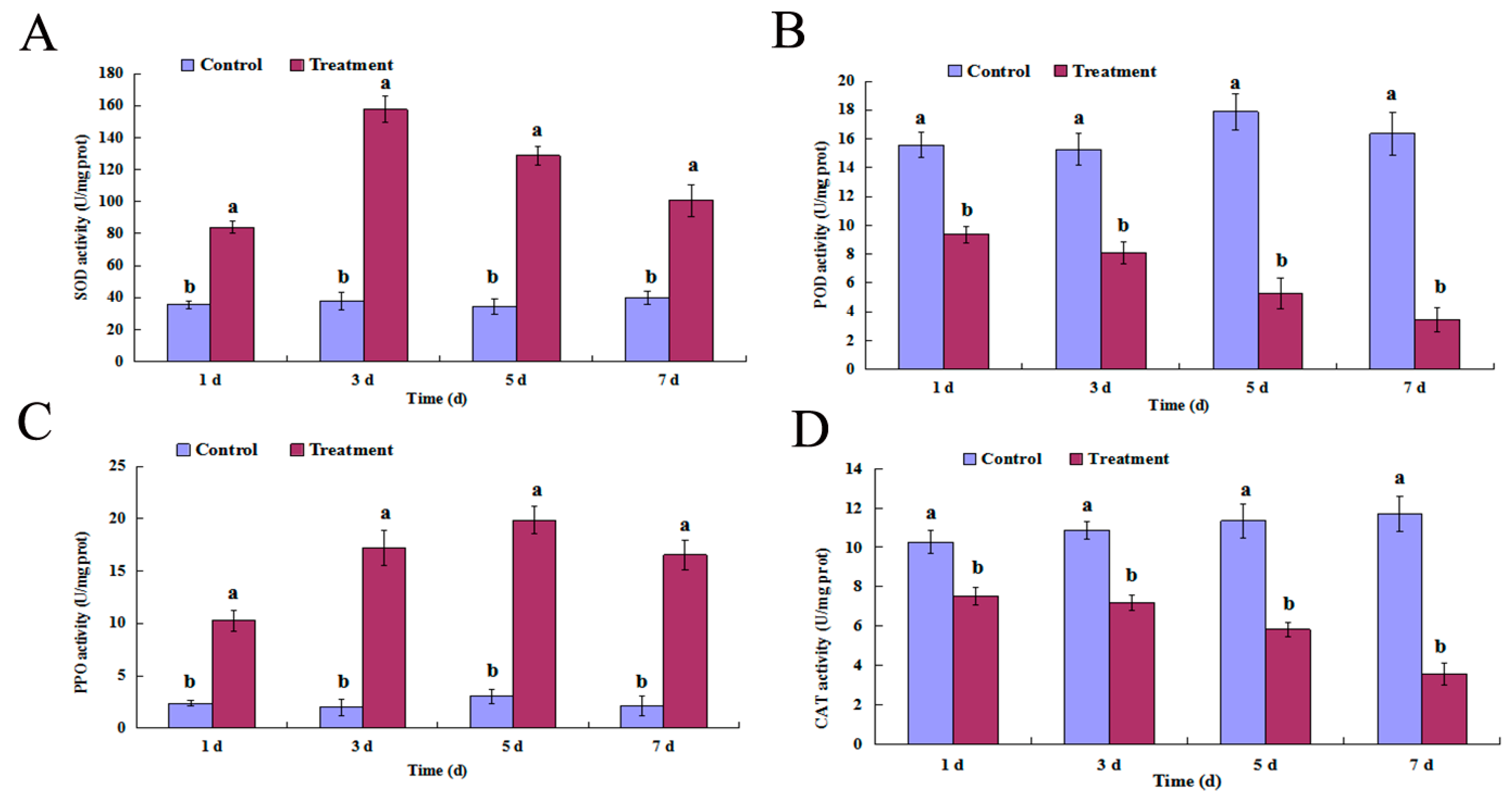
2.4. SOD, POD, PPO, and CAT Activity
3. Discussion
4. Materials and Methods
4.1. Fungicides and Strains
4.2. Sensitivity to Cuminic Acid
4.3. Protective and Curative Activity of Cuminic Acid in Greenhouse Experiments
4.4. Translocation of Cuminic Acid in Cucumber Plants
4.5. Superoxide Dismutase, Peroxidase, Polyphenol Oxidase, and Catalase Activity
4.6. Data Analysis
5. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Wang, M.; Sun, Y.; Sun, G.M.; Liu, X.K.; Zhai, L.C.; Shen, Q.R.; Guo, S.W. Water balance altered in cucumber plants infected with Fusarium oxysporum f. sp. cucumerinum. Sci. Rep. 2015, 5, 7722. [Google Scholar] [CrossRef] [PubMed]
- Kubo, Y. Studies on mechanisms of appressorial penetration by Colletotrichum lagenarium. J. Genet. Plant Pathol. 2005, 71, 451–453. [Google Scholar] [CrossRef]
- Ogura, H.; Ma, J. Persistence of Fusarium oxysporum f. sp. cucumerinum in continuous cropping fields. Ann. Phytopathol. Soc. Jpn. 1992, 58, 671–676. [Google Scholar] [CrossRef]
- Ge, Y.; Guest, D.I. Light and scanning electron microscopy studies on the infection process of melon leaves by Colletotrichum lagenarium. Physiol. Mol. Plant Pathol. 2011, 76, 67–74. [Google Scholar] [CrossRef]
- Pavlou, G.C.; Vakalounakis, D.J. Biological control of root and stem rot of greenhouse cucumber, caused by Fusarium oxysporum f. sp. radicis-cucumerinum, by lettuce soil amendment. Crop Prot. 2005, 24, 135–140. [Google Scholar] [CrossRef]
- Chung, S.; Kong, H.; Buyer, J.S.; Lakshman, D.K.; Lydon, J.; Kim, S.D.; Roberts, D.P. Isolation and partial characterization of Bacillus subtilis ME488 for suppression of soilborne pathogens of cucumber and pepper. Appl. Microbiol. Biotechnol. 2008, 80, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, W.B. Role of Research and Regulation in 50 Years of Pest Management in Agriculture Prepared for the 50th Anniversary of the Journal of Agricultural and Food Chemistry. J. Agric. Food Chem. 2002, 50, 4151–4155. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Dai, G. Antifungal activity of plant extracts against Colletotrichum lagenarium, the causal agent of anthracnose in cucumber. J. Sci. Food Agric. 2012, 92, 1937–1943. [Google Scholar] [CrossRef] [PubMed]
- Isman, M.B. Botanical insecticides, deterrents, and repellents in modern agriculture and an increasingly regulated world. Annu. Rev. Entomol. 2006, 51, 45–66. [Google Scholar] [CrossRef] [PubMed]
- Mi-Young, Y.; Byeongjin, C.; Jin-Cheol, K. Recent Trends in Studies on Botanical Fungicides in Agriculture. Plant Pathol. J. 2013, 29, 1–9. [Google Scholar]
- Copping, L.G.; Duke, S.O. Natural products that have been used commercially as crop protection agents. Pest Manag. Sci. 2007, 63, 524–554. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sun, Y.; Zhang, Y.; Zhang, X.; Feng, J.T. Antifungal activity and biochemical response of cuminic acid against Phytophthora capsici Leonian. Molecules 2016, 21, 756. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.F.; Chen, C.Z.; Yi, X.H.; Feng, J.T.; Zhang, X. Inhibition of p-isopropyl Benzaldehyde and p-isopropyl Benzoic Acid extracted from Cuminum cyminum against Plant Pathogens. Acta Bot. Boreal. Occident. Sin. 2008, 28, 2349–2354. [Google Scholar]
- Hu, L.F.; He, J.; Feng, J.T.; Zhang, X. Optimization of supercritical CO2 extraction and characterization of antifungal activity of essential oils in Cuminum cyminum L. Aust. J. Crop Sci. 2013, 7, 1809–1813. [Google Scholar]
- Hu, L.F.; Feng, J.T.; Zhang, X.; Zhang, Y.L. Isolation and Structure Detection of Fungicidal Components from Cuminum cyminum Seed. Chin. J. Pestic. Sci. 2007, 9, 330–334. [Google Scholar]
- Li, T. Environmental Safety Evalution of Cumin Fungicide. Master’s Thesis, Northwest A&F University, Yangling, China, 2012. [Google Scholar]
- Matheron, M.E.; Porchas, M. Impact of azoxystrobin, dimethomorph, fluazinam, fosetyl-Al, and metalaxyl on growth, sporulation, and zoospore cyst germination of three Phytophthora spp. Plant Dis. 2000, 84, 454–458. [Google Scholar] [CrossRef]
- Wang, L.Y.; Wang, Y.; Han, L.R.; Wang, M.; Han, X.S.; Feng, J.T. The efficacy and translocation behavior of carabrone in wheat and cucumber. Crop Prot. 2017, 100, 87–95. [Google Scholar] [CrossRef]
- Shen, Q.H.; Saijo, Y.; Mauch, S.; Biskup, C.; Bieri, S.; Keller, B.; Seki, H.; Ulker, B.; Somssich, I.E.; Schulze-Lefert, P. Nuclear activity of MLA immune receptors links isolate-specific and basal disease-resistance responses. Science 2007, 315, 1098–1103. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Singh, S.; Kumar, S.B.; Bahadur, S.H. Microbial consortium-mediated reprogramming of defence network in pea to enhance tolerance against Sclerotinia sclerotiorum. J. Appl. Microbiol. 2012, 112, 537–550. [Google Scholar] [CrossRef] [PubMed]
- Wen, P.F.; Chen, J.Y.; Kong, W.F.; Pan, Q.H.; Wan, S.B.; Huang, W.D. Salicylic acid induced the expression of phenylalanine ammonia-lyase gene in grape berry. Plant Sci. 2005, 169, 928–934. [Google Scholar] [CrossRef]
- He, Y.Q.; Zhang, H.H.; Sun, Z.T.; Li, J.M.; Hong, G.J.; Zhu, Q.S.; Zhou, X.B.; Macfarlance, S.; Yan, F.; Chen, J.P. Jasmonic acid-mediated defense suppresses brassinosteroid-mediated susceptibility to rice black streaked dwarf virus infection in rice. New Phytol. 2017, 214, 388–399. [Google Scholar] [CrossRef] [PubMed]
- Copping, L.G.; Menn, J.J. Biopesticides: A review of their action, applications and efficacy. Pest Manag. Sci. 2015, 56, 651–676. [Google Scholar] [CrossRef]
- Ujváry, I. Transforming natural products into natural pesticides-experience and expectations. Phytoparasitica 2002, 30, 439–442. [Google Scholar] [CrossRef]
- Karunanayake, A.G.; Todd, O.A.; Crowley, M.L.; Ricchetti, L.B.; Pittman, C.U., Jr.; Anderson, R.; Misan, T.E. Rapid removal of salicylic acid, 4-nitroaniline, benzoic acid and phthalic acid from wastewater using magnetized fast pyrolysis biochar from waste Douglas fir. Chem. Eng. J. 2017, 319, 75–88. [Google Scholar] [CrossRef]
- Park, J.H.; Choi, G.J.; Jang, K.S.; Lim, H.K.; Kim, H.T.; Cho, K.Y.; Kim, J.C. Antifungal activity against plant pathogenic fungi of chaetoviridins isolated from Chaetomium globosum. FEMS Microbiol. Lett. 2005, 252, 309–313. [Google Scholar] [CrossRef] [PubMed]
- Balandrin, M.F.; Klocke, J.A.; Wurtele, E.S.; Bollinger, W.H. Natural plant chemicals: Sources of industrial and medicinal materials. Science 1985, 228, 1154–1160. [Google Scholar] [CrossRef] [PubMed]
- Calsamiglia, S.; Busquet, M.; Cardozo, P.W.; Castillejos, L.; Ferret, A. Invited review: Essential oils as modifiers of rumen microbial fermentation. J. Dairy Sci. 2007, 90, 2580–2595. [Google Scholar] [CrossRef] [PubMed]
- Švecová, E.; Proietti, S.; Caruso, C.; Colla, G.; Crino, P. Antifungal activity of Vitex agnus-castus extract against Pythium ultimum in tomato. Crop Prot. 2013, 43, 223–230. [Google Scholar] [CrossRef]
- Zhang, J.; Yan, L.T.; Yuan, E.L.; Ding, H.X.; Ye, H.C.; Zhang, Z.K.; Yan, C.; Liu, Y.Q.; Feng, G. Antifungal activity of compounds extracted from Cortex pseudolaricis against Colletotrichum gloeosporioides. J. Agric. Food Chem. 2014, 62, 4905–4910. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.C.; Liu, Q.; Chen, X.B.; Zhou, L.; Liu, Z.L. Larvicidal activity of the essential oil from Tetradium glabrifolium fruits and its constituents against Aedes albopictus. Pest Manag. Sci. 2015, 71, 1582–1586. [Google Scholar] [CrossRef] [PubMed]
- Khaledi, N.; Taheri, P.; Tarighi, S. Antifungal activity of various essential oils against Rhizoctonia solani and Macrophomina phaseolina as major bean pathogens. J. Appl. Microbiol. 2015, 118, 704–717. [Google Scholar] [CrossRef] [PubMed]
- Dayan, F.E.; Cantrell, C.L.; Duke, S.O. Natural products in crop protection. Bioorg. Med. Chem. 2009, 17, 4022–4034. [Google Scholar] [CrossRef] [PubMed]
- Cantrell, C.L.; Dayan, F.E.; Duke, S.O. Natural products as sources for new pesticides. J. Nat. Prod. 2012, 75, 1231–1242. [Google Scholar] [CrossRef] [PubMed]
- Gerwick, B.C.; Sparks, T.C. Natural products for pest control: An analysis of their role, value and future. Pest Manag. Sci. 2014, 70, 1169–1185. [Google Scholar] [CrossRef] [PubMed]
- Kraiczy, P.; Haase, V.; Gencic, S.; Flindt, S.; Anke, T. The molecular basis for the natural resistance of the cytochrome bc1 complex from strobilurin-producing basidiomycetes to center Qp inhibitors. FEBS J. 1996, 235, 54–63. [Google Scholar] [CrossRef]
- Zhang, X.K.; Wu, D.X.; Duan, Y.B.; Ge, C.Y.; Wang, J.X.; Zhou, M.G.; Chen, C.J. Biological characteristics and resistance analysis of the novel fungicide SYP-1620 against Botrytis cinerea. Pestic. Biochem. Physiol. 2014, 114, 72–78. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Wang, M.; Zheng, Y.; Luo, J.M.; Yang, X.R.; Wang, X.L. Quantitative changes of plant defense enzymes and phytohormone in biocontrol of cucumber Fusarium wilt by Bacillus subtilis B579. World J. Microbiol. Biotechnol. 2010, 26, 675–684. [Google Scholar] [CrossRef]
Sample Availability: Samples of the compounds cuminic acid are available from the authors. |


| Strains a | EC50 for Cuminic Acid (μg/mL) | EC50 for Carbendazim (μg/mL) | ||
|---|---|---|---|---|
| Inhibition of Growth | Inhibition of Germination | Inhibition of Growth | Inhibition of Germination | |
| FO-1 | 21.37c b | 15.62b | 0.900b | 1.173c |
| FO-23 | 25.62c | 13.37c | 1.211b | 1.432c |
| FO-12 | 18.46d | 16.02ab | 0.794b | 0.983c |
| FO-14 | 32.34ab | 16.73ab | 0.869b | 0.874c |
| FO-8 | 30.50b | 18.22a | 1.001b | 0.992c |
| CL-32 | 22.45c | 18.36a | 7.322a | 6.406a |
| CL-12 | 35.71a | 19.43a | 6.537a | 6.035a |
| CL-7 | 30.09b | 17.44a | 5.459a | 4.941b |
| CL-11 | 29.87b | 20.45a | 6.344a | 6.075a |
| Treatment | Protective Activity | Curative Activity | ||
|---|---|---|---|---|
| Disease Index | Efficacy (%) | Disease Index | Efficacy (%) | |
| Cuminic Acid (500 µg/mL) | 66.67b a | 26.16c | 68.06b | 23.43c |
| Cuminic Acid (1000 µg/mL) | 51.39c | 43.08b | 50.00c | 43.75b |
| Cuminic Acid (2000 µg/mL) | 26.39d | 70.77a | 33.22d | 62.63a |
| Carbendazim (500 µg/mL) | 27.78d | 69.22a | 34.70d | 60.96a |
| Water Control | 90.28a | - | 88.89a | - |
| Treatment | Protective Activity | Curative Activity | ||
|---|---|---|---|---|
| Disease Index | Efficacy (%) | Disease Index | Efficacy (%) | |
| Cuminic Acid (500 µg/mL) | 42.80b a | 21.77c | 36.01b | 17.07c |
| Cuminic Acid (1000 µg/mL) | 32.92c | 39.85b | 29.01c | 33.18b |
| Cuminic Acid (2000 µg/mL) | 18.93d | 65.43a | 19.34c | 55.46a |
| Carbendazim (500 µg/mL) | 18.31d | 66.55a | 16.05c | 63.04a |
| Water Control | 54.73a | - | 43.42a | - |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, Y.; Zhang, J.; Sun, Y.; Feng, J.; Zhang, X. Evaluating the Potential Value of Natural Product Cuminic Acid against Plant Pathogenic Fungi in Cucumber. Molecules 2017, 22, 1914. https://doi.org/10.3390/molecules22111914
Wang Y, Zhang J, Sun Y, Feng J, Zhang X. Evaluating the Potential Value of Natural Product Cuminic Acid against Plant Pathogenic Fungi in Cucumber. Molecules. 2017; 22(11):1914. https://doi.org/10.3390/molecules22111914
Chicago/Turabian StyleWang, Yong, Jie Zhang, Yang Sun, Juntao Feng, and Xing Zhang. 2017. "Evaluating the Potential Value of Natural Product Cuminic Acid against Plant Pathogenic Fungi in Cucumber" Molecules 22, no. 11: 1914. https://doi.org/10.3390/molecules22111914




