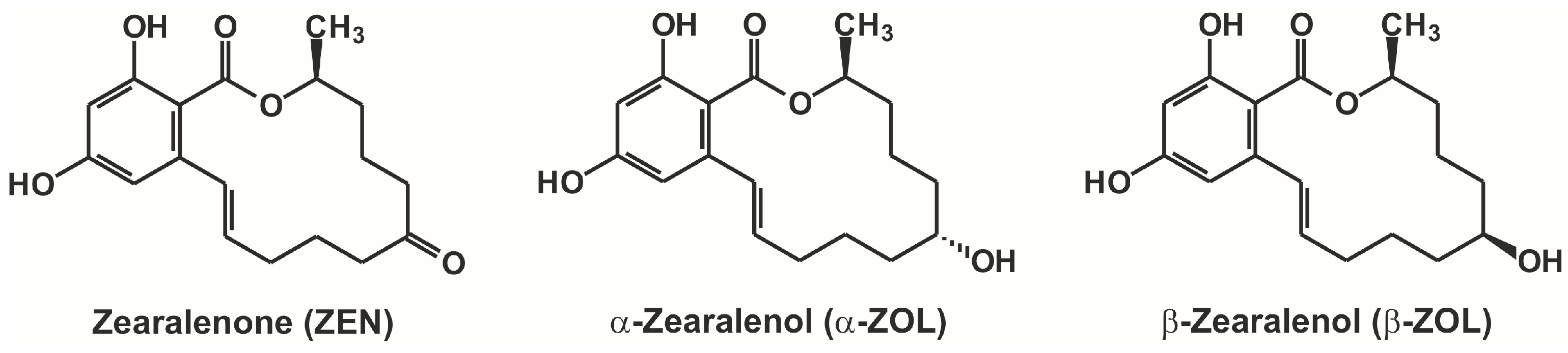
Interaction of α- and β-zearalenols with β-cyclodextrins
Abstract
:1. Introduction
2. Results and Discussion
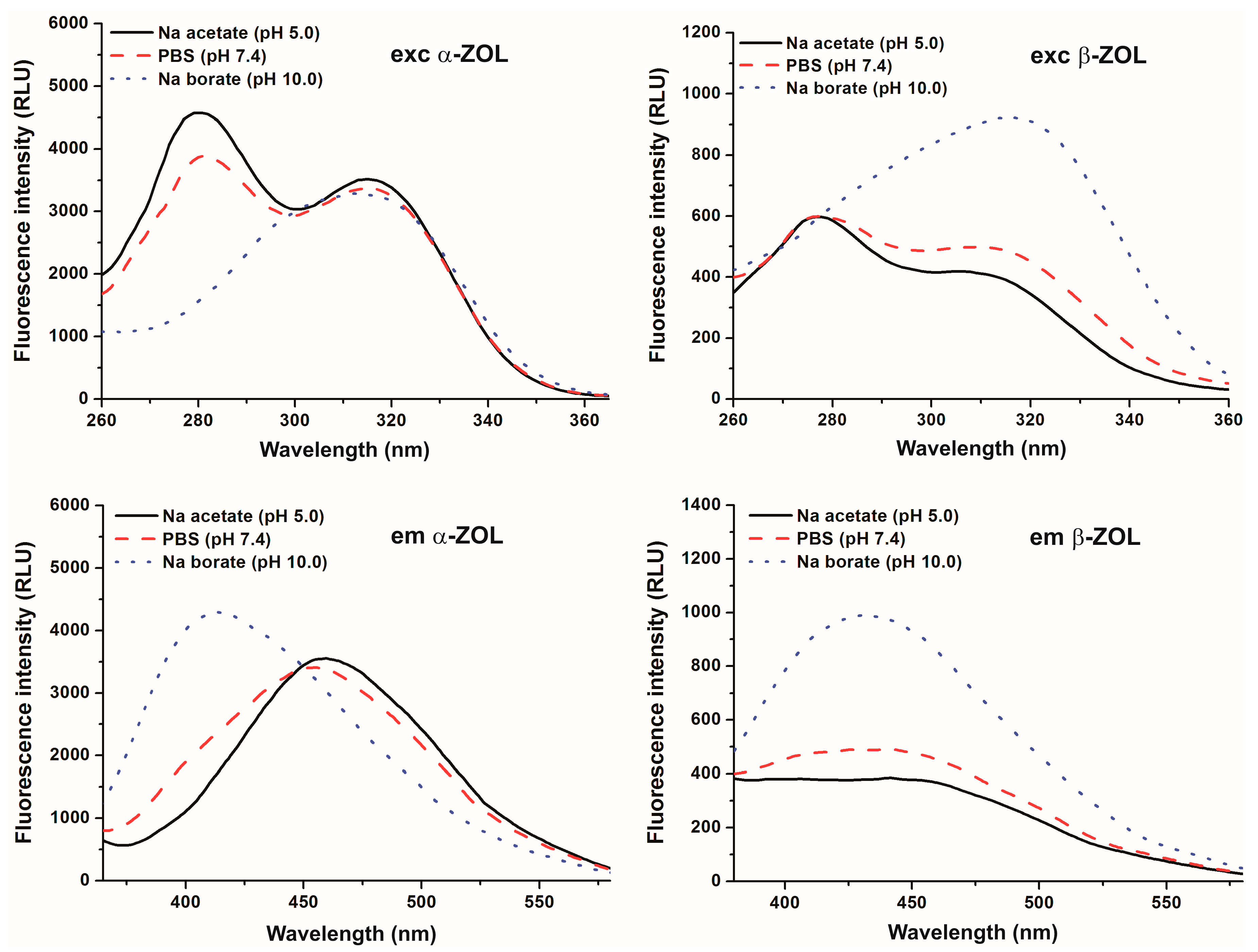
2.1. Effect of pH on the Fluorescence Spectra of α- and β-ZOLs
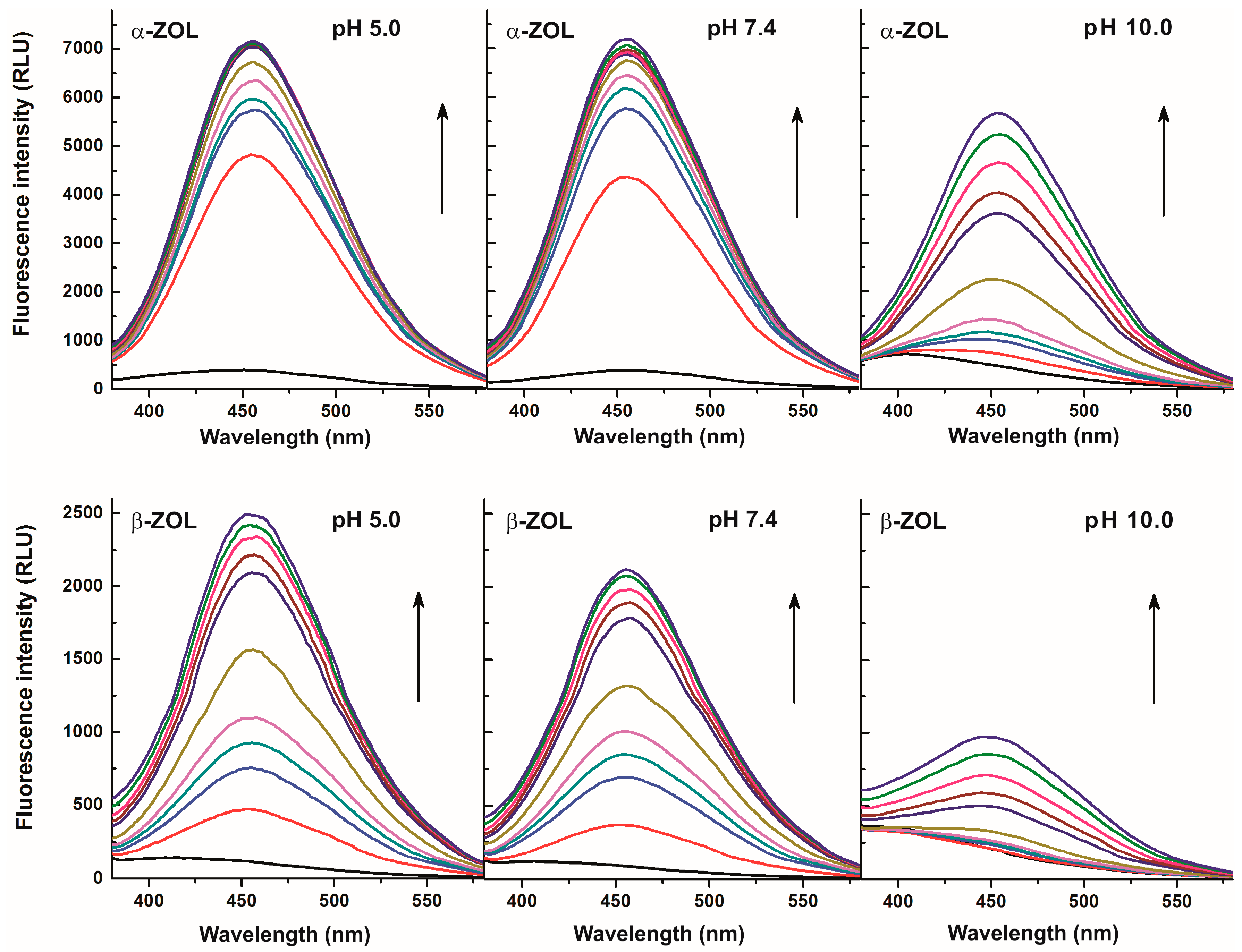
2.2. Effects of CDs on the Fluorescence Spectra of α- and β-ZOLs under Different Circumstances
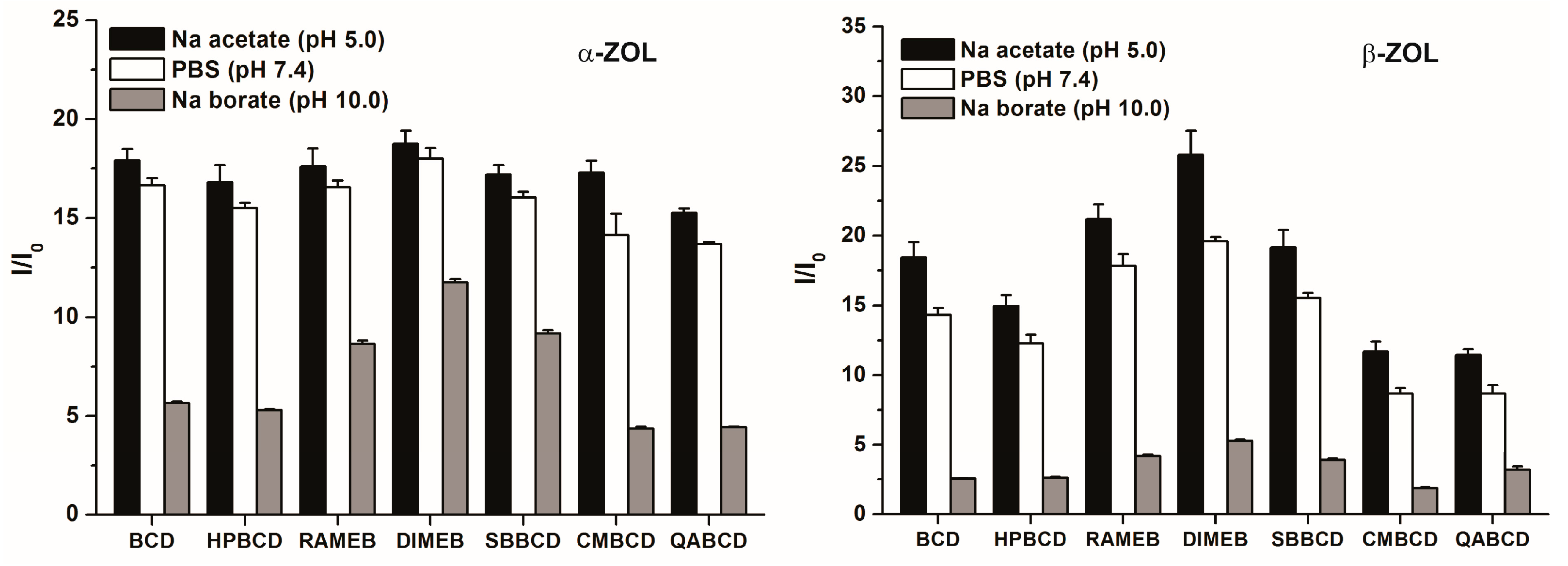
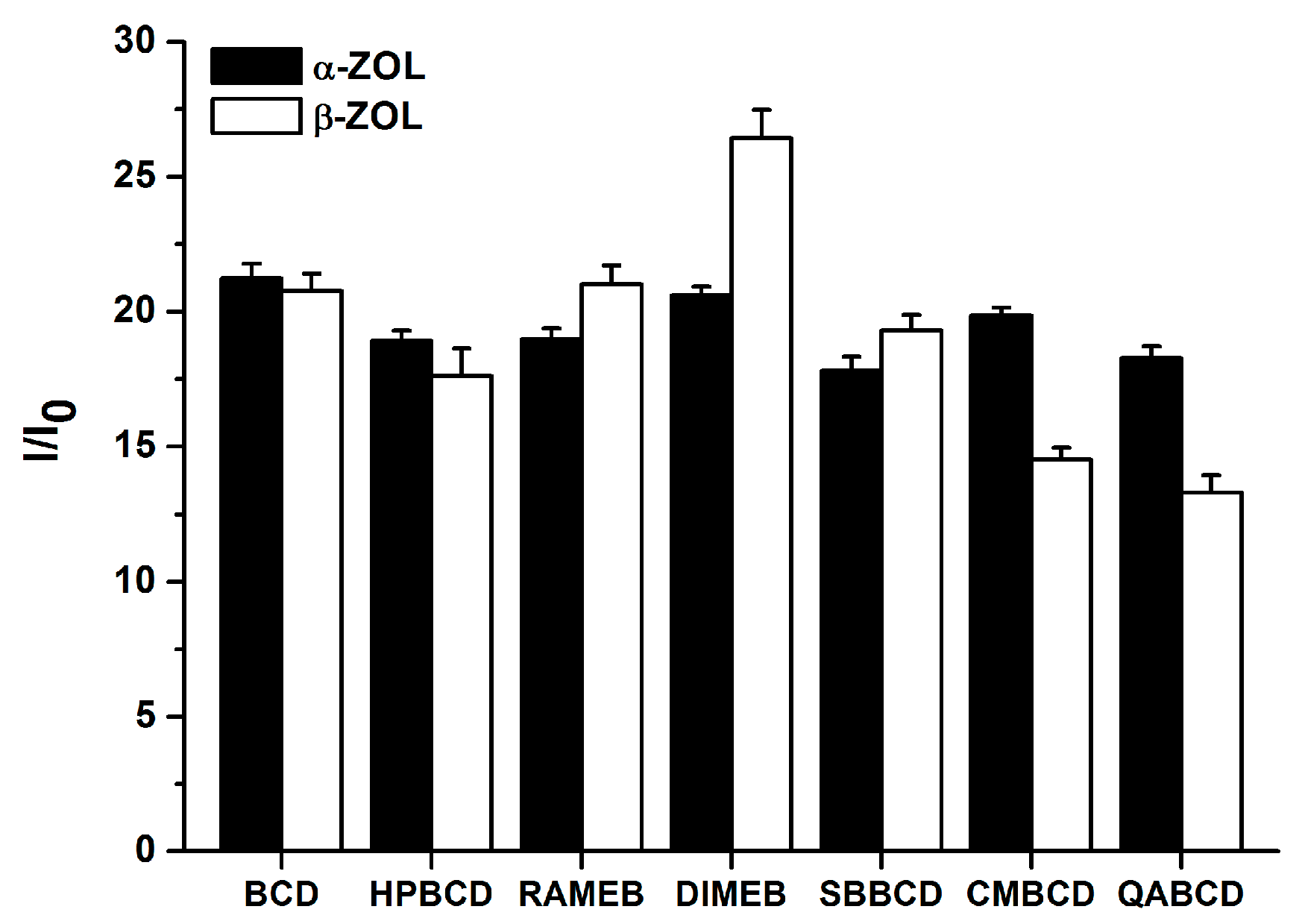
2.3. Fluorescence Enhancement of ZOLs by Native and Chemically Modified β-CDs
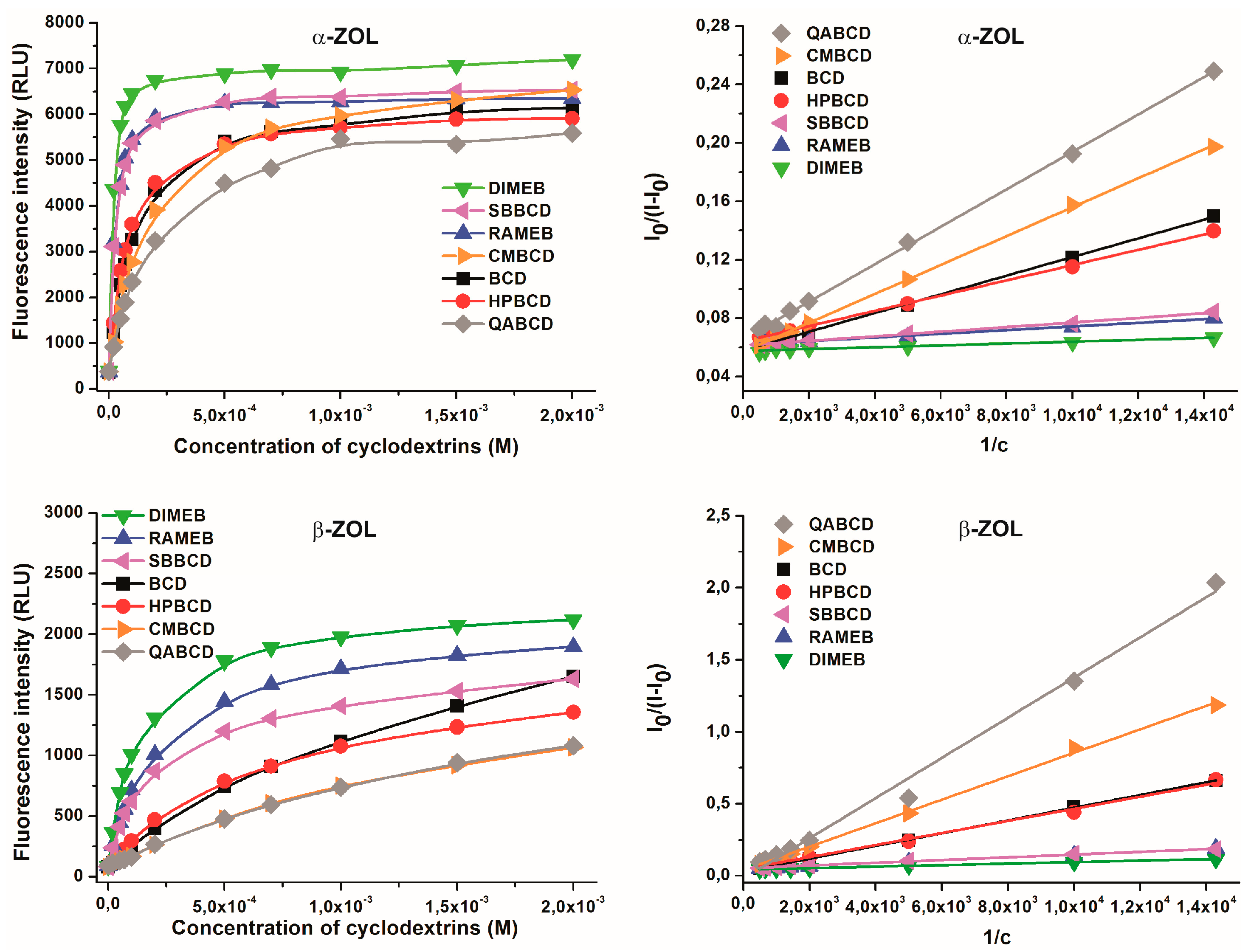
2.4. Binding Constants of ZOL-CD Complexes
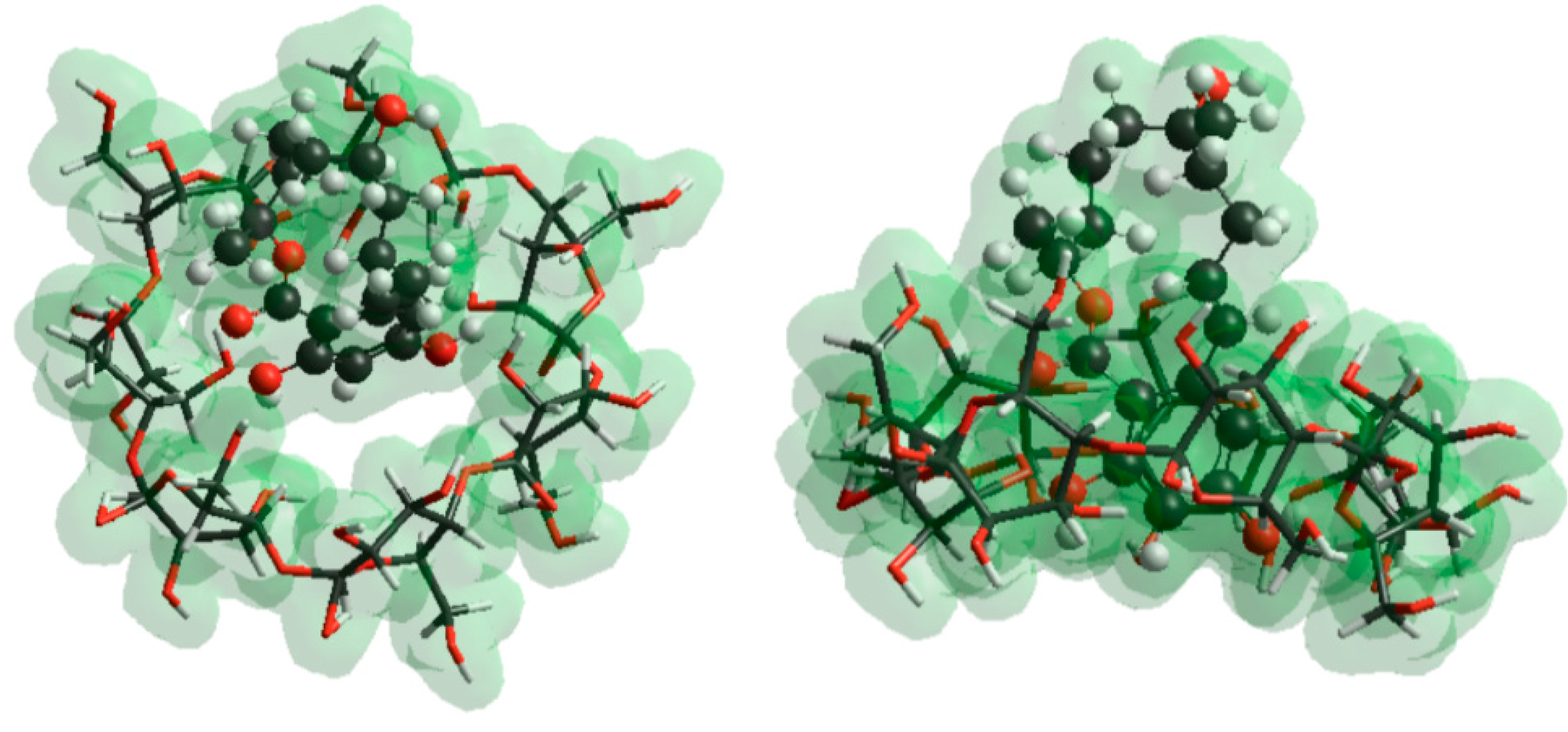
2.5. Molecular Modeling Studies
3. Materials and Methods
3.1. Reagents
3.2. Fluorescence Spectroscopic Measurements
3.3. Molecular Modeling Studies
4. Conclusions
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Zinedine, A.; Soriano, J.M.; Moltó, J.C.; Mañes, J. Review on the toxicity, occurrence, metabolism, detoxification, regulations and intake of zearalenone: An oestrogenic mycotoxin. Food Chem. Toxicol. 2007, 45, 1–18. [Google Scholar] [CrossRef] [PubMed]
- Yazar, S.; Omurtag, G.Z. Fumonisins, trichothecenes and zearalenone in cereals. Int. J. Mol. Sci. 2008, 9, 2062–2090. [Google Scholar] [CrossRef] [PubMed]
- Maragos, C.M. Zearalenone occurrence and human exposure. World Mycotoxin J. 2010, 3, 369–383. [Google Scholar] [CrossRef]
- Ryu, D.; Hanna, M.A.; Bullerman, L.B. Stability of zearalenone during extrusion of corn grits. J. Food Prot. 1999, 62, 1482–1484. [Google Scholar] [CrossRef] [PubMed]
- Poór, M.; Kunsági-Máté, S.; Sali, N.; Kőszegi, T.; Szente, L.; Peles-Lemli, B. Interactions of zearalenone with native and chemically modified cyclodextrins and their potential utilization. J. Photochem. Photobiol. B 2015, 151, 63–68. [Google Scholar] [CrossRef] [PubMed]
- Poór, M.; Kunsági-Máté, S.; Bálint, M.; Hetényi, C.; Gerner, Z.; Lemli, B. Interaction of mycotoxin zearalenone with human serum albumin. J. Photochem. Photobiol. B 2017, 170, 16–24. [Google Scholar] [CrossRef] [PubMed]
- Shier, W.T.; Shier, A.C.; Xie, W.; Mirocha, C.J. Structure-activity relationships for human estrogenic activity in zearalenone mycotoxins. Toxicon 2001, 39, 1435–1438. [Google Scholar] [CrossRef]
- Minervini, F.; Dell’Aquila, M.E. Zearalenone and Reproductive Function in Farm Animals. Int. J. Mol. Sci. 2008, 9, 2570–2584. [Google Scholar] [CrossRef] [PubMed]
- Malekinejad, H.; Maas-Bakker, R.; Fink-Gremmels, J. Species differences in the hepatic biotransformation of zearalenone. Vet. J. 2006, 172, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Videmann, B.; Mazallon, M.; Tep, J.; Lecoeur, S. Metabolism and transfer of the mycotoxin zearalenone in human intestinal Caco-2 cells. Food Chem. Toxicol. 2008, 46, 3279–3286. [Google Scholar] [CrossRef] [PubMed]
- Dellafiora, L.; Galaverna, G.; Righi, F.; Cozzini, P.; Dall’Asta, C. Assessing the hydrolytic fate of the masked mycotoxin zearalenone-14-glucoside—A warning light for the need to look at the “maskedome”. Food Chem. Toxicol. 2017, 99, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Fleck, S.C.; Churchwell, M.I.; Doerge, D.R. Metabolism and pharmacokinetics of zearalenone following oral and intravenous administration in juvenile female pigs. Food Chem. Toxicol. 2017, 106, 193–201. [Google Scholar] [CrossRef] [PubMed]
- Frizzell, C.; Ndossi, D.; Verhaegen, S.; Dahl, E.; Eriksen, G.; Sørlie, M.; Ropstad, E.; Muller, M.; Elliott, C.T.; Connolly, L. Endocrine disrupting effects of zearalenone, alpha- and beta-zearalenol at the level of nuclear receptor binding and steroidogenesis. Toxicol. Lett. 2011, 206, 210–217. [Google Scholar] [CrossRef] [PubMed]
- Filannino, A.; Stout, T.A.; Gadella, B.M.; Sostaric, E.; Pizzi, F.; Colenbrander, B.; Dell’Aquila, M.E.; Minervini, F. Dose-response effects of estrogenic mycotoxins (zearalenone, alpha- and beta-zearalenol) on motility, hyperactivation and the acrosome reaction of stallion sperm. Reprod. Biol. Endocrinol. 2011, 9, 134. [Google Scholar] [CrossRef] [PubMed]
- Schollenberger, M.; Müller, H.M.; Rüfle, M.; Suchy, S.; Plank, S.; Drochner, W. Natural occurrence of 16 fusarium toxins in grains and feedstuffs of plant origin from Germany. Mycopathologia 2006, 161, 43–52. [Google Scholar] [CrossRef] [PubMed]
- Huang, L.C.; Zheng, N.; Zheng, B.Q.; Wen, F.; Cheng, J.B.; Han, R.W.; Xu, X.M.; Li, S.L.; Wang, J.Q. Simultaneous determination of aflatoxin M1, ochratoxin A, zearalenone and α-zearalenol in milk by UHPLC-MS/MS. Food Chem. 2014, 146, 242–249. [Google Scholar] [CrossRef] [PubMed]
- Szejtli, J. Cyclodextrin Technology; Kluwer: Dordrecht, The Netherlands, 1988. [Google Scholar]
- Szente, L.; Szejtli, J. Highly soluble cyclodextrin derivatives: Chemistry, properties, and trends in development. Adv. Drug Deliv. Rev. 1999, 36, 17–28. [Google Scholar] [CrossRef]
- Dodziuk, H. Cyclodextrins and Their Complexes; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2008. [Google Scholar]
- Dall’asta, C.; Ingletto, G.; Corradini, R.; Galaverna, G.; Marchelli, R. Fluorescence enhancement of aflatoxins using native and substituted cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2003, 45, 257–263. [Google Scholar] [CrossRef]
- Poór, M.; Matisz, G.; Kunsági-Máté, S.; Derdák, D.; Szente, L.; Lemli, B. Fluorescence spectroscopic investigation of the interaction of citrinin with native and chemically modified cyclodextrins. J. Lumin. 2016, 172, 23–28. [Google Scholar] [CrossRef]
- Poór, M.; Kunsági-Máté, S.; Szente, L.; Matisz, G.; Secenji, G.; Czibulya, Z.; Kőszegi, T. Interaction of ochratoxin A with quaternary ammonium beta-cyclodextrin. Food Chem. 2015, 172, 143–149. [Google Scholar] [CrossRef] [PubMed]
- Dall’Asta, C.; Faccini, A.; Galaverna, G.; Corradini, R.; Dossena, A.; Marchelli, R. Complexation of the mycotoxin zearalenone with β-cyclodextrin: Study of the interaction and first promising applications. Mycotoxin Res. 2008, 24, 14–18. [Google Scholar] [CrossRef] [PubMed]
- Dall’Asta, C.; Faccini, A.; Galaverna, G.; Corradini, R.; Dossena, A.; Marchelli, R. Complexation of zearalenone and zearalenols with native and modified β-cyclodextrins. J. Incl. Phenom. Macrocycl. Chem. 2009, 64, 331–340. [Google Scholar] [CrossRef]
- Seidel, V.; Poglits, E.; Schiller, K.; Lindner, W. Simultaneous determination of ochratoxin A and zearalenone in maize by reversed-phase high performance liquid chromatography with fluorescence detection and β-cyclodextrin as mobile phase additive. J. Chromatogr. 1993, 635, 227–235. [Google Scholar] [CrossRef]
- Maragos, C.M.; Appell, M. Capillary electrophoresis of the mycotoxin zearalenone using cyclodextrin-enhanced fluorescence. J. Chromatogr. A 2007, 1143, 252–257. [Google Scholar] [CrossRef] [PubMed]
- Larionova, D.A.; Goryacheva, I.Y.; Van Peteghem, C.; De Saeger, S. Thin-layer chromatography of aflatoxins and zearalenones with β-cyclodextrins as mobile phase additives. World Mycotoxin J. 2011, 4, 113–117. [Google Scholar] [CrossRef]
- Appell, M.; Jackson, M.A. Sorption of ochratoxin A from aqueous solutions using β-cyclodextrin–polyurethane polymer. Toxins 2012, 4, 98–109. [Google Scholar] [CrossRef] [PubMed]
- Lemke, S.L.; Grant, P.G.; Phillips, T.D. Adsorption of zearalenone by organophilic montmorillonite clay. J. Agric. Food Chem. 1998, 46, 3789–3796. [Google Scholar] [CrossRef]
- Wang, H.; Xie, X.; Zhang, F.; Zhou, Q.; Tao, Q.; Zou, Y.; Chen, C.; Zhou, C.; Yu, S. Evaluation of cholesterol depletion as a marker of nephrotoxicity in vitro for novel β-cyclodextrin derivatives. Food Chem. Toxicol. 2011, 49, 1387–1393. [Google Scholar] [CrossRef] [PubMed]
- Yu, Y.; Chipot, C.; Cai, W.; Shao, X. Molecular dynamics study of the inclusion of cholesterol into cyclodextrins. J. Phys. Chem. B 2006, 110, 6372–6378. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Huszthy, P.; Kunsági-Máté, S. Effect of molecular vibrations on the selectivity character of pyridino-18-crown-6 derivatives towards potassium ion. Chem. Phys. Lett. 2012, 533, 45–49. [Google Scholar] [CrossRef]
- Brehm, G.; Reiher, M.; Schneider, S. Estimation of the Vibrational Contribution to the Entropy Change Associated with the Low- to High-Spin Transition in Fe(phen)2(NCS)2 Complexes: Results Obtained by IR and Raman Spectroscopy and DFT Calculations. J. Phys. Chem. A 2002, 106, 12024–12034. [Google Scholar] [CrossRef]
- Cornish-Bowden, A. Enthalpy-entropy compensation: A phantom phenomenon. J. Biosci. 2002, 27, 121–126. [Google Scholar] [CrossRef] [PubMed]
Sample Availability: Samples of the compounds are not available from the authors. |
| BCD | HPBCD | RAMEB | DIMEB | SBBCD | CMBCD | QABCD | |
|---|---|---|---|---|---|---|---|
| ZEN 1 | 3.99 1 | 3.94 1 | 4.36 1 | 4.76 1 | 4.20 1 | - 2 | 3.75 1 |
| α-ZOL | 3.90 ± 0.01 | 4.05 ± 0.02 | 4.59 ± 0.04 | 4.89 ± 0.02 | 4.58 ± 0.00 | 3.69 ± 0.02 | 3.65 ± 0.04 |
| β-ZOL | 2.79 ± 0.02 | 3.11 ± 0.04 | 3.68 ± 0.01 | 3.88 ± 0.00 | 3.64 ± 0.02 | 2.65 ± 0.05 | 2.25 ± 0.09 |
| BCD | HPBCD | RAMEB | DIMEB | SBBCD | CMBCD | QABCD | |
|---|---|---|---|---|---|---|---|
| α-ZOL | 4.03 ± 0.04 | 4.21 ± 0.01 | 4.74 ± 0.01 | 5.00 ± 0.01 | 4.68 ± 0.01 | 3.92 ± 0.02 | 3.88 ± 0.02 |
| β-ZOL | 3.08 ± 0.01 | 3.41 ± 0.02 | 3.82 ± 0.02 | 4.02 ± 0.02 | 3.85 ± 0.02 | 3.07 ± 0.01 | 2.64 ± 0.01 |
| α-ZOL | ΔH (kJ/mol) | ΔS (J/K·mol) | ΔG298K (kJ/mol) |
| BCD | −23.4 | 34.6 | −33.8 |
| DIMEB | −32.4 | 24.3 | −39.7 |
| SBBCD | −25.4 | 32.5 | −35.1 |
| β-ZOL | ΔH (kJ/mol) | ΔS (J/K·mol) | ΔG298K (kJ/mol) |
| BCD | −23.4 | 34.6 | −33.8 |
| DIMEB | −19.6 | 48.5 | −34.1 |
| SBBCD | −27.4 | 31.4 | −36.8 |
| α-ZOL | ΔH (kJ/mol) | ΔS (J/K·mol) | ΔG298K (kJ/mol) |
| BCD | −37.6 | −7.0 | −35.6 |
| DIMEB | −46.6 | −17.3 | −41.5 |
| SBBCD | −39.6 | −9.1 | −36.9 |
| β-ZOL | ΔH (kJ/mol) | ΔS (J/K·mol) | ΔG298K (kJ/mol) |
| BCD | −37.6 | −7.0 | −35.6 |
| DIMEB | −33.8 | 6.9 | −35.9 |
| SBBCD | −41.6 | −10.2 | −38.6 |
| α-ZOL | Ea (kJ/mol) | β-ZOL | Ea (kJ/mol) |
|---|---|---|---|
| BCD | −11.6 | BCD | −11.9 |
| DIMEB | −13.7 | DIMEB | −13.4 |
| SBBCD | −12.2 | SBBCD | −22.2 |
© 2017 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Poór, M.; Zand, A.; Szente, L.; Lemli, B.; Kunsági-Máté, S. Interaction of α- and β-zearalenols with β-cyclodextrins. Molecules 2017, 22, 1910. https://doi.org/10.3390/molecules22111910
Poór M, Zand A, Szente L, Lemli B, Kunsági-Máté S. Interaction of α- and β-zearalenols with β-cyclodextrins. Molecules. 2017; 22(11):1910. https://doi.org/10.3390/molecules22111910
Chicago/Turabian StylePoór, Miklós, Afshin Zand, Lajos Szente, Beáta Lemli, and Sándor Kunsági-Máté. 2017. "Interaction of α- and β-zearalenols with β-cyclodextrins" Molecules 22, no. 11: 1910. https://doi.org/10.3390/molecules22111910







