Copper Ion Attenuated the Antiproliferative Activity of Di-2-pyridylhydrazone Dithiocarbamate Derivative; However, There Was a Lack of Correlation between ROS Generation and Antiproliferative Activity
Abstract
:1. Introduction
2. Results
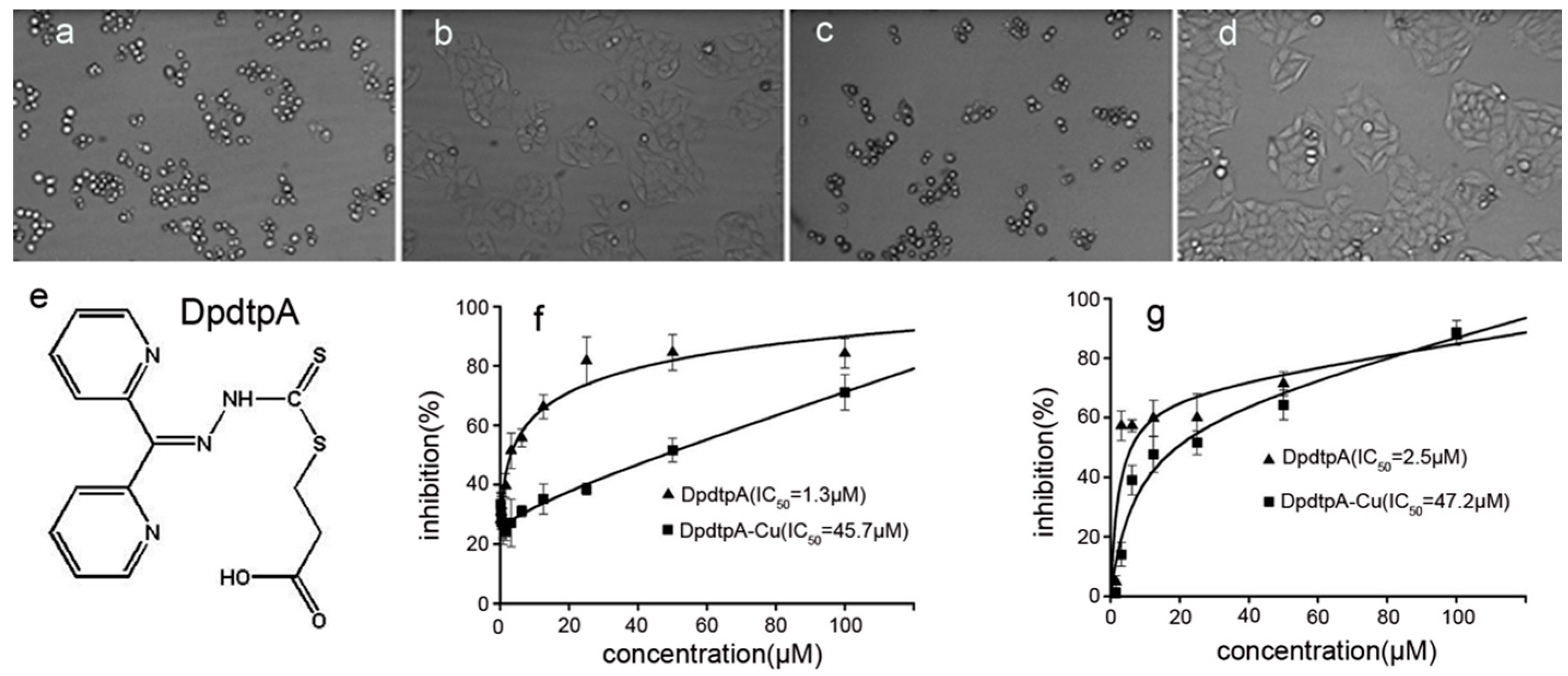
2.1. Antiproliferative Effect of DpdtpA
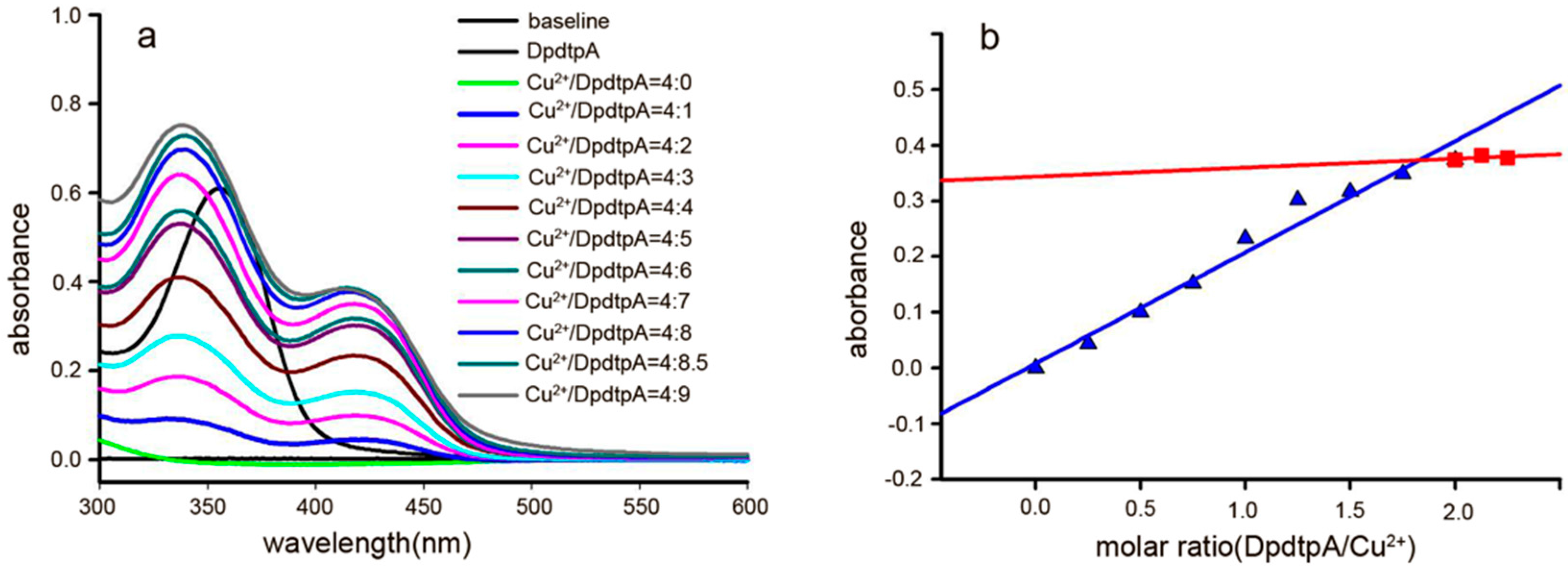
2.2. New Species Determined by the Spectral Study
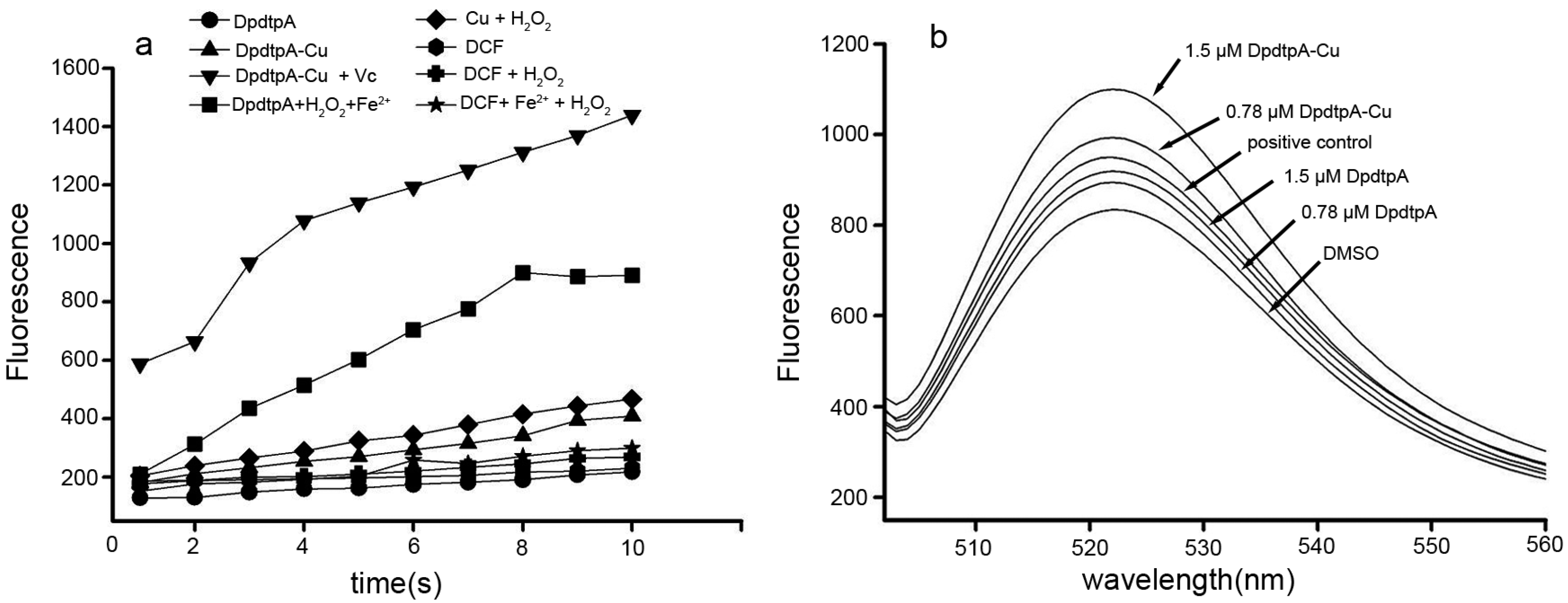
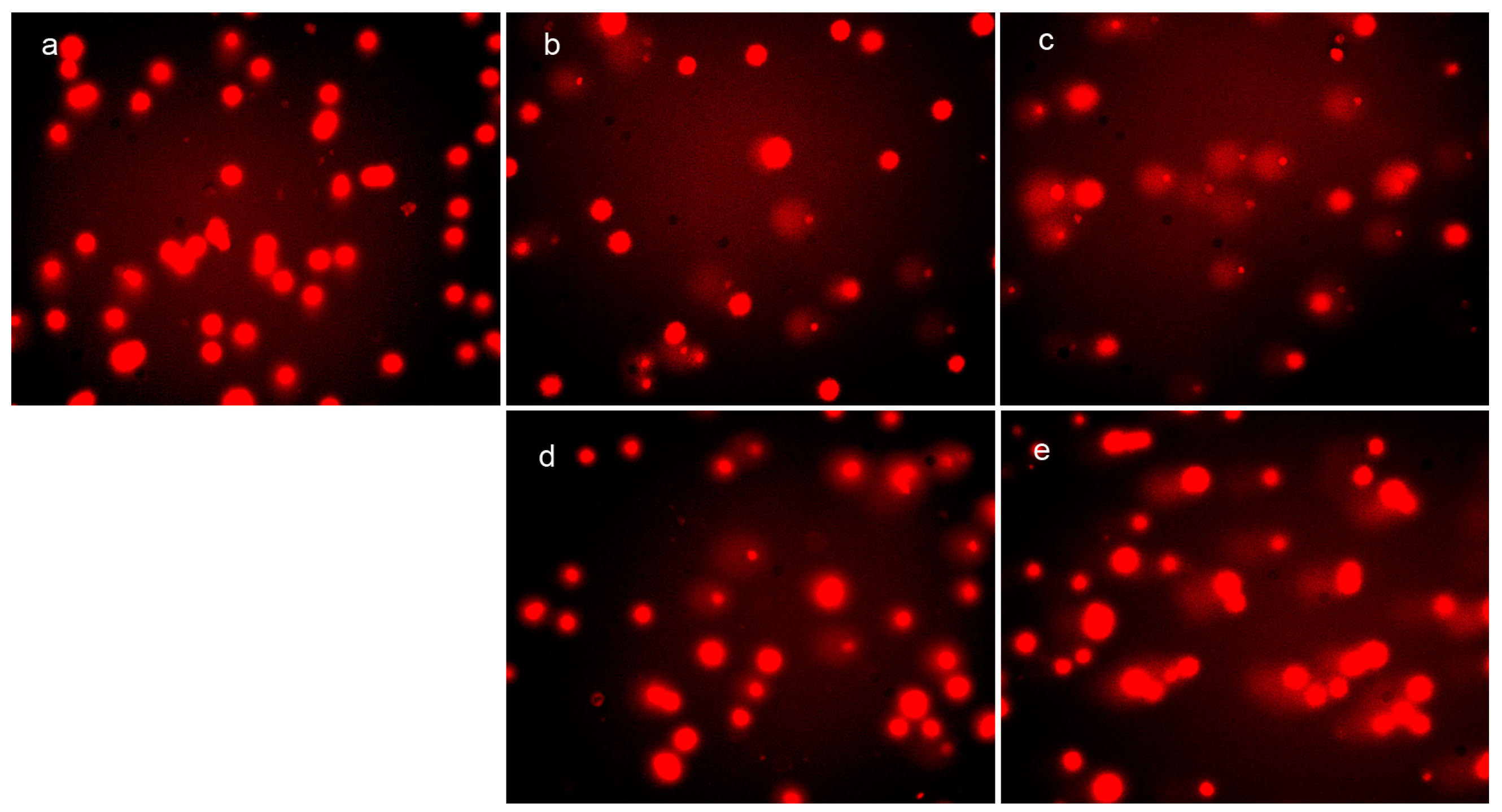
2.3. DpdtpA and Its Copper Complex (DpdtpA–Cu) Induced ROS Generation
2.4. Cellular DNA Fragmentation by DpdtpA and Its Copper Complex
2.5. DpdtpA and Its Copper Complex Induced Cellular Apoptosis
2.6. The Effect of DpdtpA and Its Copper Complex on the Cell Cycle
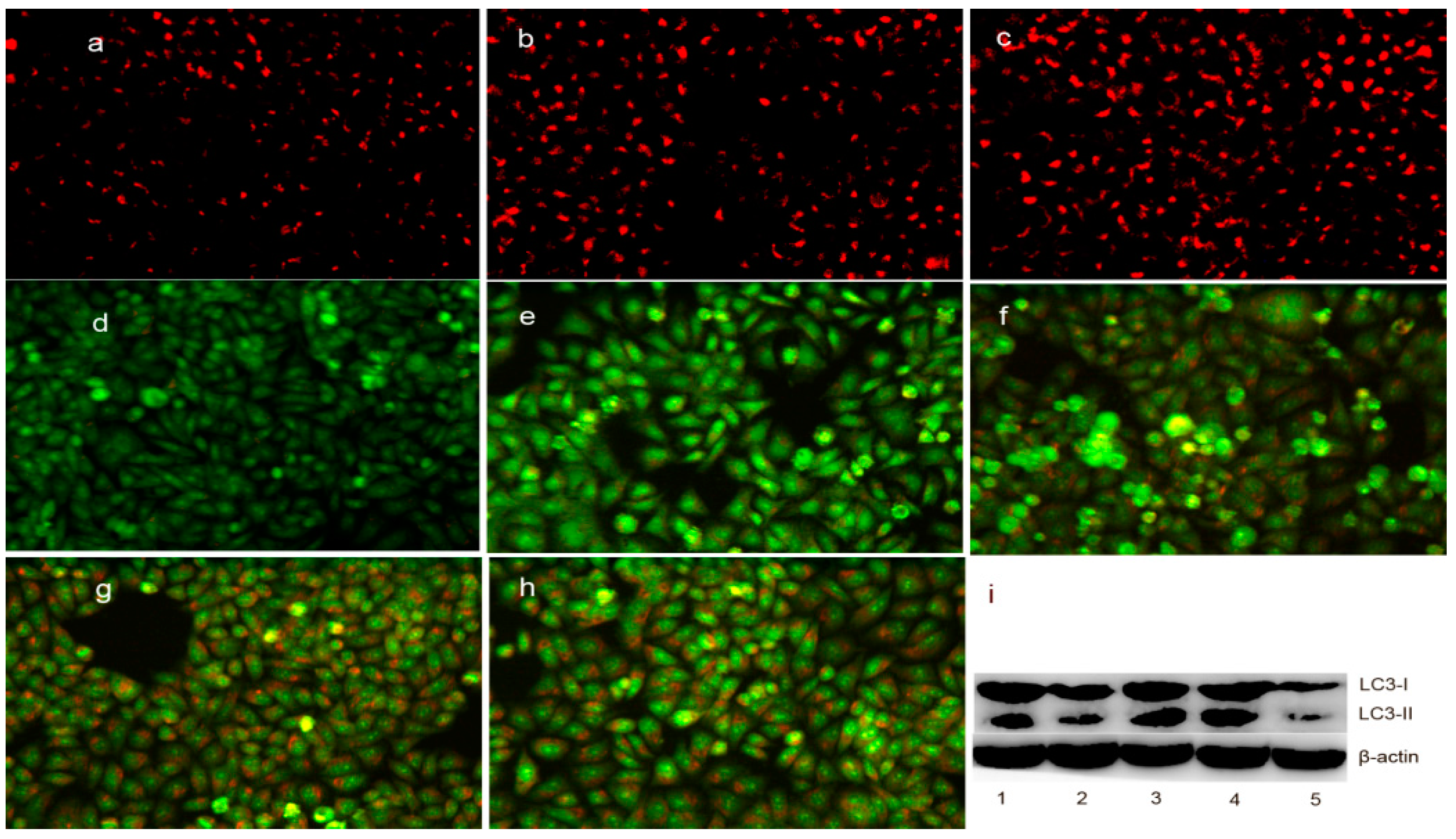
2.7. The Change in Lysosomal (Autophagosome) Membrane Permeability (LMP) in Cells upon Exposure to DpdtpA and Its Copper Complex
3. Discussion
4. Materials and Methods
4.1. General Information
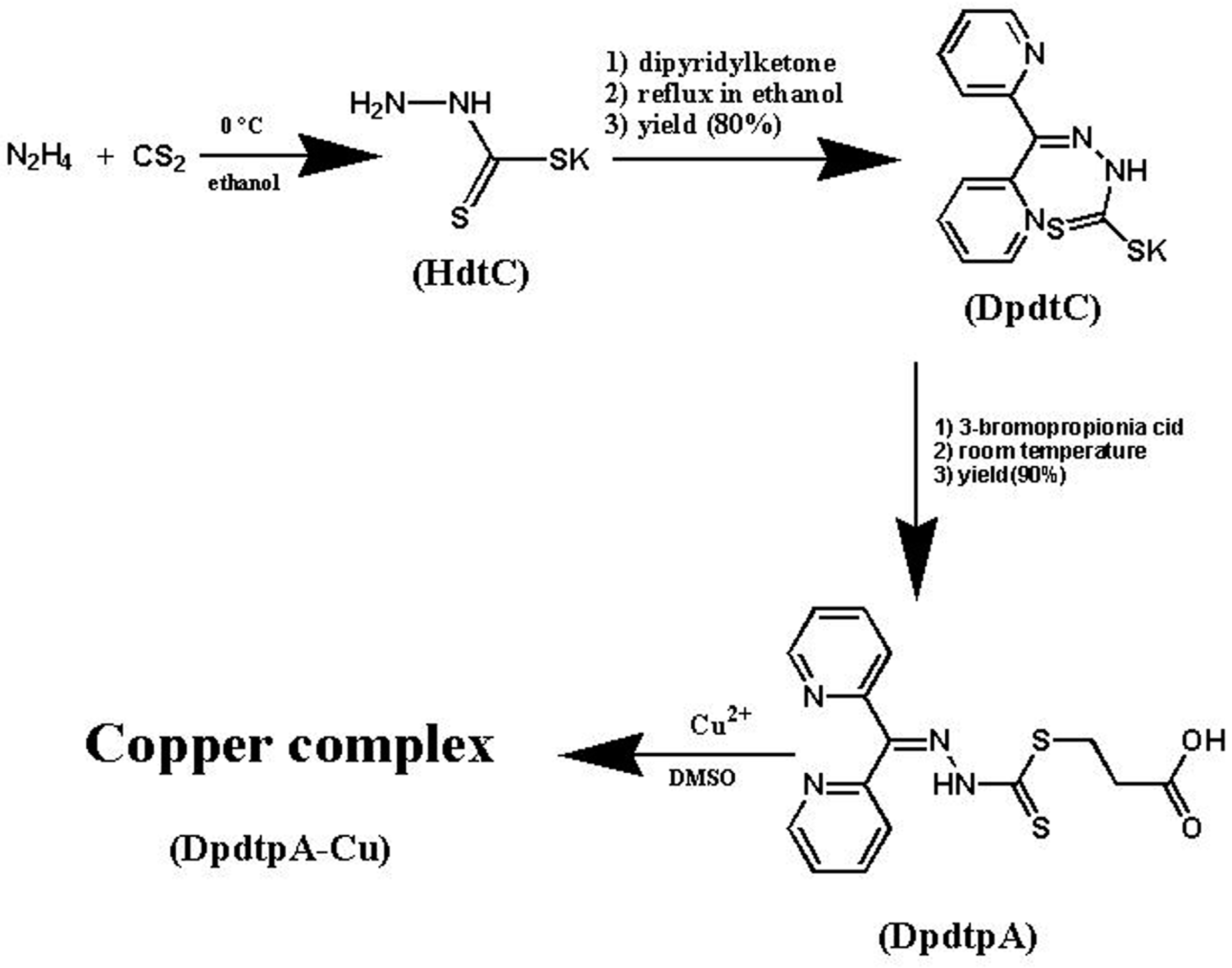
4.2. Preparation of Di-2-pyridylhydrazone Dithiocarbamate S-propionic Acid (DpdtpA)
4.3. Cytotoxicity Assay (MTT Assay)
4.4. Determination of the Molar Ratio of DpdtpA to Copper(II) Involved in the Formation of the Complex
4.5. ROS Detection in Vitro and in Vivo
4.6. Comet Assay
4.7. Western Blotting Analysis
4.8. Cell Cycle Analysis
4.9. DpdtpA and Its Copper Complex Induced Autophagy
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
Abbreviations
| MTT | 3-(4,5-dimethylthiazole-2-yl)-2,5-diphenyl tetrazoliumbromide |
| DpdtpA | Di-2-pyridylhydrazone dithiocarbamate S-propionic acid |
| DpdtpA–Cu | Copper complex of di-2-pyridylhydrazone dithiocarbamate S-propionic acid |
| ROS | Reactive oxygen species |
| ECM | Extracellular matrix |
| NMR | Nuclear magnetic resonance |
| IR | Infrared |
References
- Spano, D.; Zollo, M. Tumor microenvironment: A main actor in the metastasis process. Clin. Exp. Metastasis 2012, 29, 381–395. [Google Scholar] [CrossRef] [PubMed]
- Hiratsuka, S.; Watanabe, A.; Aburatani, H.; Maru, Y. Tumourmediated upregulation of chemoattractants and recruitment of myeloid cells predetermines lung metastasis. Nat. Cell Biol. 2006, 8, 1369–1375. [Google Scholar] [CrossRef] [PubMed]
- Catalano, V.; Turdo, A.; Di Franco, S.; Dieli, F.; Todaro, M.; Stassi, G. Tumor and its microenvironment: A synergistic interplay. Semin. Cancer Biol. 2013, 23, 522–532. [Google Scholar] [CrossRef] [PubMed]
- Sounni, N.E.; Noel, A. Targeting the tumor microenvironment for cancer therapy. Clin. Chem. 2013, 59, 85–93. [Google Scholar] [CrossRef] [PubMed]
- Goodman, V.L.; Brewer, G.J.; Merajver, S.D. Copper deficiency as an anti-cancer strategy. Endocr. Relat. Cancer 2004, 11, 255–263. [Google Scholar] [CrossRef] [PubMed]
- Khan, G.; Merajver, S. Copper chelation in cancer therapy using tetrathiomolybdate: An evolving paradigm. Expert Opin. Investig. Drugs 2009, 18, 541–548. [Google Scholar] [CrossRef] [PubMed]
- Bogaard, H.J.; Mizuno, S.; Guignabert, C.; Al Hussaini, A.A.; Farkas, D.; Ruiter, G.; Kraskauskas, D.; Fadel, E.; Allegood, J.C.; Humbert, M.; et al. Copper dependence of angioproliferation in pulmonary arterial hypertension in rats and humans. Am. J. Respir. Cell Mol. Biol. 2012, 46, 582–591. [Google Scholar] [CrossRef] [PubMed]
- Orrenius, S.; Nobel, C.S.I.; van den Dobbelsteen, D.J.; Burkitt, M.J.; Slater, A.F.G. Dithiocarbamates and the redox regulation of cell death. Biochem. Soc. Trans. 1996, 24, 1032–1038. [Google Scholar] [CrossRef]
- Buac, D.; Schmitt, S.; Ventro, G.; Kona, F.R.; Dou, Q.P. Dithiocarbamate-based coordination compounds as potent proteasome inhibitors in human cancer cells. Mini Rev. Med. Chem. 2012, 12, 1193–1201. [Google Scholar] [CrossRef] [PubMed]
- Fang, I.M.; Yang, C.H.; Lin, C.P.; Yang, C.M.; Chen, M.S. Effects of pyrrolidine dithiocarbamate, an NF-κB inhibitor, on cytokine expression and ocular inflammation in experimental autoimmune anterior uveitis. J. Ocul. Pharmacol. Ther. 2005, 21, 95–106. [Google Scholar] [CrossRef] [PubMed]
- Si, X.; McManus, B.M.; Zhang, J.; Yuan, J.; Cheung, C.; Esfandiarei, M.; Suarez, A.; Morgan, A.; Luo, H. Pyrrolidine dithiocarbamate reduces coxsackievirus B3 replication through inhibition of the ubiquitin-proteasome pathway. J. Virol. 2005, 79, 8014–8023. [Google Scholar] [CrossRef] [PubMed]
- Ozkirimli, S.; Apak, T.I.; Kiraz, M.; Yegenoglu, Y. Synthesis of new triazolyl-N, N-dialkyldithiocarbamates as antifungal agents. Arch. Pharm. Res. 2005, 28, 1213–1218. [Google Scholar] [CrossRef] [PubMed]
- Reisinger, E.C.; Kern, P.; Ernst, M.; Bock, P.; Flad, H.D.; Dietrich, M. Inhibition of HIV progression by dithiocarb. German DTC Study Group. Lancet 1990, 335, 679–682. [Google Scholar] [CrossRef]
- Li, Y.H.; Qi, H.X.; Li, X.B.; Hou, X.L.; Lu, X.Y.; Xiao, X.W. A novel dithiocarbamate derivative induces cell apoptosis through p53-dependent intrinsic pathway and suppresses the expression of the E6 oncogene of human papillomavirus 18 in HeLa cells. Apoptosis 2015, 20, 787–795. [Google Scholar] [CrossRef]
- Wang, X.J.; Xu, H.W.; Guo, L.L.; Zheng, J.X.; Xu, B.; Guo, X.; Zheng, C.X.; Liu, H.M. Synthesis and in vitro antitumor activity of new butenolide-containing dithiocarbamates. Bioorg. Med. Chem. Lett. 2011, 21, 3074–3077. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Ding, Y.; Miao, Y.; Liu, M.Z.; Li, Y.; Yang, G.F. Synthesis and antitumor activity of novel dithiocarbamate substituted chromones. Eur. J. Med. Chem. 2009, 44, 3687–3696. [Google Scholar] [CrossRef]
- Mansouri-Torshizi, H.; Saeidifar, M.; Khosravi, F.; Divsalar, A.; Saboury, A.A.; Hassani, F. DNA Binding and Antitumor Activity of α-Diimineplatinum(II) and Palladium(II) Dithiocarbamate Complexes. Bioinorg. Chem. Appl. 2011, 2011, 394506. [Google Scholar] [CrossRef]
- Milacic, V.; Chen, D.; Ronconi, L.; Landis-Piwowar, K.R.; Fregona, D.; Dou, Q.P. A novel anticancer gold (III) dithiocarbamate compound inhibits the activity of a purified 20S proteasome and 26S proteasome in human breast cancer cell cultures and xenografts. Cancer Res. 2006, 66, 10478–10486. [Google Scholar] [CrossRef]
- Nardon, C.; Schmitt, S.M.; Yang, H.; Zuo, J.; Fregona, D.; Dou, Q.P. Gold(III)-dithiocarbamato peptidomimetics in the forefront of the targeted anticancer therapy: Preclinical studies against human breast neoplasia. PLoS ONE 2014, 9, e84248. [Google Scholar] [CrossRef] [PubMed]
- Cattaruzza, L.; Fregona, D.; Mongiat, M.; Ronconi, L.; Fassina, A.; Colombatti, A.; Aldinucci, D. Antitumor activity of gold(III)-dithiocarbamato derivatives on prostate cancer cells and xenografts. Int. J. Cancer 2011, 128, 206–215. [Google Scholar] [CrossRef] [PubMed]
- Schreck, R.; Meier, B.; Männel, D.N.; Dröge, W.; Baeuerle, P.A. Dithiocarbamates as potent inhibitors of nuclear factor kappa B activation in intact cells. J. Exp. Med. 1992, 175, 1181–1194. [Google Scholar] [CrossRef]
- Ronconi, L.; Marzano, C.; Zanello, P.; Corsini, M.; Miolo, G.; Maccà, C.; Trevisan, A.; Fregona, D. Gold(III) Dithiocarbamate Derivatives for the Treatment of Cancer: Solution Chemistry, DNA Binding, and Hemolytic Properties. J. Med. Chem. 2006, 49, 1648–1657. [Google Scholar] [CrossRef] [PubMed]
- Nobel, C.S.I.; Burgess, D.H.; Zhivotovsky, B.; Burkitt, M.J.; Orrenius, S.; Slater, A.F. Mechanism of dithiocarbamate inhibition of apoptosis: Thiol oxidation by dithiocarbamate disulfides directly inhibits processing of the caspase-3 proenzyme. Chem. Res. Toxicol. 1997, 10, 636–643. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Li, C.; Fu, Y.; Liu, Y.; Zhang, Y.; Zhang, Y.; Zhou, P.; Yuan, Y.; Zhou, S.; Li, S.; Li, C. Redox cycling of a copper complex with benzaldehyde nitrogen Mustard-2-pyridine carboxylic acid hydrazone contributes to its enhanced antitumor activity, but no change in the mechanism of action occurs after chelation. Oncol. Rep. 2016, 35, 1636–1644. [Google Scholar]
- Huang, T.; Li, C.; Sun, X.; Zhu, Z.; Fu, Y.; Liu, Y.; Yuan, Y.; Li, S.; Li, C. The antitumor mechanism of di-2-pyridylketone 2-pyridine carboxylic acid hydrazone and its copper complex in ROS generation and topoisomerase inhibition, and hydrazone involvement in oxygen-catalytic iron mobilization. Int. J. Oncol. 2015, 47, 1854–1862. [Google Scholar] [CrossRef] [PubMed]
- Brodská, B.; Holoubek, A. Generation of reactive oxygen species during apoptosis induced by DNA-damaging agents and/or histone deacetylase inhibitors. Oxid. Med. Cell. Longev. 2011, 2011, 253529. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.C.; Appelqvist, H.; Nilsson, C.; Kågedal, K.; Roberg, K.; Ollinger, K. Regulation of apoptosis-associated lysosomal membrane permeabilization. Apoptosis 2010, 15, 527–540. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rehman, S.U.; Zubair, H.; Sarwar, T.; Husain, M.A.; Ishqi, H.M.; Nehar, S.; Tabish, M. Redox cycling of Cu(II) by 6-mercaptopurine leads to ROS generation and DNA breakage: Possible mechanism of anticancer activity. Tumour Biol. 2015, 36, 1237–1244. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; Zhuang, X.; Lin, L.; Yu, P.; Wang, Y.; Shi, Y.; Hu, G.; Sun, Y. New horizons in tumor microenvironment biology: Challenges and opportunities. BMC Med. 2015, 5, 45. [Google Scholar] [CrossRef] [PubMed]
- Komarov, A.M.; Mak, I.T.; Weglicki, W.B. Iron Potentiates Nitric Oxide Scavenging by Dithiocarbamates in Tissue of Septic Shock Mice. Biochim. Biophys. Acta 1997, 1361, 229–234. [Google Scholar] [CrossRef]
- Moellering, D.; McAndrew, J.; Jo, H.; Darley-Usmar, V.M. Effects of Pyrrolidine Dithiocarbamate on En-dothelial Cells: Protection against Oxidative Stress. Free Radic. Biol. Med. 1999, 26, 1138–1145. [Google Scholar] [CrossRef]
- Nicuolo, F.D.; Serini, S.; Boninsegna, A.; Palozza, P.; Calviello, G. Redox Regulation of Cell Proliferation by Pyrrolidine Dithiocarbamate in Murine Thymoma Cells Transplanted in Vivo. Free Radic. Biol. Med. 2001, 31, 1424–1431. [Google Scholar] [CrossRef]
- Fussell, K.C.; Udasin, R.G.; Gray, J.P.; Mishin, V.; Smith, P.J.; Heck, D.E.; Laskin, J.D. Redox cycling and increased oxygen utilization contribute to diquat-induced oxidative stress and cytotoxicity in Chinese hamster ovary cells overexpressing NADPH-cytochrome P450 reductase. Free Radic. Biol. Med. 2011, 50, 874–882. [Google Scholar] [CrossRef] [PubMed]
- Gillardon, F.; Wickert, H.; Zimmermann, M. Up-regulation of bax and down-regulation of bcl-2 is associated with kainate-induced apoptosis in mouse brain. Neurosci. Lett. 1995, 192, 85–88. [Google Scholar] [CrossRef]
- Chatterjee, S.; Kundu, S.; Sengupta, S.; Bhattacharyya, A. Divergence to apoptosis from ROS induced cell cycle arrest: Effect of cadmium. Mutat. Res. 2009, 663, 22–31. [Google Scholar] [CrossRef] [PubMed]
- Kumar, R.; Dwivedi, P.D.; Dhawan, A.; Das, M.; Ansari, K.M. Citrinin-generated reactive oxygen species cause cell cycle arrest leading to apoptosis via the intrinsic mitochondrial pathway in mouse skin. Toxicol. Sci. 2011, 122, 557–566. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Zhang, H.; Beach, D. D type cyclins associate with multiple protein kinases and the DNA replication and repair factor PCNA. Cell 1992, 71, 505–514. [Google Scholar] [CrossRef]
- Fu, Y.; Yang, Y.; Zhou, S.; Liu, Y.; Yuan, Y.; Li, S.; Li, C. Ciprofloxacin containing Mannich base and its copper complex induce antitumor activity via different mechanism of action. Int. J. Oncol. 2014, 45, 2092–2100. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.L.; Huang, T.F.; Zhou, S.F.; FU, Y.; Liu, Y.X.; Yuan, Y.B.; Zhang, Q.Q.; Li, S.S.; Li, C.Z. Antitumor activity of a 2-pyridinecarboxaldehyde 2-pyridinecarboxylic acid hydrazone copper complex and the related mechanism. Oncol. Rep. 2015, 34, 1311–1318. [Google Scholar] [CrossRef] [PubMed]
- Cvek, B.; Milacic, V.; Taraba, J.; Dou, Q.P. Ni(II), Cu(II), and Zn(II) diethyldithiocarbamate complexes show various activities against the proteasome in breast cancer cells. J. Med. Chem. 2008, 51, 6256–6258. [Google Scholar] [CrossRef] [PubMed]
- Cvek, B.; Dvorak, Z. Targeting of Nuclear Factor-kB and Proteasome by Dithiocarbamate Complexes with Metals. Curr. Pharm. Des. 2007, 13, 3155–3167. [Google Scholar] [CrossRef]
- Yu, Z.; Wang, F.; Milacic, V.; Li, X.; Cui, Q.C.; Zhang, B.; Yan, B.; Dou, Q.P. Evaluation of copper-dependent proteasome-inhibitory and apoptosis-inducing activities of novel pyrrolidine dithiocarbamate analogues. Int. J. Mol. Med. 2007, 20, 919–925. [Google Scholar] [CrossRef] [PubMed]
- Skrott, Z.; Cvek, B. Diethyldithiocarbamate complex with copper: The mechanism of action in cancer cells. Mini Rev. Med. Chem. 2012, 12, 1184–1192. [Google Scholar] [CrossRef] [PubMed]
- Zhang, H.; Wu, J.S.; Peng, F. Potent anticancer activity of pyrrolidine dithiocarbamate-copper complex against cisplatin-resistant neuroblastoma cells. Anticancer Drugs 2008, 19, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Zhu, C.P.; Hu, W.; Wu, H.; Hu, X. No evident dose-response relationship between cellular ROS level and its cytotoxicity—A paradoxical issue in ROS-based cancer therapy. Sci. Rep. 2014, 4, 5029. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Azad, M.B.; Gibson, S.B. Methods for detecting autophagy and determining autophagy-induced cell death. Can autophagy and determining autophagy-induced cell death. Can. J. Physiol. Pharmacol. 2010, 88, 285–295. [Google Scholar] [CrossRef] [PubMed]
- Paglin, S.; Hollister, T.; Delohery, T.; Hackett, N.; McMahill, M.; Sphicas, E.; Domingo, D.; Yahalom, J. A novel response of cancer cells to radiation involves autophagy and formation of acidic vesicles. Cancer Res. 2001, 61, 439–444. [Google Scholar] [PubMed]
- Li, L.; Ishdorj, G.; Gibson, S.B. Reactive oxygen species regulation of autophagy in cancer: Implications for cancer treatment. Free Radic. Biol. Med. 2012, 53, 1399–1410. [Google Scholar] [CrossRef] [PubMed]
- Zhen, H.B.; Xu, Q.; Hu, Y.Y.; Cheng, J.H. Characteristics of heavy metals capturing agent dithiocarbamate (DTC) for treatment of ethylene diamine tetraacetic acid–Cu (EDTA–Cu) contaminated wastewater. Chem. Eng. J. 2012, 209, 547–557. [Google Scholar] [CrossRef]
- Sample Availability: Samples of the compounds are available from the authors.


© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, T.; Fu, Y.; Huang, T.; Liu, Y.; Wu, M.; Yuan, Y.; Li, S.; Li, C. Copper Ion Attenuated the Antiproliferative Activity of Di-2-pyridylhydrazone Dithiocarbamate Derivative; However, There Was a Lack of Correlation between ROS Generation and Antiproliferative Activity. Molecules 2016, 21, 1088. https://doi.org/10.3390/molecules21081088
Wang T, Fu Y, Huang T, Liu Y, Wu M, Yuan Y, Li S, Li C. Copper Ion Attenuated the Antiproliferative Activity of Di-2-pyridylhydrazone Dithiocarbamate Derivative; However, There Was a Lack of Correlation between ROS Generation and Antiproliferative Activity. Molecules. 2016; 21(8):1088. https://doi.org/10.3390/molecules21081088
Chicago/Turabian StyleWang, Tingting, Yun Fu, Tengfei Huang, Youxun Liu, Meihao Wu, Yanbin Yuan, Shaoshan Li, and Changzheng Li. 2016. "Copper Ion Attenuated the Antiproliferative Activity of Di-2-pyridylhydrazone Dithiocarbamate Derivative; However, There Was a Lack of Correlation between ROS Generation and Antiproliferative Activity" Molecules 21, no. 8: 1088. https://doi.org/10.3390/molecules21081088





