2.2. Pd-FAU Crystals: Effect of Pd Content and Exchange Temperature
Synthesis of Pd-loaded FAU crystals resulted in a gray powder material. The Pd-FAU crystals were prepared from the corresponding Na-FAU zeolite by ion exchange by using a PdCl2 aqueous solution as palladium precursor.
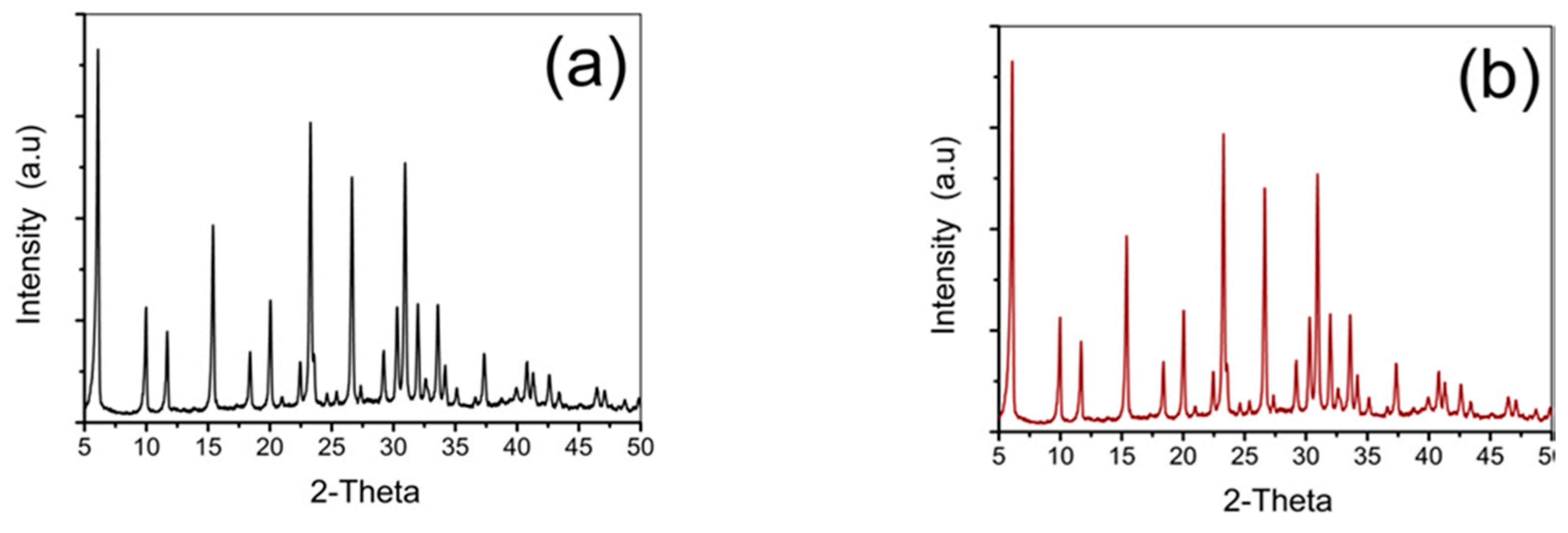
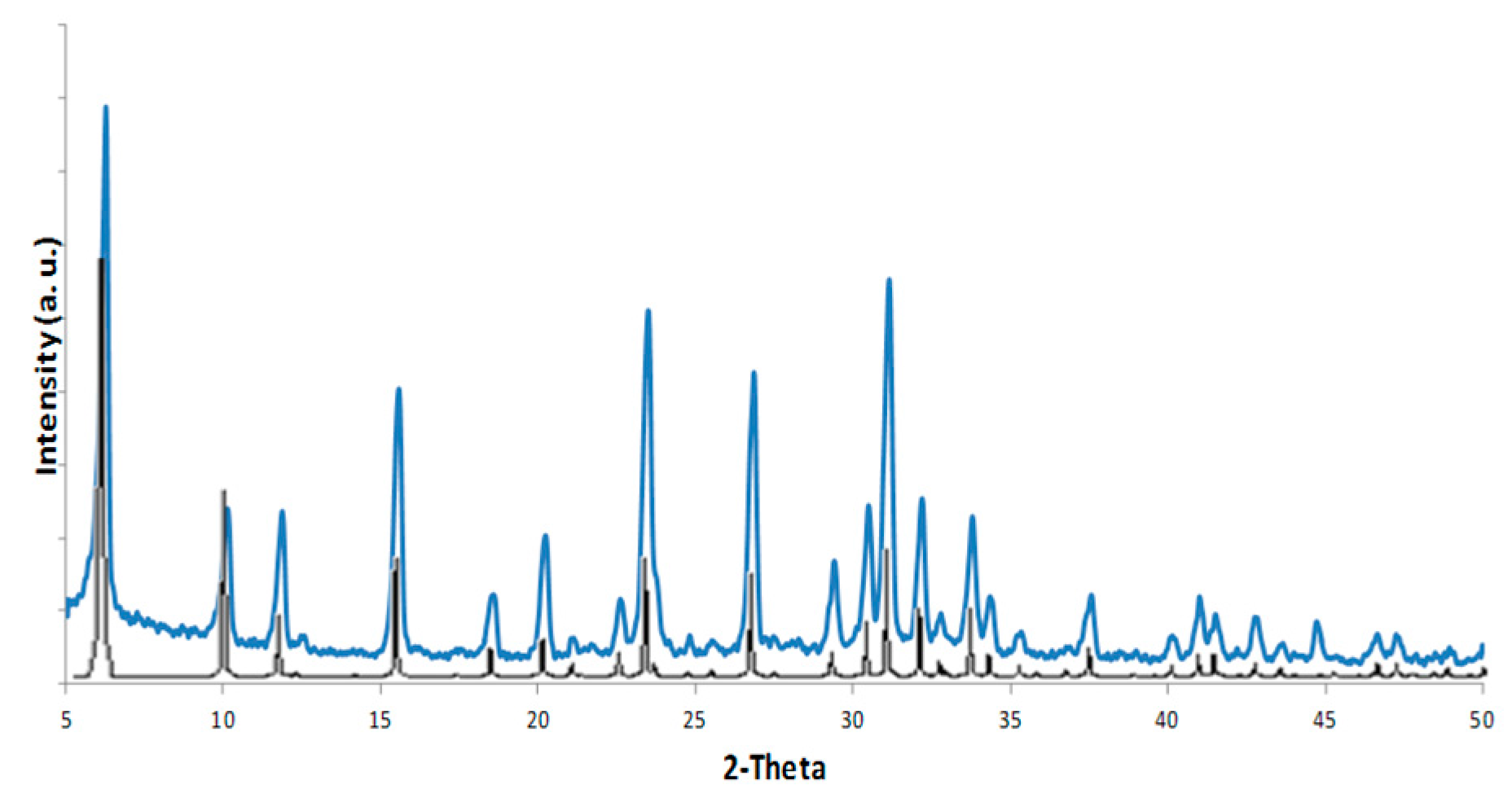
XRD patterns on the three samples: Pd-FAU(0.5-15), Pd-FAU(0.5-60) and Pd-FAU(1-60) were collected and compared with the powder pattern of the pristine FAU seeds (
Figure 1a) with similar results. As shown in
Figure 1b for Pd-FAU(0.5-15), the sample exhibits typical diffraction peaks that are characteristic of a pure FAU phase, confirming that the crystalline structure is kept unaltered after the exchange and the subsequent reduction of Pd
2+.
Moreover, for all the samples, no Pd nanocrystal diffraction peaks are observed in the XRD patterns. These results can be attributed to the presence of typical FAU peaks located at the 2-Theta values of interest, so that the eventual presence of weak diffractive peaks from Pd nanoparticles is hidden. On the other hand, it can be also a sign of the good dispersity of active Pd nanoparticles on the surface of the zeolite supports. Unfortunately, these outcomes prevented us from performing any further analysis to determine, for example, a reliable Pd mean particle size.
The texture of the samples was analyzed by SEM, using the Back Scattering Electrons (BSE) method to detect contrast between areas with different chemical compositions, while EDX analysis were performed in order to establish the effective Pd content. From
Figure 2a,b it is possible to observe visible shiny points, which are self-standing Pd particles quite big (the bar scale is 10 µm) with a heterogeneous distribution in the particle size (from
ca. 500 nm up to 1 µm). A “suffused shiny appearance” is also visible on the crystal surface, from which, as it can be seen from the TEM images of
Figure 3 described below, it can be supposed that Pd is quite homogeneously distributed on the FAU crystal surface (at a microscale level). The mean Pd content determined with EDX by mapping different areas of the samples was 1.0 and 1.5 wt % for Pd-FAU(0.5-15) and Pd-FAU(0.5-60), respectively, proving a positive effect of temperature on the efficacy of the exchange process.
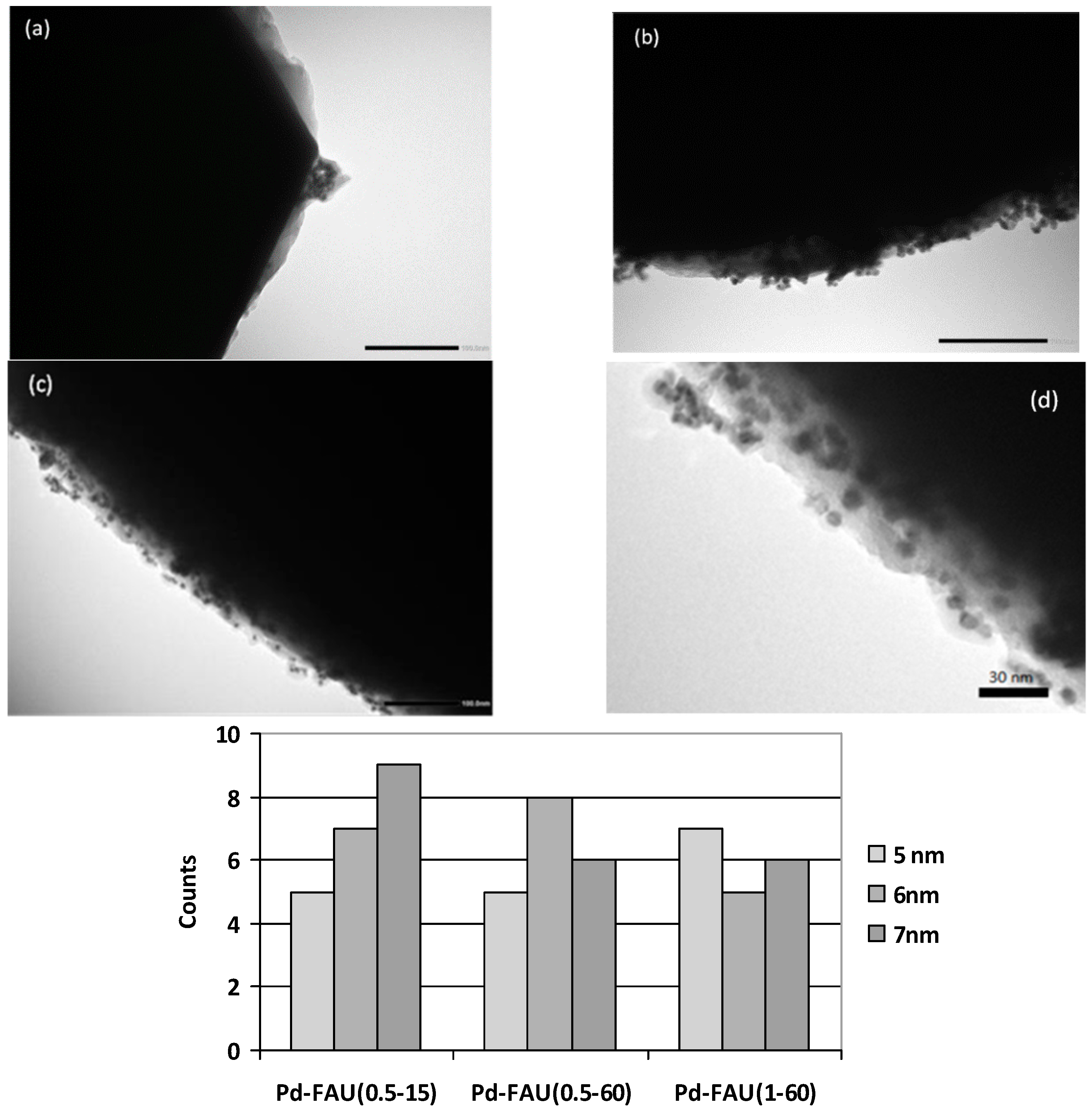
On the contrary, a careful inspection of the Pd-FAU(1-60) sample revealed the absence of any discrete self-standing Pd particles. Surprisingly, it seems that Pd species are quite uniformly distributed on the zeolite surface. The presence of palladium was confirmed by EDX analysis. The measured Pd content was 4.2 wt %, indicating a higher degree of cation exchange within the zeolite framework. The morphology and size of the Pd nanoparticles loaded on the zeolite microcrystals were investigated by Transmission Electron Microscopy (TEM) in order to get information at a nanoscale level. The transmission electron microscopy images of Pd-FAU(0.5-15) (a), Pd-FAU(0.5-60) (b) and Pd-FAU(1-60) (c) samples are shown in
Figure 3, along with the pertinent histograms showing the particle size distributions (d).
Both Pd-FAU(0.5-60) (
Figure 3b) and Pd-FAU(1-60) (
Figure 3c) samples show the presence of distinct Pd nanoparticles homogeneously dispersed on the external surface of zeolite crystals. The Pd nanoparticles feature a quasi-spherical shape and quite uniform particle size, with values ranging from 5 to 7 nm for all the samples. However, in Pd-FAU(0.5-60), some nanoparticles also form agglomerates of greater dimensions (30–50 nm) which stably adhere to the external surface of the zeolite crystals. On the contrary, in Pd-FAU(0.5-15) Pd particles are not uniformly and densely distributed on the zeolite surface (
Figure 3a). The surface area of the zeolite is only partially covered by Pd nanoparticles, which are basically present as agglomerates (100–150 nm) of particles of smaller dimensions.
The textural properties of the microcrystalline Pd-FAU samples were investigated by determining the porous volume and surface area of the host material from nitrogen adsorption isotherms, compared with data obtained for pristine FAU microcrystals (
Table 1).
As it can be seen, the Pd-FAU(1-60) shows a lower BET surface area and micropore volume if compared with samples Pd-FAU(0.5-15) and Pd-FAU(0.5-60), which can be attributed to the formation of Pd nanoclusters within the micropores of the zeolite as well as on the external surface. The nanoparticles loaded on the external surface are mostly exposed and consequently readily accessible while the Pd species included into the pores are not easily accessible to the reacting molecules and scarcely contribute to the catalytic activity. The results of the catalytic tests carried out with the three samples under the same experimental conditions are reported in
Table 2.
The degree of conversion and the selectivity values for Pd-FAU(0.5-60) are higher than Pd-FAU(0.5-15), as expected considering the higher amount of Pd catalyst loaded on the zeolite seeds. Furthermore, concerning the Pd-FAU(0.5-15) sample, the scarcity of Pd particles present on the external zeolite surface lowers the active surface area of the catalyst and, accordingly, its catalytic efficiency. On the contrary, and quite surprisingly, a lower conversion is observed for Pd-FAU(1-60). This behavior can be explained by taking into account the distribution of the active Pd metal sites deep inside the zeolite framework, which cannot be easily reached by the reacting molecules.
On the basis of the experimental results, it can reasonably hypothesized that the Pd particles on the surface of the zeolite crystals exhibit the best catalytic efficiency compared to the Pd sites confined inside the zeolite pores. The Pd-FAU(0.5-60) sample was selected for further testing of the catalytic process.
2.3. Influence of the Solvent and the Hydrogen Donor
Although the role of the catalyst is of primary importance to achieve both high activity and selectivity, the solvent selection is of great relevance, as well. Indeed, it has been demonstrated that the solvent can sensibly affect the catalyst activity and selectivity [
46] as a result of different solvent/reactant and solvent/catalyst interactions. In this work, we tested different solvents as well as different hydrogen donors. The results obtained in terms of selectivity, conversion and yield are reported in
Table 3.
As it can be seen, the catalytic activity (conversion) varies according to the following pattern: HCOONa/water > ethanol > 2-propanol and it seems to follow the decreasing polarity of the solvents as well as the decreasing dehydrogenation activity of the hydrogen donor [
47,
48]. Indeed, formic acid and its salts are strong hydrogen donors [
49]. An influence of the solvent nature on selectivity and yield was also observed. In particular, both increase with decreasing alcohol polarity, while a maximum is observed for the HCOONa/water system.
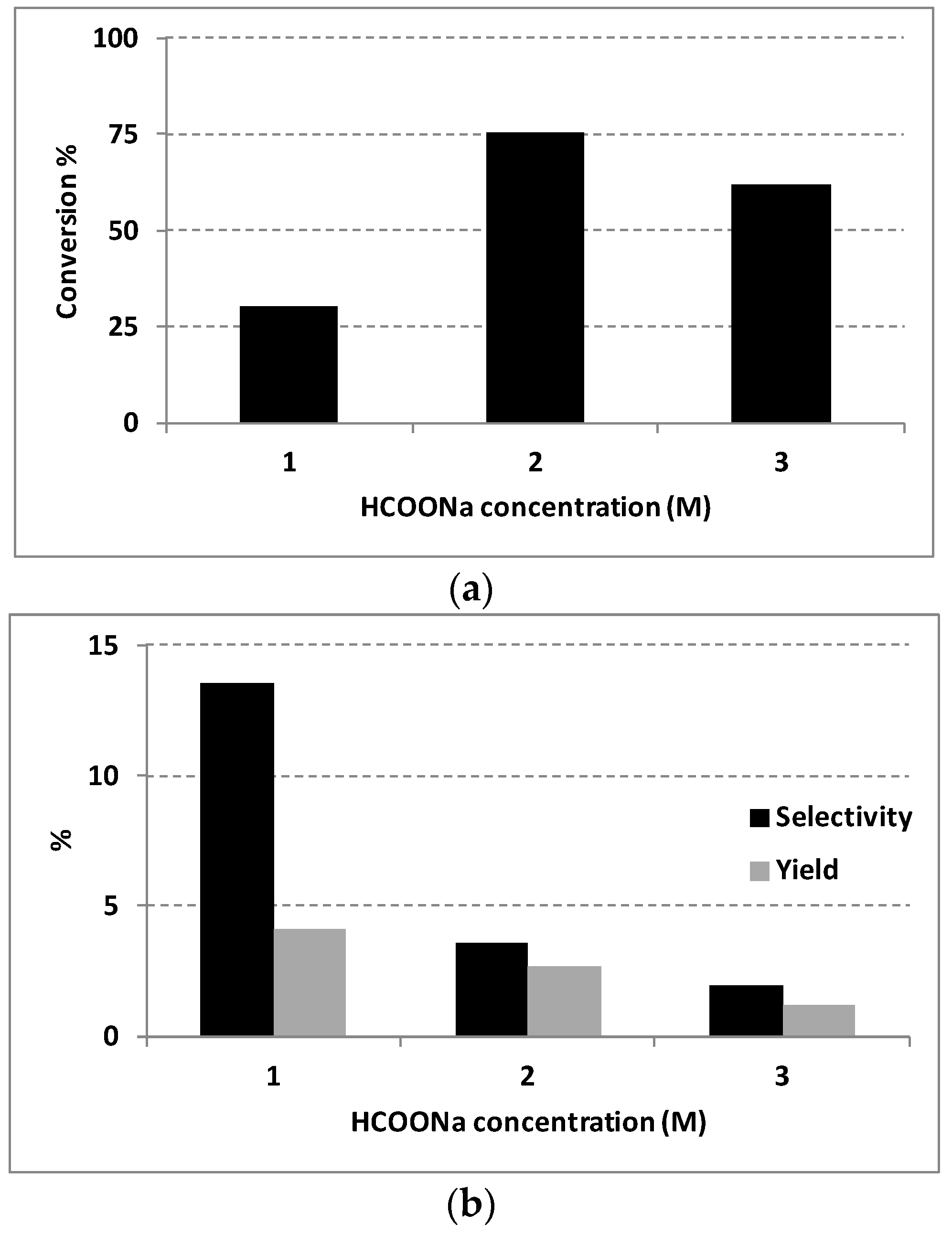
Since the HCOONa/water system seems to guarantee better results for the transfer hydrogenation process, we also investigated the effect of HCOONa concentration on the AP conversion. Results are summarized in
Figure 4.
A higher selectivity to PE was observed using HCOONa 1 M as hydrogen source, even if a lower catalytic activity is also observed, while the increase of the concentration up to 3 M significantly decreases the selectivity value.
Since formate features both a Brønsted and Lewis basicity (being an efficient hydrogen donor), we can speculate that it can also act as acidity inhibitor, especially when high concentrations are used. Indeed, the resultant pH value of the reaction media (starting pH is ca. 10) also increase by increasing the formate concentration. Moreover, the interaction of formate ions with the zeolite framework can lead to a change in the electron distribution or charge on the support, with a consequent variation of the metal/support interactions, and ultimately, of the catalyst activity. Finally, the poisoning or leaching of the Pd catalyst can also occur, caused by the increased concentration of formate molecules.
For comparative purposes, catalytic experiments were conducted in the best selected condition using pristine FAU crystals and home-made Pd particles, prepared following the procedure described for the preparation of Pd-loaded FAU crystals.
Table 4 summarizes the results. As it can be seen, even if the Pd-FAU(0.5-60) sample shows a lower catalytic activity with respect to both the unsupported Pd particles and the pristine FAU crystals, it shows a higher selectivity, yield and productivity if compared to both of them. The best performance of Pd-FAU(0.5-60) can be most likely attributed to the synergy resulting from (i) Pd particles well dispersed on the larger surface area of the zeolite support when compared to the free Pd catalyst and (ii) a selective effect of the porous zeolite framework.
2.4. Pd-FAU Membrane: Synthesis, Characterization and Catalytic Activity
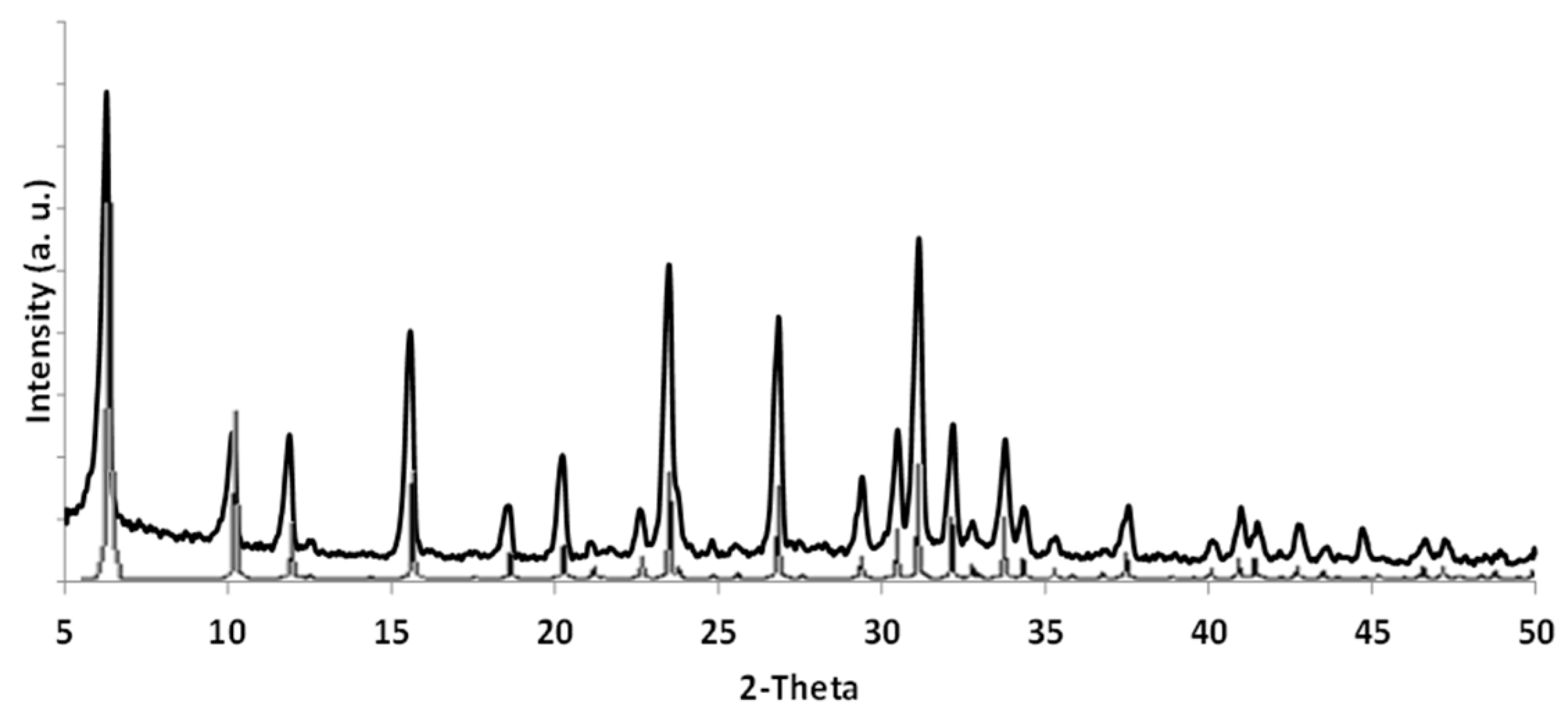
The membrane synthesis was carried out according to the procedure described in
Section 3.2 obtaining a thin and dense zeolite top layer on the support surface. XRD analysis, performed on a sample scratched from the support surface, indicated the formation of a well-crystallized, pure FAU phase (
Figure 5).
SEM images collected on the sample showed that the FAU layer has a thickness of
ca. 2 μm and presents a granule-like texture. A careful inspection revealed that the layer was constituted by closely packed intergrown nanocrystals, whose dimension was
ca. 20–30 nm (
Figure 6). EDX analysis showed that the zeolite layer had a Si/Al ratio of
ca. 2.5.
The membrane was exchanged by using the procedure described in
Section 3.3 for the FAU(0.5-60) sample, with a final Pd content of
ca. 1.4% determined by EDX. XRD analysis performed on a Pd-loaded sample indicated that crystallinity and phase purity of the zeolite layer were fully preserved upon treatment (
Figure 7).
The Pd-FAU membrane was tested under the conditions described in
Section 3.5. Typically, a lowering of the catalytic activity should be expected for the supported Pd-FAU membrane with respect to the bare Pd-FAU crystals as a result of the decrease of the active surface area, as well as more pronounced mass transport limitation effects. Indeed, even if the membrane is constituted by nanozeolites, they are not isolated domains. Because of intraparticle growth, zeolite crystals coalesce, forming a unique extended macro-structure. As a consequence, it can be seen from
Table 5 that the values of PE productivity (based on the total amount of catalyst/support and on the sole amount of loaded Pd) obtained with the Pd-FAU membrane are much higher than Pd-FAU(0.5-60).
On the basis of the obtained results and taking into account the microstructure of the zeolite membrane support, we can speculate that the nanozeolite based membrane is able to guarantee a higher surface area compared to that of the pristine FAU seeds [
50]. This surface area is thus available for palladium deposition, allowing a better metal dispersion on the zeolite membrane surface. Furthermore, the presence of a secondary mesoporosity, which can originate among crystallite domains, can also minimize the mass transport resistance, facilitating the diffusion of the AP molecules to the catalytically active zeolite wall sites. To confirm this hypothesis, TEM (
Figure 8) and BET (
Table 6) analyses were performed on the Pd-FAU membrane.
The image of Pd-FAU membrane shows the presence of well dispersed Pd nanoparticles on the external surface of the zeolite layer, with no significant variation in their particle size and shape with respect to the Pd-FAU microcrystalline samples. We did not observe the presence of any agglomerate in the samples investigated.
BET analysis (see
Table 6) indicates a high external surface area which is about 50% of the total surface area for the Pd-FAU membrane, most likely due to the presence of nanozeolites constituting the membrane layer. Moreover, the value of the median pore width of
ca. 5 nm confirms the formation of mesopores. The diffusion contribution in the zeolite pore (average pore width 1.9 nm) will be negligible compared to the diffusion in the inter-crystalline mesoporosity originated within the membrane layer. It must be observed that extra-zeolite mesoporosity is generated only in the membrane layer. Indeed, DLS measurements [
51], carried out on synthesized FAU nanocrystals, indicated mean particle size distribution values in the range of 50–59 nm. Moreover, the high pH value (
ca. 10) used in the studied catalytic reaction favors the formation of stable nanocrystals suspension, thus excluding any aggregates formation. In other words, mesoporosity will dominate the reagent diffusion to the catalytically active zeolite wall sites. This finding confirms the results recently obtained on the use of this hierarchical membrane for gas de-watering [
52].
Zeolite layers having low diffusional resistance (owing to the presence of micro-, meso- and macroporores) allow substantial improvements of selectivity when the target product is the intermediate of a successive reaction pathway or when other competitive reactions occur, since they are able to provide reduced reactant/catalyst contact-time. Furthermore, the high external surface area guaranteed by nanozeolites also provide a suitable surface for the homogeneous dispersion of the Pd catalyst (the post synthesis modification of FAU nanozeolites results into the dispersion of Pd particles mainly on the external surface of the zeolite), which in turn implies higher productivity value.
To prove the reproducibility of the results and the reliability of this catalytic system upon time, the Pd-FAU membrane was reused consecutively for three times. The results are reported in
Table 7. As it can be seen, despite a significant lowering of the conversion observed after the first cycle, the system is quite stable in terms of yield and productivity over time. Selectivity towards the formation of PE is low, but it is also preserved. These results indicate a stability of the membrane but its catalytic function must be further investigated before considering possible future industrial applications of membrane reactors manufactured with hierarchical porous membranes combining the properties of suitable zeolites and catalysts.
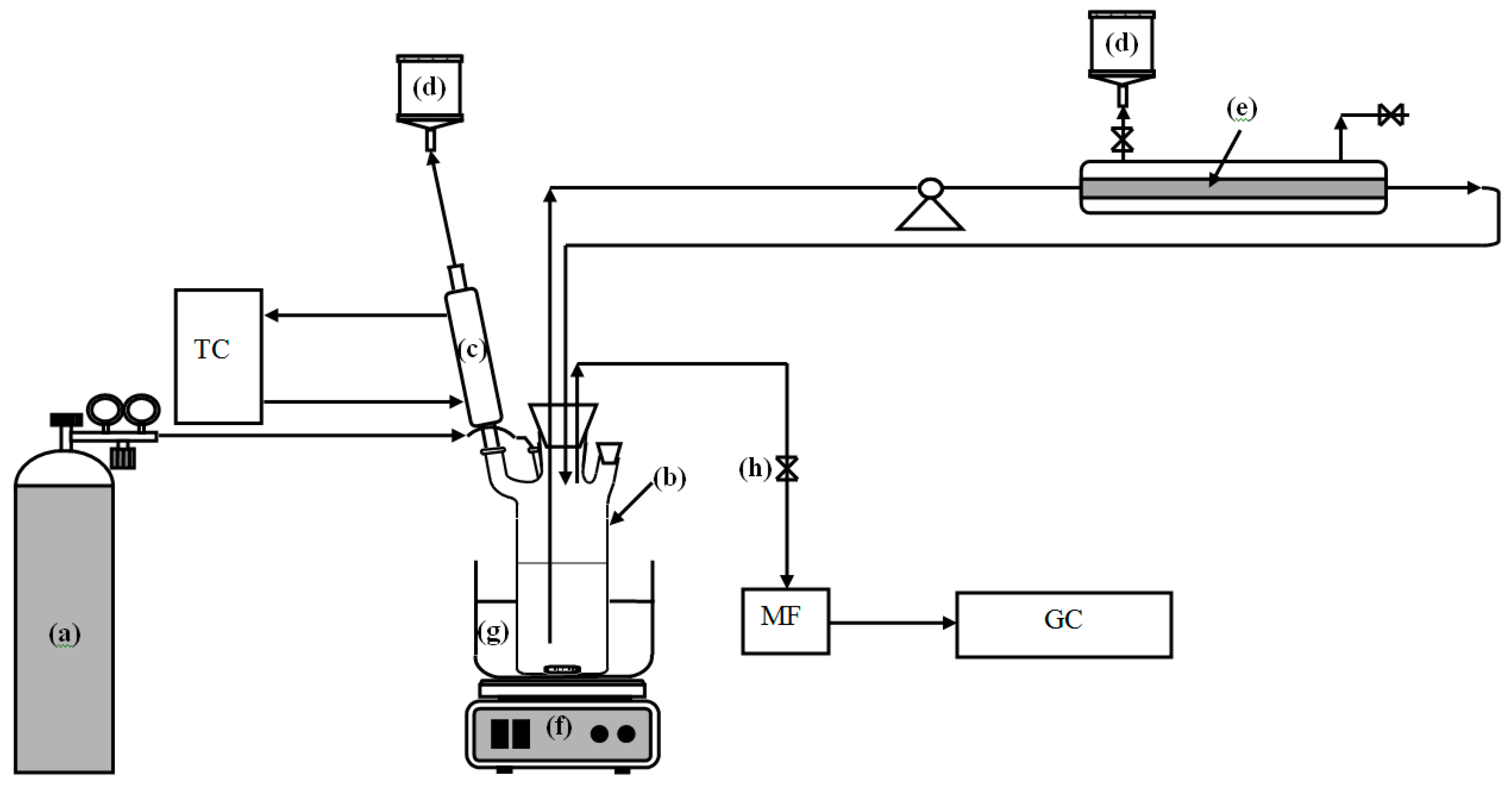
The Pd-FAU membrane was also tested in tangential-flow mode to check its adhesion on the support. To do this test the membrane was housed in the system described in
Scheme 1. The results are reported in
Table 8 and compared with that obtained in the batch system.
It can be seen that when the transfer hydrogenation reaction takes place in tangential-flow mode an increase of yield, conversion, selectivity and productivity is observed compared to the batch. This improved performance of the membrane, when operated in tangential flow mode, can be ascribed to predominance of convection in the mesoporosity over the diffusion which happens in the batch system. These results further confirm the efficacy of the proposed catalytic system for the studied reaction.
However, it is important to underline that various operating parameters (e.g., contact time of reacting solution with the catalytic membrane, pump flow rate, type of catalyst, etc.) need to be investigated and optimized in a suitable membrane reactor.
Concerning the Pd catalyst is must be observed that it was only a first choice to test the catalytic function. In
Table 9, the conversion (C), selectivity (S) and yield (Y) obtained with the Pd-FAU membrane are compared with the results obtained with another Pd catalyst [
53], supported on silica particles, used in the heterogeneous transfer hydrogenation of AP. Comparable yield (%) of PE has been obtained even if a higher ratio Pd/[AP] and higher temperature was used in ref. [
53].
However, by looking at other examples reported in literature, [
54,
55,
56] it is clear that the catalyst performance is strongly improved by the choice of catalytic system including the nature of the metal, the degree of crystallinity as well as the type of metal catalyst support. The results obtained with our catalytic system, where the micro/mesoporous zeolite membrane was used as catalyst support, could be further improved by selecting a different metal catalyst among those which appear to be more active in the transfer hydrogenation of AP.