Casbane Diterpenes from Red Sea Coral Sinularia polydactyla
Abstract
:1. Introduction
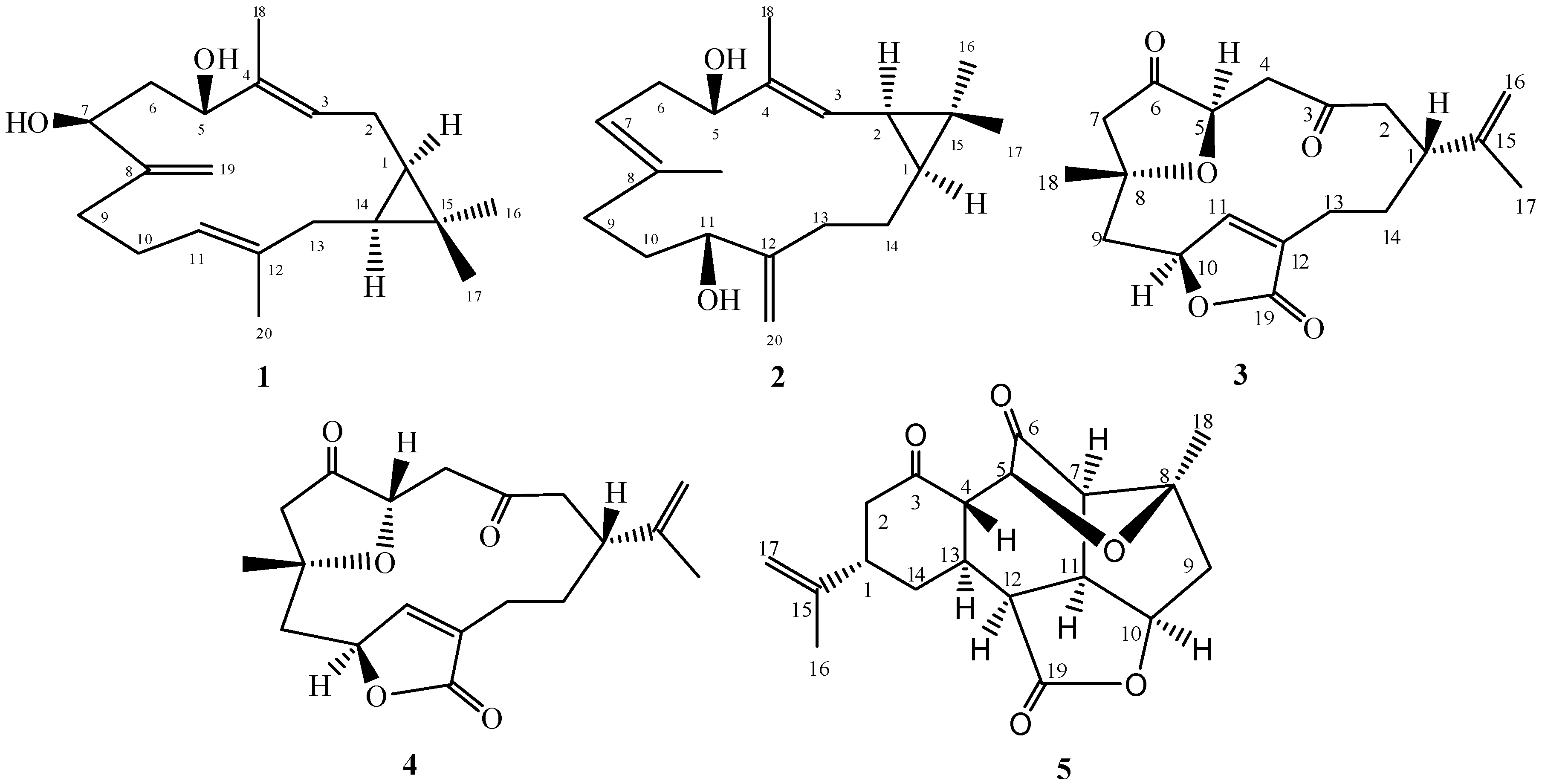
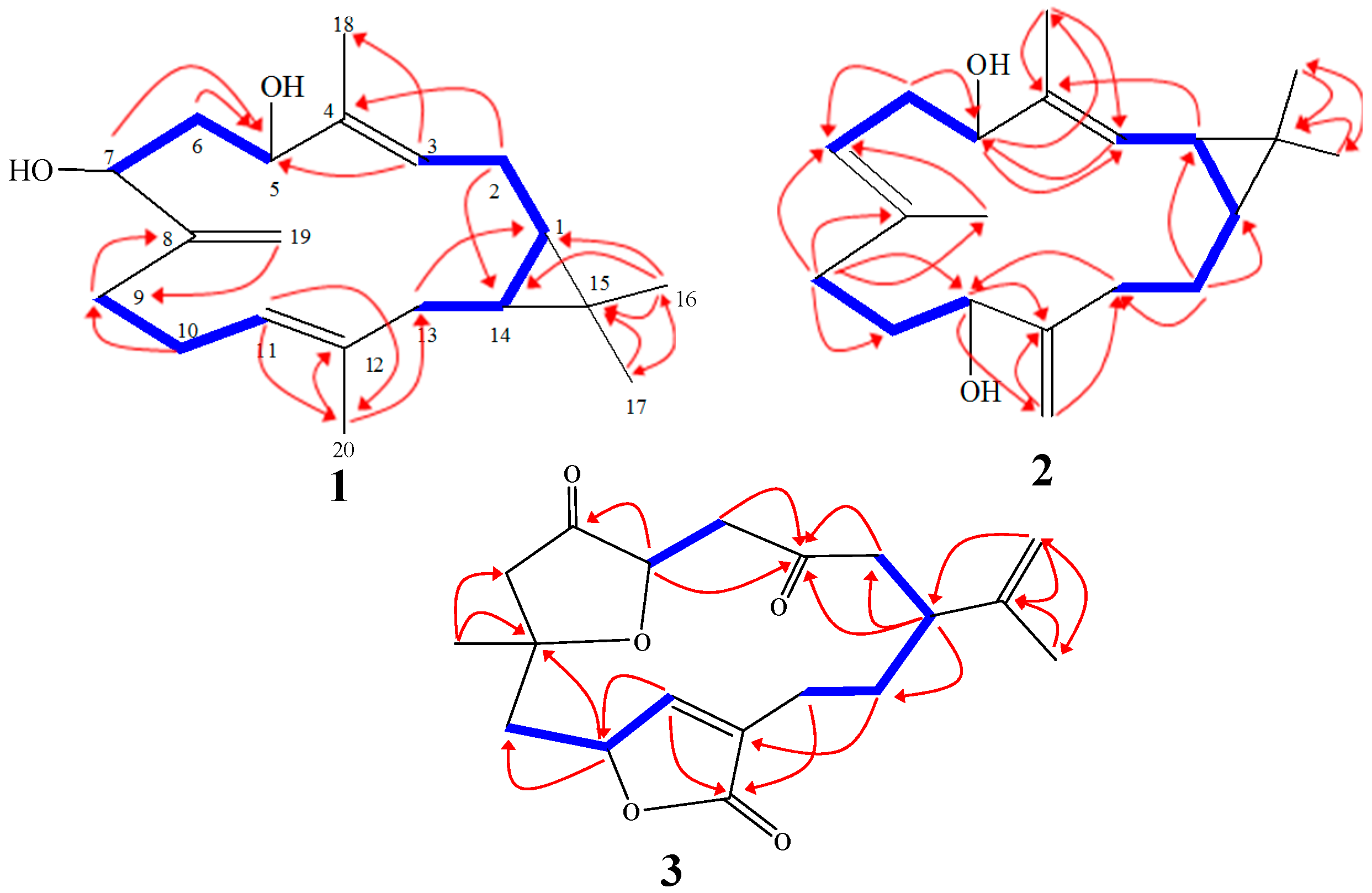
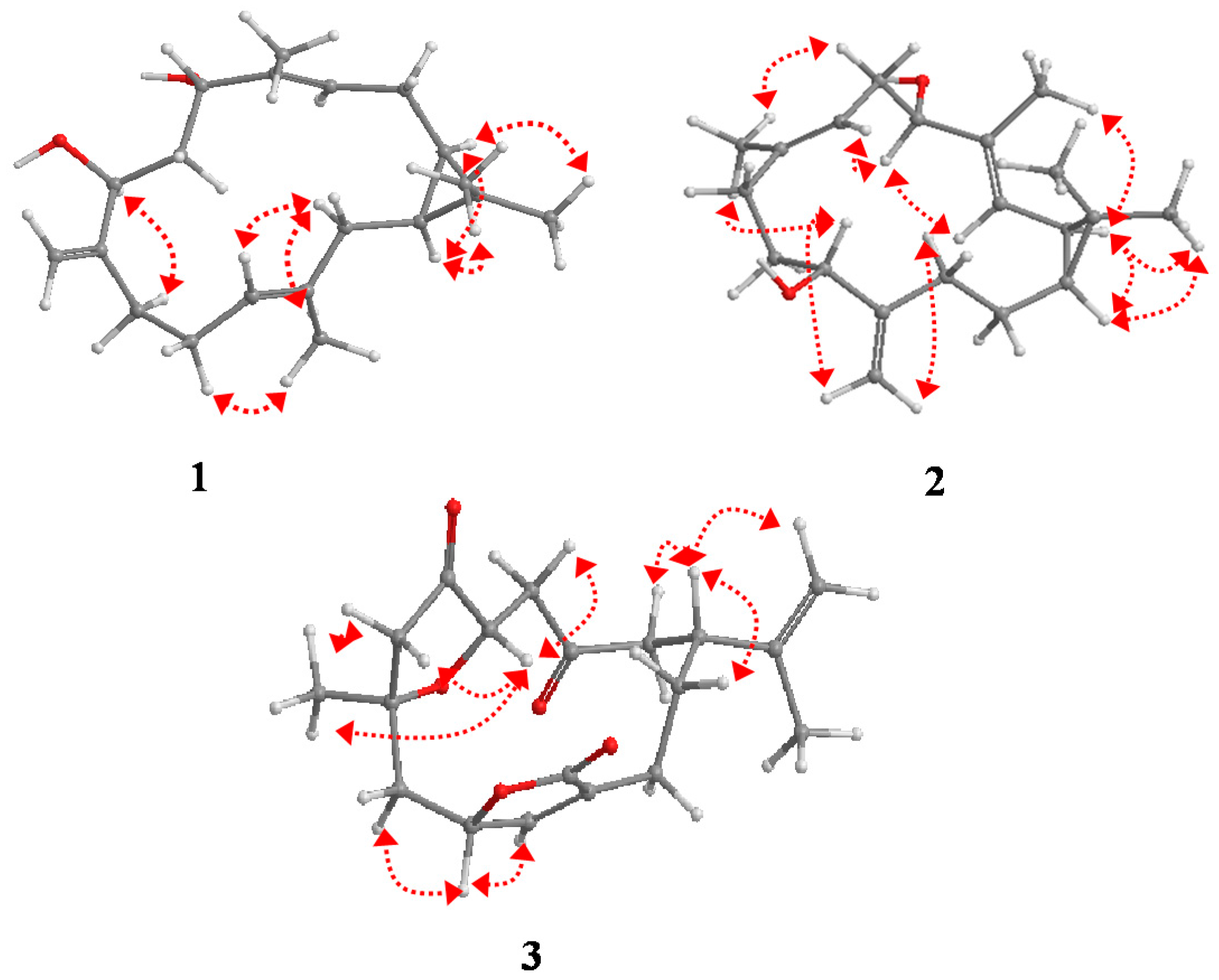
2. Results and Discussion
3. Materials and Methods
3.1. General Information
3.2. Animal Material
3.3. Extraction and Separation
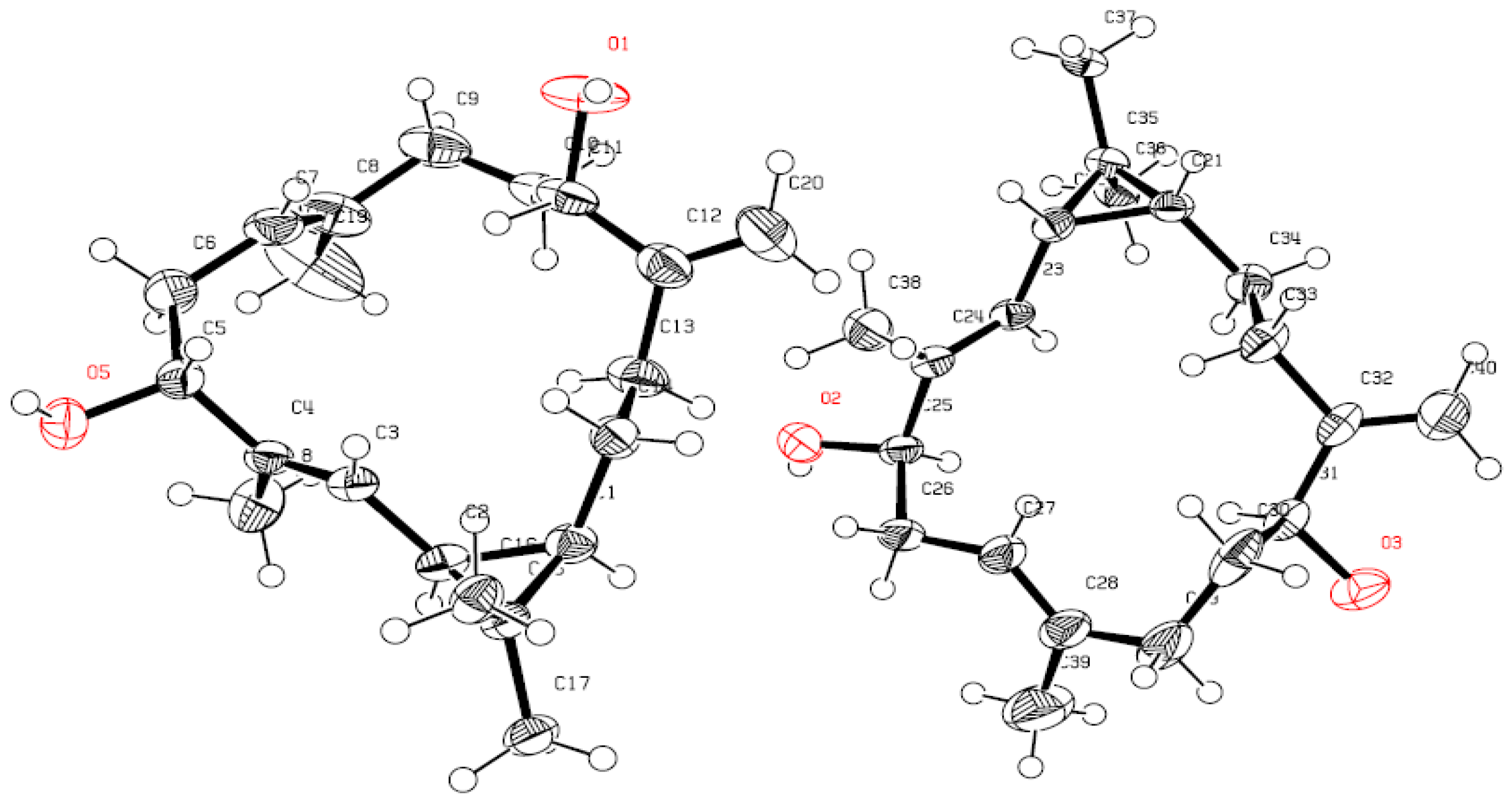
X-ray Crystallography Data
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Anjaneyulu, A.S.R.; Venkateswarlu, R. The chemical constituents of soft coral species of Sinularia genus: A review. J. Sci. Ind. Res. 1995, 54, 637–649. [Google Scholar]
- Kamel, H.N.; Slattery, M. Terpenoids of Sinularia: Chemistry and biomedical applications. Pharm Biol. 2005, 43, 253–269. [Google Scholar] [CrossRef]
- Shaaban, M.; Shaaban, K.; Ghani, M. Hurgadacin: A new steroid from Sinularia polydactyla. Steroids 2013, 78, 866–873. [Google Scholar] [CrossRef] [PubMed]
- Yin, J.; Zhao, M.; Ma, M.; Xu, Y.; Xiang, Z.; Cai, Y.; Dong, J.; Lei, X.; Huang, K.; Yan, P. New casbane diterpenoids from a south China sea soft coral, Sinularia sp. Mar. Drugs 2013, 11, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Shi, H.Y.; Yu, S.J.; Liu, D.; van Ofwegen, L.; Proksch, P.; Lin, W.H. Sinularones A–I, new cyclopentenone and butenolide derivatives from a marine soft coral Sinularia sp. and their antifouling activity. Mar. Drugs 2012, 10, 1331–1344. [Google Scholar] [CrossRef] [PubMed]
- Yamashita, T.; Nakao, Y.; Matsunaga, S.; Oikawa, T.; Imahara, Y.; Fusetani, N. A new antiangiogenic C-24 oxylipin from the soft coral Sinularia numerosa. Bioorg. Med. Chem. 2009, 17, 2181–2184. [Google Scholar] [CrossRef] [PubMed]
- Chai, M.C.; Wang, S.K.; Dai, C.F.; Duh, C.Y. A cytotoxic lobane diterpene from the formosan soft coral Sinularia inelegans. J. Nat. Prod. 2000, 63, 843–844. [Google Scholar] [CrossRef] [PubMed]
- Sheu, J.H.; Chang, K.C.; Duh, C.Y. A cytotoxic 5α,8α-epidioxysterol from a soft coral Sinularia species. J. Nat. Prod. 2000, 63, 149–151. [Google Scholar] [CrossRef] [PubMed]
- Chao, C.H.; Chou, K.J.; Huang, C.Y.; Wen, Z.H.; Hsu, C.H.; Wu, Y.C.; Dai, C.F.; Sheu, J.H. Bioactive cembranoids from the soft coral Sinularia crassa. Mar. Drugs 2011, 9, 1955–1968. [Google Scholar] [CrossRef] [PubMed]
- Wright, A.D.; Nielson, J.L.; Tapiolas, D.M.; Liptrot, C.H.; Motti, C.A. A great barrier reef Sinularia sp. yields two new cytotoxic diterpenes. Mar. Drugs 2012, 10, 1619–1630. [Google Scholar] [CrossRef] [PubMed]
- Edwards, A.J.; Head, S.M. Key Environments-Red Sea; Pergamon Press: Oxford, UK, 1987; p. 440. [Google Scholar]
- Hegazy, M.E.F.; Gamal Eldeen, A.M.; Shahat, A.A.; Abdel-Latif, F.F.; Mohamed, T.A.; Whittlesey, B.R.; Paré, P.W. Bioactive hydroperoxyl cembranoids from the Red Sea soft coral Sarcophyton glaucum. Mar. Drugs 2012, 10, 209–222. [Google Scholar] [CrossRef] [PubMed]
- Hegazy, M.E.F.; Mohamed, T.A.; Abdel-Latif, F.F.; Alsaid, M.S.; Shahat, A.A.; Paré, P.W. Trochelioid A and B, new cembranoid diterpenes from the Red Sea soft coral Sarcophyton trocheliophorum. Phytochem. Lett. 2013, 6, 383–386. [Google Scholar] [CrossRef]
- Elkhateeb, A.; El-Beih, A.A.; Gamal-Eldeen, A.M.; Alhammady, M.A.; Ohta, S.; Paré, P.W.; Hegazy, M.E. New terpenes from the Egyptian soft coral Sarcophyton ehrenbergi. Mar. Drugs 2014, 12, 1977–1986. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Carbone, M.; Vitale, R.M.; Amodeo, P.; Castelluccio, F.; Sicilia, G.; Mollo, E.; Nappo, M.; Cimino, G.; Guo, Y.W.; et al. Rare casbane diterpenoids from the Hainan soft coral Sinularia depressa. J. Nat. Prod. 2010, 73, 133–138. [Google Scholar] [CrossRef] [PubMed]
- Yang, B.; Huang, J.; Lin, X.; Liao, S.; Zhou, X.; Liu, J.; Wang, J.; Wang, L.; Liu, Y. New casbane diterpenoids from the Hainan soft coral Sinularia species. Helv. Chim. Acta 2015, 98, 834–841. [Google Scholar] [CrossRef]
- Saitman, A.; Rulliere, P.; Sullivan, S.D.E.; Theodorakis, E.A. Total synthesis of norcembrenoilide A and scabrolide D. Org. Lett. 2011, 13, 5854–5857. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, A.F.; Su, J.; Kuo, Y.; Sheu, J. Scabrolides E–G, three new norditerpenoids from the soft coral Sinularia scabra. J. Nat. Prod. 2004, 67, 2079–2082. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.T.; Li, Y.; Guo, Y.W. Terpenoids of Sinularia soft corals: Chemistry and bioactivity. Acta. Pharm. Sin. B. 2012, 2, 227–237. [Google Scholar] [CrossRef]
- Duh, C.Y.; Wang, S.K.; Chia, M.; Chiang, M.Y. A novel cytotoxic norditerpenoid from the Formosan soft coral Sinularia inelegans. Tetrahedron Lett. 1999, 40, 6033–6035. [Google Scholar] [CrossRef]
- Sample Availability: Samples of compounds 2, 4, and 5 are available from the authors.

| Position | 1 | 2 | 3 | 4 | |||
|---|---|---|---|---|---|---|---|
| δH (J in Hz) | δC | δH (J in Hz) | δC | δH (J in Hz) | δC | δC | |
| 1 | 0.69 (td, 10.9, 2.6) | 25.0 | 0.77 (ddd, 11.9, 8.9, 2.7) | 31.1 | 2.58 m | 40.8 | 38.8 |
| 2 | 1.26 (m) | 29.6 | 1.28 (dd, 10.1, 8.9) | 25.2 | 2.94 d (3.42), 2.96 d (2.04) | 50.2 | 50.2 |
| 3 | 5.19 (brd, 8.3) | 125.4 | 5.09 (dd, 10.1, 1.2) | 125.6 | 208.4 | 208.4 | |
| 4 | 137.9 | 136.8 | 2.47 m, 2.57 m | 44.0 | 44.0 | ||
| 5 | 4.39 ( dd, 10.0, 4.3) | 77.0 | 3.97 (dd, 10.7, 4.6) | 79.5 | 4.12 d (3.42), 4.14 (2.76) | 74.8 | 77.8 |
| 6 | 1.60 (ddd, 13.8, 11.1, 4.3) | 40.2 | 2.31 (ddd, 13.4, 10.7, 7.9) | 33.2 | 212.7 | 212.5 | |
| 1.94 (ddd, 13.8, 10.0, 2.2) | 2.38 (brdd, 13.4, 4.6) | ||||||
| 7 | 3.93 (brd, 11.1) | 71.2 | 4.86 (brdd, 7.9, 1.2) | 119.5 | 2.21 m, 2.39 m | 48.2 | 51.1 |
| 8 | 154.0 | 136.0 | 78.7 | 79.4 | |||
| 9 | 2.07 m | 33.4 | 1.98 (dd, 15.6, 7.6) | 34.3 | 2.11 m, 2.35 m | 43.1 | 41.7 |
| 2.19 m | 2.09 (ddd, 15.6, 13.1, 3.1) | ||||||
| 10 | 2.07 m | 24.8 | 1.52 (m) | 34.2 | 5.11 s | 78.3 | 79.0 |
| 2.19 m | 1.66 (m) | ||||||
| 11 | 5.02 (brd, 8.3) | 124.4 | 3.90 (dd, 7.6, 5.2) | 71.2 | 7.20 s | 150.7 | 151.6 |
| 12 | 136.9 | 154.5 | 131.2 | 131.1 | |||
| 13 | 1.80 m | 40.7 | 1.66 (m) | 34.1 | 1.92 m, 2.59 m | 20.8 | 20.1 |
| 2.19 m | 2.14 (ddd, 12.8, 12.8, 4.9) | ||||||
| 14 | 0.67 (td, 10.9, 4.0) | 31.5 | 1.13 (dddd, 13.4, 12.8, 11.9, 3.4) | 26.1 | 1.29 m, 1.75 m | 27.6 | 29.2 |
| 1.55 (dddd, 14.0, 13.4, 4.9, 2.7) | |||||||
| 15 | 20.1 | 20.1 | 145.8 | 145.4 | |||
| 16 | 1.00 s | 15.6 | 1.02 s | 15.5 | 4.67 s, 4.79 s | 113.1 | 113.0 |
| 17 | 1.06 s | 28.7 | 1.06 s | 29.1 | 1.64 s | 25.3 | 27.8 |
| 18 | 1.74 brs | 10.5 | 1.71 (d, 1.2) | 10.2 | 1.30 s | 18.1 | 18.4 |
| 19 | 4.89 brs | 109.8 | 1.63 (brs) | 17.1 | 174.0 | 174.4 | |
| 5.04 brs | |||||||
| 20 | 1.56 brs | 15.8 | 4.85 brs, | 108.6 | |||
| 5.02 brs | |||||||
© 2016 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons by Attribution (CC-BY) license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hegazy, M.-E.F.; Mohamed, T.A.; Elshamy, A.I.; Al-Hammady, M.A.; Ohta, S.; Paré, P.W. Casbane Diterpenes from Red Sea Coral Sinularia polydactyla. Molecules 2016, 21, 308. https://doi.org/10.3390/molecules21030308
Hegazy M-EF, Mohamed TA, Elshamy AI, Al-Hammady MA, Ohta S, Paré PW. Casbane Diterpenes from Red Sea Coral Sinularia polydactyla. Molecules. 2016; 21(3):308. https://doi.org/10.3390/molecules21030308
Chicago/Turabian StyleHegazy, Mohamed-Elamir F., Tarik A. Mohamed, Abdelsamed I. Elshamy, Montaser A. Al-Hammady, Shinji Ohta, and Paul W. Paré. 2016. "Casbane Diterpenes from Red Sea Coral Sinularia polydactyla" Molecules 21, no. 3: 308. https://doi.org/10.3390/molecules21030308











