MS-Based Metabolite Profiling of Aboveground and Root Components of Zingiber mioga and Officinale
Abstract
:1. Introduction
2. Results and Discussion
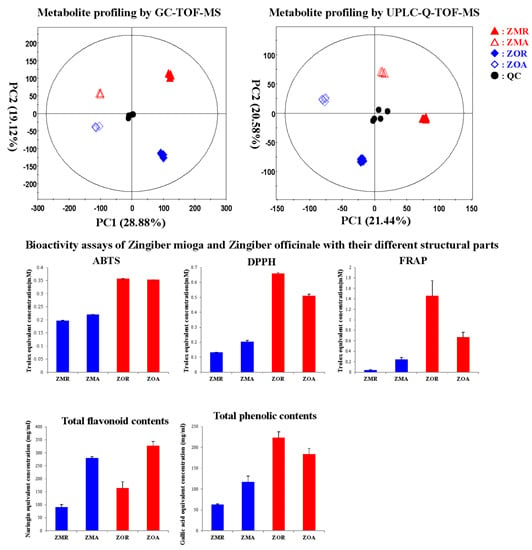
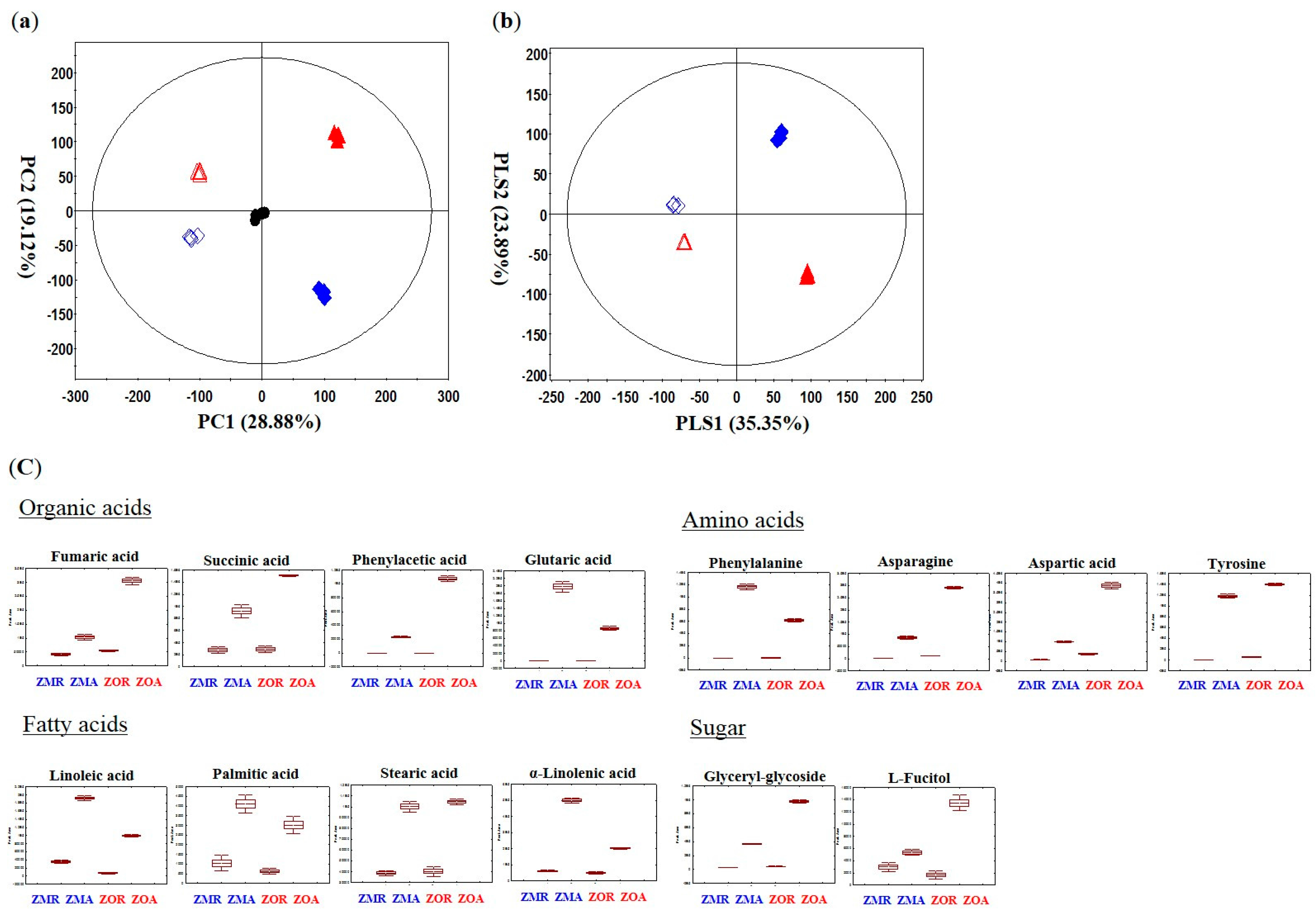
2.1. Primary Metabolites Discriminating Z. mioga and Z. officinale Analyzed by GC-TOF-MS
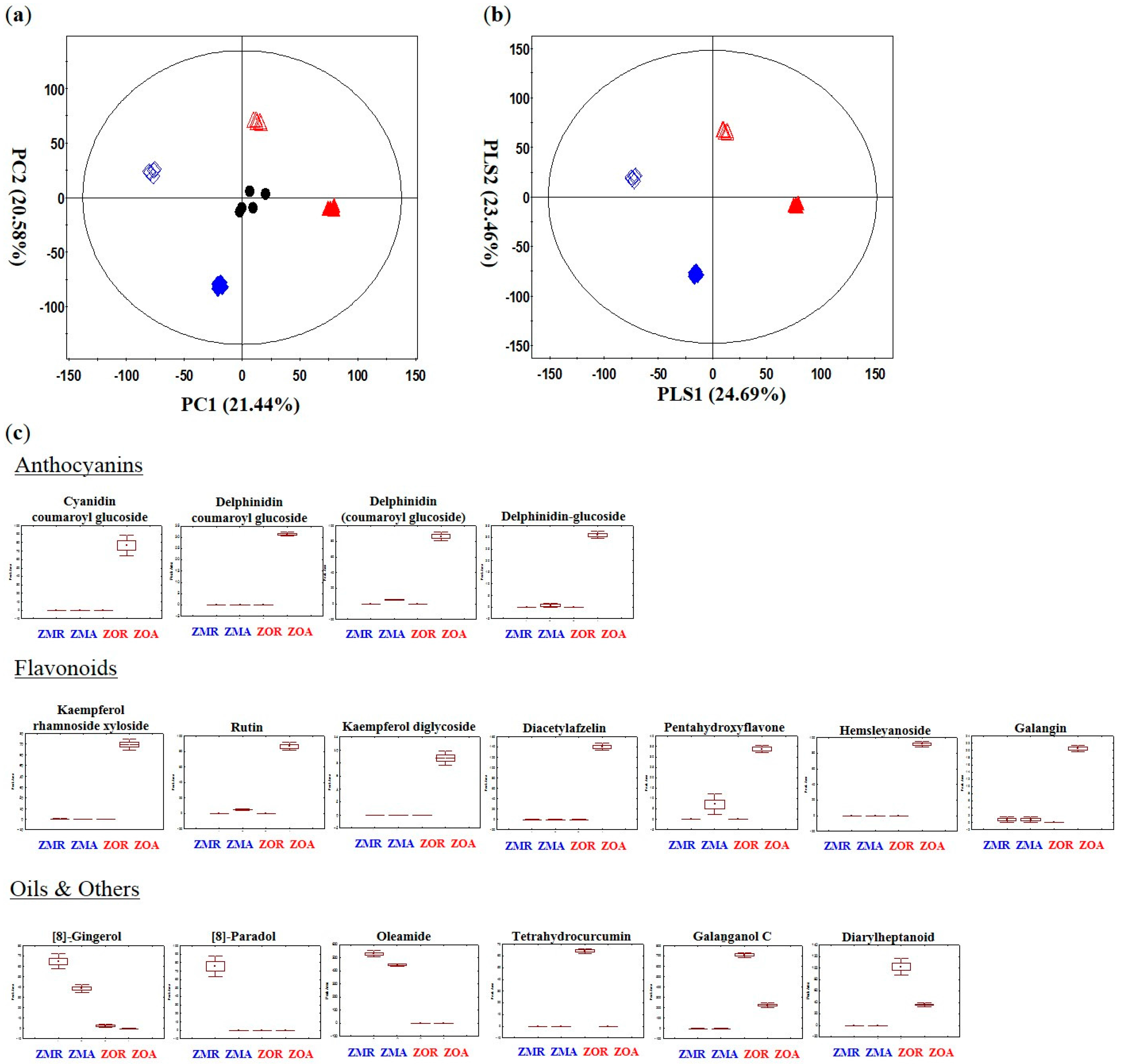
2.2. Secondary Metabolites Discriminating Z. mioga and Z. officinale Analyzed by UPLC-Q-TOF-MS
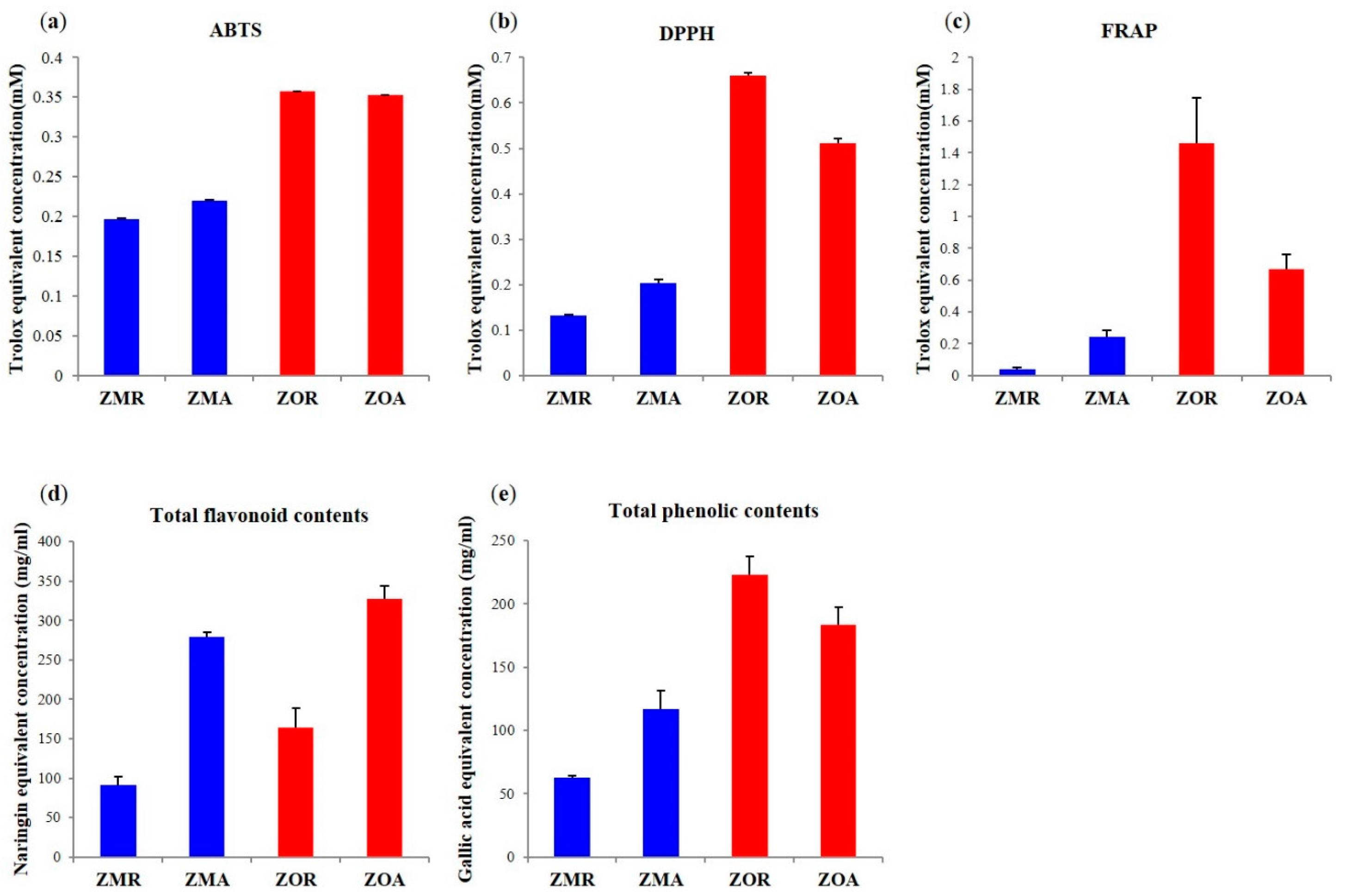
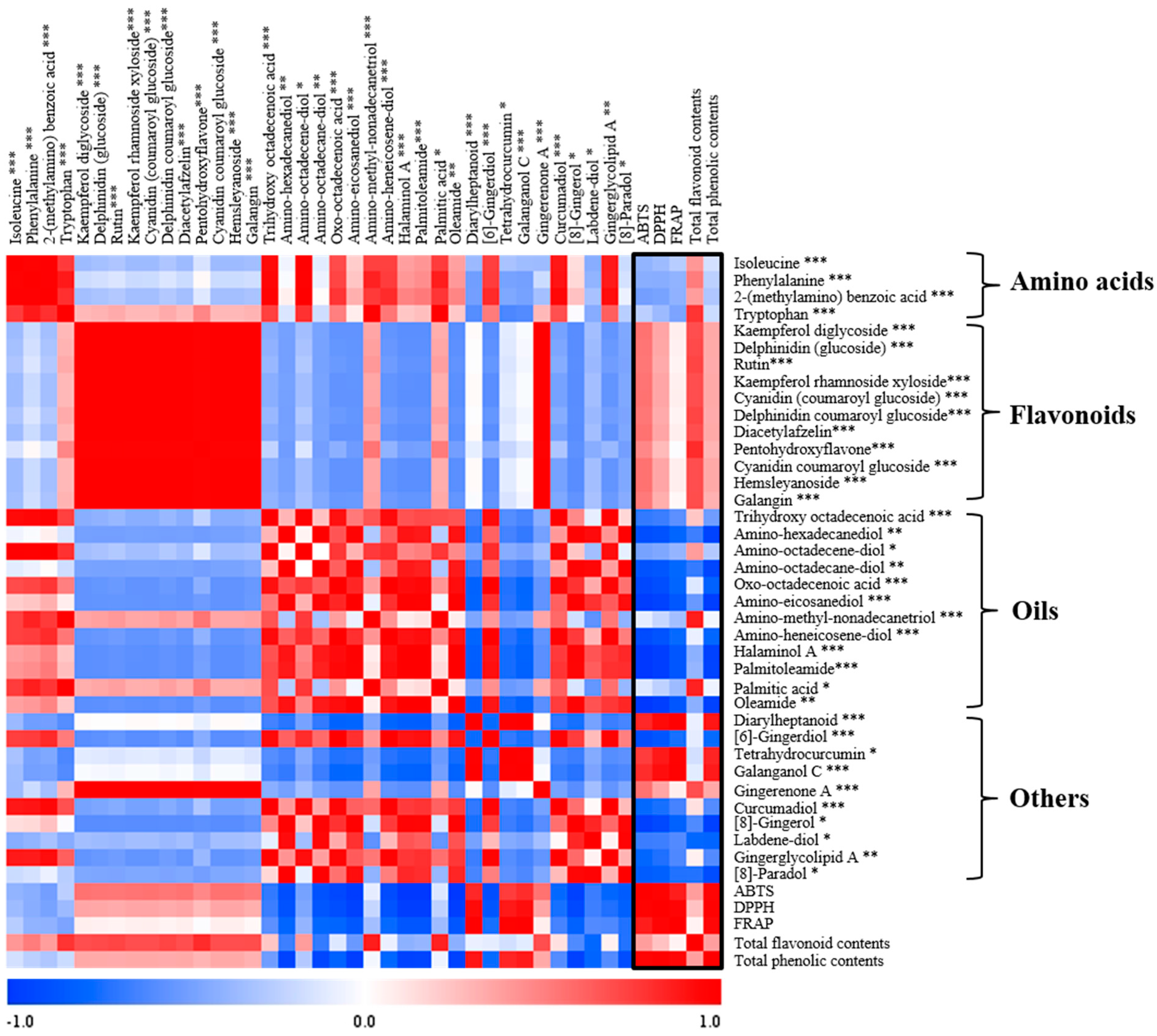
2.3. Correlation between the Significant Metabolites and Bioactivities
3. Experimental Section
3.1. Chemicals and Reagents
3.2. Plant Materials
3.3. Procedures for Preparation of Sample Solution
3.4. Instrumental Analysis
3.4.1. GC-TOF-MS Analysis
3.4.2. UPLC-Q-TOF-MS Analysis
3.5. Data Processing and Statistical Analysis
3.6. Determination of Antioxidant Activities by ABTS, DPPH, and FRAP Free Radical Scavenging Activity
3.7. Total Phenolic Content and Total Flavonoid Content
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- John, P.B.; Amanda, L.J. Pungent compounds of ginger (Zingiber officinale (L) Rosc) extracted by liquid carbon dioxide. J. Agr. Food Chem. 1986, 34, 477–480. [Google Scholar]
- Stoilova, I.; Krastanov, A.; Stoyanov, A.; Denev, P.; Gargova, S. Antioxidant activity of a ginger extract (Zingiber officinale). Food Chem. 2007, 102, 764–770. [Google Scholar] [CrossRef]
- Choi, S.K.; Suh, Y.N. Study on the clonal multiplication of Zingiber mioga Rosc. through in vitro culture of shoot apex.-(1)-Effects of basal media and growth regulators on plant regeneration and growth of plantlet. J. Korean Soc. Medical Crop Sci. 1993, 1, 38–42. [Google Scholar]
- Singh, G.; Kapoor, I.P.S.; Singh, P.; de Heluani, C.S.; de Lampasona, M.P.; Catalan, A.N.C. Chemistry, antioxidant and antimicrobial investigations on essential oil and oleoresins of Zingiber officinale. Food Chem. Toxicol. 2008, 46, 3295–3302. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, H.S.; Abbasi, K.Y.; Hammed, H.A.; Hussain, J. Survey and documentation of medicinal plants traditionally used for different aliments in district Lodhran, Punjab, Pakistan. Global J. Res. Med. Plants Indigen. Med. 2014, 3, 142–153. [Google Scholar]
- Shin, J.H.; Lee, S.J.; Sung, N.J. Effects of Zingiber mioga, Zingiber mioga Root and Zingiber officinale on the Lipid concentration in Hyperlipidimic Rats. J. Korean Soc. Food Sci. Nutr. 2002, 31, 679–684. [Google Scholar]
- Thomson, M.; Al-Qattan, K.K.; Al-Sawan, S.M.; Alanqeeb, M.A.; Khan, I.; Ali, M. The use of ginger (Zingiber officinale Rosc.) as a potential anti-inflammatory and antithrombotic agent. Prostaglandins Leukot. Essent. Fatty Acid. 2002, 67, 475–478. [Google Scholar] [CrossRef]
- Mahesh, P.; Shweta, S.; Anil, K.; Shri, T.K. Effects of elevated salinity of the growing medium on rhizome essential oil yield and fatty acid composition of Curcuma longa L. (Zingiberaceae). Facta Univ.-Ser.: Phys. Chem. Tech. 2012, 10, 45–50. [Google Scholar]
- Srivastava, N.K.; Sharma, S.; Misra, A. Influence of Zn on allocation of Leaf-assimilated 14CO2 into Primary metabolites in relation to production of essential oil and curcumin in Tumeric. J. Agr. Sci. 2006, 2, 201–207. [Google Scholar]
- Sang, S.; Hong, J.; Wu, H.; Yang, C.S.; Pan, M.H.; Badmaev, V.; Ho, C.T. Increased growth inhibitory effects on human cancer cells and anti-inflammatory potency of shogaols from Zingiber officinale relative to gingerols. J. Agric. Food Chem. 2009, 57, 10645–10650. [Google Scholar] [CrossRef] [PubMed]
- Dugasani, S.; Pichika, M.R.; Nadarajah, V.D.; Balijepalli, M.K.; Tandra, S.; Korlakunta, J.N. Comparative antioxidant and anti-inflammatory effects of [6]-gingerol, [8]-gingerol, [10]-gingerol and [6]-shogaol. J. Ethnopharmacol. 2010, 127, 515–520. [Google Scholar] [CrossRef] [PubMed]
- Chung, W.Y.; Jung, Y.J.; Surh, Y.J.; Lee, S.S.; Park, K.K. Antioxidative and antitumor promoting effects of [6]-paradol and its homologs. Mutat. Res. Gen. Tox. Enciron. Mutagen. 2001, 496, 199–206. [Google Scholar] [CrossRef]
- Connel, D.W. “Natural pungent compounds. III. The paradols and associated compounds”. Aust. J. Chem. 1970, 23, 369–372. [Google Scholar] [CrossRef]
- Mendes, P. Metabolomics and the challenges ahead. Brief. Bioinform. 2006, 7, 127. [Google Scholar] [CrossRef] [PubMed]
- Lee, S.M.; Seo, M.H.; Oh, D.K.; Lee, C.H. Targeted metabolomics for Aspergillus oryzae-mediated biotransformation of soybean isoflavones, showing variations in primary metabolites. Biosci. Biotechnol. Biochem. 2014, 78, 167–174. [Google Scholar] [CrossRef] [PubMed]
- Kim, Y.M.; Lee, J.; Park, S.H.; Lee, C.; Lee, J.W.; Lee, D.; Lee, C.H. LC-MS-based chemotaxonomic classification of wild-type Lespedeza sp. and its correlation with genotype. Plant Cell Rep. 2012, 31, 2085–2097. [Google Scholar] [CrossRef] [PubMed]
- Ku, K.M.; Choi, J.N.; Kim, J.; Kim, J.K.; Yoo, L.G.; Lee, S.J.; Lee, C.H. Metabolomics analysis reveals the compositional differences of shade grown tea (Camellia sinensis L.). J. Agr. Food Chem. 2009, 58, 418–426. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.Y.; Lee, M.Y.; Park, H.M.; Park, Y.K.; Shon, J.C.; Liu, K.H.; Lee, C.H. Urine and Serum Metabolite Profiling of Rats Fed a High-Fat Diet and the Anti-Obesity Effects of Caffeine Consumption. Molecules 2015, 20, 3107–3128. [Google Scholar] [CrossRef] [PubMed]
- Herebian, D.; Choi, J.H.; Abd El-Aty, A.M.; Shim, J.H.; Spiteller, M. Metabolite anaylsis in Curcuma domestica using GC-MS and LC-MS separation and detection techniques. Biomed. Chromatogr. 2009, 23, 951–965. [Google Scholar] [CrossRef] [PubMed]
- Gabriel, R.; Kesselmeier, J. Apoplastic solute concentrations of organic acids and mineral nutrients in the leaves of several Fagaceae. Jpn. Soc. Plant Physiol. 1999, 40, 604–612. [Google Scholar] [CrossRef]
- Lopez-Bucio, J.; Nieto-Jacobo, M.F.; Ramırez-Rodrıguez, V.; Herrera-Estrella, L. Organic acid metabolism in plants: from adaptive physiology to transgenic varieties for cultivation in extreme soils. Plant Sci. 2000, 160, 1–13. [Google Scholar] [CrossRef]
- Chia, D.W.; Yoder, T.J.; Reiter, W.D.; Gibson, S.I. Fumaric acid: An overlooked form of fixed carbon in Arabidopsis and other plant species. Planta 2000, 211, 743–751. [Google Scholar] [CrossRef] [PubMed]
- Steward, G.R.; Larher, F. Accumulation of amino acids and related compounds in relation to environmental stress. Amino acids and derivatives. In Biochemistry of Plants, 1st ed.; Paul, K.S., Eric, E.C., Eds.; Academic Press: New York, NY, USA, 1980; Volume 5, pp. 609–635. [Google Scholar]
- Chaturvedi, P.; Soundar, S.; Parekh, K.; Lokhande, S.; Chowdhary, A. Media optimization in immobilized culture to enhance the content of Kaempferol in Tylophora indica (Asclepeadaceae) and Curcumin in Curcuma longa (Zingiberaceae). Int. J. Pharm. Bio. Sci. 2014, 9, 86–90. [Google Scholar]
- Brouquisse, R.; James, F.; Pradet, A.; Raymond, P. Asparagine metabolism and nitrogen distribution during protein degradation in sugar-starved maize root tips. Planta 1992, 188, 384–395. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro, C.; Passarinho, J.A.; Ricardo, C.P. Effect of drought and rewatering on the metabolism of Lupinus albus organs. J. Plant Physiol. 2004, 161, 1203–1210. [Google Scholar] [CrossRef] [PubMed]
- Ajayi, O.B.; Akomolafe, S.F.; Akinyemi, F.T. Food value of two varieties of ginger (Zingiber officinale) commonly consumed in Nigeria. ISRN Nutr. 2013, 2013. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.Y.; Chapkin, R.S. Importance of dietary γ-linolenic acid in human health and nutrition. J. Nutr. 1998, 128, 1411–1414. [Google Scholar] [PubMed]
- Howe, G.A.; Schilmiller, A.L. Oxylipin metabolism in response to stress. Curr. Opin. Plant Biol. 2002, 5, 230–236. [Google Scholar] [CrossRef]
- Amadi, B.A.; Duru, M.K.C.; Agomuo, E.N. Chemical profiles of leaf, stem, root and flower of Ageratum conyzoides. Asian J. Plant Sci. Res. 2012, 2, 428–432. [Google Scholar]
- Olaofe, O.; Adeyeye, E.L.; Ojungbo, S. Comparative study of proximate, amino acids and fatty acids of Moringa oleifera tree. Elixir. Appl. Chem. 2013, 54, 12543–12554. [Google Scholar]
- Ludwig, L.J.; Charles-Edwards, D.D.; Withers, A.C. Tomato leaf photosynthesis and respiration in various light and carbon dioxide environments. In Environmental and Biological Control of Photosynthesis; Springer Netherland: Dordrecht, The Netherlands, 1975; pp. 29–36. [Google Scholar]
- Williams, C.A.; Harborne, J.B. The leaf flavonoids of the Zingiberales. Biochem. Syst. Ecol. 1977, 5, 221–229. [Google Scholar] [CrossRef]
- Li, X.; Yao, S.; Tu, B.; Li, X.; Jia, C.; Song, H. Determination and comparison of flavonoids and anthocyanins in Chinese sugarcane tips, stems, roots and leaves. J. Sep. Sci. 2010, 33, 1216–1223. [Google Scholar] [CrossRef] [PubMed]
- Ferreyra, M.L.F.; Rius, S.P.; Casati, P. Flavonoids: Biosynthesis, biological functions, and biotechnological applications. Front. Plant Sci. 2012, 3. [Google Scholar] [CrossRef]
- Xie, B.D.; Wang, H.T. Effects of light spectrum and photoperiod on contents of flavonoid and terpene in leaves of Ginkgo biloba L. Nanjing For. Univ. 2006, 30, 51–54. [Google Scholar]
- Harris, C.S.; Burt, A.J.; Saleem, A.; Le, P.M.; Martineau, L.C.; Haddad, P.S.; Bennett, S.A.L.; Arnason, J.T. A Single HPLC-PAD-APCI/MS method for the quantitative comparison of phenolic compounds found in leaf, stem, root and fruit extracts of Vaccinium angustifolium. Phytochem. Anal. 2007, 18, 161–169. [Google Scholar] [CrossRef] [PubMed]
- Yoshikawa, M.; Yamaguchi, S.; Kunimi, K.; Matsuda, H.; Okuno, Y.; Yamahara, J.; Murakami, N. Stomachic principles in ginger. III. An anti-ulcer principle, 6-gingesulfonic acid, and three monoacyldigalactosylglycerols, gingerglycolipids A, B, and C, from Zingiberis rhizome originating in Taiwan. Chem. Pharm. Bull. 1994, 42, 1226–1230. [Google Scholar] [CrossRef] [PubMed]
- Yoysungnoen, P.; Wirachwong, P.; Changtam, C.; Suksamrarn, A.; Patumraj, S. Anti-cancer and anti-angiogenic effects of curcumin and tetrahydrocurcumin on implanted hepatocellular carcinoma in nude mice. World J. Gastroentero. 2008, 14, 2003–2009. [Google Scholar] [CrossRef]
- Park, M.; Bae, J.; Lee, D.S. Antibacterial activity of [10]-gingerol and [12]-gingerol isolated from ginger rhizome against periodontal bacteria. Phytother. Res. 2008, 22, 1446–1449. [Google Scholar] [CrossRef] [PubMed]
- Del Carmen Ramirez-Ahumada, M.; Timmermann, B.N.; Gang, D.R. Biosynthesis of curcuminoids and gingerols in turmeric (Curcuma longa) and ginger (Zingiber officinale): identification of curcuminoid synthase and hydroxycinnamoyl-CoA thioesterases. Phytochemistry 2006, 67, 2017–2029. [Google Scholar] [CrossRef] [PubMed]
- Kim, D.O.; Jeond, S.W.; Lee, C.Y. Antioxidant capacity of phenolic phytochemicals from various cultivars of plums. Food. Chem. 2003, 81, 321–326. [Google Scholar] [CrossRef]
- Chan, E.W.C.; Tan, Y.P.; Chin, S.J.; Gan, L.Y.; Kang, K.X.; Fong, C.H.; How, Y.C. Antioxidant properties of selected fresh and processed herbs and vegetables. Free Radicals Antiox. 2014, 4, 39–46. [Google Scholar] [CrossRef]
- Gould, K.S.; Mckelvie, J.; Markham, K.R. Do anthocyanins. Plant Cell Enviorn. 2002, 25, 1261–1269. [Google Scholar] [CrossRef]
- Li, N.; Wang, L.; Zu, L.; Wang, K.; Di, L.; Wang, Z. Antioxidant and Cytotoxic Diarylheptanoids Isolated from Zingiber officinale Rhizomes. Chin. J. Chem. 2012, 30, 1351–1355. [Google Scholar] [CrossRef]
- Manjunatha, J.R.; Bettadaiah, B.K.; Negi, P.S.; Srinivas, P. Synthesis of quinoline derivatives of tetrahydrocurcumin and zingerone and evaluation of their antioxidant and antibacterial attributes. Food Chem. 2013, 136, 650–658. [Google Scholar] [CrossRef] [PubMed]
- Re, R.; Pellegrini, N.; Proteggente, A.; Pannala, A.; Yang, M.; Rice-Evans, C. Antioxidanat activity applying an improved ABTS radical cation decolorization assay. Free Radic. Biol. Med. 1999, 26, 1231–1237. [Google Scholar] [CrossRef]
- Dietz, B.M.; Kang, Y.H.; Liu, G.; Eggler, A.L.; Yao, P.; Chadwick, L.R.; Pauli, G.F.; Farnsworth, N.R.; Mesecar, A.D.; van Breemen, R.B.; et al. Xanthohumol isolated from Humulus lupulus inhibits menadione-induced DNA damage through induction of quinone reductase. Chem. Res. Toxicol. 2005, 18, 1296–1305. [Google Scholar] [PubMed]
- Benzie, I.F.F.; Strain, J.J. The ferric reducing ability if plasma (FRAP) as a measure of “antioxidant power”: The FRAP assay. Anal. Biochem. 1996, 239, 70–76. [Google Scholar] [CrossRef] [PubMed]
- Singleton, V.L.; Orthofer, R.; Lamuela-Raventos, R.M. Analysis of total phenols and other oxidation substrates and antioxidants by means of folin-ciocalteu reagent. Methods Enzymol. 1999, 299, 152–178. [Google Scholar]
- Sample Availability: No available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, J.S.; Lee, S.; Kim, H.Y.; Lee, C.H. MS-Based Metabolite Profiling of Aboveground and Root Components of Zingiber mioga and Officinale. Molecules 2015, 20, 16170-16185. https://doi.org/10.3390/molecules200916170
Han JS, Lee S, Kim HY, Lee CH. MS-Based Metabolite Profiling of Aboveground and Root Components of Zingiber mioga and Officinale. Molecules. 2015; 20(9):16170-16185. https://doi.org/10.3390/molecules200916170
Chicago/Turabian StyleHan, Ji Soo, Sunmin Lee, Hyang Yeon Kim, and Choong Hwan Lee. 2015. "MS-Based Metabolite Profiling of Aboveground and Root Components of Zingiber mioga and Officinale" Molecules 20, no. 9: 16170-16185. https://doi.org/10.3390/molecules200916170