Management of Hyperglycaemia by Ethyl Acetate Extract of Balanites aegyptiaca (Desert Date)
Abstract
:1. Introduction
2. Results
| Name of Compound | MW | Peak Area in % | RT | S. No. |
|---|---|---|---|---|
| Vanillic acid | 312 | 26.58 | 15.02 | 1 |
| unknown | -- | 8.340 | 15.30 | 2 |
| unknown | -- | 2.220 | 15.41 | 3 |
| unknown | -- | 3.210 | 15.56 | 4 |
| unknown | -- | 2.870 | 15.73 | 5 |
| Syringic acid | 342 | 24.08 | 18.04 | 6 |
| β-sitosterol | 414 | 23.94 | 24.50 | 7 |
| Groups | HA1C (%) | Glucose (mg/dL) | TNF-α (ng/dL) | IL-1β (ng/dL) |
|---|---|---|---|---|
| Control Mean ± SD | 4.2 ± 0.51 | 92.5 ± 3.8 | 0.13 ± 0.013 | 220 ± 38 |
| Diabetic Mean ± SD | 7.4 ± 0.82 a,b | 340 ± 15.8 a,b | 2.54 ± 0.122 a,b | 902 ± 56 a,b |
| Diabetic+ BE (10 mg) Mean ± SD | 6.6 ± 0.63 a,b | 280 ± 13 a,b | 0.92 ± 0.05 a,b | 637 ± 25 a,b |
| Diabetic+ BE (25 mg) Mean ± SD | 5.8 ± 0.55 a,b | 240 ± 10.7 a,b | 0.82 ± 0.05 a,b | 430 ± 33 a,b |
| Diabetic+ BE (50 mg) Mean ± SD | 5.7 ± 0.60 a,b | 140 ± 2.1 a | 0.88 ± 0.04 a,b | 312 ± 21 a |
| Animal Groups | Control | Diabetic | Dia+EAE (10 µg) | Dia+EAE (25 µg) | Dia+EAE (50 µg) |
|---|---|---|---|---|---|
| MDA(nmol/mg protein) Mean ± SD | 3.5 ± 0.1 | 13 ± 1.6 a | 9 ± 1.1 a,b | 5.0 ± 0.8 a,b | 4.5 ± 0.5 a,b |
| GSH( nmol/mg protein) Mean ± SD | 88 ± 13.2 | 43 ± 2.8 a | 45 ± 6.0 a,b | 52 ± 3 a,b | 58 ± 8 a,b |
| GST( nmol/mg protein) Mean ± SD | 19 ± 1.3 | 4 ± 0.8 a | 10 ± 1.0 a,b | 11 ± 1.0 a,b | 16 ± 2.0 a |
| SOD (U/ mg protein) Mean ± SD | 17.8 ± 1.6 | 3.1 ± 1.2 a | 10.97 ± 1.5 a,b | 11.2 ± 1.3 a,b | 14.25 ± 1 a |
| GSH-Px( U /mg protein) Mean ± SD | 49.5 ± 4.14 | 21 ± 4.1 a | 31.12 ± 7.5 a,b | 29 ± 6.1 a,b | 36 ± 9.2 a,b |
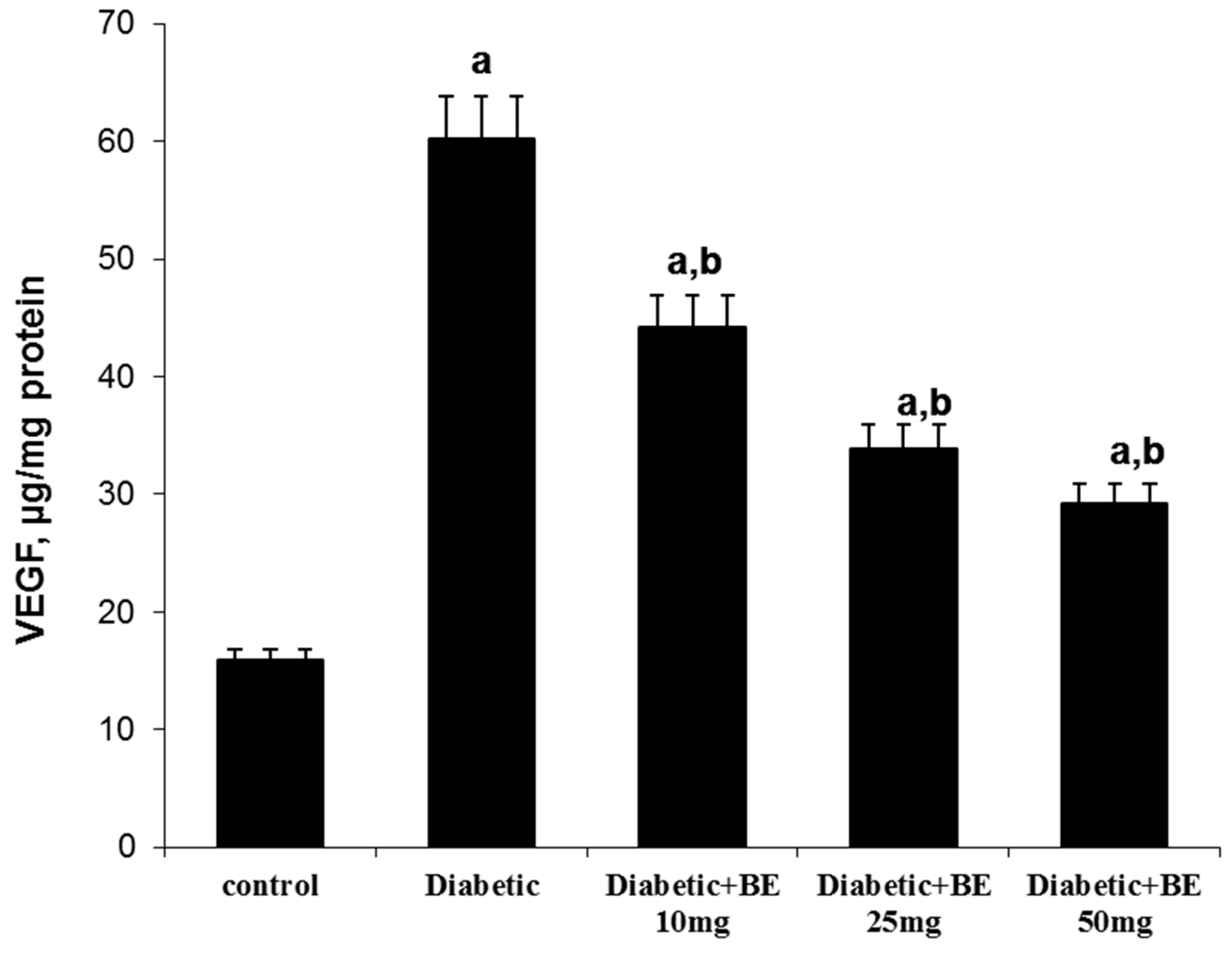
3. Discussion
4. Experimental Section
4.1. Extraction of Balanites aegyptiaca
4.2. Gas Chromatography-Mass Spectrometry Analysis (GC-MS)
4.3. Animal Experimental Design
4.4. Determination of Protein in the Retina
4.5. Determination of Malondialdhyde (MDA)
4.6. Determination of Reduced (GSH)
4.7. Assay of Superoxide Dismutase (SOD) Activity
4.8. Determination of Glutathione Peroxidase
4.9. Determination of VEGF
4.10. Statistical Analysis
Acknowledgment
Author Contributions
References
- Beit-Yannai, E.; Ben-Shabat, S.; Goldschmidt, N.; Chapagain, B.P.; Liu, R.H.; Wiesman, Z. Anti-proliferative activity of steroidal saponins from Balanites aegyptiaca—An in vitro study. Phytochem. Lett. 2011, 4, 43–47. [Google Scholar]
- Gnoula, C.; Megaliz, Z.V.; Neve, N.D.; Sauvage, S.; Ribaucour, F.; Guissou, P.; Duez, P.; Dubois, J.; Ingrassia, L.; Lefranc, F.; et al. Balanitin-6 and -7: DiosgenylSaponins Isolated from Balanites aegyptiaca Del. Display Significant Anti-Tumor Activity in Vitro and in Vivo. Int. J. Oncol. 2008, 32, 5–15. [Google Scholar]
- Hanan, A.A.; Ayman, A.; Farghaly, M.M.; Abd, E.A.; Ali, M.A. Phytochemical investigation and medicinal evaluation of fixed oil of Balanitesaegyptiacafruits (Balantiaceae). J. Ethnopharmacol. 2010, 127, 495–501. [Google Scholar]
- Jung, H.J.; Lee, C.O.; Lee, K.T.; Choi, J.; Park, H.J. Structure-Activity Relationship of Oleanane Disaccharides Isolated from Akebiaquinata vs. Cytotoxicity against Cancer Cells and NO Inhibition. Biol. Pharm. Bull. 2004, 27, 744–747. [Google Scholar] [PubMed]
- Podolak, I.; Galanty, A.; Sobolewska, D. Saponins as cytotoxic agents: A review. Phytochem. Rev. 2010, 9, 425–474. [Google Scholar] [PubMed]
- James, K. Understanding NMR Spectroscopy; University of California: Irvine, CA, USA, 2007. [Google Scholar]
- Kerwin, S.M. Soy saponins and the anticancer effects of soybeans and soy-based foods. Curr. Med. Chem. Anticancer Agents 2004, 4, 263–272. [Google Scholar] [PubMed]
- Khedr, M.G.A.; Badran, M.M.H.; El Azhari, A.A. Pitting corrosion behaviour of aluminium in water desalination plants. Desalination 1989, 72, 351–366. [Google Scholar]
- Koko, W.S.; Abdalla, H.S.; Galal, M.; Khalid, H.S. Evaluation of oral therapy on MansonialShistosomiasis using single dose of Balanitesaygyptiaca fruits and praziquantel. Fitoterapia 2005, 76, 30–34. [Google Scholar] [CrossRef] [PubMed]
- Koko, W.S.; Galal, M.; Khalid, H.S. Fasciolicidal efficacy of Albiziaanthelmintica and Balanitesaegyptiaca compared with albendazole. J. Ethnopharmacol. 2005, 71, 247–252. [Google Scholar] [CrossRef]
- Karel, L.; Roach, E.S. A Dictionary of Antibiosis; Columbia University Press: New York, NY, USA, 1951; p. 48. [Google Scholar]
- Watt, J.M.; Breyer-Brandwijk, M.G. The Medicinal and Poisonous Plants of South and East Africa; E. and S. Livingstone: Ann Arbor, MI, USA, 1962; pp. 1064–1065. [Google Scholar]
- Beentje, H.; Adamson, J.; Bhanderi, D. Kenya Trees, Shrubs, and Lianas; National Museums of Kenya: Nairobi, Kenya, 1994; p. 378. [Google Scholar]
- Khare, C.P. Indian Medicinal Plants: An Illustrated Dictionary; Springer: New York, NY, USA, 2007; pp. 77–78. [Google Scholar]
- Jagtap, S.D.; Deokule, S.S.; Pawar, P.K.; Harsulkar, A.M. Traditional Ethnomedicinal Knowledge Confined to the Pawra Tribe of Satpura Hills, Maharashtra, India. Ethnobot. Leaf. 2009, 13, 98–115. [Google Scholar]
- Al-Ghannam, S.M.; Ahmed, H.H.; Zein, N.; Zahran, F. Antitumor Activity of Balanitoside Extracted from Balanites aegyptiaca Fruit. J. Appl. Pharm. Sci. 2013, 3, 179–191. [Google Scholar]
- George, D.H.; Ali, H.K.; El Abbas, O.A. Evaluation of the biological activity of Balanites aegyptiaca Del Saponin in the control of type 11 diabetes mellitus on rats and the growth of Escherichia coli. Ahfad J. Women Change 2006, 23, 2. Available online: http://findarticles.com/p/articles/mi_hb003/is_2_23/ai_n29364027 (access on 29 June 2015). [Google Scholar]
- Mansour, H.A.; Newairy, A.A. Amelioration of impaired renal function associated with diabetes by Balanitesaegyptiaca fruits in streptozotocin-induced diabetic rats. J. Med. Res. Inst. 2000, 21, 115–125. [Google Scholar]
- Hosny, M.; Khalifa, T.; Calis, I.; Wright, A.D.; Sticher, O. Balanitoside: A furostanol glycoside, and 6-methyldiosgenin from Balanites aegyptiaca. Phytochemistry 1992, 31, 3565–3569. [Google Scholar] [CrossRef]
- Araya, Y.N. Contribution of trees for oral hygiene in East Africa. Ethnobot. Leafl. 2007, 11, 38–44. [Google Scholar]
- Yagi, K. Lipid peroxides and human diseases. Chem. Phys. Lipids 1987, 45, 337–351. [Google Scholar] [CrossRef]
- Ojo, O.O.; Nadro, M.S.; Tella, I.O. Protection of rats by extracts of some common Nigerian trees against acetaminophen-inducedhepatotoxicity. Afr. J. Biotechnol. 2006, 5, 755–760. [Google Scholar]
- Barley, S. Zygophyllaceae. In The Medicinal and poisonous plants of Southern and Eastern Africa; Watt, J.M., Breyer-Brandwijk, M.G., Eds.; Livingstone Ltd: London, UK, 1962; p. 1064. [Google Scholar]
- Zarroug, I.M.A.; Nugud, A.D.; Bashir, A.K.; Mageed, A.A. Evaluation of Sudanese plant extracts as mosquito larvicides. Int. Sci. Crude Drug Res. 1988, 26, 71–76. [Google Scholar] [CrossRef]
- Bashir, A.K.; Ahmed, G.H.M.; Suliman, S.M.; ElKheir, Y.M. Mollus-cicidal and other Biological activities of B. aegyptiaca. In The First Arab Conference on Medicinal Plants; Cairo Press: Cairo, Egypt, 1984; pp. 30–34. [Google Scholar]
- Doughari, J.M.; Pukuma, M.S.; De, N. Antibacterial effects of Balanitesaegyptiaca L. Del. and Moringaoleifera Lam. on Salmonella typhi. Afr. J. Biotechnol. 2007, 6, 2212–2215. [Google Scholar]
- Khan, F.M. Ethno-veterinary medicinal usage of flora of greater cholistan desert (Pakistan). Pak. Vet. J. 2009, 29, 75–80. [Google Scholar]
- Kokwano, J.O. Medicinal Plants in East Africa; University of Nairobi Press: Nairobi City, Kenya, 1976; p. 34. [Google Scholar]
- Kamel, M.S.; Ohtani, K.; Kurokawa, T.; Assaf, M.H.; El-Shanawany, M.A.; Ali, A.A.; Kasai, R.; Ishibashi, S.; Tanaka, O. Studies on Balanitesaegyptiaca fruits, an antidiabetic Egyptian folk medicine. Chem. Pharm. Bull. 1991, 39, 1229–1233. [Google Scholar] [CrossRef] [PubMed]
- Creach, P. Le Balanitesaegyptiaca. Ses multiples applications au Tchad; Revue de Botanique appliqué d′AgricultureTropicale. 1940, 20, 578–593. [Google Scholar]
- Okhawa, H.; Ohishi, N.; Yagi, K. Assay for lipid peroxides in animal tissue by thiobarbituric acid. Anal. Biochem. 1979, 95, 355–358. [Google Scholar]
- Moron, M.S.; Depierre, J.W.; Mannervik, B. Levels of glutathione, glutathione reductase and glutathione S-transferase activities in rat lung and liver. Biochim. Biophys. Acta 1979, 67, 582–589. [Google Scholar] [CrossRef]
- Paglia, D.E.; Valentine, W.N. Studies on the quantitative and qualitative characterisation of erythrocyte glutathione peroxidase. J. Lab. Clin. Med. 1967, 70, 158–169. [Google Scholar] [PubMed]
- Sample Availability: Not available.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al-Malki, A.L.; Barbour, E.K.; Abulnaja, K.O.; Moselhy, S.S. Management of Hyperglycaemia by Ethyl Acetate Extract of Balanites aegyptiaca (Desert Date). Molecules 2015, 20, 14425-14434. https://doi.org/10.3390/molecules200814425
Al-Malki AL, Barbour EK, Abulnaja KO, Moselhy SS. Management of Hyperglycaemia by Ethyl Acetate Extract of Balanites aegyptiaca (Desert Date). Molecules. 2015; 20(8):14425-14434. https://doi.org/10.3390/molecules200814425
Chicago/Turabian StyleAl-Malki, Abdulrahman L., Elie K. Barbour, Kalid O. Abulnaja, and Said S. Moselhy. 2015. "Management of Hyperglycaemia by Ethyl Acetate Extract of Balanites aegyptiaca (Desert Date)" Molecules 20, no. 8: 14425-14434. https://doi.org/10.3390/molecules200814425





