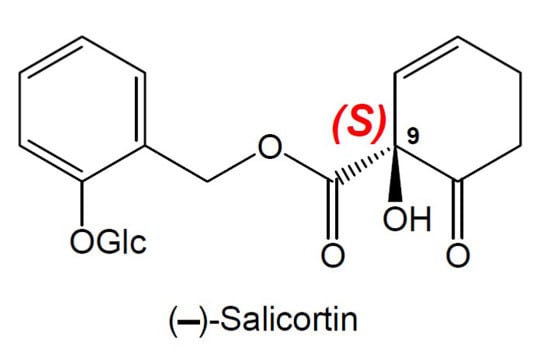
The Absolute Configuration of Salicortin, HCH-Salicortin and Tremulacin from Populus trichocarpa × deltoides Beaupré
Abstract
:1. Introduction
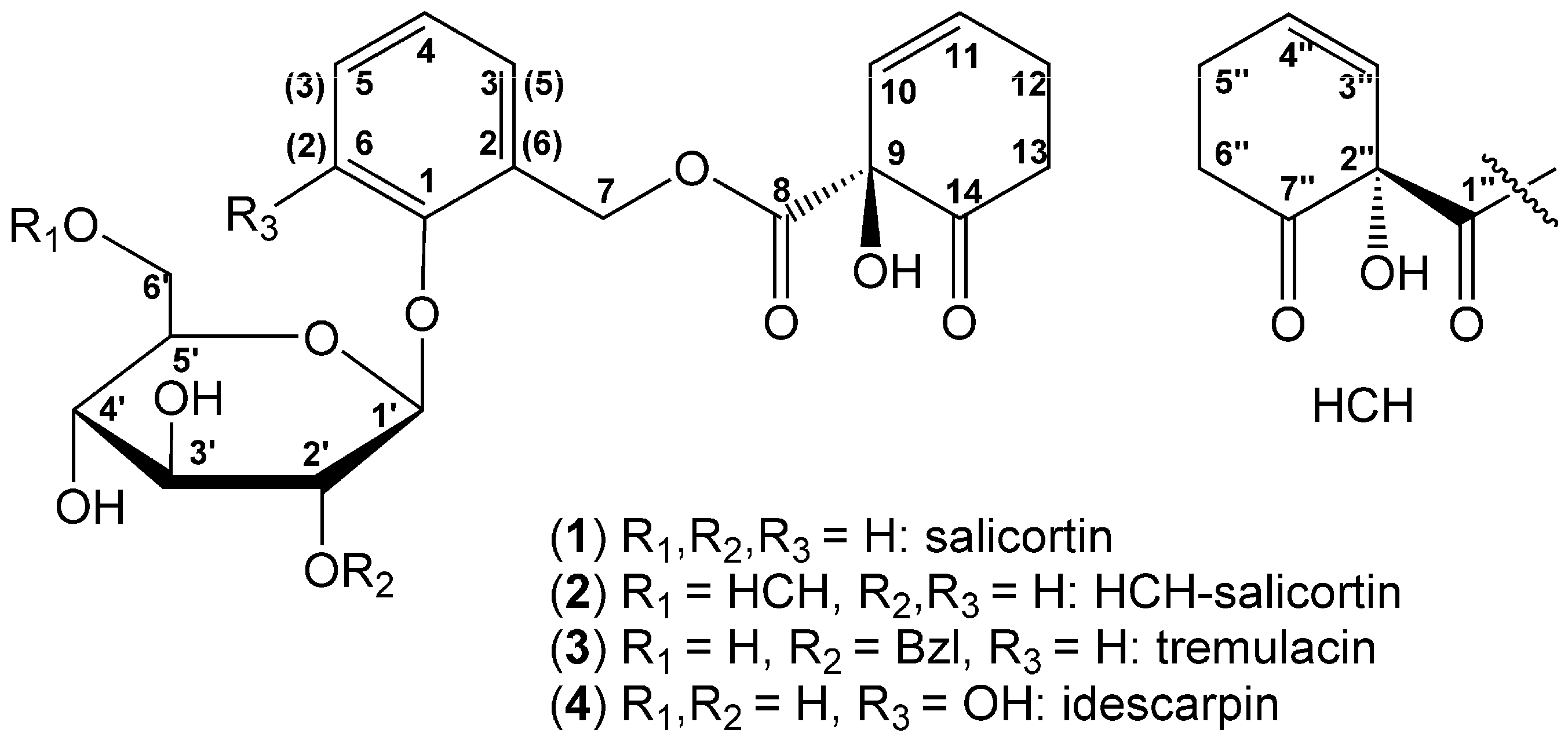
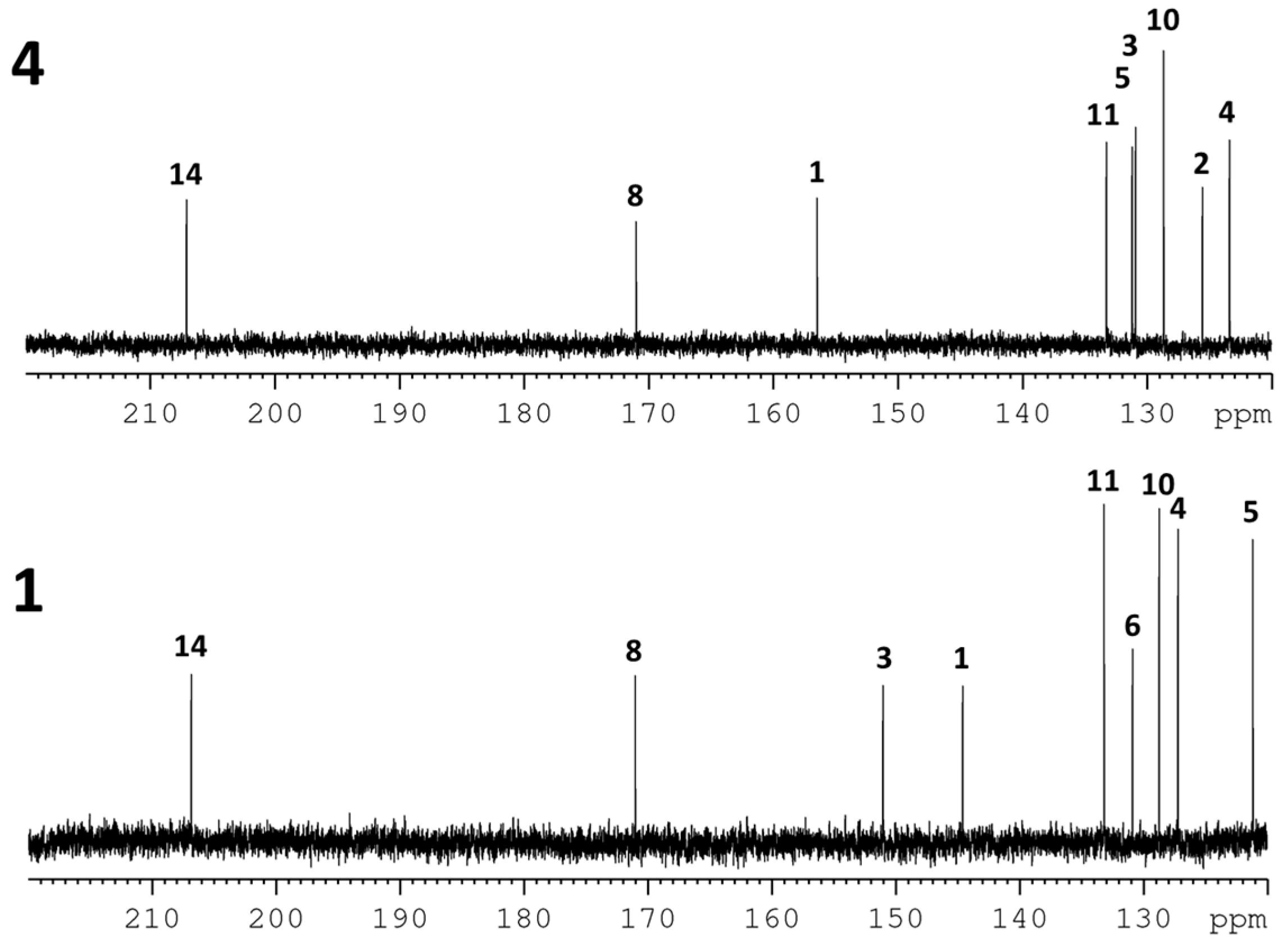
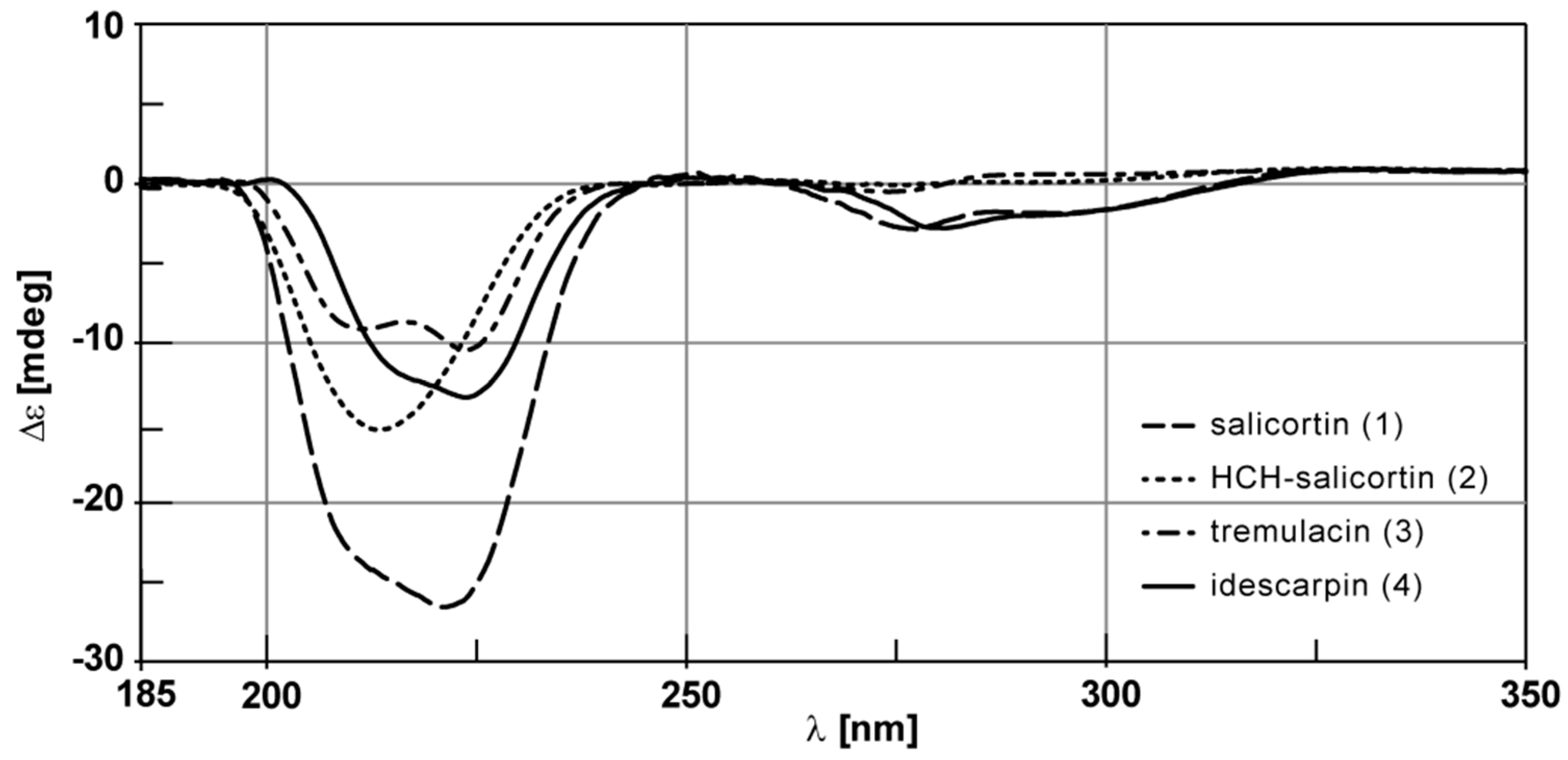
2. Results and Discussion

| Pos. | Salicortin (1) | Tremulacin (2) | HCH-salicortin (3) | Idescarpin (4) (1) | ||||
|---|---|---|---|---|---|---|---|---|
| δH, mult., J (Hz) | δC | δH, mult., J (Hz) | δC | δH, mult., J (Hz) | δC | δH, mult., J (Hz) | δC | |
| 1 | 156.3 | 155.8 | 156.3 | 144.4 | ||||
| 2 | 125.4 | 125.6 | 126.0 | 150.9 | ||||
| 3 | 7.30, dd, 7.5, 1.0 | 130.7 | 7.20, d, 7.6 | 130.0 | 7.35, d, 7.5 | 130.6 | 6.92, dd, 7.6, 1.2 | 118.3 |
| 4 | 7.05, ddd, 7.5, 7.5, 1.0 | 123.2 | 7.03, dd, 7.6, 7.6 | 123.6 | 7.11, dd, 7.5, 7.5 | 123.2 | 7.07, dd, 7.6, 7.6 | 127.0 |
| 5 | 7.33, ddd, 7.5, 7.5, 1.0 | 131.0 | 7.31, dd, 7.6, 7.6 | 130.7 | 7.41, dd, 7.5, 7.5 | 131.0 | 6.82, dd, 7.6, 1.2 | 121.0 |
| 6 | 7.15, dd, 7.5, 1.0 | 116.1 | 7.17, d, 7.6 | 116.2 | 7.13, d, 7.5 | 116.0 | 130.7 | |
| 7a | 5.30, d, 12.3 | 64.2 | 5.02, d, 12.5 | 63.5 | 5.30, d, 12.3 | 64.0 | 5.45, d, 12.6 | 64.5 |
| 7b | 5.26, d, 12.3 | 4.78, d, 12.5 | 5.26, d, 12.3 | 5.23, d, 12.6 | ||||
| 8 | 170.8 | 170.4 | 170.3 | 170.9 | ||||
| 9 | 78.9 | 78.7 | 78.7 | 79.0 | ||||
| 10 | 5.74, ddd, 9.9, 1.8, 1.8 | 128.5 | 5.67, d, 9.8 | 128.4 | 5.69, ddd, 9.8, 1.6, 1.6 | 128.3 | 5.73, ddd, 9.9, 2.0, 2.0 | 128.6 |
| 11 | 6.11, ddd, 9.9, 3.4, 3.4 | 133.1 | 6.11, ddd, 9.8, 4.2, 4.2 | 132.9 | 6.09, ddd, 9.8, 3.7, 3.7 | 132.8 | 6.12, ddd, 9.9, 3.5, 3.5 | 133.0 |
| 12a | 2.61, m/2.49, m | 27.2 | 2.63, m/2.50, m | 27.2 | 2.61, m/2.50, m | 27.1 | 2.62, m/2.52, m | 27.2 |
| 13a | 2.85, m/2.52, m | 36.2 | 2.83, m/2.50, m | 36.0 | 2.83, m/2.52, m | 36.0 | 2.90, m/2.54, m | 36.2 |
| 14 | 206.9 | 206.8 | 206.7 | 206.7 | ||||
| 1' | 4.92, d, 7.5 | 101.6 | 5.25, d, 8.0 | 99.9 | 4.93, d, 7.8 | 101.5 | 4.57, d, 7.7 | 106.7 |
| 2' | 3.42, dd, 9.0, 7.5 | 74.3 | 5.16, d, 8.0, 9.5 | 74.9 | 3.38, dd, 9.5, 7.8 | 74.0 | 3.45, dd, 8.8, 7.7 | 74.8 |
| 3' | 3.46, dd, 9.0, 9.0 | 77.2 | 3.78, m | 75.2 | 3.42, dd, 9.5, 9.5 | 76.9 | 3.39, dd, 8.8, 8.8 | 77.0 |
| 4' | 3.39, dd, 9.0, 9.0 | 70.8 | 3.54, m | 71.0 | 3.30, dd, 9.5, 9.5 | 70.6 | 3.34, dd, 8.8, 8.8 | 70.6 |
| 5' | 3.42, ddd, 9.0, 5.4, 1.8 | 77.1 | 3.54, m | 77.7 | 3.68, ddd, 9.5, 6.5, 2.0 | 74.7 | 3.29, ddd, 8.8, 5.2, 2.4 | 77.0 |
| 6'a | 3.77, dd, 12.0, 1.8 | 62.2 | 3.84, m | 62.1 | 4.53, dd, 12.0, 2.0 | 65.7 | 3.75, dd, 12.0, 2.4 | 62.1 |
| 6'b | 3.61, dd, 12.0, 5.4 | 3.69, m | 4.24, dd, 12.0, 6.5 | 3.62, dd, 12.0, 5.2 | ||||
| 1'' | 166.3 | 170.7 | ||||||
| 2'' | 130.3 | 78.7 | ||||||
| 3'' | 8.05, d, 7.6 | 130.3 | 5.74, ddd, 9.8, 1.7, 1.7 | 128.3 | ||||
| 4'' | 7.50, dd, 7.6, 7.6 | 129.4 | 6.12, ddd, 9.8, 3.5, 3.5 | 132.9 | ||||
| 5'' | 7.63, dd, 7.6, 7.6 | 134.2 | 2.61, m/2.50, m | 27.1 | ||||
| 6'' | 7.50, dd, 7.6, 7.6 | 129.4 | 2.83, m/2.52, m | 36.0 | ||||
| 7'' | 8.05, d, 7.6 | 130.3 | 207.1 | |||||
3. Experimental Section
3.1. General Information
3.2. Isolation and Identification of Salicortin (1), HCH-Salicortin (2), Tremulacin (3) and Idescarpin (4)
4. Conclusions
Supplementary Materials
Acknowledgments
Author Contributions
Conflicts of Interest
References
- Massad, T.J.; Trumbore, S.E.; Ganbat, G.; Reichelt, M.; Unsicker, S.; Boeckler, A.; Gleixner, G.; Gershenzon, J.; Ruehlow, S. An optimal defense strategy for phenolic glycoside production in Populus trichocarpa—Isotope labeling demonstrates secondary metabolite production in growing leaves. New Phytol. 2014, 203, 607–619. [Google Scholar] [CrossRef] [PubMed]
- Haruta, M.; Pedersen, J.A.; Constabel, C.P. Polyphenol oxidase and herbivore defense in trembling aspen (Populus tremuloides): cDNA cloning, expression, and potential substrates. Physiol. Plantarum. 2001, 112, 552–558. [Google Scholar] [CrossRef]
- Julkunen-Tiitto, R.; Hakulinen, J.; Meier, B. The response of growth and secondary metabolism to Melampsora rusts in field cultivated willow (Salix) clones. Acta Hortic. 1994, 381, 679–682. [Google Scholar]
- Boeckler, G.A.; Gershenzon, J.; Unsicker, S.B. Phenolic glycosides of the Salicaceae and their role as anti-herbivore defenses. Phytochemistry 2011, 72, 1497–1509. [Google Scholar] [CrossRef] [PubMed]
- Pentzold, S.; Zagrobelny, M.; Rook, F.; Bak, S. How insects overcome two-component plant chemical defence: Plant β-glucosidases as the main target for herbivore adaptation. Biol. Rev. 2014, 89, 531–551. [Google Scholar] [CrossRef] [PubMed]
- Clausen, T.P.; Reichardt, P.B.; Bryant, J.P.; Werner, R.A.; Post, K.; Frisby, K. Chemical model for short-term induction in quaking aspen (Populus tremuloides) foliage against herbivores. J. Chem. Ecol. 1989, 15, 2335–2346. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Lee, S.H.; Kang, J.; Yang, H.; Jeong, E.J.; Kim, H.P.; Kim, Y.C.; Sung, S.H. Salicortin-derivatives from Salix pseudo-lasiogyne twigs inhibit adipogenesis in 3T3-L1 cells via modulation of C/EBPα and SREBP1c dependent pathway. Molecules 2013, 18, 10484–10496. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.; Lee, H.H.; Lee, J.-K.; Ye, S.-K.; Kim, S.H.; Sung, S.H. Anti-adipogenic activity of compounds isolated from Idesia polycarpa on 3T3-L1 cells. Bioorg. Med. Chem. Lett. 2013, 23, 3170–3174. [Google Scholar] [CrossRef] [PubMed]
- Martineau, L.C.; Muhammad, A.; Saleem, A.; Herve, J.; Harris, C.S.; Arnason, J.T.; Haddad, P.S. Anti-adipogenic activities of Alnus incana and Populus balsamifera bark extracts, part II: Bioassay-guided identification of actives salicortin and oregonin. Planta Med. 2010, 76, 1519–1524. [Google Scholar] [CrossRef] [PubMed]
- Baek, S.; Kim, D.; Lee, C.; Kho, Y.; Lee, C. Idescarpin isolated from the fruits of Idesia polycarpa inhibits melanin biosynthesis. J. Microbiol. Biotechnol. 2006, 16, 667–672. [Google Scholar]
- Kim, S.H.; Jang, Y.P.; Sung, S.H.; Kim, Y.C. Inhibitory activity of phenolic glycosides from the fruits of Idesia polycarpa on lipopolysaccharide-induced nitric oxide production in BV2 microglia. Planta Med. 2006, 72, 167–169. [Google Scholar]
- Thieme, H. Isolierung eines neuen Phenolglucosids aus Salix purpurea. Pharmazie 1964, 11, 681. [Google Scholar]
- Babst, B.A.; Harding, S.A.; Tsai, C.-J. Biosynthesis of phenolic glycosides from phenylpropanoid and benzenoid precursors in Populus. J. Chem. Ecol. 2010, 36, 286–297. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.B.; Kim, H.W.; Lee, M.; Lee, H.H.; Kim, S.H.; Kang, S.K.; Sung, S.H. Isolation and structure elucidation of (–)-idescarparide, a new spiro compound from Idesia polycarpa. Tetrahedron Lett. 2014, 55, 5447–5449. [Google Scholar] [CrossRef]
- Rehill, B.; Clauss, A.; Wieczoek, L.; Whitham, T.; Lindroth, R. Foliar phenolic glycosides from Populus fremontii, Populus angustifolia and their hybrids. Biochem. Syst. Ecol. 2005, 22, 125–131. [Google Scholar] [CrossRef]
- Rasmussen, B.; Nkurunziza, A.-J.; Witt, M.; Oketch-Rabah, H.A.; Jaroszewski, J.W.; Stærk, D. Dovyalicin-type spermidine alkaloids from Dovyalis species. J. Nat. Prod. 2006, 69, 1300–1304. [Google Scholar] [CrossRef] [PubMed]
- Domisse, R.A.; van Hoof, L.; Vlietinck, A.J. Structural analysis of phenolic glycosides from Salicaceae by NMR spectroscopy. Phytochemistry 1986, 25, 1201–1204. [Google Scholar] [CrossRef]
- Maansson, M.; Phipps, R.K.; Gram, L.; Munro, M.H.G.; Larsen, T.O.; Nielsen, K.F. Ex-plorative solid-phase extraction (E-SPE) for accelerated microbial natural product discovery, dereplication, and purification. J. Nat. Prod. 2010, 73, 1126–1132. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.R.; Aichinger, C.; Rinner, U.; Hudlicky, T. Chemoenzymatic synthesis of idesolide from benzoic acid. Synlett 2011, 5, 725–729. [Google Scholar]
- Sample Availability: Samples of the compounds salicortin, HCH-salicortin, tremulacin and idescarpin are available from the authors.
© 2015 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license ( http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Feistel, F.; Paetz, C.; Lorenz, S.; Schneider, B. The Absolute Configuration of Salicortin, HCH-Salicortin and Tremulacin from Populus trichocarpa × deltoides Beaupré. Molecules 2015, 20, 5566-5573. https://doi.org/10.3390/molecules20045566
Feistel F, Paetz C, Lorenz S, Schneider B. The Absolute Configuration of Salicortin, HCH-Salicortin and Tremulacin from Populus trichocarpa × deltoides Beaupré. Molecules. 2015; 20(4):5566-5573. https://doi.org/10.3390/molecules20045566
Chicago/Turabian StyleFeistel, Felix, Christian Paetz, Sybille Lorenz, and Bernd Schneider. 2015. "The Absolute Configuration of Salicortin, HCH-Salicortin and Tremulacin from Populus trichocarpa × deltoides Beaupré" Molecules 20, no. 4: 5566-5573. https://doi.org/10.3390/molecules20045566